Abstract
The purpose of this study was to investigate whether a polymorphism in the matrix metalloproteinase-12 gene (MMP-12 −82A/G) is correlated with serum protein levels or with the susceptibility for carotid plaques in the Chinese Han population. Polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) was performed on the −82A/G polymorphism in the MMP-12 gene for 1314 patients with acute cerebral infarctions; 710 of these cases were diagnosed with stable plaques, 340 cases were diagnosed with vulnerable plaques and 264 cases had no plaques. At the same time, serum MMP-12 levels were measured using the enzyme-linked immunosorbent assay (ELISA). Compared to the AA genotype, the frequency of the AG+GG genotypes was not significantly different between the three groups (χ2 = 1.242, p = 0.537), and the frequency of the G allele of the MMP-12 gene was not different within the three subgroups (χ2 = 1.218, p = 0.544). There were no significant differences in MMP-12 protein levels among the three groups (F = 0.675, p = 0.510); similarly, there was no difference in MMP-12 protein levels between the stable plaque group and the vulnerable plaque group (p = 0.755). There was also no difference between the vulnerable plaque group and the no plaque group (p = 0.420). The current data suggest that the inter-individual variability in the MMP-12 gene variation may not be a risk factor for vulnerable plaques in the Chinese Han population.
Introduction
Cerebrovascular disease has become the leading cause of disability and death in urban and rural populations, and the trends in incidence have increased year by year. Epidemiological studies indicate that there are approximately 150–200 million new cases of stroke each year in China; the incidence rate of stroke is approximately (116–219)/10 million people after adjusting ages in the population, and the mortality rate is approximately (58–142)/10 million people each year. 1 Currently, there are more than 700 million patients with cerebrovascular disease; ischemic stroke accounts for approximately 70% of these cases. If we do not take timely and effective interventions, heavy burdens will result for our society and economy. With the rapid rise of the economy and changes in lifestyle, the incidence rate of ischemic stroke, which arises from atherosclerosis, is increasing rapidly. Carotid atherosclerosis and the recurrence of ischemic stroke are closely related. Much evidence has proved that most lesions causing ischemic events are not in the narrowing of blood vessels, but are instead due to atherosclerotic plaque rupture or fissure and thrombosis, resulting in blood vessel blockage. Therefore, the biological characteristics of atherosclerosis, including plaque vulnerability, deserve widespread attention.
In a histological classification system proposed by the American Heart Association (AHA), atherosclerotic lesions are divided into six types representing different stages of the disease. A modification of this system has been proposed by Morgan et al., differentiating advanced lesions into those with a thick fibrous cap and those with a thin one. 2 Stable atherosclerotic plaque with a thick cap that is rich in collagen and smooth muscle cell, whereas a large lipid core covered by a thin fibrous cap with dense inflammatory infiltrates characterizes the vulnerable plaque.
Matrix metalloproteinases (MMPs) are a super family of zinc-dependent proteases, which play a major role in degrading the extracellular matrix (ECM). MMPs can digest the composition of the fibrous plaque cap, resulting in damage to its structure and accelerating plaque rupture, which is an important reason for plaque instability.3,4 MMP-12 is a key member of the family of MMPs and is secreted by macrophages. MMP-12 displays broad substrate specificity, including extracellular matrix proteins such as fibronectin, laminin, vitronectin, type IV collagen, and heparan sulfate.5,6 Thus, MMP-12 not only digests elastin, but also degrades the basement membrane, which enables macrophages to penetrate injured tissues during inflammation. In a previous study, 7 MMP-12 expression was found to be correlated with advanced and ruptured human atherosclerotic plaques. In addition, a common functional MMP-12 ( −82A/G) promoter polymorphism that increases its expression is associated with increased coronary artery stenosis in diabetic patients. A common polymorphism was identified within the promoter of the MMP-12 gene (an adenosine [A]-to-guanosine [G] substitution at position −82) that influences the binding of the transcription factor activator protein-1 (AP-1). The greater binding affinity of AP-1 to the A allele is associated with higher MMP-12 promoter activity in vitro. Therefore, it is interesting to investigate whether the MMP-12 −82A/G polymorphism is associated with carotid plaque formation and progression in the Chinese Han population.
