Abstract
We aimed to test the association between matrix metalloproteinase (MMP) genetic polymorphisms and (a) intima–media thickness in the common carotid (IMTcc) and (b) the presence of plaques in the carotid and femoral bifurcations. Carotid and femoral bifurcations were scanned with ultrasound in 762 Cypriot community dwellers (46% men) over the age of 40 years. IMTcc and the presence of plaques were recorded. The MMP1 1G/2G, MMP3 5A/6A, MMP7 -181A>G, MMP9 R279Q, and MMP12 -82A>G polymorphisms were determined with the TaqMan method. In men, the presence of plaques in any bifurcation was associated with the MMP9 279Q allele (ORadjusted=4.50; 95% CI=2.0 to 10.1; p<0.001) and the MMP7 -181A allele was associated with the presence of femoral plaques (ORadjusted=2.61; 95% CI=1.36 to 4.99; p=0.004). In women, the presence of femoral plaques was associated with the MMP12 -82G allele (ORadjusted=1.9; 95% CI=1.14 to 3.16; p=0.014). Our results suggest that the effect of common MMP genotypes on plaque presence may be site- and sex-dependent.
Keywords
Introduction
Carotid intima–media thickness (IMTc)1–3 and the presence and number of carotid plaques4–7 as measured with high-resolution ultrasound, are surrogate markers of atherosclerosis and have been shown to be associated with the extent and severity of coronary atherosclerosis 8 as well as future episodes of myocardial infarction and stroke.1–4,6,7 Although IMTc is most often measured in studies, doubts have been raised recently as to its clinical utility, whereas the presence of carotid plaques has been shown to have a greater utility in estimating risk for future cardiovascular events, both in observational studies and a meta-analysis.9,10 In addition to carotid atherosclerosis, atherosclerosis in the femoral arteries (i.e. presence and number of femoral plaques) has also been shown to predict cardiovascular disease (CVD), independent of carotid atherosclerosis,10–12 and may represent a different pathophysiological process with structural differences.13–15
Metalloproteinases (MMPs) are a family of endopeptidases with the ability to degrade the extracellular matrix (ECM), which have been shown to be involved in the remodelling of atherosclerotic plaques. 16 Currently, matrix remodelling is thought to contribute to plaque instability, rupture and thrombus formation 17 and is considered a dynamic process. Current concepts of atherosclerosis, which emphasize the remodelling of the arterial wall, provide a framework for understanding how, through weakening and destabilization, angiographically undetectable moderate lesions could become culprits for acute coronary syndromes. 16 Increasing evidence indicates that genetic variability (single nucleotide polymorphisms; SNPs) in the genes coding for MMPs may alter their transcriptional activity and contribute to susceptibility to cardiovascular disease.16,18
The aim of this study was to test the association between ultrasonically determined measures of carotid and femoral subclinical atherosclerosis (i.e. IMT in the common carotid, carotid and femoral plaque presence) and functional genetic polymorphisms in genes encoding five MMPs (MMP1 1G/2G (rs1799750), MMP3 5A/6A (rs3025058), MMP7 -181A>G (rs11568818), MMP9 R279Q (rs17576), and MMP12 -82A>G (rs2276109)) in community-dwelling middle-aged men and women.
Methods
Subjects
The Cyprus Study is a population-based cohort study of cardiovascular disease and atherosclerosis in 1106 individuals aged 40 years or more from two areas in Cyprus.19,20 Baseline data have been collected from residents of two randomly selected areas in Cyprus and their relatives living in any major town between 2003 and 2008. All inhabitants were identified through the population list held at the Mayor’s office and all those over the age of 40 years were invited to participate. The overall participation rate of those invited was 95%. The Ethics Committee of the Cyprus Institute of Neurology and Genetics approved the study. All participants provided written informed consent. For this analysis, baseline data of the first 762 subjects recruited, who had complete data on intima–media thickness in the common carotid (IMTcc), plaque and MMP genotypes, were used.
Cardiovascular disease status, risk factors and ultrasonic measurements
Cardiovascular disease status (prevalent cases), risk factors and ultrasonic measurements have been described previously. 11 Type 2 diabetes (T2D) was considered present if subjects were taking oral hypoglycemic agents, insulin or had a fasting blood sugar in excess of 7.0 mmol/L. Hypertension was considered present if subjects were on treatment or had a systolic blood pressure (BP) ≥ 140 mmHg or a diastolic BP ≥ 90 mmHg.
