Abstract
The aim of the present study was to investigate the differences in digital reactive hyperemic index (RHI) in men and postmenopausal women. We investigated the differences in and correlates of RHI, measured by peripheral artery tonometry (PAT), in a group of 82 men (mean age ± SD: 55.6 ± 8.2 years; body mass index: 29.0 ± 4.2 kg/m2) and 125 postmenopausal women (58.9 ± 5.2 years; 27.7 ± 4.1 kg/m2). We also examined fRHI values (natural log of the PAT ratio of the 90–120 seconds post-occlusion interval) and augmentation index (AIx) as a measure of arterial stiffness. We found that RHI, fRHI and AIx were significantly lower in men compared to postmenopausal women (p<0.0001). We also found that fRHI values were significantly lower in individuals with (MetS+) versus without (MetS–) the metabolic syndrome (MetS). Endothelial inflammation was present in MetS+ subjects as indicated by increased plasma soluble intercellular adhesion molecule-1 (sICAM-1) (p<0.001) and E-selectin (p=0.0519) concentrations compared to MetS– individuals. No significant difference was found in RHI or AIx between MetS+ versus MetS– individuals. In summary, our study reveals that men have an impairment of endothelial function, assessed by digital PAT, compared to postmenopausal women. Furthermore, we show that the presence of the MetS is characterized by endothelial dysfunction, as suggested by lower fRHI, as well as by endothelial inflammation, which likely contributes to the increased cardiovascular disease risk associated with the MetS.
Introduction
Cardiovascular disease (CVD) is among the leading causes of death worldwide. Many risk factors for CVD have been identified including age, hyperlipidemia, hypertension, abdominal obesity and insulin resistance. 1 In recent years, the clustering of some of these metabolic alterations (abdominal obesity, dyslipidemia, insulin resistance), the so-called metabolic syndrome (MetS), 2 has also been identified as a condition associated with increased risk of chronic diseases. Women have a lower risk of CVD compared to men, 3 a protection that progressively disappears at menopause most likely due to increases in circulating low-density lipoprotein (LDL)-cholesterol and triglyceride (TG) concentrations, abdominal fat deposition and insulin resistance, as well as a decrease in high-density lipoprotein (HDL)-cholesterol.4,5
Endothelial dysfunction (ED) (i.e. impairment of the normal functions of the vascular endothelium) occurs in the presence of vasoconstrictive, growth-promoting, procoagulant and proinflammatory conditions. 1 As ED is often present long before any obvious clinical signs of CVD can be noted, 6 the measurement of endothelial function is now recognized as an important variable to consider in the assessment of CVD risk. 7 In a recent study looking at the effect of age and sex on endothelial function in a healthy population, arterial flow-mediated dilation (FMD), a correlate of endothelial function, was reported to be better in women compared to men before the age of 70. 8 Other studies also revealed that men show a significantly deteriorated endothelial function when compared to pre- and postmenopausal women.9,10 Interestingly, no difference in FMD was noted between men and women after the age of 70 years, 8 suggesting that age or menopause-related metabolic disturbances may have an effect on endothelial function.
In recent years, reactive hyperemic index (RHI) calculated from digital peripheral arterial tonometry (PAT) measurements has been increasingly used as a noninvasive measure of endothelial function and studies have shown that it is associated with numerous traditional CVD risk factors.11–13 Female sex and age have also been associated with higher RHI10,12 and a large range of RHI values has been reported in menopausal women. 5 The present study was therefore undertaken in order to investigate the differences in and correlates of RHI in men and postmenopausal women.
Methods
Subjects
The present study was performed using baseline data from subjects enrolled in a 4-week intervention study investigating the impact of dietary spices and herbs on vascular function. Eighty-two middle-aged men and 125 postmenopausal women were recruited in Québec City (Québec, Canada) and Winnipeg (Manitoba, Canada). In order to be included in the study, subjects had to be aged between 40 and 70 years, have a waist circumference ≥ 94 cm for men or ≥ 80 cm for women, as well as a Framingham 10-year risk score under 20%. 14 All women had to be menopausal with menopause being defined as the absence of menses for at least 1 year. Only nine women were using hormonal replacement therapy. Individuals were excluded from the study if they were undergoing treatment for hypertension, CVD, type 2 diabetes mellitus (T2DM) or other endocrine disorders, if they were smokers or consumed more than one drink a day (15 g). Those with specific dietary habits (e.g. vegetarianism), using supplements (vitamins, minerals, flavonoids) or with an aversion towards dietary spices and/or herbs were also excluded from the study. Each participant signed a consent form which was approved by the Human Research Ethics Committee of Université Laval and the Biomedical Research Ethics Board of the University of Manitoba.
