Abstract
The objective of this study was to determine the association between a specialist clinical pharmacist working in collaboration with medical staff and prescribing in peripheral arterial disease (PAD). A retrospective cohort study was conducted comparing the influence of a dedicated clinical pharmacist on two samples of patients admitted to a single vascular surgery unit in either 2007 (control group) prior to implementation of a comprehensive clinical pharmacy service or 2009 (comparison group) post implementation. Data were obtained via review of medical records and electronic reports. A total of 685 patients were identified, resulting in 964 admissions. The patient to pharmacist ratio decreased from 62 to 33 patients per day in 2009. More patients were initiated on an antiplatelet (OR 4.6, 95% CI 2.26 to 9.53, p<0.001) and statin (OR 3.4, 95% CI 1.97 to 6, p<0.001) in 2009 compared to 2007. Risk factor modification increased in 2009, resulting in action being taken more often for HbA1c>7% (OR 3.45, 95% CI 1.64 to 7.27, p=0.001), total cholesterol >4 mmol/L in females (OR 14.5, 95% CI 2.67 to 78.6, p=0.002) and blood pressure above target (OR 1.9, 95% CI 1.01 to 3.73, p=0.05) when a comprehensive clinical pharmacist service was available. There was a non-significant reduction in mortality (18.7% (65) to 14.2% (46), p=0.13) and cardiovascular outcomes (5.5% (19) to 4.3% (14), p=0.44) within 12 months of discharge. In conclusion, prescribing of evidence-based medication for PAD and risk factor modification increased with a comprehensive clinical pharmacist service. This study provides important insight into optimising treatment in this patient group and how a pharmacist can be a helpful addition to the multidisciplinary team.
Introduction
Peripheral arterial disease (PAD) is a highly prevalent condition worldwide, particularly in the elderly population. Both symptomatic and asymptomatic PAD is associated with increased cardiovascular risk and PAD is now considered a coronary artery disease (CAD) risk equivalent in terms of cardiac events and mortality. 1
Prevention of CAD events must therefore be a major factor in the management of all patients presenting with lower extremity atherosclerosis, with rigorous assessment of cardiovascular risk factors routinely performed. Subsequent implementation of risk factor modification strategies including pharmacotherapy is also essential and is supported by multiple international guidelines.2–7 Risk factor modification is, however, not performed optimally in patients with PAD. The REACH 8 registry and PARTNERS 9 study have shown that cardiovascular risk factor modification and control in patients with PAD is not achieved as frequently as those with primary CAD or cerebrovascular disease.
Various studies have shown an association between clinical pharmacy services and a reduced length of hospital stay and mortality.10–12 A clinical pharmacist specialising in the management of cardiovascular disease may be able to improve uptake of cardiovascular risk factor modification strategies. The Pharmacist Influence on Prescribing in Peripheral Arterial Disease (PIPER) study was proposed to examine the impact of a clinical pharmacist, working in collaboration with medical staff, on the level of risk factor modification in the inpatient setting for patients with arterial disease.
Method
PIPER was a retrospective cohort study aiming to determine the influence of a dedicated vascular unit clinical pharmacist on prescribing. Two samples of patients admitted to a vascular surgery unit in either 2007 (control group), prior to implementation of a comprehensive clinical pharmacy service, or 2009 (comparison group), after implementation, were compared. The primary outcome measures were threefold: (1) antiplatelet prescribing; (2) statin prescribing; (3) appropriate cardiovascular risk factor management compared to internationally recognised targets. Secondary outcome measures were cardiovascular events and mortality.
Patient samples from 2007 and 2009 were studied due to the expected difference in patient to pharmacist ratio arising from the full-time employment of a pharmacist specialising in the management of cardiovascular disease. The year 2008 represented the transition (washout) period between the comparison groups with reference to patient load.
Patient selection
The Adelaide Vascular Audit database was used to identify patients admitted with arterial disease from January to December in 2007 and 2009. Eligible patients were those admitted for treatment or investigation of aneurysmal disease (abdominal aortic aneurysm repair or size >5.5 cm in men or >5 cm in women), carotid disease (undergoing carotid endarterectomy) or PAD (documented history of PAD, ankle–brachial pressure index <0.9 or undergoing a revascularisation intervention). Patients were excluded if they were admitted as a planned day case without complications or were admitted for less than 24 hours.
