Abstract
Acute limb ischemia (ALI) is defined as a sudden decrease (<14 days) in limb perfusion causing a potential threat to limb viability. Endovascular treatment of ALI is an effective and appropriate primary treatment strategy in patients with a viable acutely ischemic limb due to a recent arterial occlusion. We present a case of acute limb ischemia and discuss percutaneous interventional treatment strategies including catheter-directed thrombolysis, rheolytic thrombectomy, and utilization of distal protection devices.
Keywords
Background
Acute limb ischemia (ALI) is defined as a sudden decrease (<14 days) in limb perfusion causing a potential threat to limb viability. Endovascular treatment of ALI is an effective and appropriate primary treatment strategy in patients with a viable acutely ischemic limb due to a recent arterial occlusion. We present a case of acute limb ischemia and discuss percutaneous interventional treatment strategies.
Clinical history
The patient is an 84-year-old female complaining of severe left calf pain at rest. She presented with worsening claudication (Fontaine IIb) that began a week prior to admission and had increased in intensity. The patient also reported associated left lower extremity pallor, but denied paresthesias or motor dysfunction.
She had undergone percutaneous transluminal angioplasty and stenting of the left superficial femoral artery (SFA) with Viabahn® (Gore, Flagstaff, AZ, USA) stents at an outside facility 5 months previously for an episode of critical limb ischemia (CLI), manifested as ischemic rest pain involving her left calf. Her medications included aspirin 81 mg daily. She had discontinued clopidogrel after 2 months following her lower extremity intervention.
On examination, her right common femoral, popliteal, posterior tibial and dorsalis pedis pulses were normal (2+). Her left common femoral pulse was 2+ but the left popliteal, posterior tibial and dorsalis pedis pulses were present by Doppler ultrasound only. There was no motor weakness or sensory loss. Arterial ultrasound demonstrated an occluded left superficial femoral artery (SFA). Her ankle–brachial index (ABI) was 0.9 on the right and 0.46 on the left.
Procedure
Based upon her clinical presentation and suspected left proximal SFA occlusion, the patient was brought urgently to the catheterization laboratory for percutaneous intervention. Contralateral common femoral artery (CFA) access was obtained with a 4 Fr × 11 cm sheath.
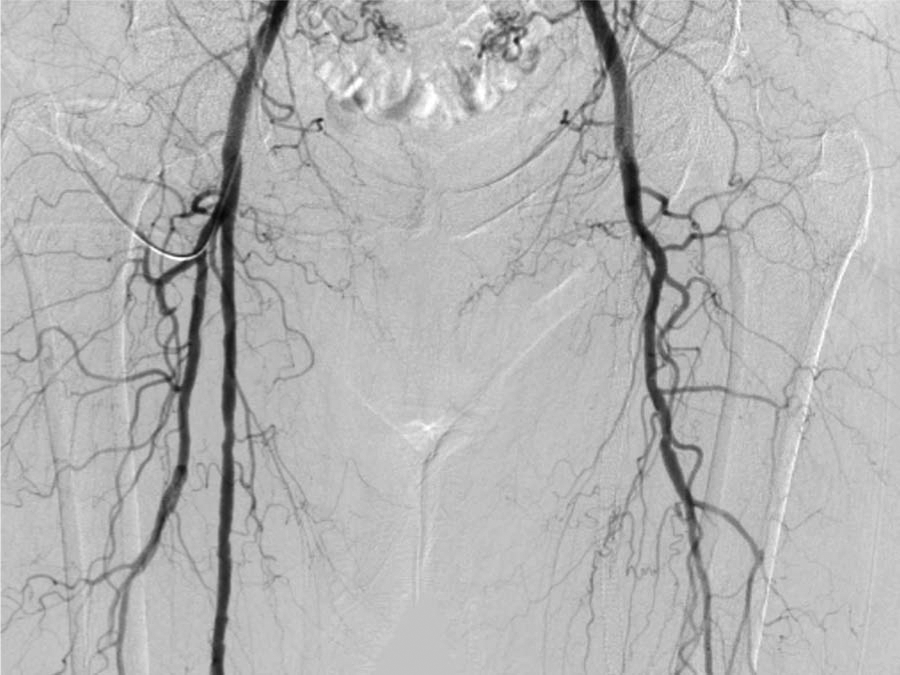
Digital subtraction angiography of the abdominal aorta at the level of the renal arteries, as well as an infrarenal abdominal aortogram with runoff, was performed using a 4 Fr pigtail catheter (Figures 1–4). Aortography revealed no significant aortoiliac disease. The proximal left SFA was occluded. The left profunda femoris (deep femoral) artery was patent, which is important for lower limb viability. There was reconstitution of the distal SFA above the knee via collaterals from the profunda femoris artery. The left popliteal artery was patent with luminal irregularities. Three-vessel infrapopliteal runoff was present.