Methods
Study population
All of the subjects with cerebral infarctions were patients who were hospitalized in the Department of Neurology for the first time from March 2010 to June 2011. The diagnosis of the atherothrombotic cerebral infarction was in line with the newly classified subtype of ischemic stroke determined by Han. 8 Subjects were confirmed for atherothrombotic cerebral infarctions with head CT and/or MRI; asymptomatic cerebral infarctions, infarctions with bleeding and cerebral hemorrhages were excluded. Carotid artery ultrasonic scanning was performed on patients who met the requirements within 48 hours of patient admission. 8 Based on the ultrasonic criteria for the classification of the carotid plaques, patients who had carotid plaques with echogenic characteristics were classified into the stable plaque group (n = 710; 392 male cases, 318 female cases; ages ranging from 43 to 91 years, with an average age of 69.6 ± 9.4 years). Patients who had plaques with echolucent or mixed echo characteristics were classified into the vulnerable plaque group (n = 340; 218 male cases, 122 female cases; ages ranging from 44 to 89 years, with an average age of 68.8 ± 10.5 years). Patients with no plaques or plaques less than 1 mm thick intima were classified into the no plaque group (n = 264; 138 male cases, 126 female cases; ages ranging from 43 to 85 years, with an average of 67.8 ± 9.9 years). All of the subjects were of Han Chinese heritage without a history of cerebral apoplexy. No consanguinity relationship existed among the cases in the study. None of the subjects had any of the following conditions: tumors, systematic inflammatory disease and autoimmune disease, severe trauma (within 6 months), serious infection (within 6 months), liver and kidney disease, blood disease, thyroid disease, tuberculosis, malignant tumors, heart valve disease, atrial fibrillation, heart failure, acute coronary syndrome, peripheral vascular disease, or pregnancy. Additionally, none of the subjects had recently taken glucocorticoids or immunosuppressants (within 6 months), and none of the subjects had taken antiplatelet agents or cholesterol drugs for 1 month prior to admission. There were no significant differences in the mean age or sex among the three groups. The Medical Ethics Committee of Taizhou Hospital approved the study, and all patients provided written informed consent.
Ultrasound assessment of carotid plaques
Carotid ultrasound examinations were performed in the following pre-defined areas: right distal common carotid artery, left distal common carotid artery, right bulb, left bulb, right proximal internal carotid artery, and left proximal internal carotid artery. Atherosclerotic plaques were defined as a local thickening of the intima ≥ 1 mm. The measurement was performed by a 7–12 MHz scanning frequency ultrasound machine in B-mode (Sonos 5500; Agilent, Santa Clara, CA, USA). All measurements of carotid plaques in this study were made by the same certified vascular ultrasound technologist.
Based on previous ultrasonic criteria 9 for the classification of carotid plaques, patients were divided into three groups: stable plaque group (echogenic plaque), vulnerable plaque group (echolucent plaque) and no plaque group (local thickening of the intima < 1 mm).
Polymerase chain reaction-restriction fragment length polymorphisms analysis (PCR-RFLP)
Genomic DNA was extracted from whole-blood samples using the Blood Genomic DNA Isolation Mini Kit (TaKaRa Biotechnology Co., Ltd, Dalian, China) according to the manufacturer’s protocol. The DNA concentration was determined by the absorption at 260 nm in a BioPhotometer Plus (Eppendorf, Hamburg, Germany). All DNA samples were frozen at −80°C until use.
The specific primers used to amplify the MMP-12 polymorphism were 5′-GTCAAGGGATGATATCAGCT-3′ (forward) and 5′-CTTCTAAACGGATCAATTCAG-3′ (re-verse), synthesized by GeneCore Biotechnologies Co., Ltd (Shanghai, China). The PCR reaction was performed in a 25 µl volume containing 0.1 µg of DNA, 2.5 µl of 10× buffer, 1 µl of 25 mM MgCl2, 0.5 µl of 10 mM dNTP, 0.5 µl of 20 µM primers (forward and reverse), 0.5 µl of 5 U/µl Taq DNA polymerase (Promega, Madison, WI, USA) and the appropriate volume of sterile water. The cycling parameters for the amplification of the MMP-12 gene were the following: pre-denatured at 95°C for 5 min, followed by 28 cycles at 94°C for 45 s, 53°C for 30 s and 72°C for 45 s, and a final extension period at 72°C for 10 min. PCR was performed with a Thermo P×2 instrument (Waltham, MA, USA). PCR products were purified (Agarose Gel DNA Fragment Recovery Kit; TaKaRa) and digested with the Pvu II restriction enzyme (Ferments, Glen Burnie, MD, USA) for 16 h at 37°C. The digested products were electrophoresed on a 2.5% agarose gel. Ten percent of each PCR sample was sequenced by a Prism 3100 DNA Genetic Analyzer (Applied Biosystems, Carlsbad, CA, USA) in order to validate the result of the PCR-RFLP.
For the determination of serum protein levels of MMP-12, the human MMP-12 enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems China Co. Ltd, Shanghai) were used according to the manufacturer’s instructions.
Statistical analysis
Statistical analyses were performed with SPSS version 17.0 software. Measurement data are presented as the mean ± standard deviation (SD). We used an ANOVA analysis with an LSD post-hoc test to compare the measurement data among the three groups. Countable data were assessed using the R × C chi-squared test (χ2) and partitions of the χ2 method. Hardy–Weinberg analysis was performed to compare the observed and expected genotype frequencies using the χ2 test. Logistic regression analysis was performed to test for interactions between genotypes and clinical parameters and to extract the most prognostic factors for vulnerable plaques. Two-sided probability values of p < 0.05 or p < 0.0125 were considered significant.