All scans were performed using a Philips (ATL) HDI 5000 duplex scanner (Seattle, WA, USA). The IMT complex of the far wall of the common carotid artery was measured at its thickest part (mean of three readings) on both transverse and longitudinal sections 1.5–2.0 cm proximal to the bifurcation which was free from any focal thickening (i.e. atherosclerotic plaques). The mean of the measurements from both carotid arteries was used in the analysis (IMTcc). The extracranial vessels (common carotid artery (CCA), external carotid artery (ECA), internal carotid artery (ICA)) and approximately 1.5–2 cm proximal and distal to the femoral bifurcations were carefully examined in both transverse and longitudinal section for the presence of plaques. An arterial bifurcation (internal carotid or common femoral bifurcation) was classified as being affected by plaque if there was a focal thickening greater than 1.2 mm that did not uniformly involve the whole arterial wall. 5 The two ultrasonographers who performed the ultrasonic scans were not aware of the clinical, biochemical or genetic risk factors of the subjects. The inter- and intra-observer variabilities of the two ultrasonographers have been reported previously. 11
Biochemical measurements
A venous blood sample was obtained from all subjects after a 6–12-hour fast. After at least 15 minutes at room temperature, the samples were centrifuged and serum and plasma were immediately aliquoted and frozen to −80°C until analysis. Serum fasting glucose, total cholesterol (TChol), high-density lipoprotein-cholesterol (HDL-C), low-density lipoprotein-cholesterol (LDL-C) and triglycerides (TG) were quantitatively determined with enzymatic color tests. All tests were performed with the Olympus AU640 automatic analyzer and all reagents were from Olympus Diagnostics (Beckman Coulter International SA, Nyon, Switzerland). Coefficients of variation were less than 5% for all measurements.
DNA was extracted from whole blood in EDTA anticoagulant with the use of commercial kits (Qiagen maxiBlood extraction kit; Qiagen Hilden, Germany) according to the manufacturer’s instructions. The MMP1 1G/2G (rs1799750), MMP3 5A/6A (rs3025058), MMP7 -181A>G (rs11568818), MMP9 R279Q (rs17576), and MMP12 -82A>G (rs2276109) single nucleotide polymorphisms were determined with the TaqMan method. The primers and probes used are shown in the Supplementary Table. Oligonucleotides were synthesized by Applied Biosystems® (ABI), (Paisly, UK). Amplification was performed in a 5 μl volume with 4.25 ng of genomic DNA, 2.5 μl of TaqMan® Universal PCR Master Mix, 0.125 μl of 40× Assay Mix and 2.375 μl of DNase-free dH2O according to the ABI protocol for dry template dispersion. The polymerase chain reaction (PCR) conditions were according to the ABI TaqMan® protocol and 384-well plates were used. After amplification, the plates were read with the ABI Prism 7900HT Sequence Detection System for allelic discrimination and results were analyzed using the SD 2.1 software. In some cases allelic discrimination was not possible due to poor DNA quality, resulting in slightly different numbers for genotypes. The MMP3 5A/6A polymorphism was additionally analyzed with the Restriction Fragment Length Polymorphism (RFLP) method, thus resulting in no missing genotypes. Missing cases were <5% for all genotypes and therefore no missing data methods were used. A random number generator was used for duplicates and four no template controls (NTCs) were used in every plate to assure quality control. Genotyping was performed at the Centre for Cardiovascular Genetics, UCL, London, UK.
Statistical methods
We tested the Hardy–Weinberg equilibrium on a contingency table of observed-versus-predicted genotypic frequencies with an exact test.
Subclinical atherosclerosis outcomes were classified as binary variables: IMTcc greater or less than 0.07 cm (population mean); presence versus absence of carotid or femoral plaques and presence versus absence of plaques in any of the four bifurcations scanned (0 vs 1–4).
Associations between subclinical atherosclerosis outcomes and MMP genotypes were investigated by computing age- and sex-adjusted basic (Model 1) and multivariable adjusted (Model 2: Model 1 + smoking in pack-years, hypertension, T2D, body mass index (BMI) and TChol) odds ratios using logistic regression. We also conducted sex-specific analyses because of recent evidence of sex-specific associations between the MMP9 (-1562C>T) polymorphism and carotid artery stiffness. 21 Formal testing for sex interaction was done by computing the interaction term in the multivariable model, as appropriate. To adjust for multiple testing, the Bonferroni correction was used, raising the significance level from 0.05 to 0.01 (0.05/5 SNPs) for each outcome as well as step-wise methods.
All statistical analyses were computed using Stata v.12.0 (StataCorp, College Station, TX, USA).