Physical measurements
Body weight, height, and waist and hip circumferences were measured using standardized procedures. 15 Blood pressure was measured at rest using an automated sphygmomanometer (HEM-907XL; Omron Healthcare Inc., Bannockburn, IL, USA).
Endothelial function
Digital pulse amplitude was measured using a PAT device placed on the tip of each index finger (EndoPAT2000; Itamar Medical, Caesarea, Israel). The PAT device included a pneumatic plethysmograph that applied uniform pressure to the surface of the distal finger, allowing measurements of pulse volume changes. The inflation pressure of the digital device was electronically set to 10 mmHg below diastolic blood pressure or 70 mmHg (whichever value was lower). Baseline amplitude was measured from each fingertip for 5 minutes. Arterial flow was then interrupted for 5 minutes by a cuff placed on a proximal forearm and inflated at a pressure of 200 mmHg or 60 mmHg higher than the systolic blood pressure, unless the occlusion appeared incomplete, at which time the cuff pressure was increased to 250 mmHg (whichever value was lower). The cuff was then rapidly deflated and the pulse amplitude was recorded electronically in both fingers and analyzed using the automated algorithm provided by Itamar Medical, which provided the average PAT measure for each 30-second interval after forearm cuff deflation up to 5 minutes. RHI was then calculated as the ratio of the post- to pre-occlusion PAT amplitude of the tested arm, divided by the post- to pre-occlusion PAT values ratio of the control arm. We also looked at fRHI values, which correspond to the natural log of the PAT ratio of the 90–120 seconds post-occlusion interval, a variable that has been found to be more closely associated with CVD risk factors. 12 This fRHI was first reported in the Framingham Heart Study 12 and is now automatically calculated by the EndoPAT software. Arterial stiffness, expressed as the augmentation index (AIx) calculated by the EndoPAT software, was also examined.
Inflation and deflation of the cuff was performed using an E20 Rapid Cuff Inflator (DE Hokanson Inc., Bellevue, WA, USA). PAT measurements were performed in the morning (after 7 a.m. but no later than 10 a.m.). Subjects were in the fasting state, resting in a supine position in a room at constant temperature (22°C/72°F).
Blood measurements
Traditional cardiometabolic profile variables were assessed. Total and HDL-cholesterol as well as TG levels were determined in plasma from 12-hour fasting blood samples, as previously described,16,17 while LDL-cholesterol was calculated. 18 Plasma-oxidized LDL particles (OxLDL; Alpco Diagnostics, Windham, NH, USA), soluble forms of the vascular cell adhesion molecule-1 (sVCAM-1) and E-selectin (sE-selectin; Abcam, Cambridge, MA, USA), as well as of the intercellular adhesion molecule-1 (sICAM-1; R&D Systems, Hornby, Ontario, Canada), were measured in plasma using commercial assays. Serum glucose was measured enzymatically, 19 whereas serum insulin was measured by radioimmunoassay with polyethylene glycol separation. 20 The homeostasis model assessment index of insulin resistance (HOMA-IR) value was then calculated. 21
Statistical procedures
Data are presented as means ± standard deviation (SD) unless stated otherwise. Differences between groups were tested with general linear model (GLM) procedures with adjustments for age and recruitment center as well as sex when appropriate. Spearman’s correlation coefficients were calculated to test for associations between the different variables. Individuals with the MetS were characterized as having at least three of the following criteria 2 : waist circumference ≥ 102 cm for men or ≥ 88 cm for women; fasting triglycerides ≥ 1.7 mmol/L; fasting HDL-cholesterol ≤ 1.03 mmol/L for men or ≤ 1.29 mmol/L for women; blood pressure ≥ 130/85 mmHg; and fasting plasma glucose of ≥ 5.6 mmol/L. A total of 26 men (32% of men) and 28 women (22% of women) met the criteria for MetS. When needed, values were log10 transformed for analysis, but for practical reasons raw data are presented in tables and figures. Regression analyses were also conducted in order to identify the significant contributors to the variance of RHI, fRHI and AIx values. In these analyses, only the variables that showed significant univariate associations with RHI, fRHI or AIx were included in the model. All analyses were conducted using the SAS statistical package (version 9.3; SAS Institute, Cary, NC, USA). Throughout the analyses, a p-value ≤ 0.05 was considered as statistically significant.