Results
Demographic and clinical features of the subjects are summarized in Table 1. Of the 1314 patients, 710 patients with stable carotid plaques, 340 patients with vulnerable carotid plaques and 264 control subjects were enrolled in the study. Statistical analyses indicated that except for levels of high-density lipoprotein-cholesterol (HDL-C), fibrinogen (FG) and homocysteine (Hcy), none of the clinical parameters showed any significant differences among the three groups.
Demographic and clinical characteristics of the subjects (sample size = 1314)
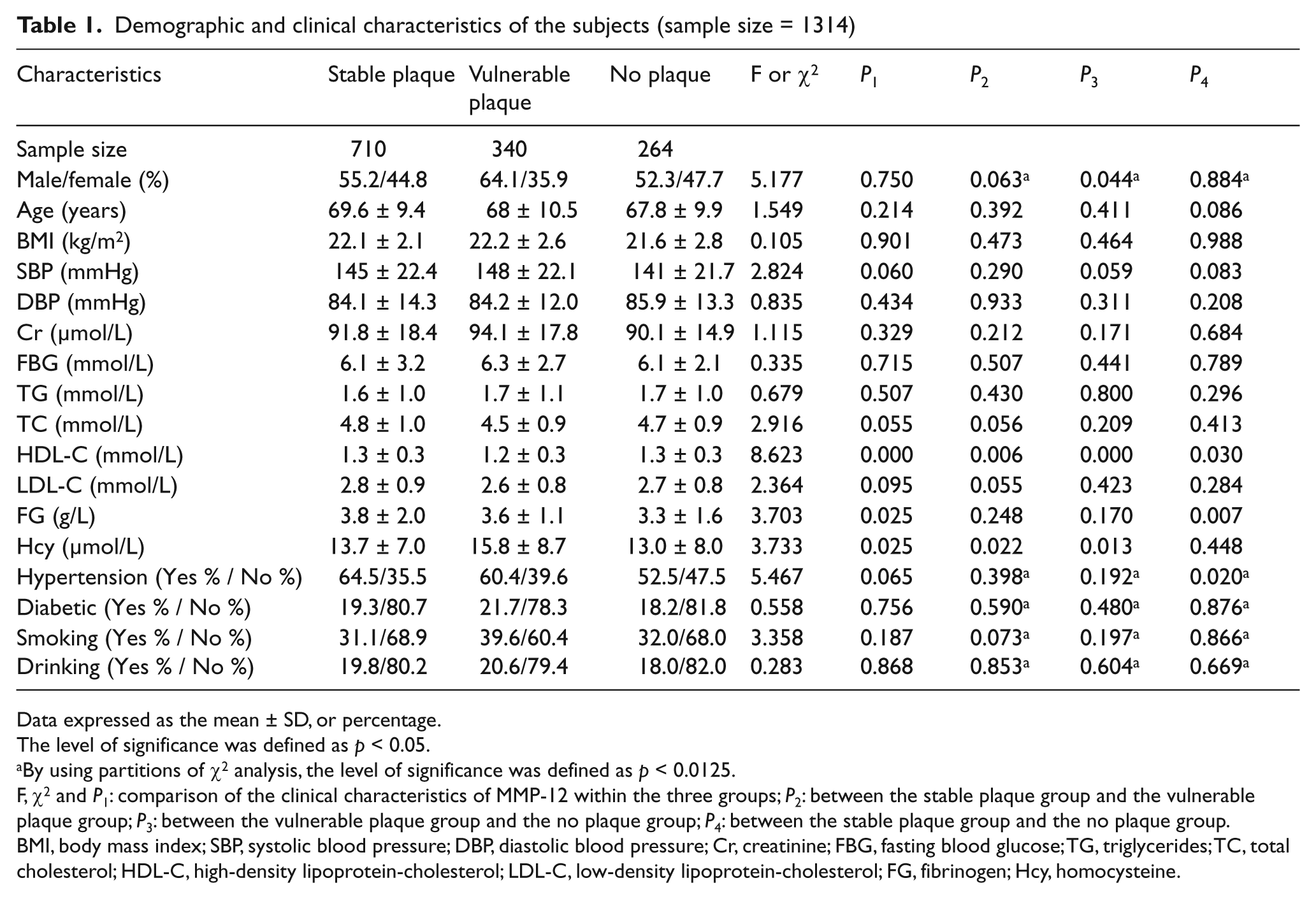
Data expressed as the mean ± SD, or percentage.
The level of significance was defined as p < 0.05.
By using partitions of χ2 analysis, the level of significance was defined as p < 0.0125.
F, χ2 and P1: comparison of the clinical characteristics of MMP-12 within the three groups; P2: between the stable plaque group and the vulnerable plaque group; P3: between the vulnerable plaque group and the no plaque group; P4: between the stable plaque group and the no plaque group.