Data collection
Retrospective data collection involved examination of inpatient medical records and electronic reports to record patient demographics, diagnosis, past medical history, previous admissions, risk factor control, vital signs (blood pressure and heart rate), medications on admission and discharge, contraindications to indicated medications, relevant laboratory data, pharmacist medication history and pharmacist interaction with patients during admission. Data collection was undertaken by two authors (LBB, MS) accredited in the use of the hospital clinical information system. Both were non-blinded due to the nature of the demographics collected.
Analysis of antiplatelet and statin prescribing focused on the patients not already prescribed an antiplatelet agent or a statin on admission. It was assumed that unless a patient had a contraindication to therapy (according to the product information) or was prescribed warfarin that they should be considered for antiplatelet therapy.
Risk factor targets were based on national and international guidelines.3,13 Blood pressure was recorded as the average of the blood pressure readings during the first 24 hours. If this appeared to differ significantly from the rest of the admission, another 24-hour average was taken. Target blood pressure was less than 140/90 mmHg, or for those with diabetes or renal disease it was less than 130/80 mmHg. 3 For diabetic control the target glycosylated haemoglobin (HbA1c) was <7%, 3 total cholesterol (TC) target was <4 mmol/L. 13 HbA1c and lipids were obtained from the state-wide public health pathology database in the 6 and 12 months prior to admission, respectively, due to time frame recommendations for monitoring from national guidelines.13,14 Each risk factor was analysed independently; only patients above each individual target were analysed for appropriate response. Action taken in response to a risk factor above the recommended target was defined as appropriate or inappropriate. Appropriate action was a change in therapy; for example, an increase in statin dose in response to a TC above target, or a valid documented reason for not changing therapy such as a dose change since the last result or already prescribed maximal therapy. Inappropriate action was when no action was taken and no rationale documented.
Cardiovascular outcomes were defined as a myocardial infarct or ischaemic stroke. They were determined through the International Classification of Diseases (ICD) of primary and secondary diagnosis on the state-wide public health information database for subsequent admissions within 12 months from the date of first discharge. Mortality data were obtained via linkage with the Australian Institute of Health and Welfare’s National Death Index (NDI). All analyses were adjusted for appropriate confounders.
A pharmacist interaction was deemed to have occurred when there was a medication history documented, a clinical chart review which was determined from a pharmacist signature on the chart, or documentation of discharge counselling. Clinical pharmacy services operated as a 5-day per week service; therefore, if a patient was admitted for 7 days they could have a pharmacist interaction a maximum of five times (i.e. on 70% of admitted days). The patient to pharmacist ratio was the number of patients the pharmacist was allocated to review each day.
The study was approved by the Southern Adelaide Clinical Human Research and Ethics Committee.
Statistics
Chi-squared tests were used to compare differences in proportions. The t-test or Mann–Whitney U-test were used to compare differences in means according to whether the variable was normally distributed or not. Logistic regression analysis was used to examine the possible associations between study years, prescribing of medications at discharge, and appropriate action taken when not at risk factor targets. Clustering over individuals and using robust standard errors to account for correlated readings, adjusting for age, sex, CAD and cerebrovascular disease. Hosmer–Lemeshow tests were performed to assess goodness-of-fit. Cox proportional hazards modelling was used to assess differences in cardiovascular events and mortality between years. The proportional hazards assumption was verified for all survival models.
A p-value less than 0.05 (two-tailed) was considered statistically significant. All statistical analyses were performed on Intercooled Stata 12.0 for windows (StataCorp LP).
Results
In 2007 there were 351 patients with 485 admissions and in 2009 there were 334 patients with 479 admissions. The two groups were well matched in terms of baseline characteristics (Table 1).
Baseline characteristics.