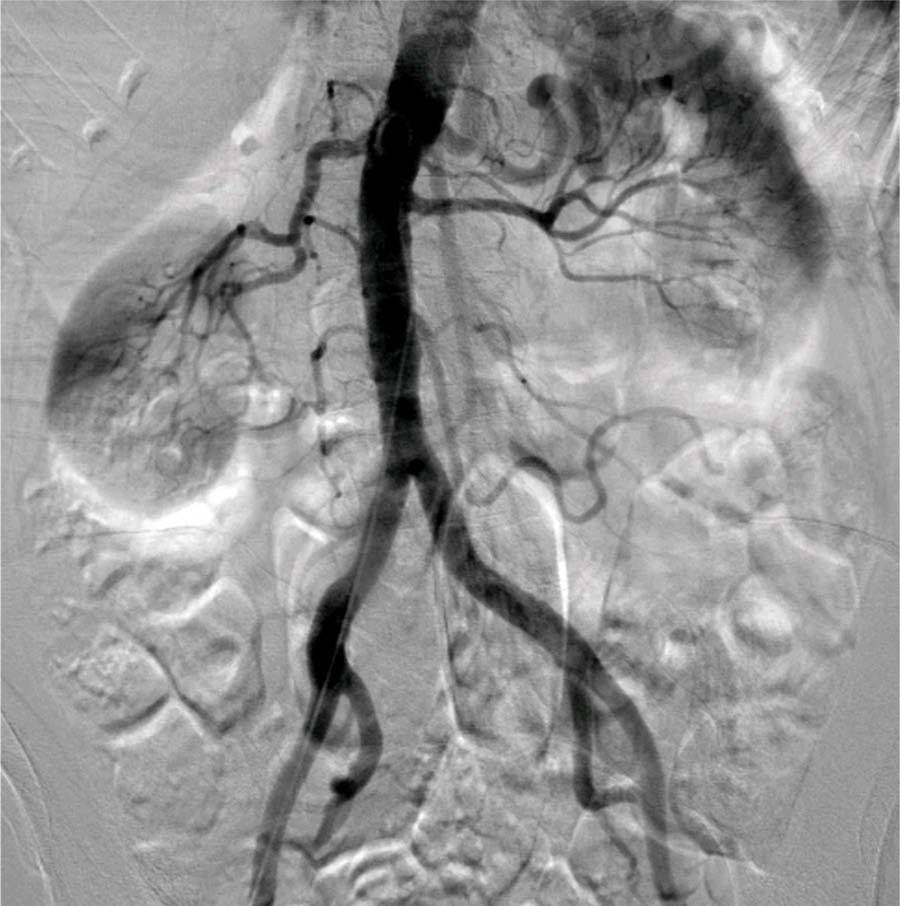
Digital subtraction angiography of the abdominal aorta with pigtail catheter.
Digital subtraction angiography showing proximal left superficial femoral artery occlusion.
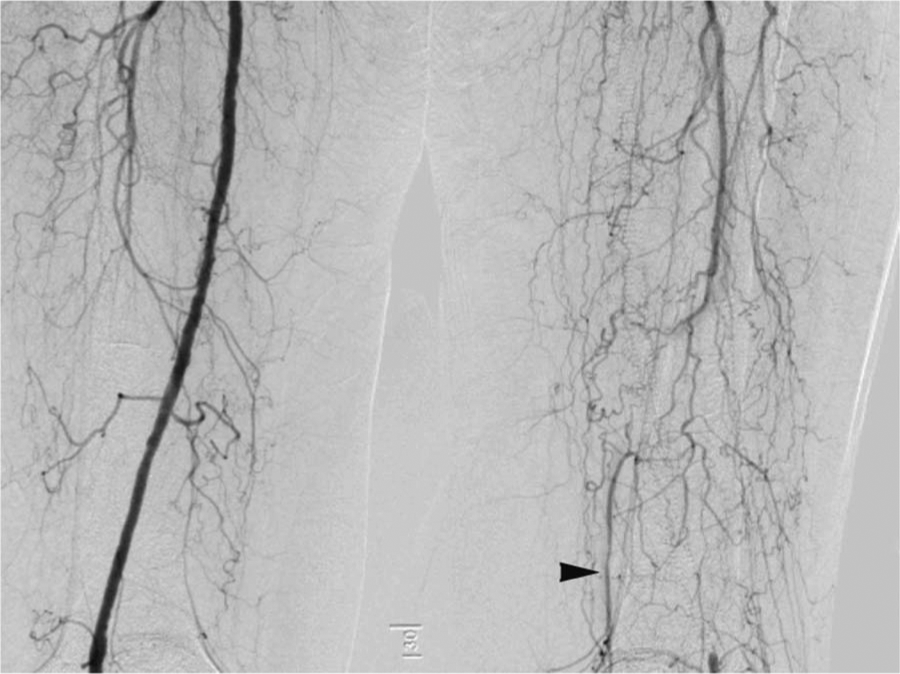
Digital subtraction angiography showing mid superficial femoral artery occlusion with geniculate collaterals (arrow).