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; Cr, creatinine; FBG, fasting blood glucose; TG, triglycerides; TC, total cholesterol; HDL-C, high-density lipoprotein-cholesterol; LDL-C, low-density lipoprotein-cholesterol; FG, fibrinogen; Hcy, homocysteine.
The binary logistic regression analysis (Table 2) shows that low-density lipoprotein-cholesterol (LDL-C) levels are a risk factor for the vulnerable plaque group compared to the stable plaque group (p = 0.040, OR = 1.809, 95% CI (1.027, 3.187)). The p-value for the Hcy levels is less than 0.05, and the odds ratio (OR) is less than 1.
Binary logistic regression estimating the risk factors for vulnerable plaque
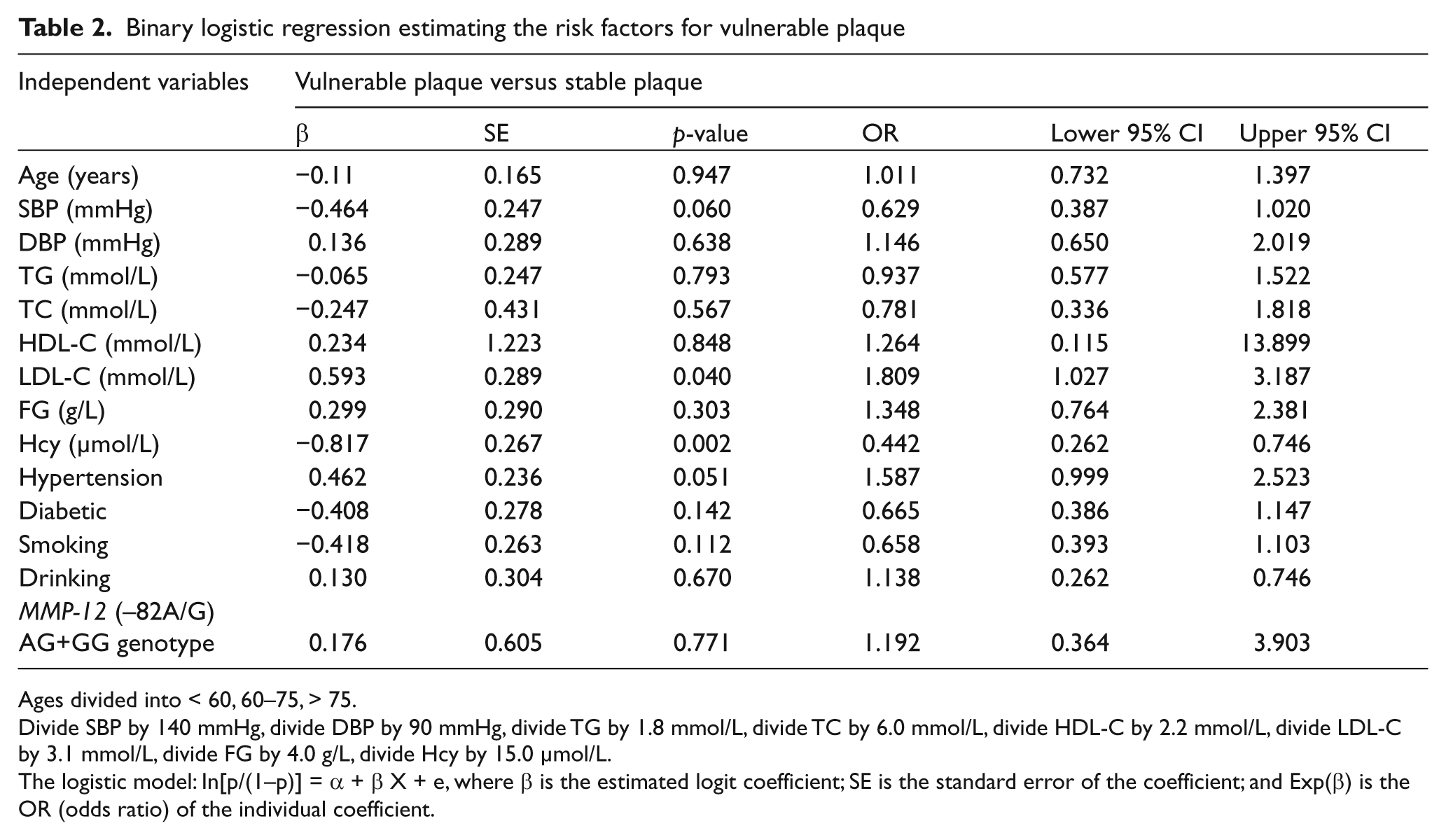
Ages divided into < 60, 60–75, > 75.