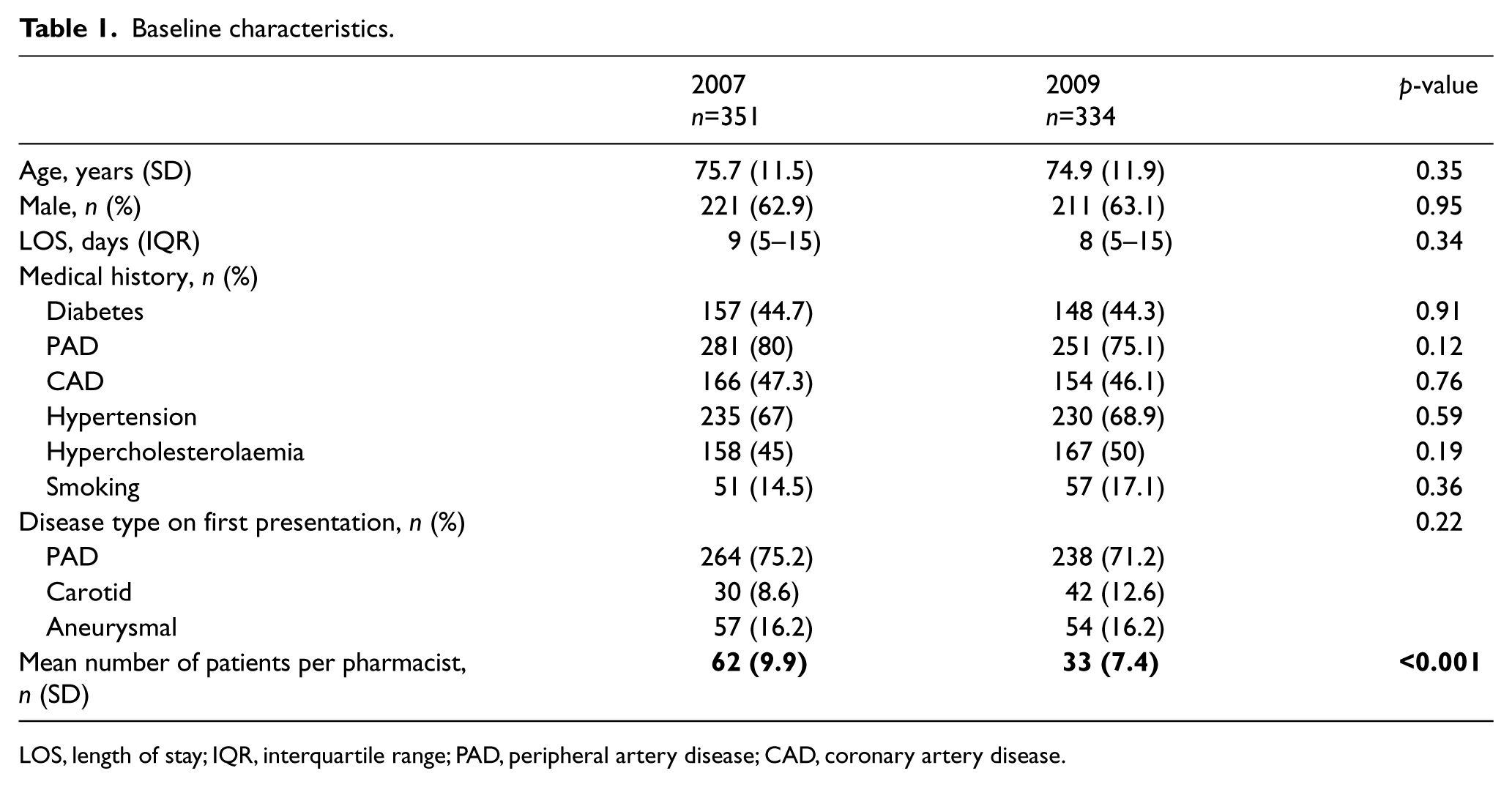
LOS, length of stay; IQR, interquartile range; PAD, peripheral artery disease; CAD, coronary artery disease.
The patient to pharmacist ratio was reduced from a mean of 62 patients per day in 2007 to 33 patients per day in 2009 (p<0.001). In 2009, the number of patients experiencing at least one interaction with the pharmacist was 97% (465) compared to 72% (349) in 2007 (p<0.001). On average, a patient to pharmacist interaction occurred on 44% of admitted days in 2009 compared to 16% of admitted days in 2007 (p<0.001). Furthermore, a documented medication history was prepared for the majority of patients in 2009 (0% vs 97%, p<0.001) and more patients were reviewed on discharge in 2009 (9.3% vs 73.5%, p<0.001) (Table 2).
Proportion of patients receiving pharmacist review.