Results
Of the 762 subjects studied, 113 (14.8%) had clinical evidence of prevalent cardiovascular disease; documented myocardial infarction with hospital admission and/or presence of infarct on electrocardiogram (ECG) in 58, presence of angina or coronary intervention and/or myocardial ischemia on ECG in 43, ischemic hemispheric neurological events in nine (five strokes, four TIAs) and lower limb ischemia in three.
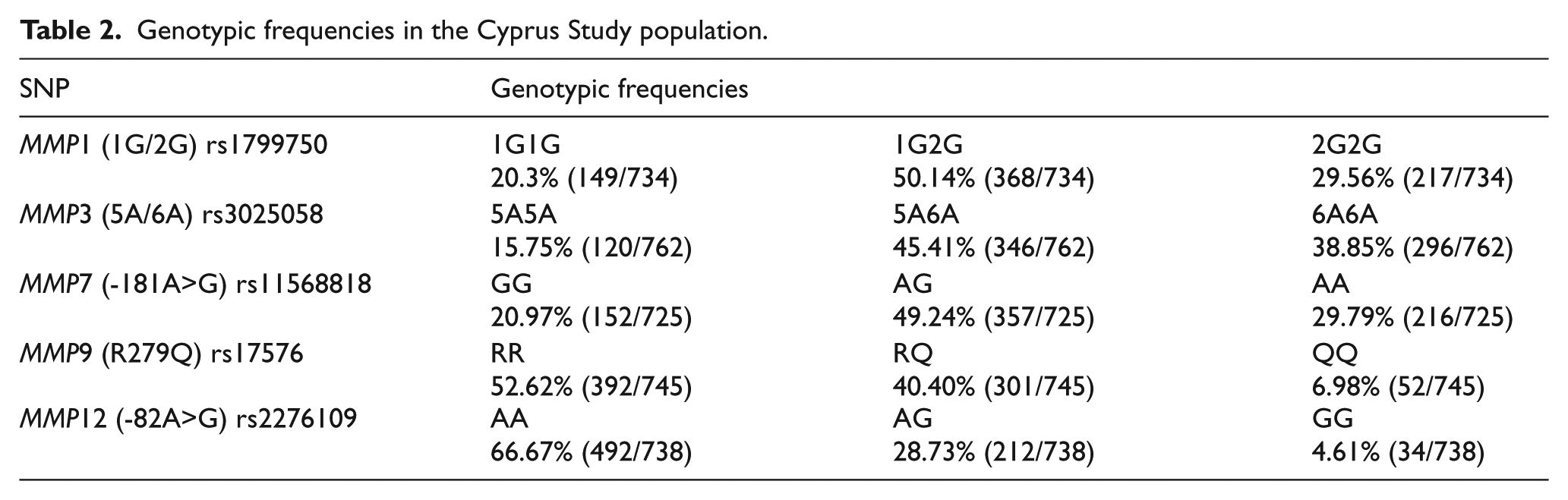
All genotypes were in Hardy–Weinberg equilibrium. Baseline characteristics of the population according to MMP genotype are shown in Table 1 and genotypic frequencies for the Cypriot population studied are shown in Table 2.
Baseline characteristics of the population in all subjects and according to MMP genotypes.
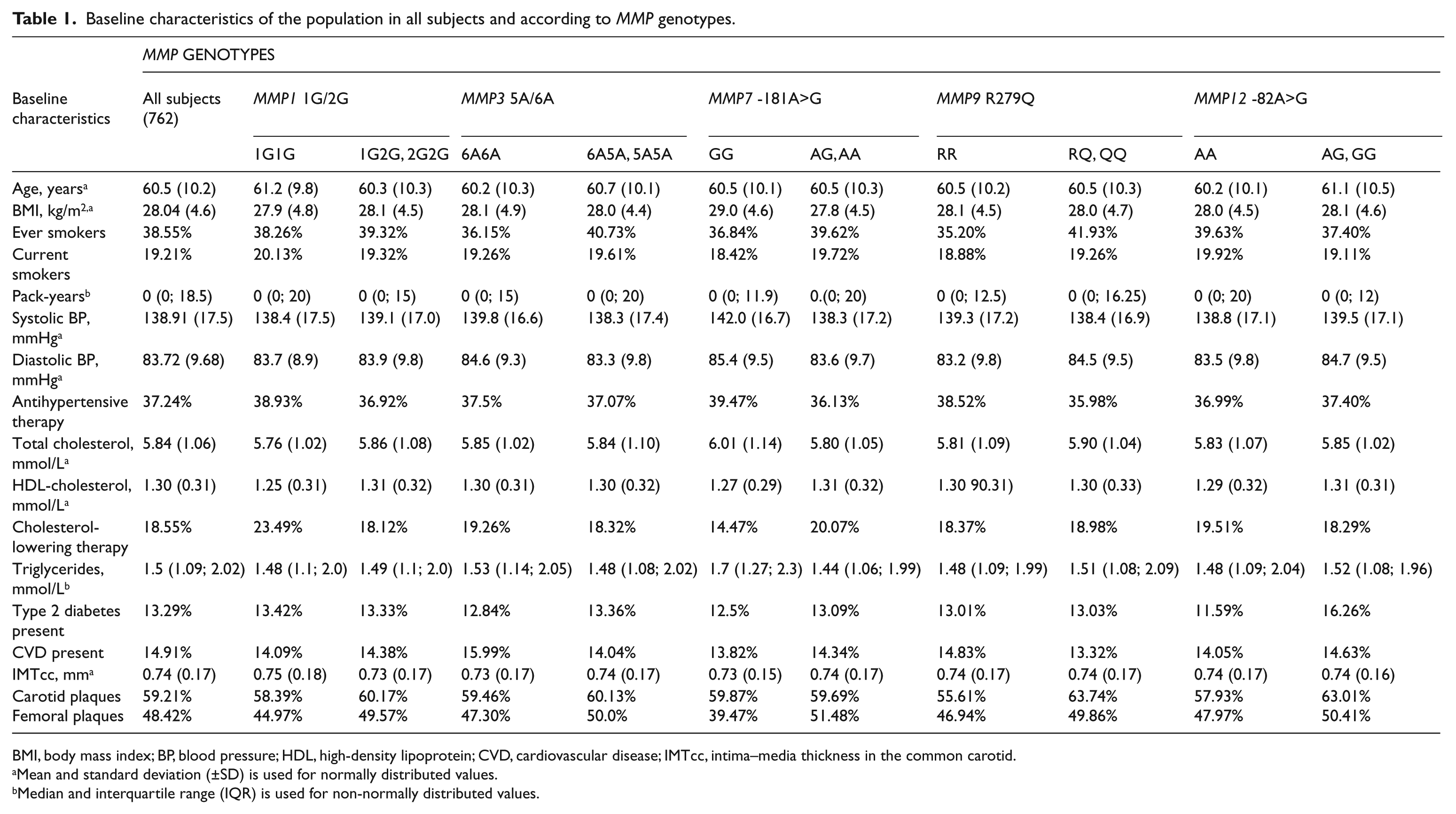
BMI, body mass index; BP, blood pressure; HDL, high-density lipoprotein; CVD, cardiovascular disease; IMTcc, intima–media thickness in the common carotid.
Mean and standard deviation (±SD) is used for normally distributed values.
Median and interquartile range (IQR) is used for non-normally distributed values.
Genotypic frequencies in the Cyprus Study population.
IMTcc and MMP genotypes
In our study population, there was some weak evidence that the MMP12 -82A>G genotype (AA vs AG, GG) was associated with IMT greater than 0.07 cm (population mean) especially before (Model 1: OR=1.51; 95% CI=1.06 to 2.14; p=0.02) adjusting for additional risk factors (Model 2: OR=1.45; 95% CI=0.99 to 2.10; p=0.05). In a step-wise regression, the best model explaining the variation in IMT > 0.07 cm included age, BMI, sex, smoking in pack-years and the MMP12 -82A>G genotype (R2 for the model=0.22). Results are shown in detail in Table 3.
Associations between subclinical atherosclerosis outcomes and MMP genotypes in all subjects: (a) Model 1 and (b) Model 2.