Divide SBP by 140 mmHg, divide DBP by 90 mmHg, divide TG by 1.8 mmol/L, divide TC by 6.0 mmol/L, divide HDL-C by 2.2 mmol/L, divide LDL-C by 3.1 mmol/L, divide FG by 4.0 g/L, divide Hcy by 15.0 µmol/L.
The logistic model: In[p/(1–p)] = α + β X + e, where β is the estimated logit coefficient; SE is the standard error of the coefficient; and Exp(β) is the OR (odds ratio) of the individual coefficient.
Moreover, age, systolic blood pressure (SBP) and FG are risk factors for the vulnerable plaque group compared to the no plaque group (Table 3). HDL-C is a protective factor in Table 3 (p = 0.014, OR = 0.289, 95% CI (0.107, 0.780)).
Binary logistic regression estimating the risk factors for vulnerable plaque
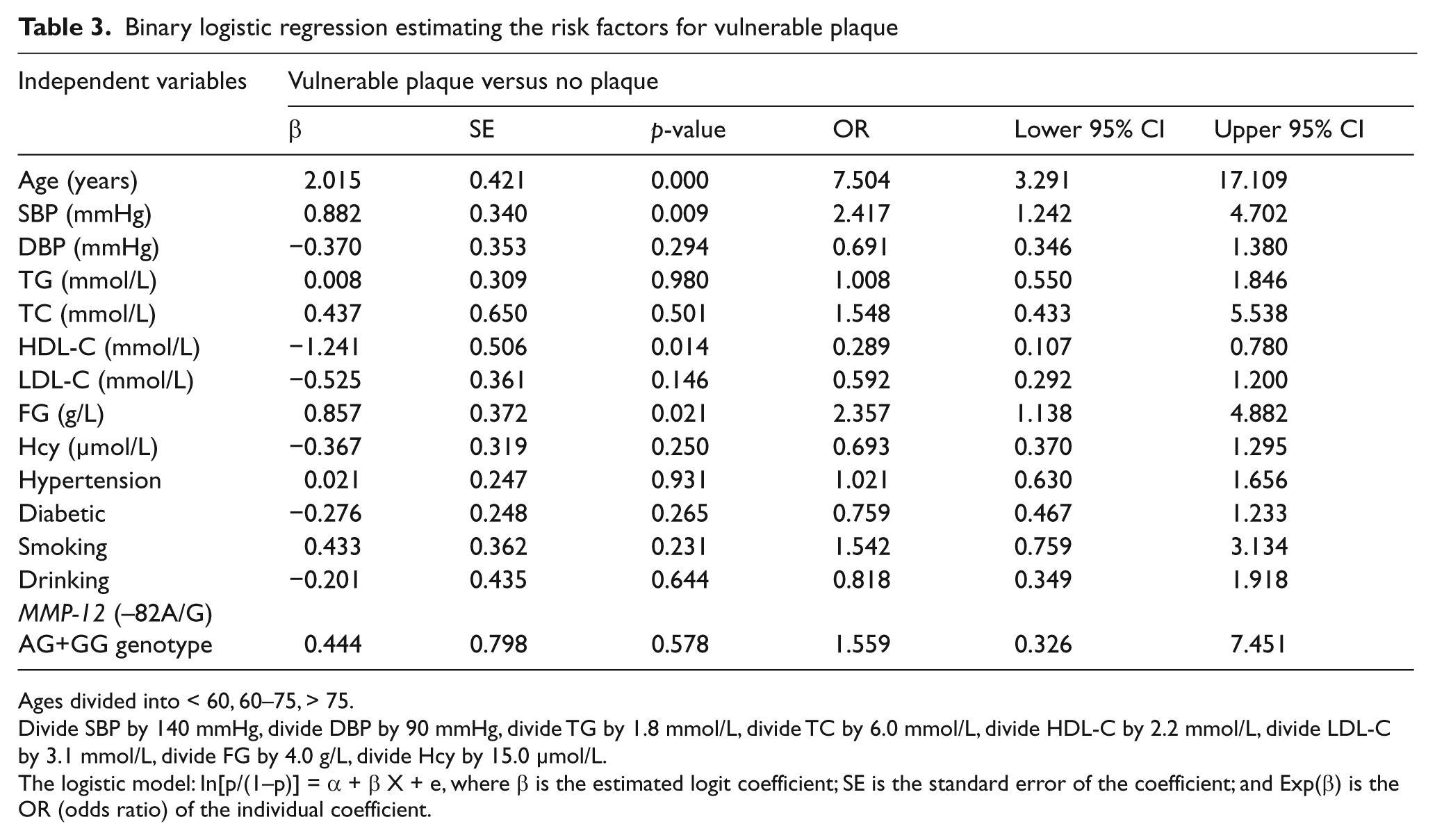
Ages divided into < 60, 60–75, > 75.
Divide SBP by 140 mmHg, divide DBP by 90 mmHg, divide TG by 1.8 mmol/L, divide TC by 6.0 mmol/L, divide HDL-C by 2.2 mmol/L, divide LDL-C by 3.1 mmol/L, divide FG by 4.0 g/L, divide Hcy by 15.0 µmol/L.
