Abstract
Fluorescence microlymphography (FML) is used to visualize the lymphatic capillaries. A maximum spread of the fluorescence dye of ≥ 12 mm has been suggested for the diagnosis of lymphedema. However, data on sensitivity and specificity are lacking. The aim of this study was to investigate the accuracy of FML for diagnosing lymphedema in patients with leg swelling. Patients with lower extremity swelling were clinically assessed and separated into lymphedema and non-lymphatic edema groups. FML was studied in all affected legs and the maximum spread of lymphatic capillaries was measured. Test accuracy and receiver operator characteristic (ROC) analysis was performed to assess possible threshold values that predict lymphedema. Between March 2008 and August 2011 a total of 171 patients (184 legs) with a median age of 43.5 (IQR 24, 54) years were assessed. Of those, 94 (51.1%) legs were diagnosed with lymphedema. The sensitivity, specificity, positive and negative likelihood ratio and positive and negative predictive value were 87%, 64%, 2.45, 0.20, 72% and 83% for the 12-mm cut-off level and 79%, 83%, 4.72, 0.26, 83% and 79% for the 14-mm cut-off level, respectively. The area under the ROC curve was 0.82 (95% CI: 0.76, 0.88). Sensitivity was higher in the secondary versus primary lymphedema (95.0% vs 74.3%, p = 0.045). No major adverse events were observed. In conclusion, FML is a simple and safe technique for detecting lymphedema in patients with leg swelling. A cut-off level of ≥ 14-mm maximum spread has a high sensitivity and high specificity of detecting lymphedema and should be chosen.
Introduction
Lymphedema is a disease caused by the anomalous development of the lymphatic system or injury to the lymphatic vascular system resulting in interstitial accumulation of protein-enriched fluid. 1 Millions of people are affected worldwide by lymphedema, which is a chronic, debilitating and common but often overlooked condition. 2 Moreover, lymphedema is frequently misdiagnosed, treated too late or not treated at all. 3 Complications resulting from lymphedema include functional disability, psychosocial disorders, infection, skin changes and, rarely, malignant transformation. 1 The prevalence of primary lymphedema is epidemiologically poorly documented. Estimates vary between one case per 6000 and 1.15 cases per 100,000.4,5 In persons younger than 20 years of age, the incidence of primary (idiopathic) lymphedema is estimated to be two cases per 100,000. 5 Secondary lymphedema is more common and results from injury, surgery, chronic infection or functional failure of the lymphatic system owing to fluid overload. Filarial infection is worldwide the most frequent cause of secondary lymphedema. 6
Early recognition, diagnosis and correct management of the swollen extremity remain a challenge. Fluorescence microlymphography (FML) is a useful test in evaluating different forms of edema of the lower extremities and helps characterize lymphedema. Several studies have shown its value in differentiating between healthy legs and lymphedema.7,8 FML involves intradermal injection of a fluorescent marker, a FITC-labeled (fluorescein isothiocyanate) high molecular dextran that can be visualized with fluorescence microscopy. The spread of the fluorochrome has been measured as the maximum distance of the visualized capillary network from the dye deposit. A visible capillary network around the dye deposit with a maximum spread of ≥ 12 mm has been set as a cut-off value to discriminate between normal and pathological dye spread suggesting the presence of lymphedema. 9 However, the accuracy of this test has not been evaluated in patients with leg swelling. Thus, our aim was to prospectively assess the accuracy of FML for diagnosing lymphedema in patients with leg swelling.
Patients and methods
Patients with leg swelling and the suspicion of lymphedema were referred to our tertiary vascular center for further evaluation. In all patients a thorough history and clinical examination were assessed by two experienced vascular physicians. Recognition of cutaneous findings of lymphedema includes peau d’orange, pitting and non-pitting edema, and the presence of a positive Stemmer sign. Lymphedema was diagnosed clinically according to the criteria of the International Lymphoedema Framework 10 and after elimination of other non-lymphatic causes of edema at the bedside. This study focused on leg swelling. Referral for suspicion of lymphedema of the upper extremity was very rare because the diagnosis is often obvious. To establish the role of FML in the diagnosis of the swollen lower extremity, the patients were divided into two distinct groups: lymphedema and non-lymphatic limb swelling.
All data were collected and entered into a prospectively maintained database. All patients underwent duplex ultrasound imaging to confirm or rule out venous incompetence. In this study the anonymity of the patient’s data was retained and written informed consent has been received according to the requirements of the institutional ethical review board.