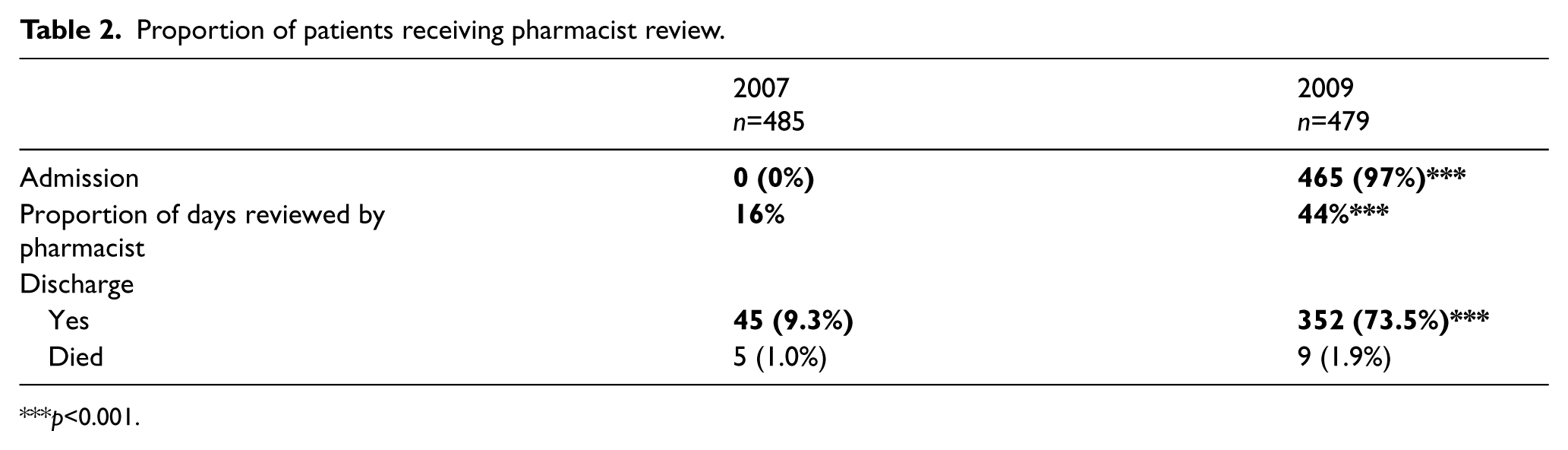
p<0.001.
Those who were not already prescribed an antiplatelet prior to admission were more likely to be initiated on therapy during their admission in 2009 (OR 4.6, 95% CI 2.26 to 9.53, p<0.001) independent of age, sex, CAD and cerebrovascular disease (Table 3). The prescribing of antiplatelets overall on discharge was 80.4% (315/392) in 2007 and 94% (328/349) in 2009.
Initiation of essential medication.
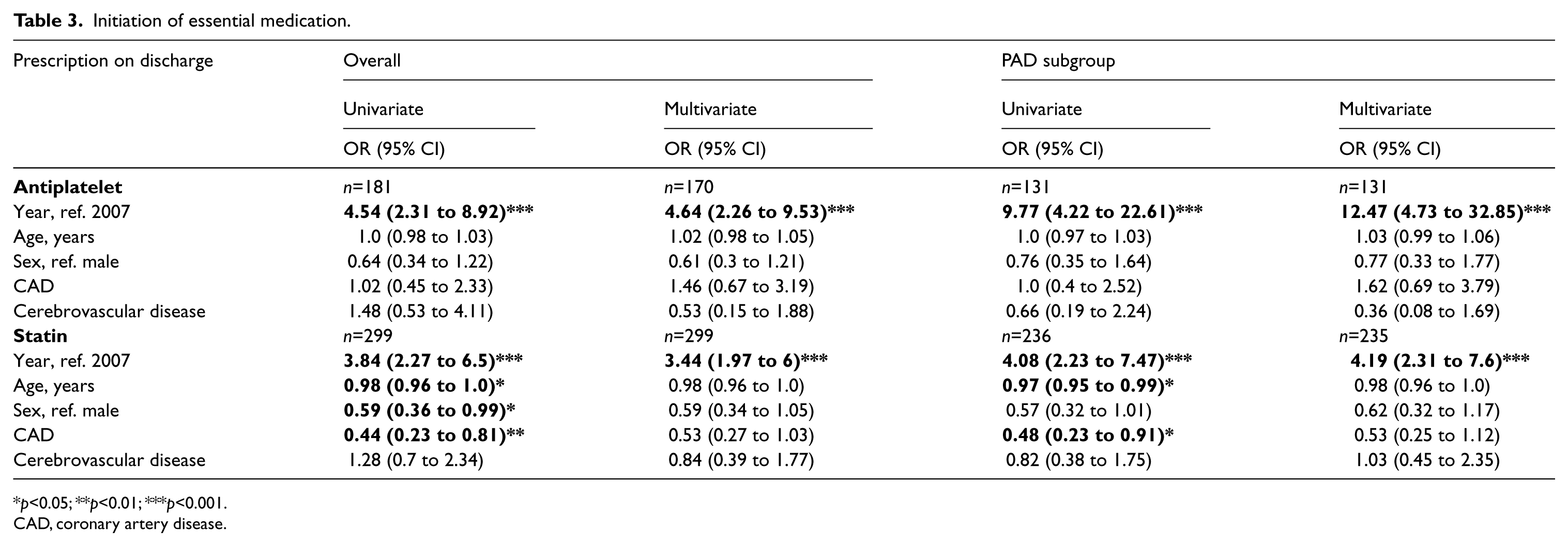
p<0.05; **p<0.01; ***p<0.001.