The logistic model: In[p/(1–p)] = α + β X + e, where β is the estimated logit coefficient; SE is the standard error of the coefficient; and Exp(β) is the OR (odds ratio) of the individual coefficient.
The size of the PCR products that were amplified by the MMP-12 gene-specific primers was 137 bp. The PCR products were digested with the Pvu II restriction enzyme, and the results indicate that the AA genotype of the −82A/G polymorphism in the MMP-12 gene produced a single band (137 bp), whereas the AG genotype produced three bands (137 bp, 119 bp, 18 bp). We did not find the GG genotype in this study, as shown in Figure 1. Because the molecular weight of 18 bp is smaller, and the electrophoretic speed obviously quickened in agarose gel, it was not shown in Figure 1.
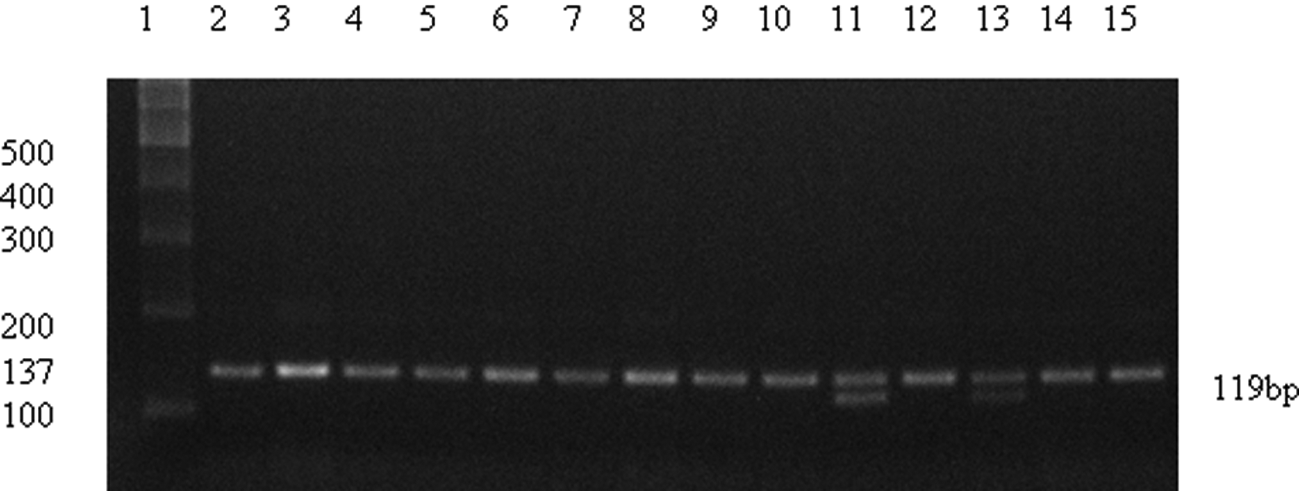
Electrophoresis of the MMP-12 ( −82A/G) products digested with the Pvu II restriction enzyme. Lane 1 represents the 100 bp DNA ladder; lanes 2, 3, 4, 5, 6, 7, 8, 9, 10, 12, 14 and 15 represent the AA genotype (a single band of 137 bp); lanes 11 and 13 represent the AG genotype (three bands of 137 bp, 119 bp and 18 bp (not visible)).
The genotype and allele frequencies of the MMP-12 ( −82 A/G) polymorphism in the three groups are displayed in Table 4. The genotype frequencies in the three groups were in accordance with the Hardy–Weinberg equilibrium (χ2 = 0.144, p = 0.705 for the stable group; χ2 = 0.099, p = 0.753 for the vulnerable group; χ2 = 0.989, p = 0.845 for the no plaque group), suggesting that our sample was representative of the population. Compared to the AA genotype, the frequency of the AG+GG genotypes was not significantly different among the three groups (χ2 = 1.242, p = 0.537). In addition, the frequency of the G allele was 2% in the stable plaque group, 2.4% in the vulnerable plaque group and 1.1% in the no plaque group. There were also similar trends in the allele frequencies when multiple comparisons of the G allele frequency were done among the three subgroups.