FML was performed in all patients by one research nurse (MS) with more than 10 years’ experience in FML diagnostics. The contrast medium was produced by our pharmacy by reconstituting FITC-dextran (molecular weight 150,000 Da) (Bio Chemica, Germany) with 0.9% sodium chloride solution (MiniPlasco Connect; B Braun Medical, Switzerland) to a 25% solution in a sterile fashion and passed through a bacteria filter. Next, 0.1 ml of the solution of FITC-labeled dextran was injected into the intradermal layer of the skin using a tuberculin syringe and a 25-gauge needle. The most swollen part of the leg was selected as the site of injection. FITC is excited by light with a wavelength of approximately 495 nm and emits light with a wavelength of approximately 521 nm. A fluorescent light microscope (Leica Microsystems AG, Switzerland) was used to visualize the lymphatic capillaries. Ten minutes after the injection the maximum extension of the fluorescent contrast medium in the lymph capillaries from the border of the original dye deposit was measured in four directions and the maximum spread was documented.
This manuscript was prepared in compliance with the STARD Initiative checklist for complete and accurate reporting of studies of diagnostic accuracy. 11
Statistical analysis
Descriptive statistics were performed using non-parametric statistics. Basic statistical parameters such as sensitivity, specificity, positive and negative predictive values and accuracy for lymphedema detection were calculated. Positive likelihood ratio (LR+) was defined as sensitivity/ 1-specificity and negative likelihood ratio (LR–) as specificity/1-sensitivity. Receiver operator characteristic (ROC) curves were calculated to identify the potential cut-off value that has the highest sensitivity and specificity for diagnosing lymphedema. A Pearson chi-squared statistic was used to compare the positive test results of primary and secondary lymphedema. A two-sided p-value of < 0.05 was considered statistically significant. Data analysis was performed using Stata/IC software version 10 (Stata, Inc., College Station, TX, USA).
Results
Between March 2008 and August 2011 a total of 171 patients (184 legs) with FML were studied. The final diagnosis of lymphedema was retained in 94 (51.1%) legs. Detailed clinical data and patient characteristics are shown in Table 1. Women were more prevalent than men in patients with lymphedema and non-lymphatic edema, respectively. In those with lymphedema, primary lymphedema was more prevalent than secondary lymphedema. The lymphedema group and the control group were well balanced in terms of sex and age.
Clinical data of 171 patients with lower extremity swelling separated by lymphedema and non-lymphatic edema.
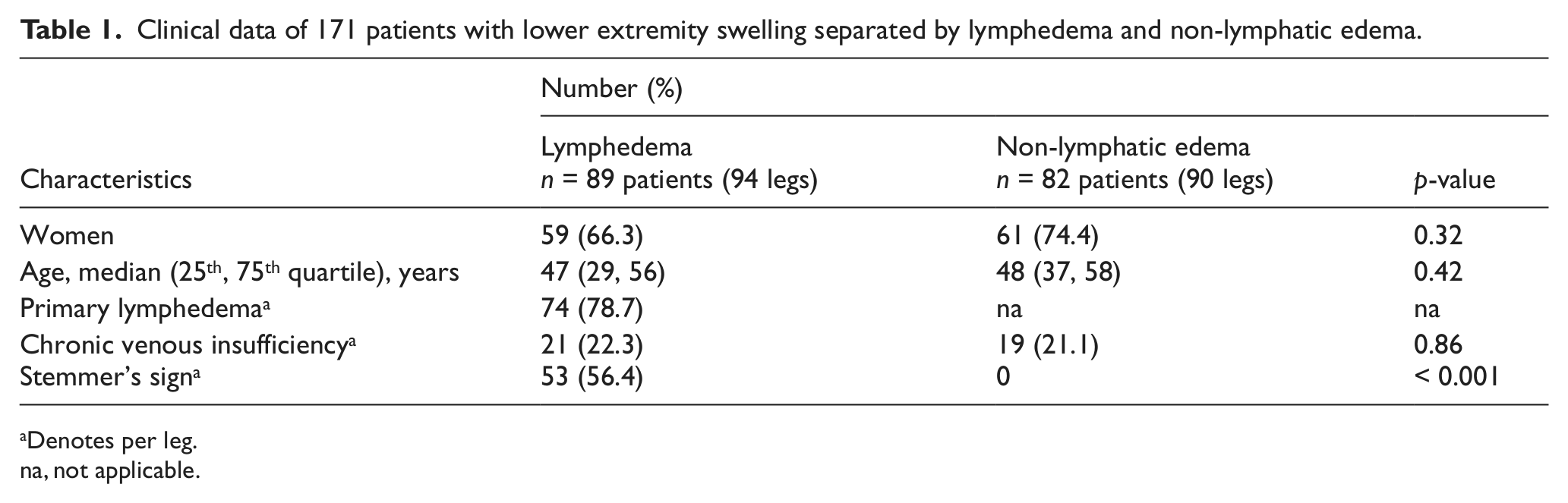
Denotes per leg.