Results
Table 1 shows the physical and metabolic characteristics of study participants. On average, men were younger than postmenopausal women (p<0.001) and were also characterized with a higher BMI (p<0.05), larger waist circumference (p<0.0001) and higher diastolic blood pressure (p<0.05) compared to postmenopausal women. On the other hand, postmenopausal women had significantly higher HDL-cholesterol levels (p<0.0001) which led to a significantly lower total/HDL-cholesterol ratio (p<0.0005) compared to men. Circulating glucose concentrations and HOMA-IR values were found to be significantly higher in men (p<0.05). Table 1 also shows that men were characterized by lower RHI, fRHI and AIx values compared to postmenopausal women (p<0.0001). Furthermore, ED identified as a RHI value < 1.6722,23 was more prevalent in men (50% of men) than in postmenopausal women (33% of women). No significant difference was observed in plasma oxidized LDL, sICAM-1, sVCAM-1 or sE-selectin concentrations between men and postmenopausal women.
Physical and metabolic characteristics of the study participants.
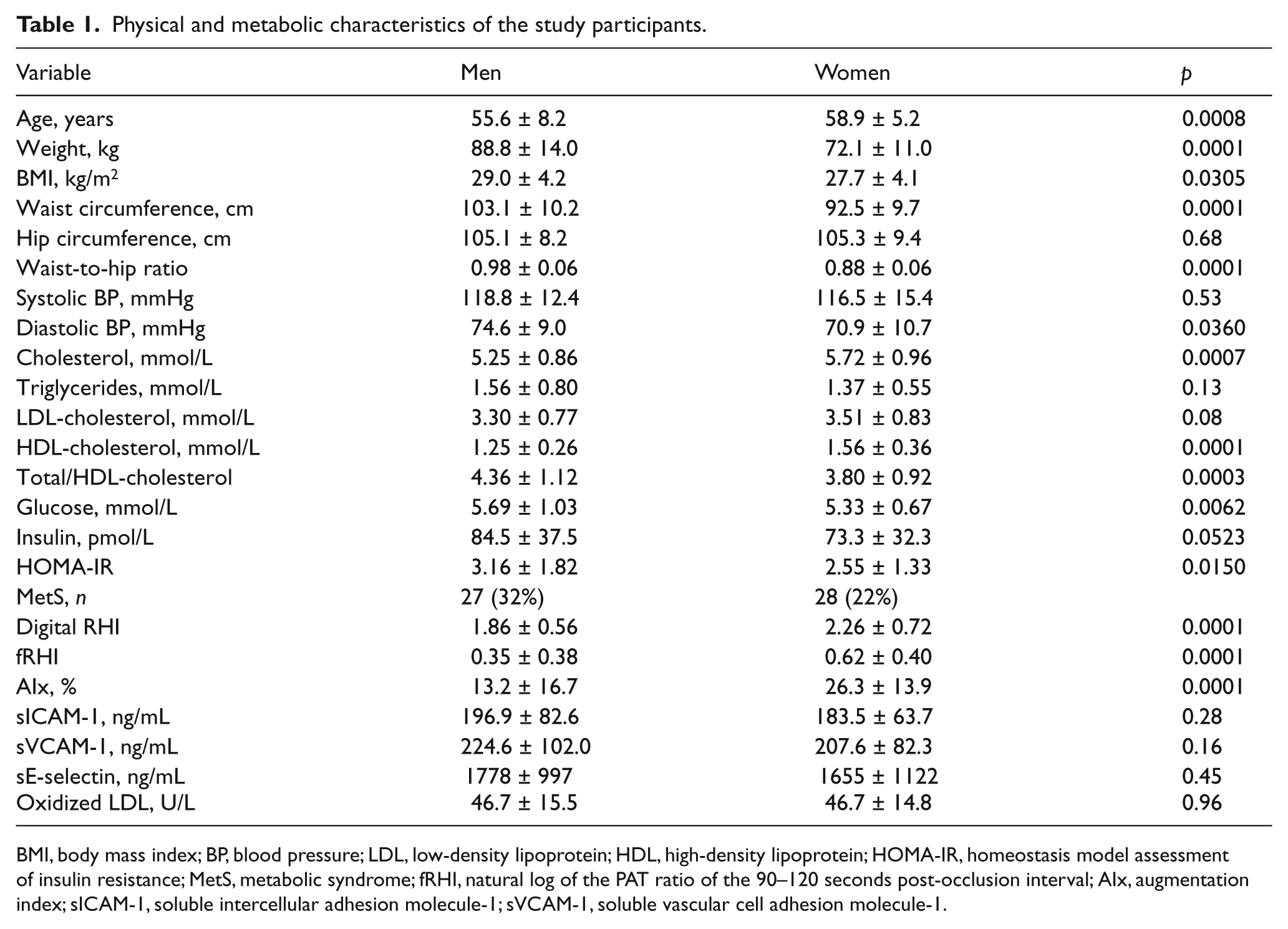
BMI, body mass index; BP, blood pressure; LDL, low-density lipoprotein; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment of insulin resistance; MetS, metabolic syndrome; fRHI, natural log of the PAT ratio of the 90–120 seconds post-occlusion interval; AIx, augmentation index; sICAM-1, soluble intercellular adhesion molecule-1; sVCAM-1, soluble vascular cell adhesion molecule-1.