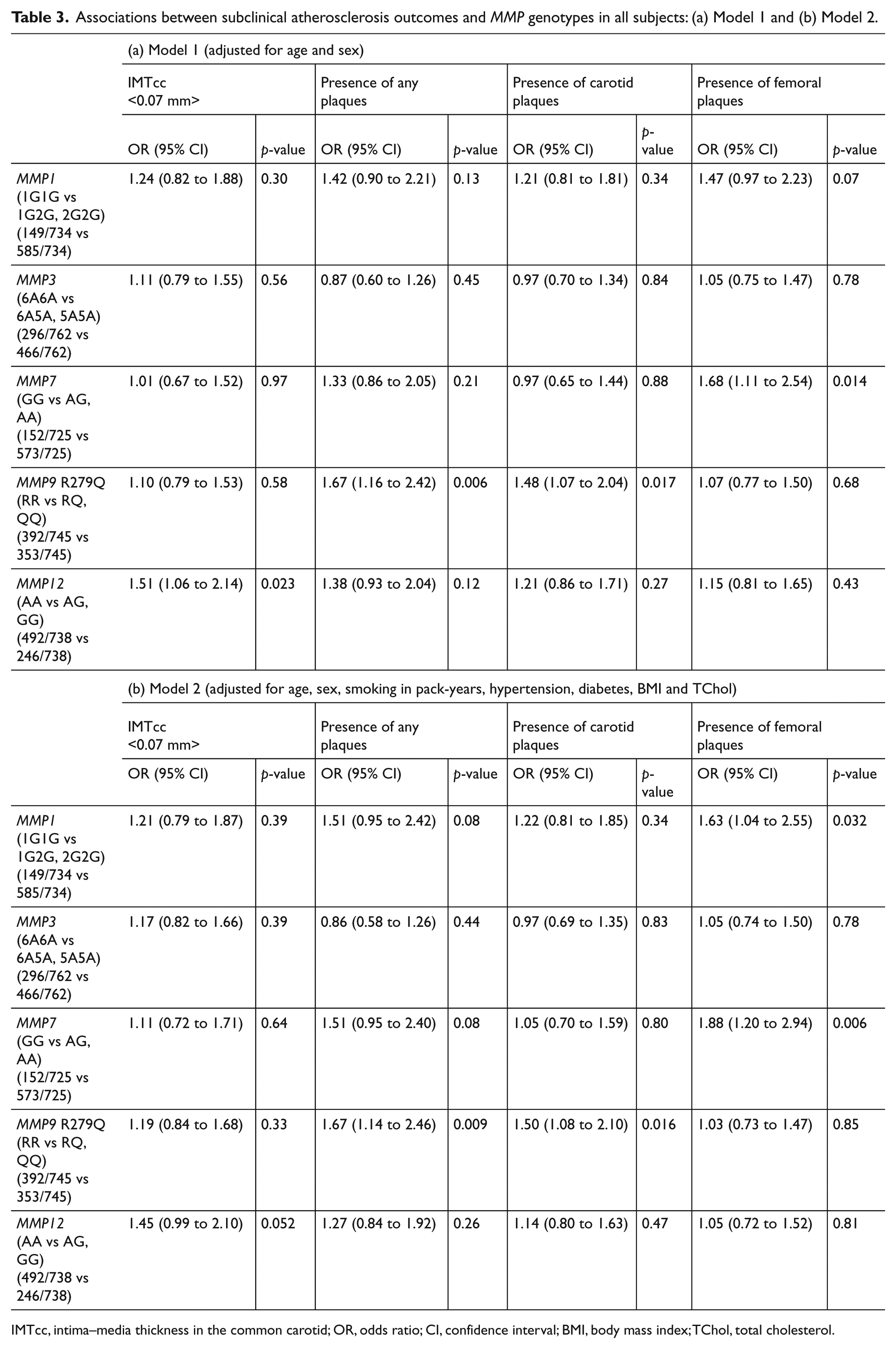
IMTcc, intima–media thickness in the common carotid; OR, odds ratio; CI, confidence interval; BMI, body mass index; TChol, total cholesterol.
Plaque presence and MMP genotypes
Presence of plaques (0 versus 1–4 bifurcations with plaque) was strongly associated with the MMP9 R279Q polymorphism (RR versus RQ, QQ) both before (Model 1: OR=1.67; 95% CI=1.16 to 2.42; p=0.006) and after controlling for CVD risk factors (Model 2: OR=1.67; 95% CI=1.14 to 2.46; p=0.009) with no change in the effect estimate, indicating that the association was not significantly confounded by the risk factors in the model. Further analysis showed that the association was driven by the presence of plaques in the carotid (Model 1: OR=1.5; 95% CI=1.07 to 2.04; p=0.017) rather than femoral bifurcations (Model 1: OR=1.07; 95% CI=0.77 to 1.50; p=0.68), and the association persisted even after further adjustment (Model 2: OR=1.5; 95% CI=1.08 to 2.10; p=0.016 for carotids). In a step-wise regression, the best model explaining the variance in carotid plaque presence included age, sex, hypertension, the MMP9 R279Q genotype and diabetes (R2 for the model=0.14).
There was a trend between presence of femoral plaques and the MMP7 -181A>G polymorphism (GG vs AA, AG) before (Model 1: OR=1.68; 95% CI=1.11 to 2.54; p=0.014) but especially after further adjustment, with carriers of the A allele having nearly twofold increased odds for the presence of femoral plaques (Model 2: OR=1.88; 95% CI=1.20 to 2.94; p=0.006). Results are shown in detail in Table 3.