na, not applicable.
The injection sites and the maximal spread of the fluorescence dye are given in Table 2. The deposit of the dye and the measurement of the maximal spread of the fluorescence dye after 10 minutes are shown in Figures 1 and 2. The injection for FML was mostly performed at the ankle and the dorsum pedis of patients with lymphedema and patients with non-lymphatic edema. Figure 3 represents the distribution of the maximal spread of the fluorochrome in patients with chronic leg swelling (i.e. lymphedema and non-lymphatic leg swelling).
Injection sites of FITC-labeled dextran and maximum spread of the dye separated by lymphedema and non-lymphatic edema.

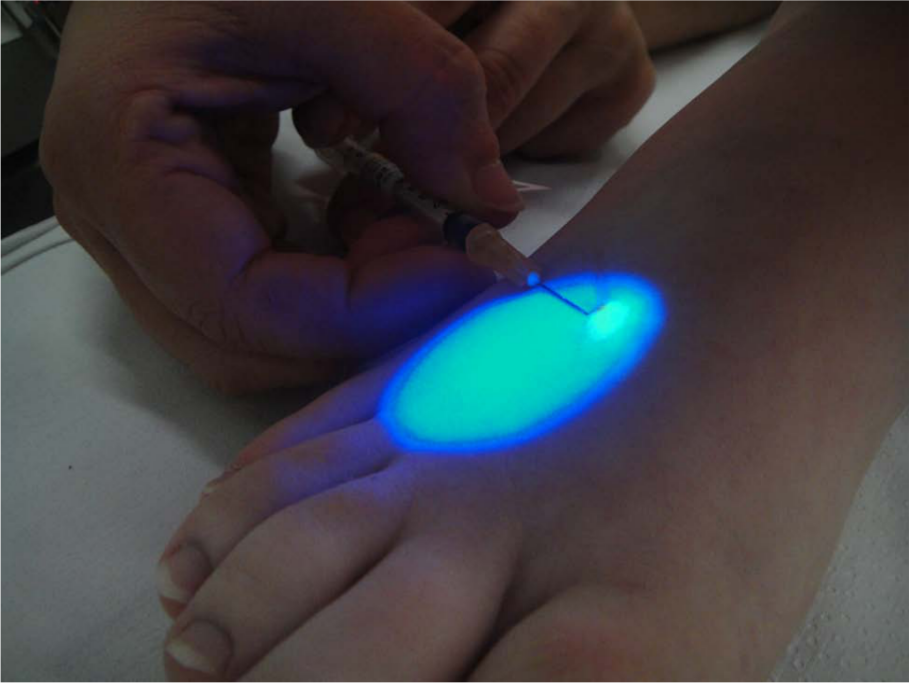
Subepidermal injection of 0.1 ml of 25% solution of FITC-dextran in the dorsum of the foot and fluorescence illumination of the dye.