Table 2 shows the associations between RHI values and the physical as well as cardiometabolic variables of the participants. In the entire group, we found that RHI was negatively correlated with overall adiposity and abdominal fat accumulation as suggested by significant negative associations with BMI (r = −0.17, p<0.05) and waist circumference (r = −0.21, p<0.005). Circulating plasma TG (r = −0.18, p<0.01) and sICAM-1 (r = −0.15, p<0.05) levels as well as total/HDL-cholesterol ratio (r = −0.24; p<0.001) (Table 2) were also associated with RHI. Similar correlations were noted when fRHI and AIx were examined (Table 2).
Spearman correlations of physical and metabolic characteristics with RHI, fRHI and AIx in study participants.
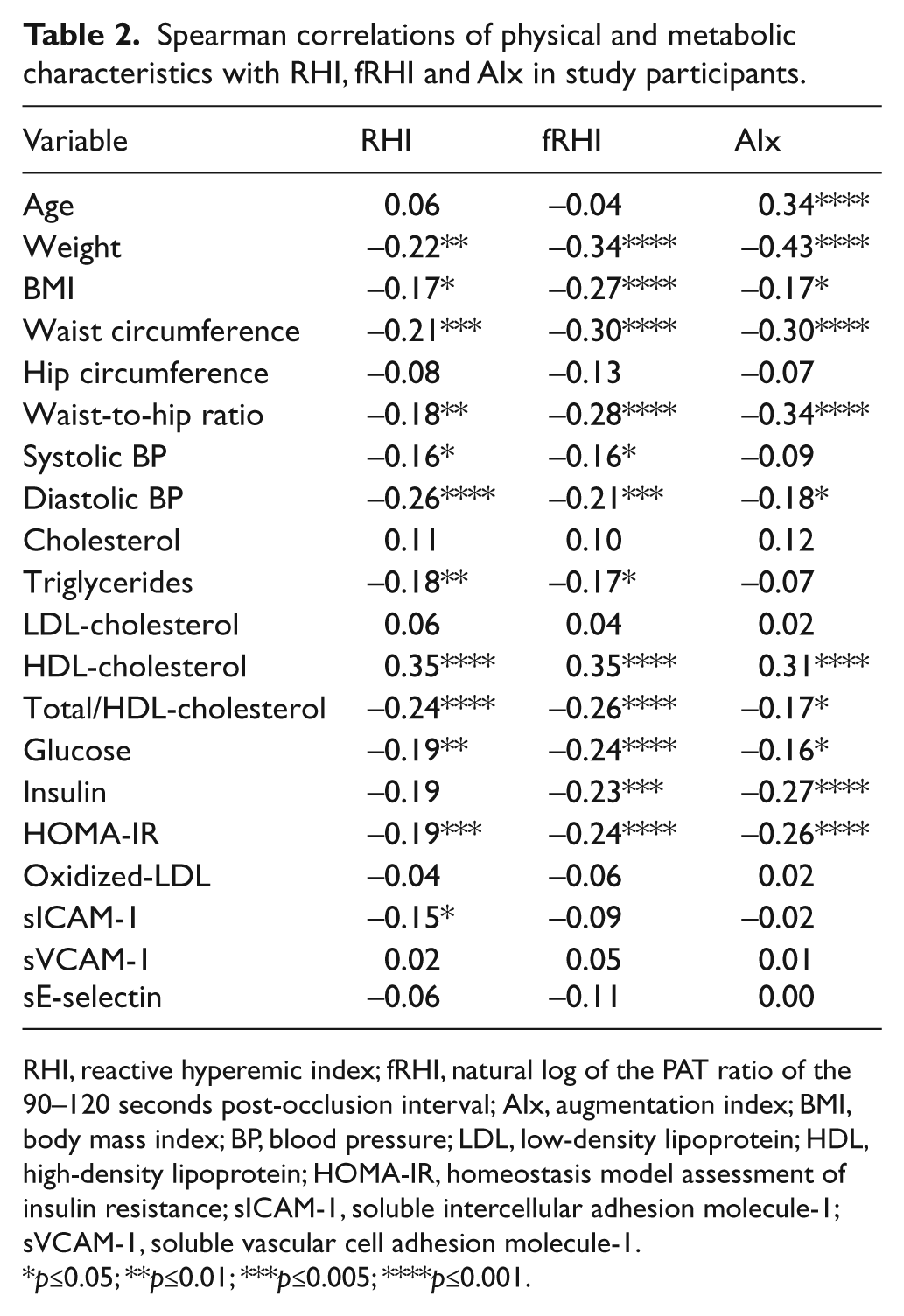
RHI, reactive hyperemic index; fRHI, natural log of the PAT ratio of the 90–120 seconds post-occlusion interval; AIx, augmentation index; BMI, body mass index; BP, blood pressure; LDL, low-density lipoprotein; HDL, high-density lipoprotein; HOMA-IR, homeostasis model assessment of insulin resistance; sICAM-1, soluble intercellular adhesion molecule-1; sVCAM-1, soluble vascular cell adhesion molecule-1.
