Abstract
Pentraxin 3 (PTX3) is a novel inflammatory cytokine produced in atherosclerotic plaque. We hypothesized that this marker may be a better predictor of peripheral artery disease (PAD) than C-reactive protein (CRP) in hemodialysis (HD) patients. The ankle–brachial index (ABI) was measured in HD patients. PTX3 levels in 116 HD patients and 30 healthy blood donors were measured by ELISA. A total of 116 HD patients (age: 56.44 ± 14.08 years) were enrolled; 21 (18%) patients had PAD. PTX3 was significantly higher in PAD versus non-PAD patients (5.55 ± 2.63 vs 2.32 ± 1.29 ng/mL; p < 0.001). In a univariate analysis, ABI correlated significantly with age, blood glucose and triglycerides, and plasma PTX3 (r = -0.548, p < 0.001) and high-sensitivity (hs)CRP (r = -0.495, p < 0.001). Using ROC curve analysis for PAD, PTX3 (cut-off value 4.06 ng/mL, AUC 0.901, p < 0.0001) showed a significantly better positive predictive value than hsCRP (cut-off value 3.33 mg/L, AUC 0.640, p < 0.05). Logistic regression analysis further confirmed that PTX3 (OR = 9.755, p = 0.001) was an independent predictor of PAD. In conclusion, we demonstrated that PTX3 may be a better marker of PAD than hsCRP, and independently correlated with PAD in HD patients.
Introduction
Cardiovascular disease (CVD) is the primary cause of morbidity and premature mortality in end-stage renal disease (ESRD), accounting for about 50% of deaths. 1 Some experts have begun to pay attention to the importance of peripheral artery disease (PAD) and its biomarkers. 2 Recent data suggest that PAD is prevalent in hemodialysis (HD) patients and is a strong predictor for subsequent cardiovascular and overall mortality. 3 Traditional risk factors alone do not explain the unacceptably high prevalence and incidence of CVD in this population. 4 The search for non-traditional risk factors that may be involved in the pathogenesis of CVD in ESRD patients, especially for chronic inflammation, has become an area of intense investigation. Moreover, it is increasingly recognized that chronic kidney disease (CKD) is associated with a state of persistent microinflammation. 5
Pentraxin 3 (PTX3) is a recently discovered mediator of acute inflammation. PTX3 is a long pentraxin, structurally related to, although distinct from, classic short pentraxins, such as C-reactive protein (CRP) and serum amyloid P.6 –8 PTX3 expression occurs in a variety of cell types, including endothelial cells and mononuclear phagocytes. 9 While it is structurally related to CRP, PTX3 is expressed more in peripheral tissues, with recent evidence demonstrating a marked upregulation of PTX3 in atherosclerotic plaques. 10 In HD patients, Boehme et al. 11 found that PTX3 levels were markedly elevated. Suliman et al. 12 showed that high PTX3 levels were independently associated with all-cause mortality. Given the importance of microinflammation of arteries in PAD, as well as the strong correlation between inflammation and CVD in HD patients, we hypothesized that local inflammation, as reflected by PTX3, could be a marker of PAD, and a better predictor of this comorbidity than high-sensitivity (hs)CRP.
Materials and methods
Participants
Patients
We enrolled HD patients from the dialysis center of Renji Hospital, Shanghai, China, between May 2008 and July 2008. There were 167 patients in our dialysis center (91 males, 76 females; mean age: 57.00 ± 14.65 years).
Inclusion criteria were stable HD for at least 3 months at the time of evaluation and the ability and willingness to provide informed consent. In total, 116 patients were enrolled. All patients were dialysed on synthetic (polysulfone) membranes with bicarbonate dialysate. Diagnosis of renal failure was as follows: glomerulonephritis in 40.5%, diabetes mellitus in 6.9%, hypertension in 6.9%, obstructive uropathy in 2.6%, tubulo-interstitial nephropathy in 1.7%, and other unknown.
Exclusion criteria included severe heart failure; severe unrelated medical conditions (e.g. cancer, sepsis, advanced liver or lung disease); active tuberculosis, systemic lupus erythematosus, systemic vasculitis, or rheumatoid arthritis; peritoneal dialysis or kidney transplantation patients who converted to HD; and patients with an ankle–brachial index (ABI) of ≥ 1.3 (because this indicates poorly compressible leg arteries and an inability to gauge arterial obstruction accurately).
Thirty healthy volunteers of the same ethnic origin who were frequency-matched with respect to age and gender were also studied. None had any evidence of CKD, diabetes, or high blood pressure.
Informed consent was obtained from each patient and control subject. The study was approved by the Ethical Committee of Shanghai Jiaotong University School of Medicine.