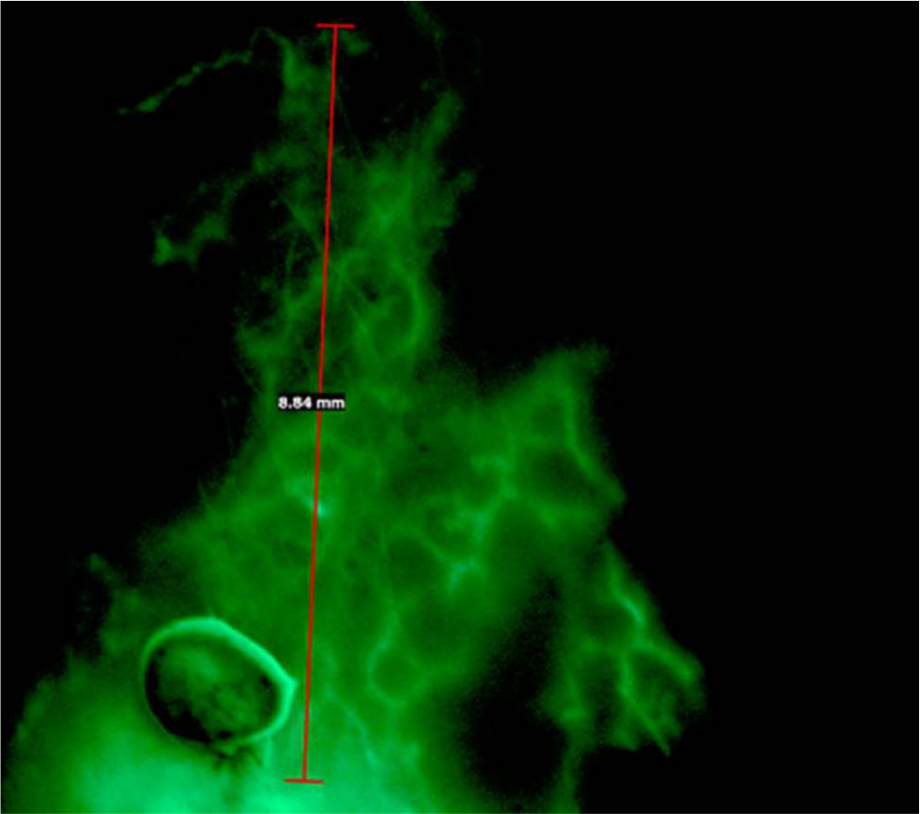
Reticular network of lymphatic microvessels arising from the dye deposit in the dorsum of the foot region magnified by microscope.
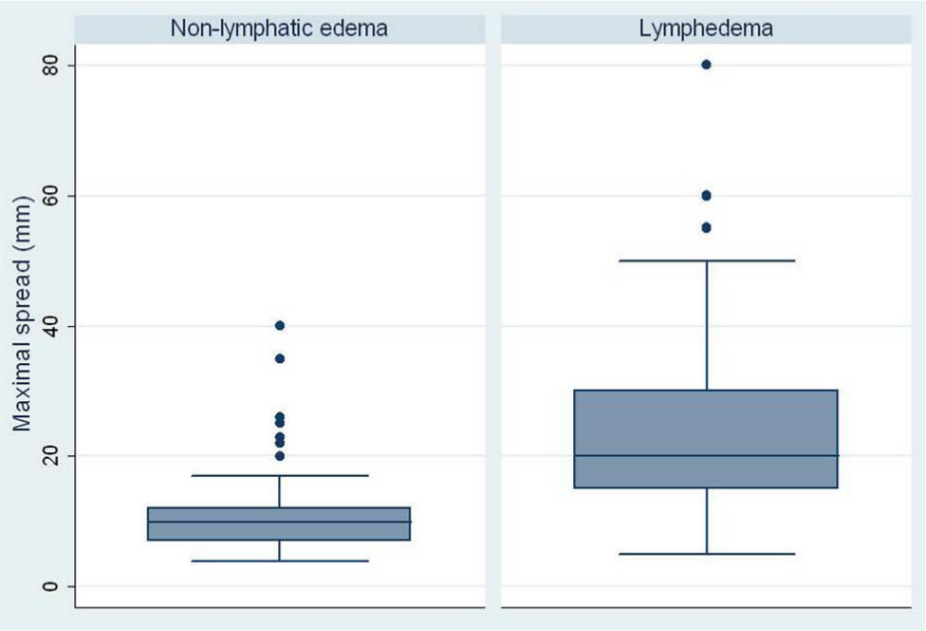
Box plot graphic of the maximal spread of the fluorescence dye in patients with lymphedema and non-lymphatic swelling.
Table 3 represents the test accuracy at different cut-off levels. The 14-mm spread is the overall threshold value of FML that is associated with 79% sensitivity and 83% specificity, 4.72 LR+ and 0.26 LR– for detecting lymphedema. The positive and negative predictive value was 83% and 79%, respectively. The area under the ROC curve was 0.82 (95% CI: 0.76, 0.88).
Sensitivity (true-positive), specificity (true-negative) positive and negative predictive values and likelihood ratios at different cut-off levels.