Genotype and allele frequencies of the MMP-12 ( −82A/G) polymorphism
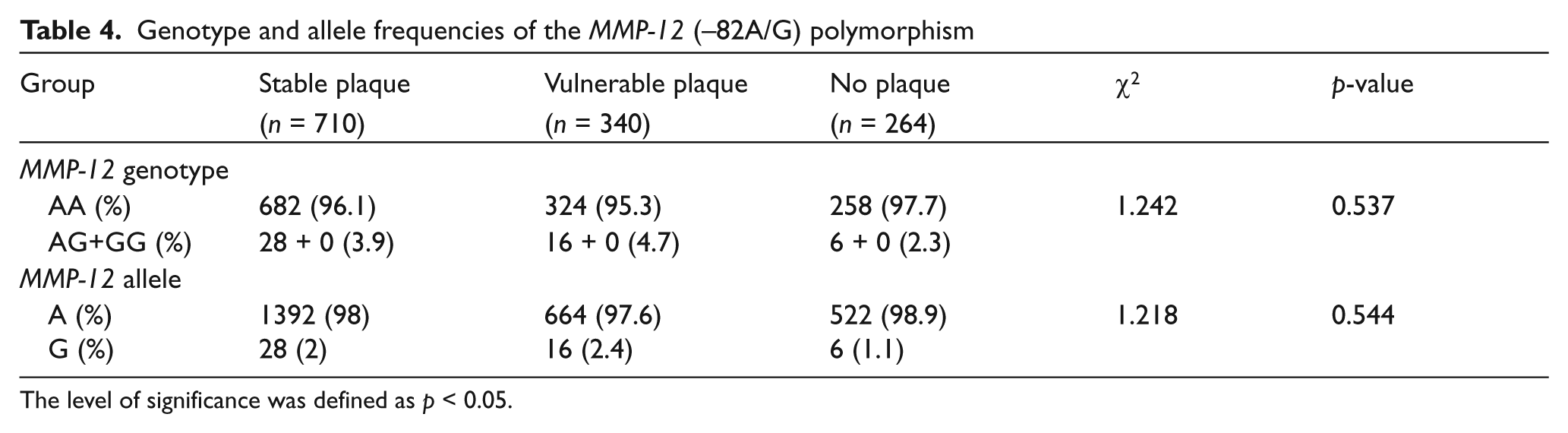
The level of significance was defined as p < 0.05.
The serum MMP-12 protein levels in the three groups are shown in Table 5. MMP-12 protein levels were not significantly different among the three groups (F = 0.675, p = 0.510); likewise, serum MMP-12 protein levels were not significantly different between the stable plaque group and the vulnerable plaque group or between the vulnerable plaque group and the no plaque group (p = 0.755 and p = 0.420).
Comparison of serum MMP-12 levels among the three groups

Data are expressed as the mean ± SD.
The level of significance was defined as p < 0.05.
F and P1: comparison of serum level with three groups; P2: comparison of stable plaque with vulnerable plaque; P3: comparison of vulnerable plaque with no plaque; P4: comparison of stable plaque with no plaque.
Discussion
Atherothrombotic cerebral infarction is primarily related to the rupture of vulnerable plaques in the carotid artery. The rupture of a plaque gives rise to the exposure of thrombogenic materials (e.g. tissue factors and lipids) located in the lipid core to the blood. This leads to thrombus formation and, consequently, to the ischemic event. ‘Vulnerable plaque’ is a term used to identify all thrombosis-prone plaques and plaques with a high probability of undergoing rapid progression, thus becoming culprit plaques. 10 The internal portion of these plaques contains large amounts of lipid, hemorrhage and ulceration, which are echolucent in the ultrasound test.9,10 Because a vulnerable plaque has a thin fibrous cap, it is considered to be prone to rupture, which can result in an acute ischemic event, whereas a plaque with a thick cap is more stable and has a lower complication rate. 11 The family of MMPs consists of over 20 secreted or cell surface enzymes that are capable of degrading extracellular matrix proteins, as well as clotting factors, lipoproteins, latent growth factors, and chemotactic and cell adhesion molecules.12–14 Overexpression of these enzymes in advanced lesions may contribute to the thinning of the plaque cap and to the development of ischemic events resulting from plaque rupture.
MMP-12 (macrophage elastase) was first identified as an elastolytic metalloproteinase secreted by activated macrophages.15,16 In addition to elastase activity, MMP-12 displays a broad substrate specificity, including ECM proteins such as fibronectin, laminin, vitronectin, type IV collagen, and heparan sulfate.5,6 Thus, MMP-12 not only digests elastin but also degrades the basement membrane, which enables macrophages to penetrate injured tissues during inflammation. In a previous study, it was shown that MMP-12 can accelerate the initiation of atherosclerosis and stimulate the progression from fatty streaks to fibrous plaques in transgenic rabbits. 17 In a study by Johnson et al., 18 the data demonstrated that a selective MMP-12 inhibitor (RXP470.1) retards atherosclerosis development and results in a more fibrous plaque phenotype in mice. RXP470.1 treatment results in less complex plaques, an increased smooth muscle cell/macrophage ratio, less macrophage apoptosis, increased cap thickness, smaller necrotic cores, and a decreased incidence of calcification. MMP-12 expression, in particular, has been correlated with advanced and ruptured human atherosclerotic plaques. In addition, a common functional MMP-12 ( −82A/G) promoter polymorphism that increases expression of MMP-12 is associated with increased coronary artery stenosis in diabetic patients. 7 According to the above paper, a common polymorphism was identified within the MMP-12 gene promoter (an adenosine [A]-to-guanosine [G] substitution at position −82) that influences the binding of the transcription factor activator protein-1 (AP-1). The greater binding affinity of AP-1 to the A allele is associated with higher MMP-12 promoter activity in vitro. Furthermore, in a study by Morgan et al., 2 the authors report that MMP-12 transcript levels, which were quantified by real-time reverse-transcriptase polymerase chain reaction, determined atherosclerotic plaque stability in histopathological tissues removed from patients undergoing carotid endarterectomy. However, in a previous study, 19 the MMP-12 ( −82A/G) polymorphism was not associated with coronary aneurysms. In another study, 20 MMP-12 ( −82A/G) gene variations did not influence the development of intracranial aneurysms in the population studied.