CAD, coronary artery disease.
Subgroup analysis showed that the majority of patients initiated on an antiplatelet during admission were patients with PAD. There was no difference in antiplatelet prescribing in the aneurysmal subgroup (OR 0.34, 95% CI 0.07 to 1.65, p=0.18), and all patients not already on an antiplatelet in the carotid group were initiated on therapy in 2007 (one patient) and 2009 (10 patients). Therefore, the potential influence of a clinical pharmacist working with the medical team was more pronounced when analysing antiplatelet prescribing in the PAD subgroup alone (OR 12.47, 95% CI 4.73 to 32.85, p<0.001) (Table 3). The proportion of patients prescribed an antiplatelet on discharge in the PAD group increased from 79.3% (234/295) in 2007 to 97.7% (259/265) in 2009.
Patients discharged on a statin increased from 66% (239/362) in 2007 to 88% (298/336) in 2009. There was a significant increase in the proportion of patients initiated on therapy in 2009 (OR 3.44, 95% CI 1.97 to 6, p<0.001) (Table 3). The impact was most marked in the PAD subgroup (OR 4.19, 95% CI 2.31 to 7.6, p<0.001).
With respect to diabetic control, for patients not at target (HbA1c >7%) where an adjustment in therapy was warranted, appropriate action was taken more often in 2009 (OR 3.45, 95% CI 1.64 to 7.27, p=0.001). It was also more likely that appropriate action was taken in response to an elevated blood pressure in 2009 (OR 1.9, 95% CI 1.01 to 3.73, p=0.05). When lipids were not at target there was an interaction between sex and year. There was no difference in the treatment of men between years (OR 1.9, 95% CI 0.78 to 4.67, p=0.15), whereas women were much more likely to be initiated on therapy in 2009 (OR 14.5, 95% CI 2.67 to 78.6, p=0.002) compared to 2007 (Table 4).