Patient groups
Participants were divided into three groups:
PAD group: HD patients with an ABI of < 0.9 (PAD can be determined with high sensitivity and specificity using ABI – a simple, non-invasive procedure. PAD was defined based on clinical symptoms (intermittent claudication, which evolves to pain at rest and a risk of tissue necrosis, and even to amputation), an ABI of < 0.9 in either leg, 13 previous history of lower extremity revascularization procedures and/or angiography findings).
Non-PAD group: HD patients with an ABI of ≥ 0.9.
Controls: healthy volunteers with no evidence of CKD, diabetes, or high blood pressure.
Clinical data
Demographic and medical data were obtained from medical records and interviews with patients at study entry. These included age, gender, smoking history (ever vs never), diabetes history, pre-existing CVD (defined as previous angiogram showing significant occlusive disease, a history of a myocardial infarction, or a history of coronary artery bypass surgery or angioplasty, a history of cerebrovascular accident including cerebral bleeding and infarction), blood pressure (systolic blood pressure, diastolic blood pressure, and mean arterial pressure) and body mass index (BMI) (weight/height2), and dialysis-related information, including underlying renal diagnosis, dialysis age, and Kt/V.
Laboratory measurements
Blood was drawn with EDTA anticoagulant in the morning after an overnight fast of at least 12 h before a dialysis session. The samples were separated via centrifugation (4000 rpm, 10 min) and immediately stored at −80°C for subsequent assays. The plasma concentrations of PTX3 were evaluated using an ELISA kit (R&D Systems, USA). Regarding the reproducibility of the PTX3, we repeated the experiment twice. The averages of the two experiments were used as the value of the PTX3.
Other laboratory tests were conducted in our central testing laboratory. Hemoglobin, creatinine, albumin, cholesterol, triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), and glucose were evaluated using routine biochemical methods. Concentrations of hsCRP were measured by an immune turbidity method (Orion Diagnostica, Finland). Levels of parathyroid hormone (PTH) were measured using an isotopic chemiluminescence method (Siemens Medical Solutions Diagnostics, USA).
Measurement of ankle–brachial index
The ABI was measured in all HD patients using an ABI device (MICROLITE 84M/0289, Sony) by a professional technician; the device measures arm and ankle (brachial and posterior tibial arteries, respectively) blood pressure by an ultrasonic technique. The measurement was obtained after completion of the dialysis treatment and after allowing patients to rest in a supine position at least for 5 minutes. The ABI was calculated by the ratio of the ankle systolic pressure divided by the arm systolic pressure. The systolic pressure of the arm without dialysis access and the lower value of the ankle pressure were used for the calculation. The criterion for the diagnosis of PAD was an ABI of < 0.9.
Statistical analyses
Data are reported as mean ± SD or as median and interquartile range, as appropriate. CRP and PTH, which did not demonstrate a Gaussian distribution, were transformed into natural logarithms for analysis. Comparison between groups was performed using an unpaired t-test for mean (SD) data, non-parametric Wilcoxon rank sum test for median (interquartile range) data, and the chi-squared test for categorical variables. Correlations were reported as the Pearson correlation coefficient. Sensitivity, specificity, and cut-off levels for PTX3 and hsCRP as predictors of the presence of PAD were analyzed using receiver-operating-characteristic (ROC) curves. Additionally, logistic regression analysis was conducted to examine associations between PTX3, hsCRP, lipid profiles, blood pressure, diabetic status, age, and pre-existing CVD, and the presence of PAD. All p-values were two-tailed, and values of < 0.05 were considered to indicate statistical significance. All confidence intervals (CIs) were calculated at the 95% level. All calculations were performed using the SPSS software (version 11.0 for Windows; SPSS Inc., Chicago, IL, USA).
Results
Incidence of PAD in HD patients
In total, 116 HD patients (62 males, 54 females; mean age: 56.44 ± 14.08 years) who had been treated with regular HD for at least 3 months in our center were enrolled. The numbers of patients with and without PAD were 21 and 95, respectively. The incidence of PAD was 21/116 (18%). In PAD patients, six cases presented with claudication, one with claudication and rest pain, and three with angiography findings, who underwent limb vascularization procedures.
Clinical and biochemical parameters at enrollment
Baseline characteristics of the study subjects are shown in Table 1. The mean age of the HD patients was 56.44 ± 14.08 years. Among the 116 patients, 21 subjects fulfilled the clinical diagnosis of PAD. The PAD and non-PAD groups were matched for age, gender, smoking status, BMI, and several established cardiovascular risk factors. The patients in the PAD group were older (64.95 ± 9.69 vs 54.56 ± 14.32 years, p = 0.002), and had higher prevalences of diabetes mellitus, smoking histories, and pre-existing cardiovascular events than subjects without the PAD. The PAD group also had significantly higher serum TG, blood glucose, and hsCRP levels. BMI, prevalence of hypertension, duration of dialysis, hemoglobin levels, calcium levels, phosphorus levels, and Kt/V were similar in both groups.