p≤0.05; **p≤0.01; ***p≤0.005; ****p≤0.001.
We also conducted multiple regression analyses (Table 3) in order to quantify the independent contributions of physical and metabolic variables to RHI values and found that HDL-cholesterol, diastolic BP and sex were all significant predictors of the variance in digital RHI. Sex and HDL-cholesterol were also identified as independent predictors along with BMI of fRHI values. In addition, Table 3 shows that AIx was best predicted by age, sex and insulin resistance expressed as the HOMA-IR.
Multiple regression analysis of digital reactive hyperemic index (RHI) as well as the natural log of RHI of the 90–120 seconds post-occlusion interval (fRHI) 12 and arterial stiffness (augmentation index, AIx).
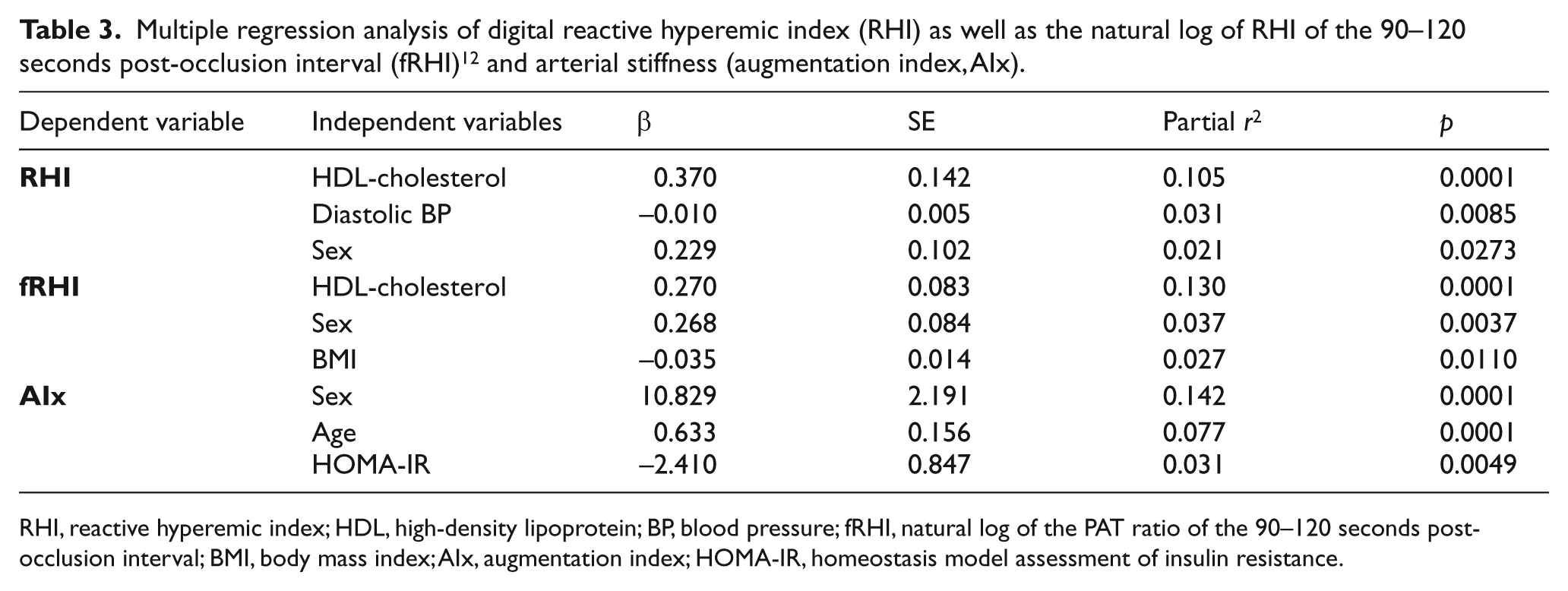
RHI, reactive hyperemic index; HDL, high-density lipoprotein; BP, blood pressure; fRHI, natural log of the PAT ratio of the 90–120 seconds post-occlusion interval; BMI, body mass index; AIx, augmentation index; HOMA-IR, homeostasis model assessment of insulin resistance.