Risk factor modification for patients above risk factor targets.
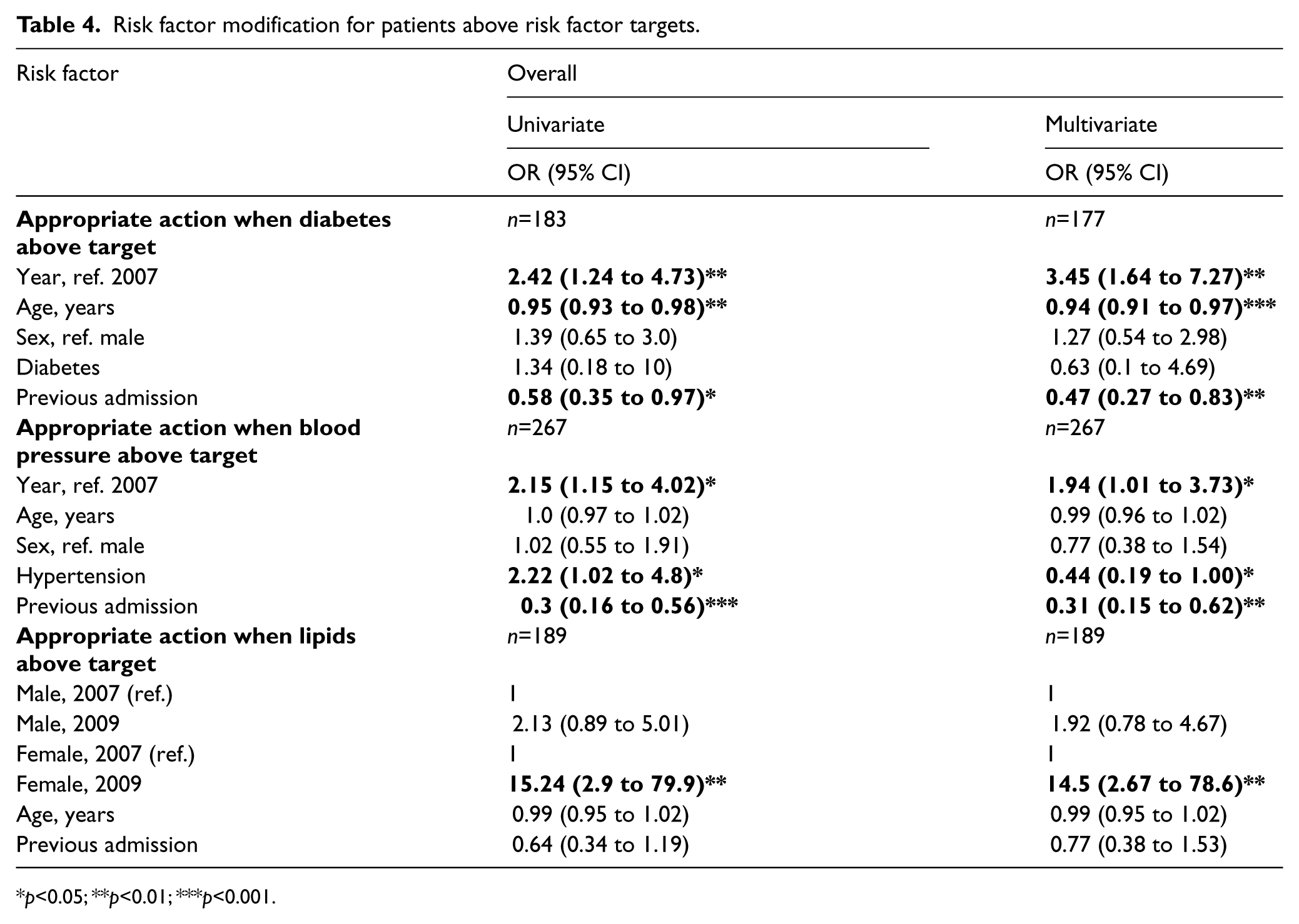
p<0.05; **p<0.01; ***p<0.001.
There was no reduction in non-fatal cardiovascular events within 12 months of first discharge, with 19 events in 2007 and 14 events in 2009 (p=0.44). While there was a trend towards a reduction in mortality within 12 months from the date of first discharge (18.7% (65) in 2007 compared to 14.2% (46) in 2009), it was not statistically significant (p=0.13) (Table 5).
Secondary outcomes.
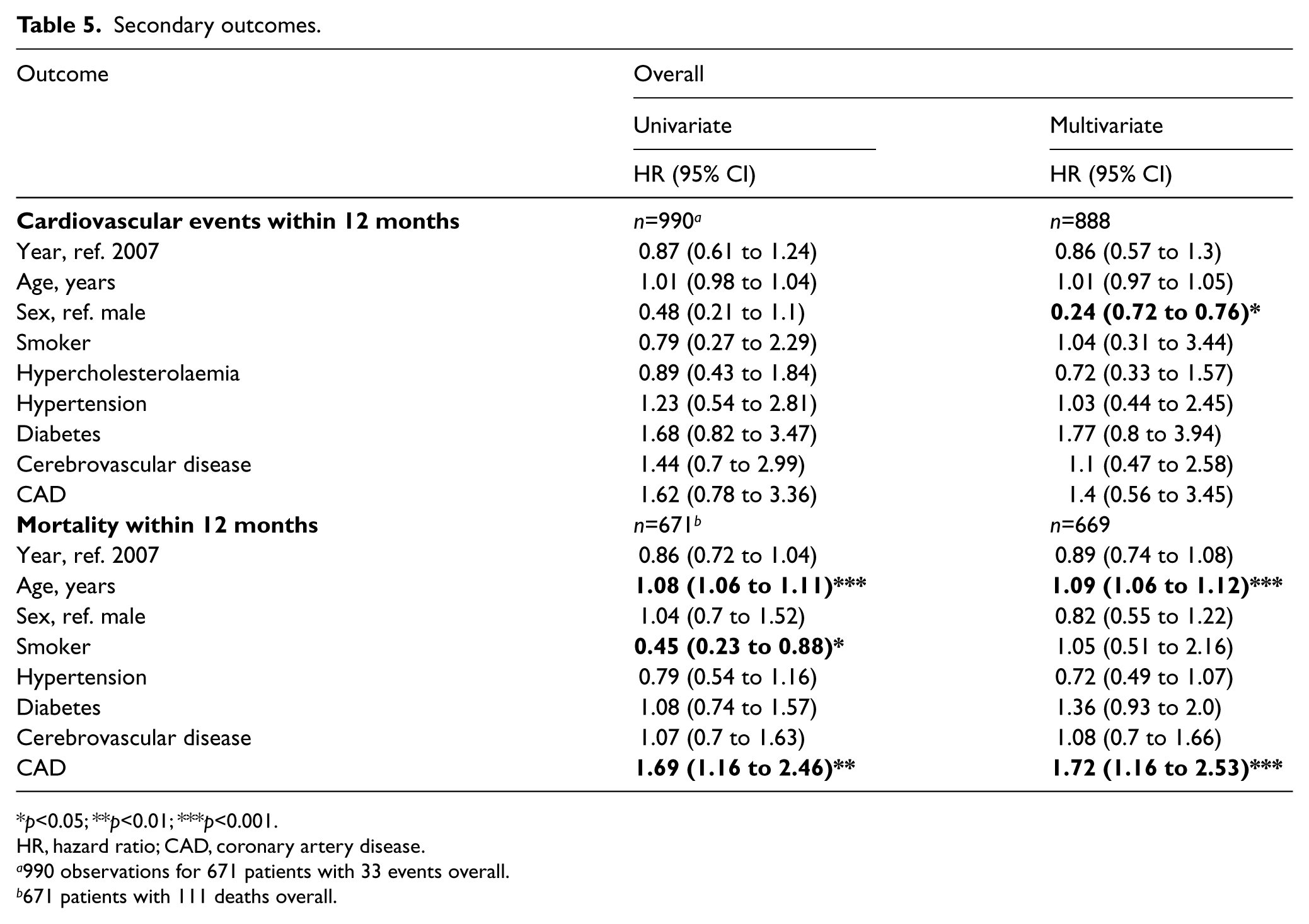
p<0.05; **p<0.01; ***p<0.001.
HR, hazard ratio; CAD, coronary artery disease.
990 observations for 671 patients with 33 events overall.
671 patients with 111 deaths overall.
Discussion
The PIPER study revealed that patients were reviewed more often and more comprehensively by pharmacists in 2009 compared to 2007. This was in part brought about by a reduction in the patient to pharmacist ratio. The result was an increase in prescribing of antiplatelets and statins as well as initiation of therapeutic changes for patients not reaching accepted targets for diabetes, hyperlipidaemia and hypertension. While there was a trend suggesting a possible reduction in cardiovascular events and mortality within 12 months in 2009, this did not reach statistical significance.
Comparisons with other studies
The need for medical management of cardiovascular risk factors in those with PAD is well established and supported by international guidelines.2–7 Although definitive guidelines exist, PAD patients continue to be undertreated in terms of medical management and risk factor modification. The REACH registry showed that in the outpatient setting patients with PAD alone were prescribed antiplatelets (76% vs 84%) and statins (50% vs 73%) less often than those with PAD and CAD or cerebrovascular disease. 8 Overall and individual risk factor control was also lower in the PAD only group compared to those without PAD.
The PARTNERS 9 study also demonstrated that in primary care PAD patients were treated less aggressively than those with cardiovascular disease. Antiplatelets were prescribed less often (54% vs 71%, p<0.001), while hypertension (88% vs 95%, p<0.001) and hyperlipidaemia (56% vs 73%, p<0.001) were treated less frequently in those with established PAD. This was demonstrated more recently in a larger study in the primary care setting. 15 The significance of these results should not be understated as the primary care setting may represent the ideal environment for early prophylactic intervention in this cohort of patients.
Less evidence exists for the level of risk factor management in the inpatient setting where one would expect intervention to be greatest if not already achieved, particularly when admitted under specialist care. Inpatients admitted for carotid intervention or PAD under a vascular surgery unit were analysed. 16 Carotid patients were more likely to be treated with an antithrombotic (97% vs 75%) and a cholesterol-lowering medication (63% vs 36%) than PAD patients. Those with PAD received these therapies more frequently if they had previous CAD or cerebrovascular disease (polyvascular disease). Antiplatelet use was 2.93 (1.56–5.51) times more likely and statin therapy was 2.41 (1.32–4.39) times more likely in polyvascular disease patients than those with PAD alone. This demonstrates that under-treatment occurs not only in primary care but also in the acute care setting.