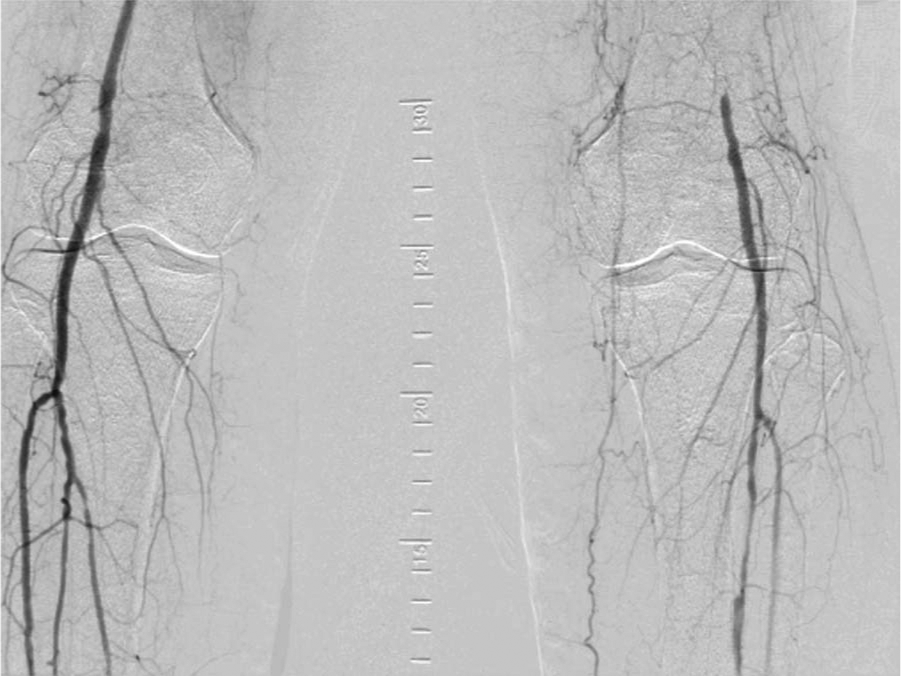
Digital subtraction angiography showing three-vessel runoff.
A 4 Fr internal mammary catheter (IMA) was advanced over a 0.035-in × 180-cm J-wire into the abdominal aorta. The J-wire was removed and the IMA catheter withdrawn to engage the ostium of the left common iliac artery. A 0.035-in × 180-cm Stiff Angled Glidewire® (Terumo, Ann Arbor, MI, USA) was advanced to the left common femoral artery. Next, the IMA catheter was advanced over the Glidewire and positioned in the left CFA. The Glidewire was removed and selective angiography revealed the left proximal SFA occlusion (Figure 5). A 0.035-in × 260-cm Amplatz Extra Stiff guidewire (Cook Medical, Bloomington, IN, USA) was inserted into the IMA catheter and positioned in the left CFA. The IMA catheter was removed and the 4 Fr sheath was exchanged over the Extra Stiff Amplatz guidewire for a 6 Fr 55-cm sheath (Raabe; Cook Medical) placed in the left CFA.
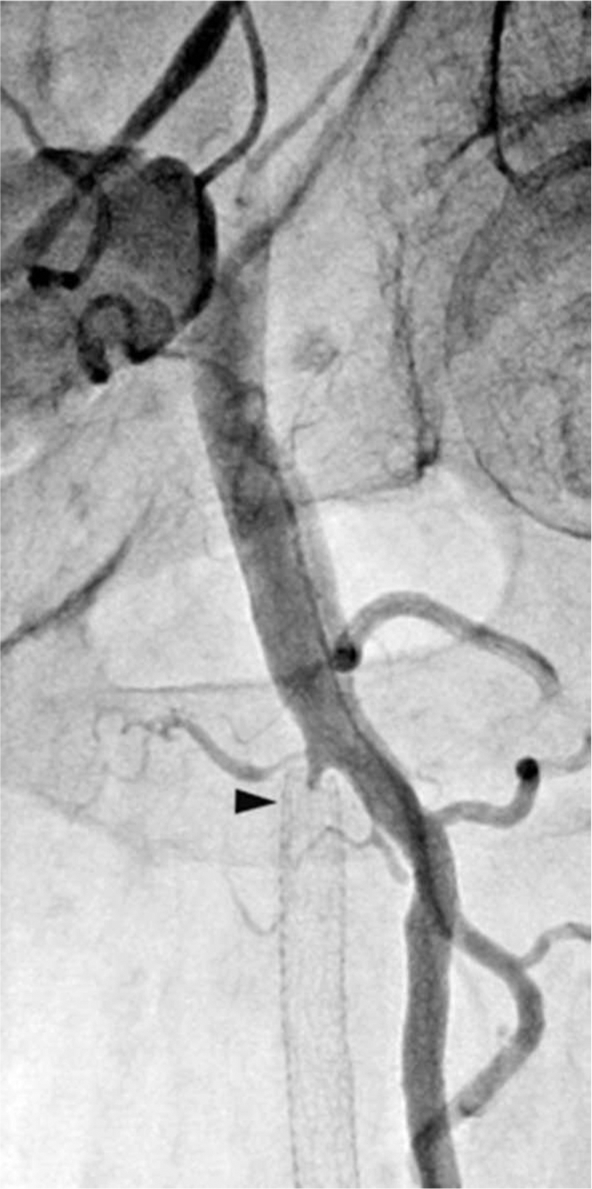
Selective angiography with left internal mammary catheter showing left superficial femoral artery occlusion (arrow).
The Glidewire was reinserted and used to cross the SFA occlusion and advanced to the left popliteal artery. A 4 Fr 125-cm multipurpose (MP) catheter was advanced into the left popliteal artery over the Glidewire. The Glidewire was removed and angiography through the catheter confirmed the intraluminal location of the catheter. The MP catheter was exchanged for a 4 Fr 135-cm × 20-cm McNamara infusion catheter (ev3, Irvine, CA, USA). Subsequent angiography revealed evidence of significant thrombus burden within the Viabahn stents in the left SFA (Figure 6).