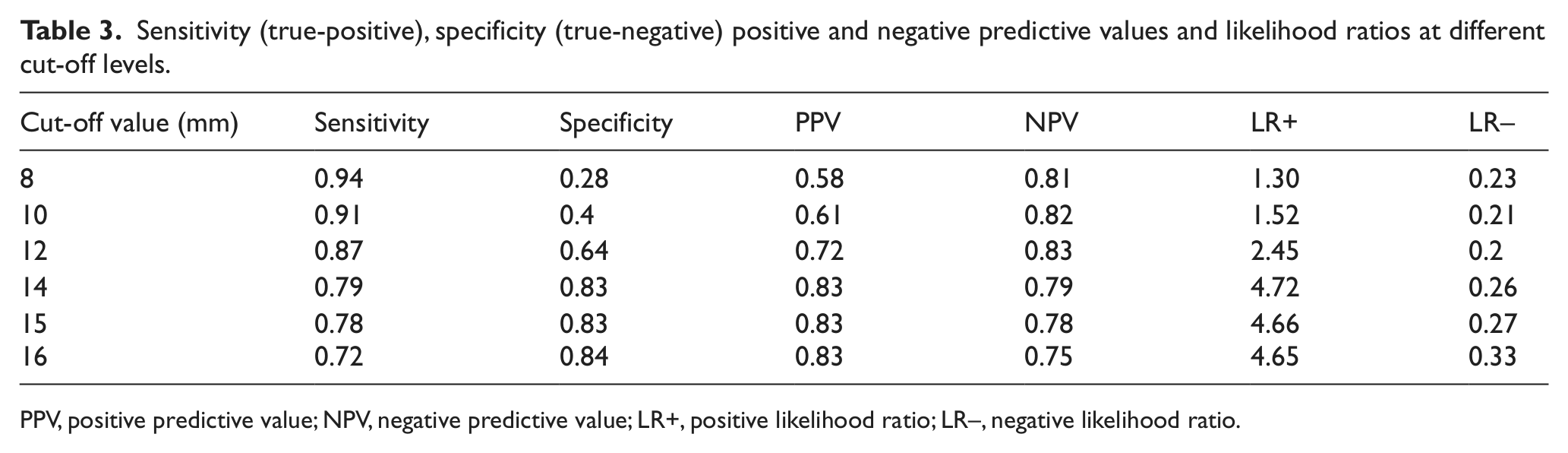
PPV, positive predictive value; NPV, negative predictive value; LR+, positive likelihood ratio; LR–, negative likelihood ratio.
When using the 12-mm cut-off level, 83.8% (n = 62) of primary lymphedema had a positive test. In secondary lymphedema, all of the 20 legs had a positive test, resulting in a trend towards a significant difference (p = 0.054). Comparing primary lymphedema with secondary lymphedema using the 14-mm cut-off level, there was a 74.3% (n = 55) and 95% (n = 19) positive test, respectively (p = 0.045).
Intradermal injection of FITC-labeled dextran in the most swollen part of the lower extremity was well tolerated. Severe complications and severe adverse events caused by FITC-labeled dextran were not observed in any patients. Of particular importance, no apparent edema or anaphylactic reaction was observed in any patients throughout the study. There were no signs of systemic or local inflammatory reactions throughout the examination period. Overall, a total of three minor side effects (1.8%) were observed. Two patients (1.1%) with non-lymphatic limb swelling and one patient (0.5%) with lymphedema developed a self-limiting minor rash at the injection site. No death was observed during the study.
Discussion
This study assessed the accuracy of FML in patients with lower extremity swelling. Our results suggest that FML is an expedient additional test for assessing lymphedema with a high sensitivity but low specificity for the actual cut-off value of ≥ 12 mm. A higher cut-off value of ≥ 14 mm has a high sensitivity and high specificity for detecting lymphedema and should be chosen.