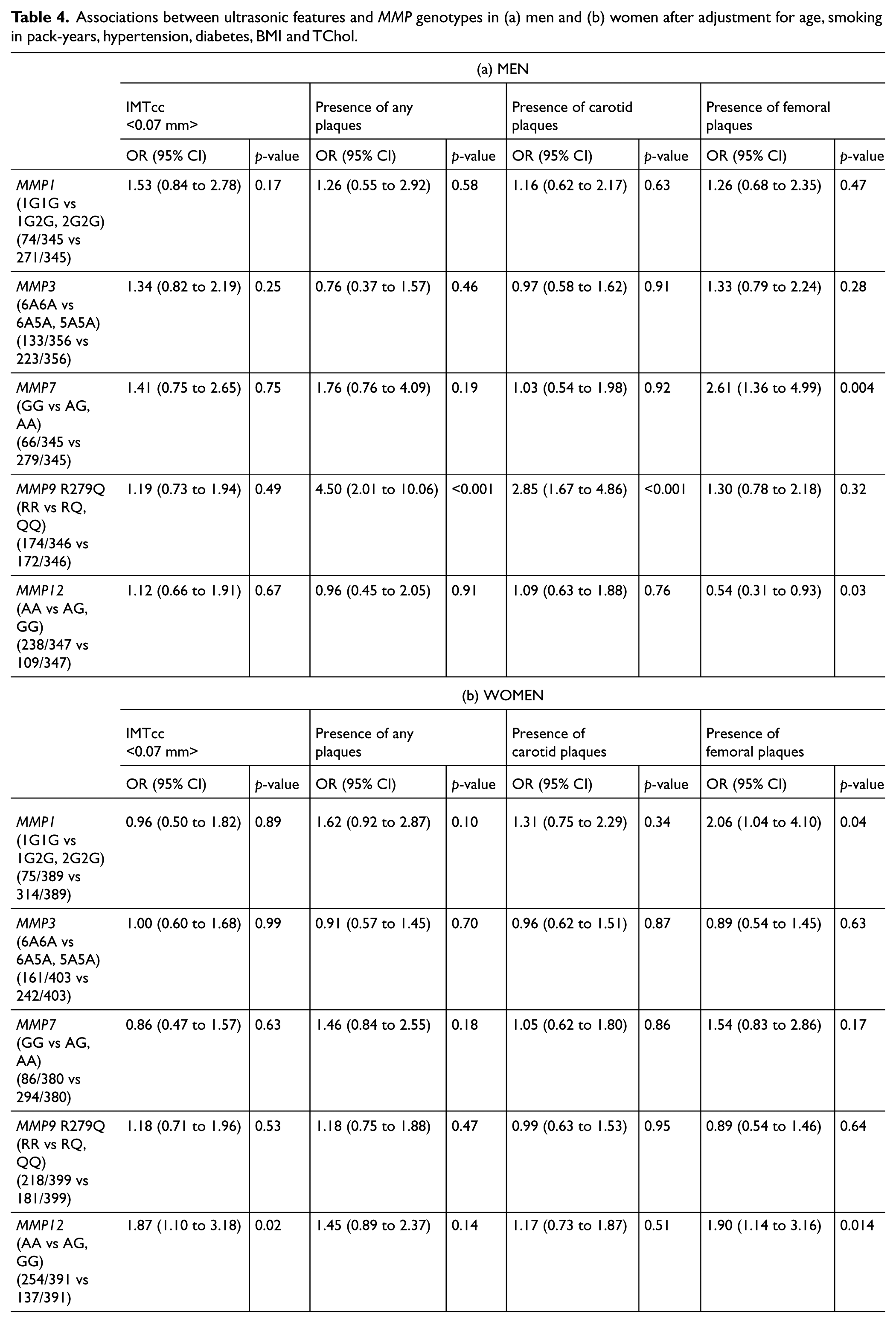
Given the differences between men and women in subclinical atherosclerosis shown by our team 20 and others,21,22 a sex-specific analysis was also carried out, which showed that both the association between the MMP9 R279Q genotype and plaque presence in the carotids (Model 2: ORmen=2.85; 95% CI=1.67 to 4.86; p<0.001; p for sex interaction=0.003) and the MMP7 -181A>G genotype and plaque presence in the femoral bifurcations were driven by men (Model 2: ORmen=2.61; 95% CI=1.36 to 4.99; p=0.004; p for sex-interaction<0.001). The trend between the MMP12 -82A>G genotype and IMT greater than 0.07 cm was more evident in women (Model 2: ORwomen=1.87; 95% CI=1.10 to 3.18; p=0.02; p for sex interaction=0.33) although it did not reach statistical significance. In women, the MMP12 -82AG and GG genotypes showed a weak association with the presence of femoral plaques after adjustment (Model 2: ORwomen=1.90; 95% CI=1.14 to 3.16; p=0.014; p for sex interaction<0.001). Results for men and women separately are shown in detail in Table 4.