Demographic and clinical data of hemodialysis patients.
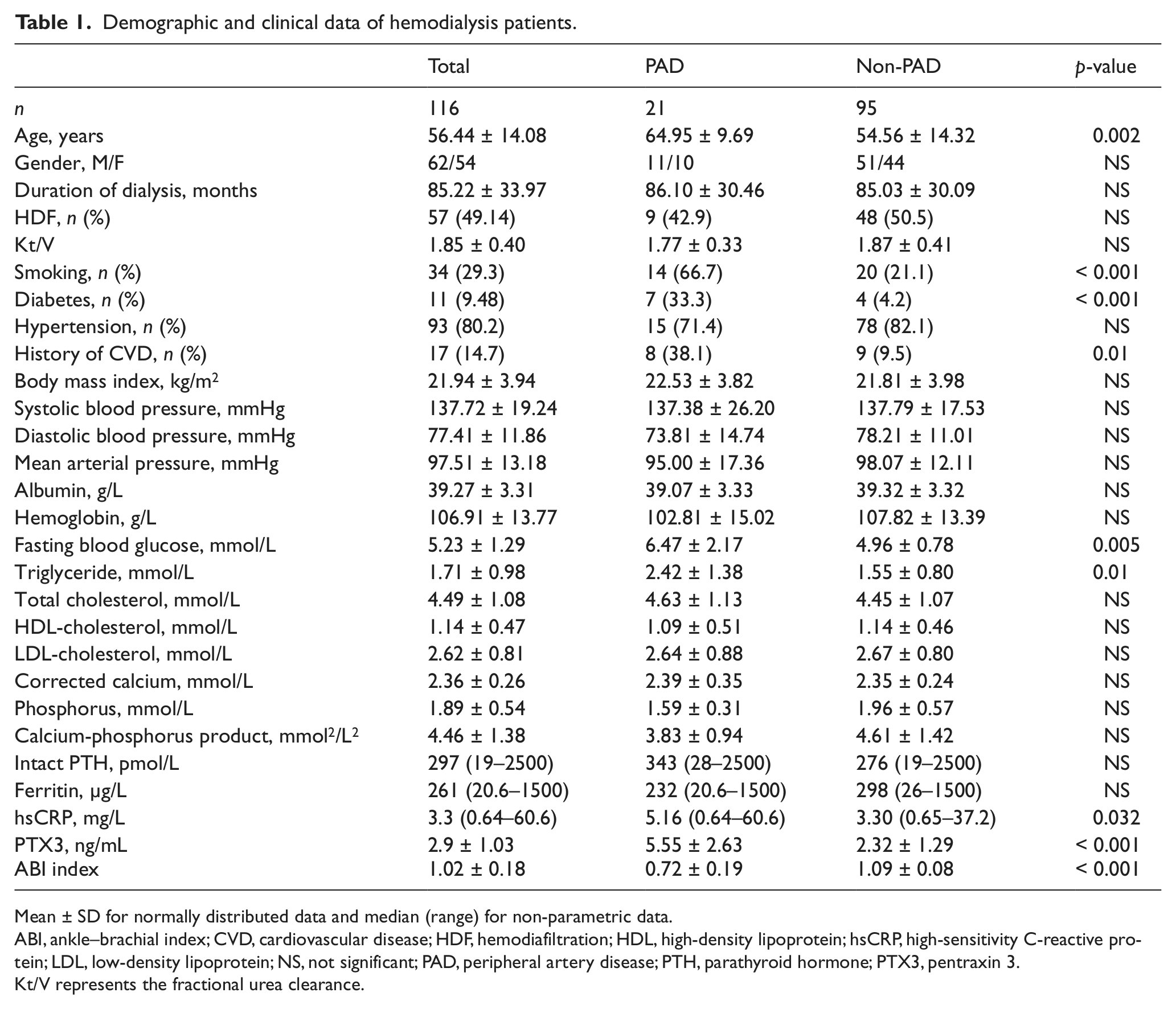
Mean ± SD for normally distributed data and median (range) for non-parametric data.
ABI, ankle–brachial index; CVD, cardiovascular disease; HDF, hemodiafiltration; HDL, high-density lipoprotein; hsCRP, high-sensitivity C-reactive protein; LDL, low-density lipoprotein; NS, not significant; PAD, peripheral artery disease; PTH, parathyroid hormone; PTX3, pentraxin 3.
Kt/V represents the fractional urea clearance.