Finally, in an effort to better characterize the impact of the MetS on endothelial function, we compared RHI, fRHI and AIx values as well as circulating adhesion molecule concentrations in individuals with (MetS+) versus without (MetS–) the MetS. Figure 1 shows that RHI and AIx values were not significantly different between MetS+ versus MetS– individuals (p>0.05). However, the presence of the MetS was associated with a significantly lower fRHI (p<0.01). As illustrated in Figure 2, fasting plasma sICAM-1 concentrations were significantly increased in MetS+ versus MetS– individuals (p<0.001) and a tendency for higher sE-selectin concentrations was also noted in MetS+ versus MetS– individuals (p = 0.0519). The impact of the MetS on circulating sICAM-1 and sE-selection levels remained significant when men and women were analyzed separately. Plasma sVCAM-1 concentrations were not different between MetS+ versus MetS– individuals.
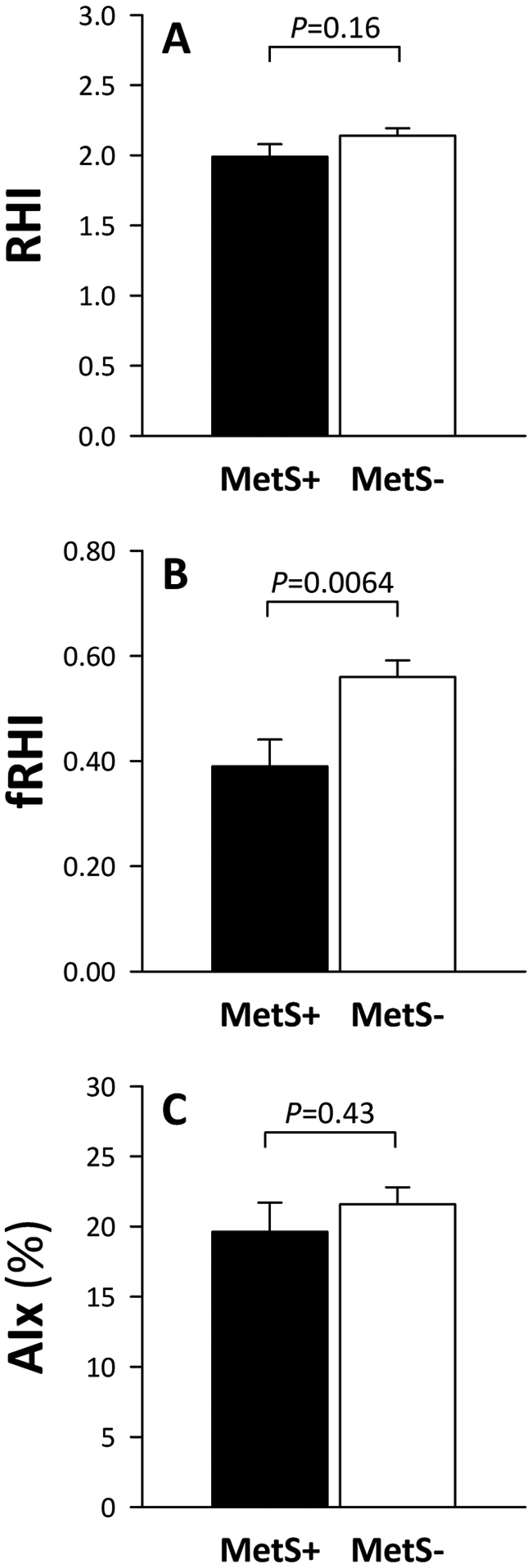
(A) Digital pulse amplitude tonometry (PAT) waveform reactive hyperemic index (RHI); (B) natural log of RHI of the 90–120 seconds post-occlusion interval (fRHI); 12 and (C) PAT-derived augmentation index (AIx) in individuals with (MetS+, black bars) or without (MetS–, white bars) the metabolic syndrome (MetS). (Data presented as means ± SEM.)