Associations between ultrasonic features and MMP genotypes in (a) men and (b) women after adjustment for age, smoking in pack-years, hypertension, diabetes, BMI and TChol.
Discussion
Several matrix metalloproteinases (MMPs) have been reported to be expressed in atherosclerotic lesions; however, the exact role of each one in the formation and progression of atherosclerosis remains to be fully elucidated. We studied functional genetic polymorphisms in genes encoding for five MMPs (MMP1 (1G/2G), MMP3 (5A/6A), MMP7 (-181A>G), MMP9 (R279Q) and MMP12 (-82A>G)) in relation to subclinical atherosclerosis phenotypes as assessed by ultrasound, in a large (n=762), middle-aged, general population cohort.
We report a strong association between the MMP9 279Q allele and a three- to fourfold increase in risk of having any bifurcation affected by plaque or having a plaque in the carotid bifurcation; this association was mainly driven by men (Model 2: ORmen=4.5; p<0.001 for any plaque and ORmen=2.85; p<0.001 for carotid plaques). Men were also more likely to have a plaque in the femoral arteries if they carried the MMP7 -181A allele (Model 2: OR=2.6; p=0.004 and p for sex interaction<0.001). There was some evidence for association between having a femoral plaque and carrying the MMP12 -82G allele in women (Model 2: OR=1.9; p=0.014 and p for sex-interaction=0.01).
A study by Armstrong et al. looking at six polymorphisms in four MMP genes in a similar-sized community cohort, in relation to IMT, reported an association between the MMP9 R279Q genotype and internal carotid artery bulb IMT; however, the authors suggest this may have been a spurious finding, as no association was seen with common carotid or bulb IMT. 23 In the Framingham study, circulating MMP9 was not associated with IMTcc or IMT in the bulb but was, however, associated with internal carotid stenosis <25% 24 (i.e. plaque presence). We also show no association between the MMP9 R279Q and IMTcc (>0.07 cm) but report a strong association between the MMP9 279Q allele and presence of plaques in men; especially presence of carotid plaques. These results provide further support to the notion that IMT and atherosclerotic plaque may represent different pathophysiological mechanisms.25,26 Our results are also in agreement with previously reported sex differences in atherosclerosis phenotypes (carotid plaque and stenosis) 27 as well as with a recently published study reporting a sex-specific effect of another MMP9 functional polymorphism and carotid arterial stiffness. 21
Both MMP9 and MMP12 degrade components of the basement membrane, thus allowing macrophages to penetrate injured tissues during inflammation, as well as promoting vascular smooth muscle cell migration. Jormsjo and colleagues 28 reported an association between the MMP12 -82A>G polymorphism and coronary luminal diameter in diabetic patients, while findings by Orbe et al. 29 show higher expression of MMP9 in occlusive lesions from carotid but not femoral arteries and further support differential expression of MMPs in different plaque types and arterial beds. A recently published study also did not find any association between the MMP12 -82G allele and carotid plaque susceptibility in Chinese patients with acute cerebral infarctions; 30 however, both the differences in the populations studied and the allele frequencies found (3.8% for the G allele in the Chinese population versus 18.9% in the Cypriot population studied) make these two studies difficult to compare. We show an association between the MMP12 -82G allele and femoral plaques in women (p=0.014) but given that this association was in the opposite direction in men (although not reaching statistical significance after adjustment for multiple testing), this may have been a spurious finding.
MMP7 has been found to be expressed in human atherosclerotic lesions but its exact role has not been defined. One recent study reported an association between the MMP7 -181G allele and presence of vulnerable plaques – but not stable plaques – in the carotids in a Chinese population. 31 We report for the first time an association between the MMP7 -181A>G polymorphism and presence of femoral plaques in men. Femoral plaques have been previously reported to show a different morphology (more fibrocalcific plaques) compared to carotid plaques (more fibrous cap atheroma) 15 and the pattern of disease progression was shown to be sex-dependent.13,14
In our study population, no association was seen between the MMP1 2G allele and carotid plaques, in contrast to a recent case–control study in a Serbian population. 32 However, the association shown was with severe carotid plaques in patients undergoing endarterectomy, which may not be comparable to our general population subjects, most of whom had non-severely stenotic plaques. We show a weak association in women between the MMP1 2G allele and presence of femoral plaques but this result could be due to chance, as it did not reach statistical significance after adjustment for multiple comparisons.