Selective angiography via the infusion catheter (McNamara) of the superficial femoral artery. The Raabe sheath is positioned in the ostial left superficial femoral artery.
The patient received catheter-directed thrombolysis with alteplase (Activase®; Genentech, San Francisco, CA, USA) at 2 mg/h divided between the Raabe sheath and the infusion catheter for 8 hours with planned relook angiography. Unfractionated heparin was administered through the Raabe sheath at a constant infusion rate of 500 units/h. The Raabe sheath was positioned in the proximal left SFA and the multi-holed infusion catheter was placed in the distal SFA.
The next day the patient was brought back to the catheterization laboratory and angiography through the McNamara catheter revealed significant improvement but persistent thrombus within the SFA stents (Figure 7). The patient complained about remaining supine, making continued low-dose lysis unattractive. Mechanical thrombectomy (rheolytic thrombectomy) was chosen to expedite the thrombus removal and complete the case. A 0.014-in × 300-cm Platinum Plus™ (Boston Scientific, Natick, MA, USA) guidewire was advanced into the left popliteal artery and an embolic protection device (Spider; ev3) was advanced over the Platinum Plus guidewire.
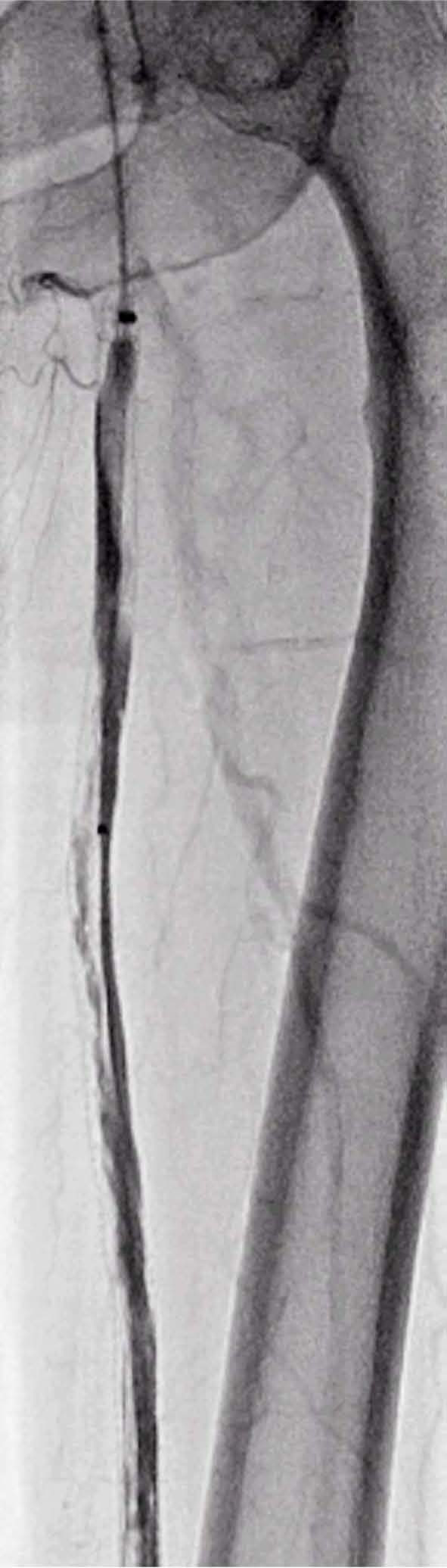
Relook angiography via the infusion catheter (McNamara).
The Platinum Plus guidewire was removed and the Spider filter positioned in the popliteal artery. The McNamara catheter was removed and thrombectomy of the SFA was performed with the Possis AngioJet (Medrad, Minneapolis, MN, USA) thrombectomy catheter. The thrombectomy catheter was removed and balloon angioplasty performed (5 × 40 mm) in the proximal and distal left SFA (10 atm for 10 sec) (Figures 8 and 9). The angioplasty balloon was removed and a Zilver (Cook Medical) 6 × 20 mm self-expanding nitinol stent was inserted and deployed across the distal left SFA lesion. Post-dilation of the distal left SFA stent (5 × 20 mm at 7 atm for 12 sec) and proximal left SFA stent was performed.