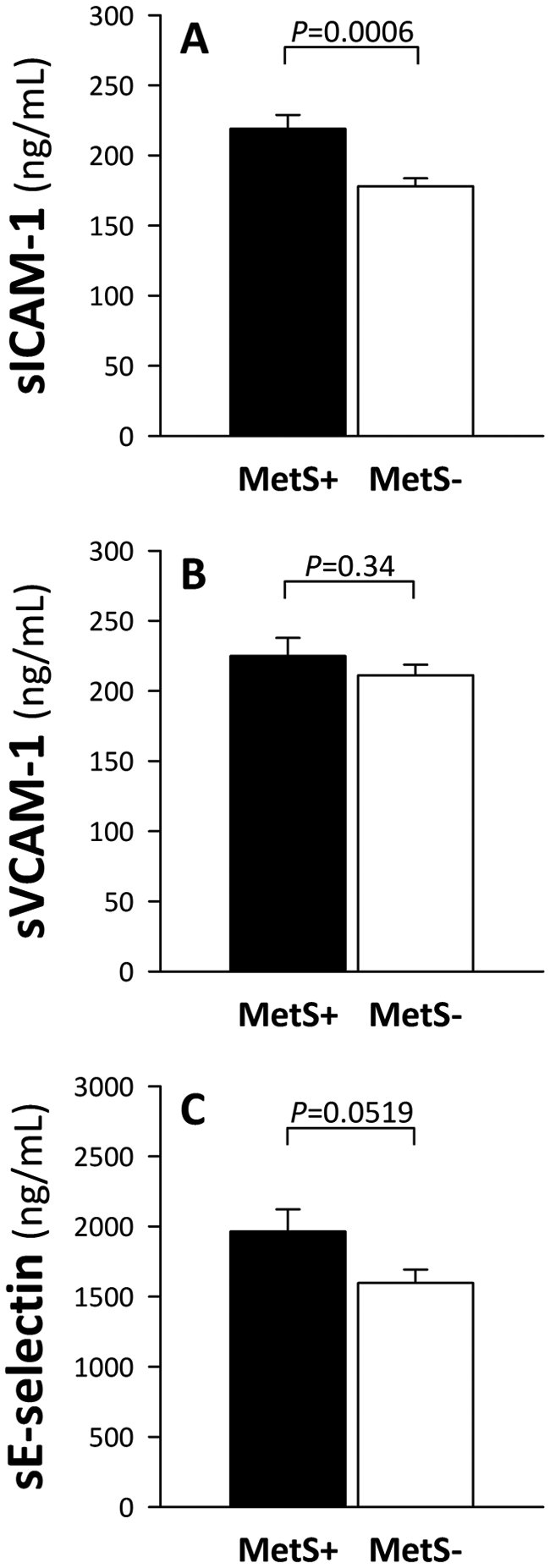
Fasting plasma (A) soluble intercellular adhesion molecule-1 (sICAM-1), (B) soluble vascular cell adhesion molecule-1 (sVCAM-1) and (C) sE-selectin in individuals with (MetS+, black bars) or without (MetS–, white bars) the metabolic syndrome (MetS). (Data presented as means ± SEM.)
Discussion
In the present study, we show that there is a clear difference in the endothelial function of middle-aged men and postmenopausal women, with men displaying lower RHI and fRHI values derived from digital PAT measurements. This sex difference in endothelial function is in accordance with previous observations10,12 and concordant with the higher incidence of CVD in men compared to women of a similar age. 4 The independent contribution of this sex difference in endothelial function in CVD risk stratification, however, remains the subject of thorough investigation. 24 On the other hand, our results are in conflict with those from a recent study that reported no difference in the RHI values of men and women. 25 Differences in the populations of the latter study (i.e. younger men and premenopausal women) may account for this discrepancy.
There is a well-described sex difference in CVD risk as women show a more favorable risk profile than men that is explained, at least partly, by differences in sex hormone profiles. Indeed, estrogens have been characterized with antioxidant properties as well as being able to favorably modulate the lipid profile and endothelial function.26,27Accordingly, hormonal changes occurring at menopause in women (i.e. loss of estrogens) have been shown to be related to the increased CVD risk in menopausal women. 28 However, as we tested postmenopausal women, their better endothelial function compared to men is not likely to be largely influenced by circulating estrogens.
Age has also been associated with the impairment of endothelial function 29 and this effect has been reported to occur earlier in men than in women, with men showing a progressive impairment of arterial FMD around 40 years of age, while a steep decline in endothelial function is observed in women 10 years later – consistent with the onset of menopause and progressive loss of the protective effect of estrogens on the vasculature. 27 Digital fRHI has also been positively associated with age, 12 which may appear to be in apparent contradiction with our observations as we did not find such an association. However, although a significant predictor of fRHI in the Framingham Study, 12 the association with age was weak and may only be revealed in large population studies. On the other hand, the fact that the age-related decline in endothelial function was revealed by arterial FMD measures in most previous studies rather than digital RHI, could also suggest potential differences in the effect of age on micro and macrovascular regulation.