We did not find any association between the MMP3 5A6A polymorphism and the atherosclerotic phenotypes studied, despite some previous reports for an association between the 6A6A genotype and increased IMT.33–35 These discrepancies could be explained by the much smaller size of those studies as well as different protocols of measuring IMT at different sites. In a recent meta-analysis of the effect of the MMP3 5A6A polymorphism on coronary disease using data from 15 studies, differential effects were suggested, with studies in subjects of European descent showing no association between myocardial infarction and the MMP3 5A allele, and studies in subjects of Asian descent suggesting a positive association. 36
It is worth noting that ~19% of our population is on anti-hyperlipidemic treatment and statins have been found to reduce plasma activity of MMP2 and cell death and increase collagen content in human carotid plaques 37 with cardioprotective effects. It is possible that those with plaques and/or prevalent CVD have other risk factors as well and are receiving treatment; however, when repeating the analysis, excluding those on anti-hyperlipidemic treatment, results did not change. Contacting an additional sensitivity analysis by excluding those with prevalent CVD from the analysis resulted in no significant difference in the results either (results not shown).
A strength of our study is the community-based sample and the detailed phenotypic characterization of the subjects with regards to ultrasonically determined atherosclerosis, as well as having data from both carotid and femoral bifurcations. A limitation is the use of baseline, cross-sectional data in the analysis, restricting the possibility of causal inferences from our data and allowing for bias. However, we adjusted our findings for common risk factors for atherosclerosis and conducted relevant sensitivity analyses with no changes in our results. Prospective studies measuring progression of IMT and plaque development are still warranted. An additional limitation is that while we assume that the effect of the MMP7, MMP9 and MMP12 variants on plaque is due to their influence on tissue levels of the respective enzymes, we do not have any direct measure of these in order to investigate this.
Our findings are in agreement and provide further support to previous reports,24,29 suggesting that the proteolytic mechanisms governed by MMPs may differ between carotid and femoral arteries and between sexes 32 and suggesting a complex role of MMPs in plaque formation and development. This is in agreement with the theory of disease susceptibility versus disease-modifying genes, where the former may contribute to the initiation of disease while the latter may influence progression and outcome. 38
The MMP7 -181A and the MMP9 279Q alleles were associated with a greater than 2.5 times the risk of having an atherosclerotic plaque in the femoral and carotid arteries (respectively) after adjusting for common risk factors in middle-aged men. Our findings are consistent with site-specific matrix remodeling and support a role for the use of common MMP genotypes in studies of subclinical atherosclerosis.
Footnotes
Appendix
Primers and probes used for genotyping.
| SNP | Forward primer and VIC-probe | Reverse primer and FAM-probe |
|---|---|---|
| MMP1 (1G/2G) rs1799750 |
F-ACATGTTATGCCACTTAGATGAGGAAA | R-GCGTCAAGACTGATATCTTACTCATAAACAATA |
| VIC-TTGAGATAAGTCATATCCTTTCTA | FAM-TTGAGATAAGTCATATCTTTCTA | |
| MMP3 (5A/6A) | F-TCAATGTGGCCAAATATTTTCCCTGTA | R-ATTCTATGGTTCTCCATTCCTTTGATGG |
| rs3025058 | VIC-ACATGGTTTTTTCCCC | FAM-ACATGGTTTTTCCCC |
| MMP7 (-181A>G) |
F-GGAGTCAATTTATGCAGCAGACAGA | R-AGTGTTTTCTTTCTTTTTAGAGTCTACAGAACT |
| VIC-CTTTGAAAGACAAATACA | FAM-CTTTGAAAGACGAATACA | |
| MMP9 (R279Q) | F-TCCCCCTTTCCCACATCCT | R-CAGGGTTTCCCATCAGCATTG |
| rs17576 | VIC-CTCTACACCCAGGACGG | FAM-TCTACACCCGGGACGG |
| MMP12 (-82A>G) |
F-TGCTTTTGTTTGCATGTTTTTGAGATAGA | R-CCGGGTTCTGTGAATATGAATCCT |
| VIC-ATGATATCAACTATGAGTCACT | FAM-ATATCAACTGTGAGTCACT |

SNP, single nucleotide polymorphism.
Acknowledgements
The authors express their gratitude to all the participants of the Cyprus Study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by a Cyprus Cardiovascular Disease Educational and Research Trust (CCDERT) and Joint Cyprus Research Promotion Foundation, Ministry of Health and Cyprus Heart Foundation grant (No. 41/5PE) and a Research Promotion Foundation grant (PENEK-05/04). SEH is funded by the British Heart Foundation (RG2008/015).