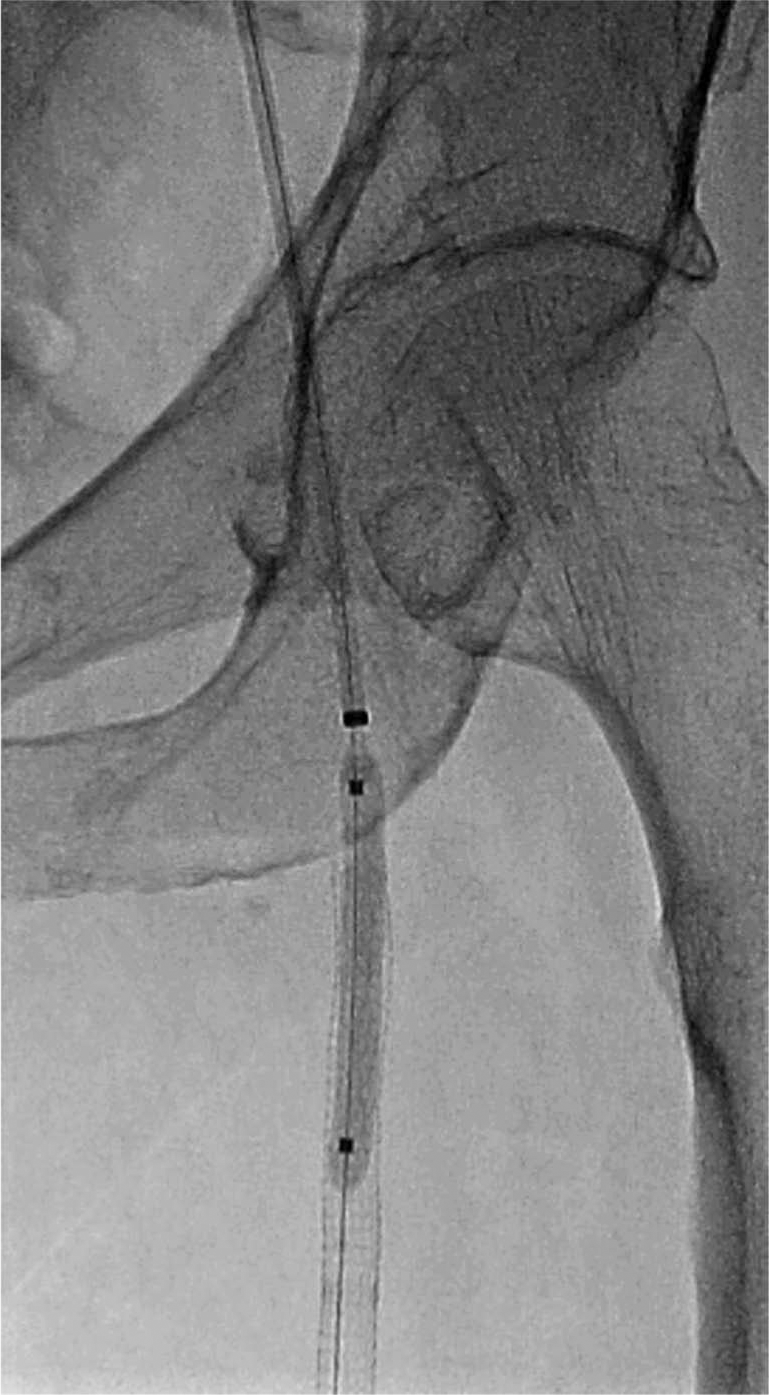
Digital angiography of percutaneous transluminal angioplasty of the proximal left superficial femoral artery.
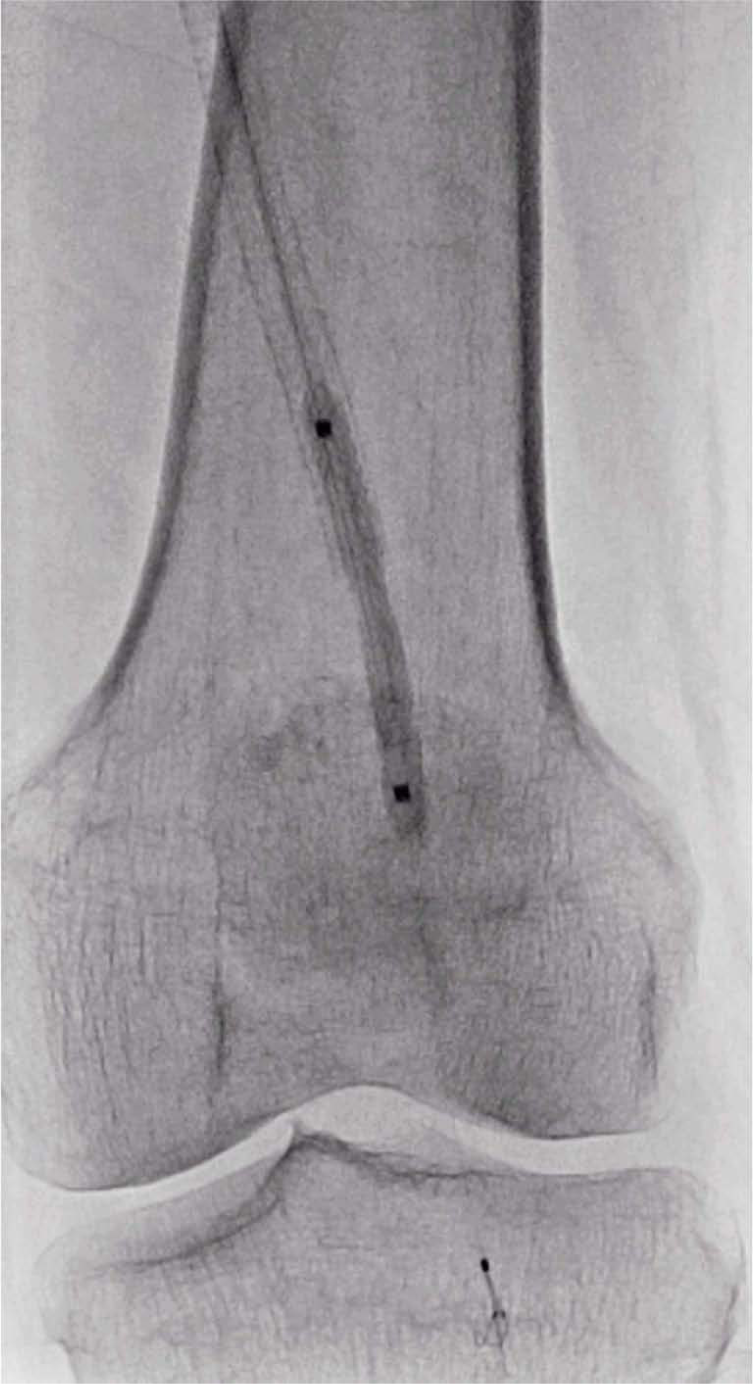
Digital angiography of percutaneous transluminal angioplasty of the distal left superficial femoral artery with the filter used for embolic protection.