The rate of prescribing antiplatelets in PIPER in the PAD subgroup increased from around 80% to over 97%. This is superior to the results reported by the REACH registry 8 at 84% in those with polyvascular disease and 75% by Gen Teh et al. 16 Similarly, statin prescribing for PAD in PIPER exceeded that of the previously mentioned studies, increasing from 66% to 88% compared to 73% for polyvascular disease in REACH 8 and 36% in Gen Teh et al. 16 This shows prescribing in PIPER was slightly below that of the REACH registry in 2007 and there was significant improvement with the introduction of a full-time clinical pharmacist service.
The benefits of a clinical pharmacy service on medication errors, medication costs and mortality have been well documented.10–12 In the acute care setting, time constraints, particularly for junior medical staff, can result in risk factor management not being a major priority. Pharmacists are in a prime position to ensure the quality use of medicines (QUM) and patient safety due to their in-depth knowledge of medications and to focus on these aspects of patient care. While other professionals are able to follow algorithms for prescribing, pharmacists can provide advice on interactions with other drugs or disease, contraindications and adverse reactions. Despite this there is little evidence assessing the impact of a specialist pharmacist on inpatients with vascular disease.
The influence of a pharmacist on risk factor management in CAD has been studied in the outpatient setting. A recent meta-analysis of 30 trials showed that pharmacist care resulted in significant reductions in blood pressure, total cholesterol, low-density lipoproteins (LDLs) and smoking cessation. 17 Seeing as the risk factors for PAD and CAD are the same, there is no reason why PAD patients could not gain the same benefit from such interventions. This influence is not restricted to outpatient clinics as was demonstrated in PIPER, where benefit was shown for inpatients. While these studies were in different care settings they all support the theory of improved prescribing with pharmacist involvement. Of course, consideration should also be given to the potential influence of greater awareness of the need to manage cardiovascular risk factors leading to improvements in care over time.
Strengths and weaknesses
The level of adherence by patients and compliance of general practitioners to risk factor management changes was not able to be determined due to the retrospective nature of the study. Compliance can be an issue in this patient group 18 due to the perception that PAD is not as severe as CAD, which is evident in the under-treatment of PAD.8,9,15,16 In addition to compliance, the time taken to gain benefit from treatment changes must be considered. Risk factor management and prescribing of antiplatelets and statins have a wealth of evidence to show that they decrease cardiovascular outcomes. This is supported by the multiple guidelines2–7 that exist for the treatment of PAD, but there is undoubtedly a time lag between treatment change or medication initiation and an improvement in adverse outcomes. The Heart Protection Study 19 showed the benefit of statin therapy on major vascular events in those with any cardiovascular disease (including coronary, cerebral or peripheral), diabetes or hypertension. This difference was not statistically significant at 1 year but was during each subsequent year – showing that it may take time to attain benefit from statin therapy. Therefore, the follow-up period for PIPER may not have been long enough to allow for the full benefit of medication changes on cardiovascular outcomes to be shown.
While retrospective studies have the benefit of reducing certain bias such as the Hawthorne effect (e.g. increasing prescribing of antiplatelets while study data are collected), they carry with them limitations. One such limitation was the lack of follow-up data on adverse reactions to medications initiated. Part of the pharmacist’s role is to promote and ensure the quality use of medicines, which is defined as the judicious, appropriate, safe and efficacious use of medicines. Therefore, all medication adjustments or initiations suggested by the pharmacist were made with this in mind and the risk versus benefit for each patient considered. While this study focuses on risk factor management medications, QUM applies to all areas of medication use. The pharmacist’s role has been sustained and expanded to include quality improvement research.
Conclusion
The results of PIPER show that prescribing of essential medications and risk factor modification increased in 2009 when a comprehensive clinical pharmacist service worked in collaboration with the medical team. This would be expected over time to have an impact on cardiovascular events and mortality but the follow-up period may not have been long enough for the difference to reach statistical significance.
Considering the high prevalence of PAD in the elderly community, only a small proportion of patients have a hospital admission and it is usually those with severe disease. This raises the question of whether our focus should shift towards intervening earlier in the disease process in order to gain maximal benefit.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Presented at the Australia and New Zealand Society for Vascular Surgery Conference, 12–15 November 2011, Brisbane, Australia.