Impaired endothelial function has been associated with larger vessel size 29 and, in this regard, blood vessel diameter could contribute to the sex difference in RHI we report herein. Indeed, it has been previously shown that the resting brachial artery diameter (BAD) is smaller in women compared to men, while RHI values are higher in women than in men. 10 Interestingly, when women were individually matched with men for resting BAD, the RHI values of men and women were found to be comparable. 10 Unfortunately, we did not measure resting BAD in the present study and, thus, we were not able to examine its contribution in the sex differences in RHI and fRHI we report herein.
Obesity has also been shown to be associated with ED as there is an inverse relationship reported between FMD and BMI.30,31 Abdominal obesity has been suggested to be more important in the relationship of obesity to endothelial dysfunction as the impairment of FMD is more closely associated to the waist-to-hip ratio than BMI. 32 Results of the present study give further support to this association between endothelial dysfunction and abdominal fat accumulation as waist circumference was negatively associated with RHI and fRHI, an observation which is concordant with previous observations. 12
The detrimental impact of a dysmetabolic profile on endothelial function has been previously revealed by an impaired FMD 33 and higher RHI 34 in individuals with the MetS. The peculiar metabolic and hormonal milieu of the MetS has been suggested to contribute to endothelial dysfunction through the impairment of nitric oxide synthesis and degradation. 35 Our results are in contradiction with these previous observations as we report that RHI values are not significantly different between MetS+ versus MetS– individuals. It must be pointed out that the significant association between a low RHI and the MetS that has been recently reported 34 was noted in a larger (n~1000) and more heterogeneous population of MetS+ individuals that included smokers as well as patients with diabetes, hypercholesterolemia or with a previous history of heart disease, whereas the metabolic alterations of subjects included in the present study were of a far lesser extent. We, however, found that fRHI (i.e. the natural log of the PAT ratio of the 90–120 seconds post-occlusion interval) was different between MetS– versus MetS+ individuals, with the latter showing a lower fRHI. This gives more support to the suggestion that fRHI could be a better correlate of cardiometabolic disturbances than RHI. Furthermore, PAT-derived AIx has been identified as a correlate of CVD risk factors, 36 and we also noted that PAT-derived AIx was associated with obesity and insulin resistance. However, there was no significant difference in AIx between MetS+ versus MetS– individuals. Finally, we found that circulating sICAM-1 concentrations were increased in MetS+ individuals, supporting the notion that the MetS is associated with endothelial activation and inflammation. 37
A potential limitation of the present study is that we only tested middle-aged men and postmenopausal women. Although we report a sex difference in RHI and fRHI, we were not able to thoroughly investigate the impact of age and menopause on endothelial function as we would have with the inclusion of younger men and premenopausal women in the study. Furthermore, while FMD is still considered the gold standard technique for the measurement of endothelial function, we used digital PAT in our assessment of endothelial function. In this regard, there are equivocal observations about the association between FMD and digital PAT, with some finding a significant association between both techniques 38 while others failed to find such an association.39,40 Nonetheless, and as previously suggested, 41 determination of digital PAT remains relevant as it may give information on the response of small blood vessels (capillary bed), which may react differently from larger vessels, such as those evaluated with arterial FMD. In this sense, although not interchangeable, FMD and digital PAT measures may be complementary methods that can be used in the assessment of endothelial function. The relevance and informative potential of FMD and digital PAT will need to be examined in future studies looking at changes in vascular health and endothelial function that occur with age and menopause.
In summary, our study shows that compared to postmenopausal women, middle-aged men are characterized by an impairment of the peripheral blood vessel endothelial response to hyperemic stimulus as suggested by significantly lower digital RHI and fRHI values. Our study also reinforces the relationship between the MetS and alterations of the vascular function and, in this regard, fRHI may be a better correlate of cardiometabolic disturbances than RHI alone. Further studies need to be undertaken in order to better understand the etiology of endothelial dysfunction and arterial stiffness in ageing men and women as well as in the context of the MetS.
Footnotes
Acknowledgements
We acknowledge the contributions of nurses and research professionals, as well as of the subjects who participated in the study – without whom, no clinical research would be possible.
Declaration of conflicting interest
The authors have no conflict of interest to disclose.
Funding
This study was supported by McCormick Science Institute (MSI).