Angiography through the Raabe sheath revealed a filling defect consistent with thrombus within the Spider filter (Figure 10). An aspiration catheter (Pronto Lp®; Vascular Solutions Inc., Minneapolis, MN, USA) was used to remove thrombus from the filter. The aspiration catheter was removed and angiography confirmed resolution of the filling defect within the filter. The filter was retrieved. Final angiography revealed technical success with a patent SFA stent graft with no residual thrombus and three-vessel runoff without distal embolization (Figure 11).
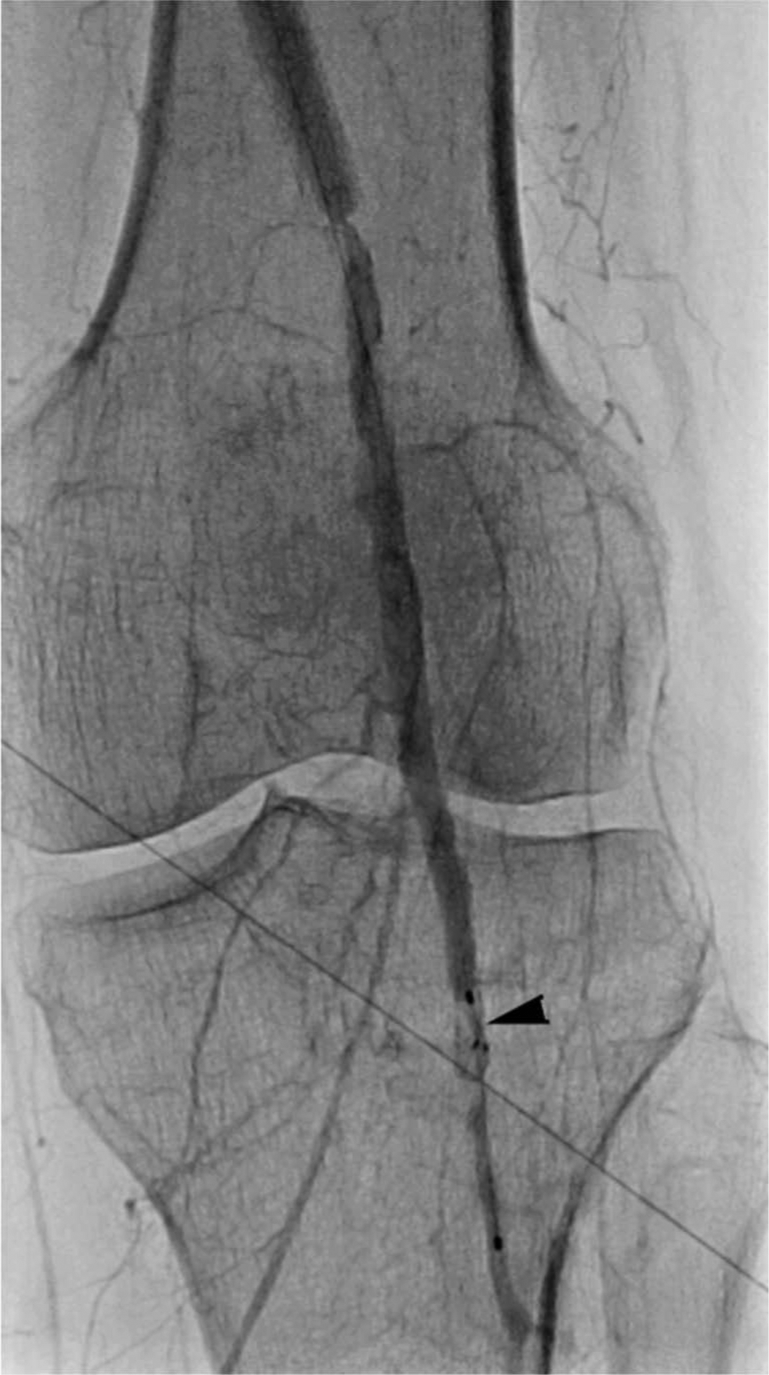
Digital angiography of the distal left superficial femoral artery. A filling defect is seen within the filter (arrow).