The aims of this study were to compare the genotype and allele frequencies of the MMP-12 ( −82A/G) polymorphism along with the serum protein levels of MMP-12 among the stable plaque, vulnerable plaque and no plaque groups. Using the PCR-RFLP method, we found that the frequency of the AG+GG genotypes was not significantly different among the three groups (χ2 = 1.242, p = 0.537); this is in contradiction to the report that the MMP-12 ( −82A/G) promoter polymorphism increases its expression and is associated with increased coronary artery stenosis in diabetic patients. In addition, the frequency of the G allele was extremely low. Only 2% of the stable plaque group, 2.4% of the vulnerable plaque group and 1.1% of the no plaque group have the G allele. There were also similar trends in allele frequencies when multiple comparisons of the G allele frequency were done among the three subgroups. In addition, we also observed that the differences in the serum protein levels of MMP-12 between groups do not reach statistical significance. A comparison of the stable plaque group with the vulnerable plaque group revealed that there is no difference in the serum levels of MMP-12 protein. There was no difference in serum levels of MMP-12 protein between the vulnerable plaque group and the no plaque group as well. In contrast, Jia et al. 21 showed that the serum levels of MMP-12 are markedly increased in an unstable angina pectoris group and an acute myocardial infarction group compared to the control group (stable angina pectoris group).
According to the studies of Jormsjö et al. 7 and Jia et al., 21 the differences in genotype frequency, allele frequency and serum level of MMP-12, which were measured in acute ischemic infarction patients in the present study, are not in accord with those found for coronary artery disease and heart diseases. Although cerebral atherosclerosis and coronary atherosclerosis may have many similarities, at the molecular genetic level the incidence of cerebral infarction may have many of its own characteristics. As there are many neurotransmitters expressed in the brain, and signal transduction and gene expression are extraordinarily complex, the genetic susceptibility to myocardial infarctions due to gene polymorphisms may not hold true for cerebral infarctions. A study from the UK, 2 in which histopathological characteristics were reported, suggests that MMP-12 transcript levels are correlated with carotid atherosclerosis plaque stability. However, the MMP-12 transcript levels are not different in the Chinese Han population, which is perhaps due to the different ethnic background. In addition, we identified LDL-C and SBP as risk factors for carotid plaque formation. Therefore, we also proved that statin and antihypertensive treatment and/or preventing plaque rupture may be effective.
On the other hand, the risk profile of early atherogenesis consists of traditional risk factors, such as age hypertension, hyperglycemia, hyperlipidemia, and cigarette smoking, supplemented by a variety of less well-established risk conditions, including high FG and high Hcy. A variety of cross-sectional surveys has revealed a strong relationship between high FG levels and atherosclerosis.22,23 FG and its degradation products can stimulate smooth muscle cell migration from the arterial media to the intima, compromising vessel lumen. FG can promote platelet activation and aggregation, accelerating the atherosclerotic process. 24 Our study also confirmed that FG is a risk factor during binary logistic regression analysis between the vulnerable plaque and no plaque groups. In addition, we observed that HDL-C is a protective factor in binary logistic regression between the vulnerable plaque group and no plaque group (OR = 0.289, 95% CI (0.107, 0.780)). In contrast to Sasaki et al., 25 who reported a positive relationship between plasma total homocysteine (tHcy) levels and carotid atherosclerosis and cerebral infarction, we observed that it is a protective factor (OR = 0.442, 95% CI (0.262, 0.746)) for vulnerable carotid plaque. Possible explanations include that our study examined the relationship between Hcy and carotid plaque as compared to cerebral infarction. In addition, our survey population sample is small and confined to the Chinese Han population as compared to Caucasians in the prior reported study.
In summary, the current data suggest that the inter- individual variability in MMP-12 gene variation may not be a risk factor for the development of vulnerable plaques, which are known to have a multifactorial etiology.
Footnotes
Funding
This work was supported by Zhejiang Provincial Natural Science Foundation (Y2080618) and Zhejiang Provincial Traditional Chinese Medicine Research Foundation (2011ZA111).
Conflict of interest
None declared.