FML can be an office-based procedure using a relatively simple technique, yielding real-time valuable clinical information that can be used for counseling the patient. FML was performed according to an almost atraumatic technique as described by Bollinger et al. 8 It can even be applied in regions with indurated skin or near venous ulcers. The only known complication is a local itching reaction to the fluorescent macromolecular dye resembling the sting of a mosquito. 12 If lymphatic flow into precollectors and collectors is not impeded, FITC-labeled dextran will be drained by these precollectors and collectors and the superficial lymph capillaries network is only filled to a minor extent. 12 In lymphedema patients, the dye expands rapidly into the capillary network system as a result of lymph drainage obstruction and can be visualized with fluorescent light. The current study shows that when clinical criteria were used as the gold standard for diagnosing lymphedema, the area under the ROC curve was 0.82 (95% CI: 0.76, 0.88), which is a good overall discriminative value of the diagnostic test. We also analyzed the positive and negative predictive values and positive and negative LR of different cut-off levels of FML. Once the test has been performed, the sensitivity and specificity do not indicate whether a positive result truly means the presence of the disease. That information is given by the predictive values. When using a cut-off level of 14-mm spread of FML, our LR for a positive test substantially increases from 2.45 (cut-off level of 12 mm) to 4.72 without substantial loss of the negative LR. Furthermore, the PPV increases from 0.72 to 0.83, whereas the NPV only decreases from 0.83 to 0.79; thus suggesting a more accurate diagnostic test when using the 14-mm spread cut-off level. When comparing primary with secondary lymphedema using the 14-mm spread as the cut-off level, we found a significant higher sensitivity in secondary lymphedema patients than in primary lymphedema. This might be due to the fact that the 14-mm cut-off level is more accurate than the 12-mm cut-off level.
Lymphedema is diagnosed clinically and, when necessary, the diagnosis can be confirmed through lymphoscintigraphic imaging. 13 It is performed using the intra- or subcutaneous administration of a radiolabeled large molecule (typically 99m-technetium-labeled sulfur colloid or human serum albumin). The agent is administered parenterally into the interdigital space of the hands or feet. The limbs are serially imaged with a scintigraphic camera. 14 Lymphoscintigraphy has been reported to have a 92% sensitivity and a 100% specificity for lymphedema. 15 The high sensitivity and specificity of this study is based by the fact that the control group consisted of 17 healthy non-swollen legs diagnosed clinically, which does not reflect our target population and therefore might overestimate the diagnostic accuracy of this test.15 –17 We chose our control group patients with non-lymphatic limb swelling to study FML as the test must discriminate lymphedema from non-lymphatic edema to reflect the real-world application of the test. If we had chosen healthy legs without any swelling as the control group we believe that the sensitivity and specificity of our study would be much higher. Moreover, lymphoscintigraphy involves radiation exposure, is time-consuming, technically challenging and is not available at all facilities. This diagnostic test is based on a qualitative or semi-quantitative evaluation of the lymphatic drainage and description of the image patterns. Furthermore, the protocol for lymphoscintigraphy is not standardized and differs among diagnostic centers. 3 Although there are some limitations, lymphoscintigraphy remains the standard investigation for identifying edema of lymphatic origin. Direct contrast lymphangiography has been largely abandoned because of the technical complexity of the procedure, the challenge to repeat the test and the unacceptable complications.18,19 Magnetic resonance imaging and computed tomography can be used to evaluate the presence and severity of edema, but differentiation of lymphedema from edema associated with other internal medical diseases is not possible.
Despite the large sample size, there are some limitations to this study. The currently accepted ‘gold standard’ for the diagnosis of lymphedema is based on history and clinical examination. Thus, misclassification might happen and may attenuate our results. However, as senior fellows with at least 10 years of experience in vascular medicine evaluated these patients and the fact that there is a lack of definite cause of limb swelling, we believe that the rate of misclassification of lymphedema is low.
Despite these limitations, our results provide valuable information about the accuracy of FML. Second, this study provides information in terms of real-time study and the practice-based usability of FML in detecting lymphedema. The widespread use of this repeatable test to confirm the involvement of the lymphatic system as a factor in the development of chronic edema of the extremities would help to enhance the diagnosis of patients with chronic leg swelling. The role of FML is not as a substitute for lymphoscintigraphy but should be integrated as a complementary tool in lymphedema diagnosis because still too many patients with this vascular dysfunction are frequently misdiagnosed, treated too late or not treated at all. Recently, therapeutic lymphangiogenesis to reverse lymphedema by targeting VEGF receptor-3 or cell therapy to augment lymphatic neovascularization in animal models have been shown to be promising.20,21 FML could be used as a baseline and outcome measure in these studies.
In conclusion, our results support the usefulness of FML in the evaluation of the chronically swollen extremity. The examination has no known contraindications and the technique is simple and safe. The cut-off value of ≥ 14 mm is the threshold dye spread that has a high sensitivity and high specificity and should be chosen.
Footnotes
Conflict of interest
All authors reported no conflict of interest associated with this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