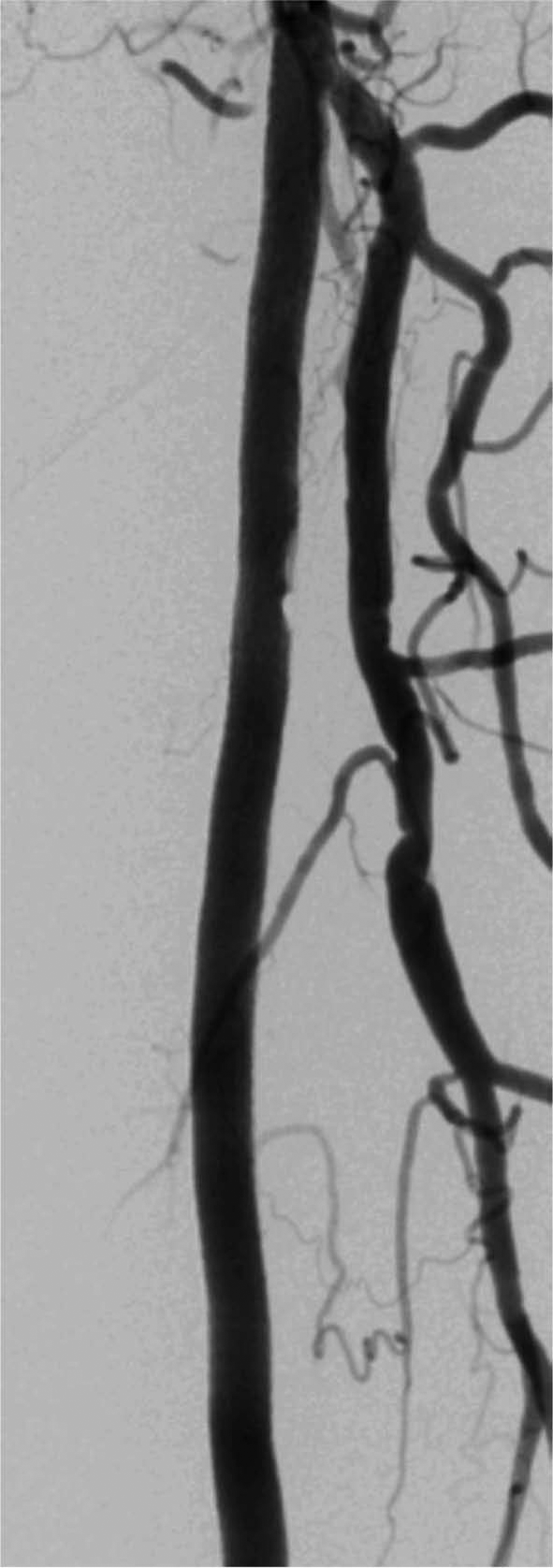
Digital angiography of the left superficial femoral artery post intervention.
Discussion
ALI affects as many as 13–17 cases per 100,000 people per year and is associated with significant morbidity, mortality and limb loss rates. 1 2 As such, early diagnosis and management are crucial in obtaining the best patient outcomes. The optimal management strategy depends on the clinical presentation of the patient, which is contingent on the amount of ischemia and the presence of collateral flow. Additionally, the comorbid conditions influence the choice of therapy and expected outcomes.
Endovascular revascularization is an appropriate strategy in patients with ALI with a viable or marginally threatened limb (Table 1) (Rutherford class I and IIa).3–5 Patients presenting with a severely threatened limb (Rutherford class IIb) require emergent surgical revascularization (within 3–6 hours) for limb salvage. Furthermore, patients with irreversible injury due to ALI manifesting as major tissue loss or profound sensory loss require primary amputation. Revascularization of non-viable limbs may precipitate a reperfusion syndrome with release of potassium, myoglobin and other toxic elements from the non-viable limb leading to multiorgan damage and death.
Acute limb ischemia.
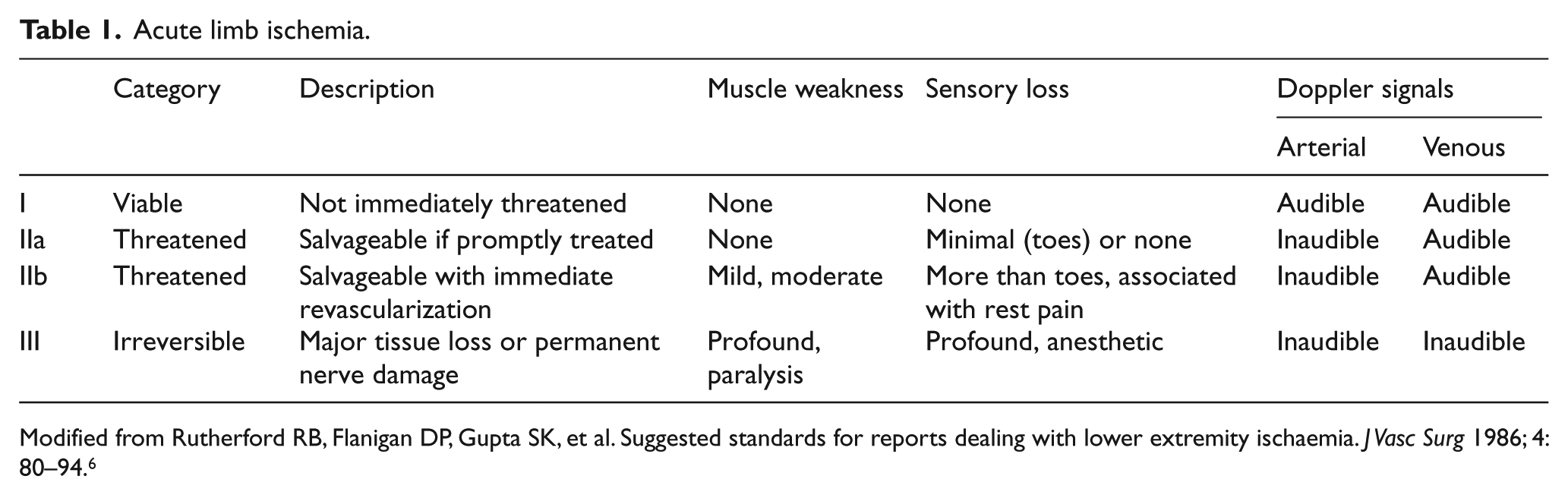
Modified from Rutherford RB, Flanigan DP, Gupta SK, et al. Suggested standards for reports dealing with lower extremity ischaemia. J Vasc Surg 1986; 4: 80–94. 6
The contralateral femoral artery was chosen as the access site to discern the extent of the occlusion and determine the inflow anatomy of the left SFA culprit lesion. Furthermore, this allowed access to the proximal vascular bed with an adequate working distance from the suspected occlusion of the SFA. Owing to the acuity of the occlusion with fresh thrombus, it was felt that catheter-directed thrombolysis would be an appropriate initial treatment strategy and that contralateral access would be associated with lower access site complications than with antegrade access. 7 Antegrade ipsilateral access would also have been problematic when dealing with a proximal SFA occlusion. A brachial artery approach may be considered in cases where a crossover approach is not feasible due to iliac anatomy. However, the need for long catheter length may preclude treatment depending on the location of the occlusion. In addition, brachial artery catheterization has been associated with significant rates of brachial artery thrombosis and pseudoaneurysm formation. 8 Radial access would demand even longer length catheters, which are not readily available.
Our patient presented with a threatened limb and was an appropriate candidate for percutaneous intervention as she had no evidence of motor or sensory loss and presented within 7 days of symptom onset. She had extensive thrombus burden within the previously placed Viabahn stents. Percutaneous intervention of lesions with heavy thrombus burden can be particularly challenging. Thrombolysis has been shown to be similar to surgery in terms of amputation-free survival in patients with ALI.4,5,9 Although surgical thrombectomy was a viable alternative, we considered endovascular treatment to be an appropriate initial strategy. Endovascular treatment would avoid the morbidity of open surgery, but if not effective would not preclude surgery. Owing to the acuity of the occlusion and the extent of the thrombus burden, we proceeded with catheter-directed thrombolysis. Mechanical thrombectomy was considered as an alternative treatment. However, we were concerned about the risk for distal embolization with rheolytic thrombectomy. The previously placed stents were ‘covered’ stents and the amount of thrombus made direct stenting unreasonable as an initial strategy.
Patients with acute limb ischemia may not be candidates for thrombolysis because of excessive major bleeding risks. Absolute contraindications to thrombolysis have been well documented and include cerebrovascular events within 2 months, active bleeding diathesis, recent gastrointestinal bleeding, and intracranial trauma within the last 3 months. Other limitations of thrombolytic therapy can include persistent occlusion from organized thrombus or embolized plaque despite complete lysis of amenable thrombus, major bleeding and progression to irreversible ischemia. The risk of systemic bleeding complications including major intracranial hemorrhage is 5–15%.5,9
Pharmacologic thrombolysis is infused via catheters with multiple side holes to improve surface contact of the drug. In this case, the Raabe sheath was positioned in the proximal left SFA and the multi-holed infusion catheter in the distal left SFA with the intent that the thrombolytic infusion along the length of occlusion would help lyse the thrombus, provide dissolution of downstream embolization and potentially uncover a treatable culprit lesion. Mechanical thrombectomy may be an alternative or a useful adjunct to lysis and has been demonstrated to be effective in restoring immediate blood flow in acute limb-threatening ischemia. 10 In patients with limb-threatening ischemia due to thrombotic arterial occlusions presenting within 14 days, mechanical thrombectomy has been shown to result in favorable long-term limb salvage, and amputation-free success. 11
The utility of mechanical thrombectomy in ALI due to stent or graft thrombosis has been described in case reports. 12 The filter wire and the aspiration catheter were used to reduce the risk of distal embolization with rheolytic thrombectomy. The rate of distal embolization with thrombolytic therapy has been reported at a rate of 3.8% to 37% and for rheolytic thrombectomy devices from 25% to 56%.13 –17 One review of acute limb ischemia patients treated with rheolytic thrombectomy found that distal embolization occurred in 9.8% of patients, with clinically significant distal embolization requiring further mechanical and/or pharmacologic management strategy being reported in various studies.13,18
Conclusion
Endovascular revascularization is an appropriate strategy in patients with ALI with a viable or marginally threatened limb. Optimal percutaneous management may require a combination of techniques including catheter-directed thrombolysis, rheolytic thrombectomy, and utilization of distal protection devices.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