Plasma PTX3 levels among the three groups
The concentration of plasma PTX3 in the PAD group was higher than that of the non-PAD subjects (5.55 ± 2.63 vs 2.32 ± 1.29 ng/mL, p < 0.001). The concentration of plasma PTX3 in the HD patients was higher than that of the control group (2.9 ± 1.03 vs 1.7 ± 0.85 ng/mL, p < 0.001). The concentration of plasma PTX3 of the non-PAD group was also higher than that of the control group (2.32 ± 1.29 vs 1.7 ± 0.85 ng/mL, p = 0.003; Figure 1).
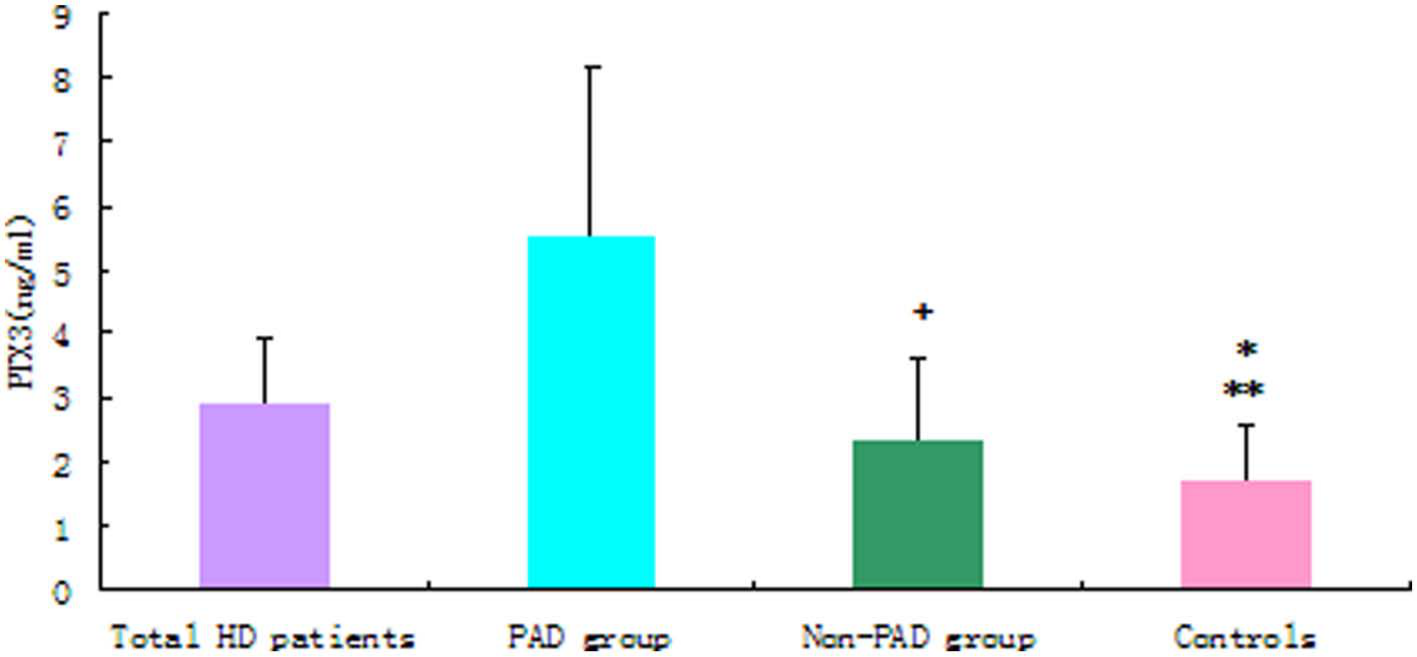
Comparison of plasma PTX3 levels among the groups. (*Total HD patients compared with controls: p < 0.001; **non-PAD group compared with controls: p = 0.003; +PAD group compared with non-PAD group: p < 0.001.)
Correlations between ABI and other factors
The univariate analysis (Table 2) showed a negative correlation in the patients between ABI values and plasma PTX3 levels (r = −0.548, p < 0.001; Figure 2), hsCRP (r = -0.495, p < 0.001), age (r = −0.254, p = 0.006), blood glucose (r = −0.313, p = 0.001), and TG levels (r = −0.321, p = 0.0004). After adjustment for age and gender, significant negative correlations between ABI measurements and PTX3 persisted (r = −0.528, p < 0.001). After further adjustment for age, gender, BMI, hsCRP, blood glucose, TG, and Kt/V, the association remained (r = −0.410, p < 0.001). On the other hand, there was no significant association between ABI and blood pressure, serum albumin, serum PTH, or serum cholesterol.
Correlation coefficients for ankle–brachial index (ABI) and other variables in hemodialysis patients.

BMI, body mass index; HDL, high-density lipoprotein; hsCRP, high-sensitivity C-reactive protein; LDL, low-density lipoprotein; log-PTH, log-transformed parathyroid hormone; PTX3, pentraxin 3.
Kt/V represents the fractional urea clearance.
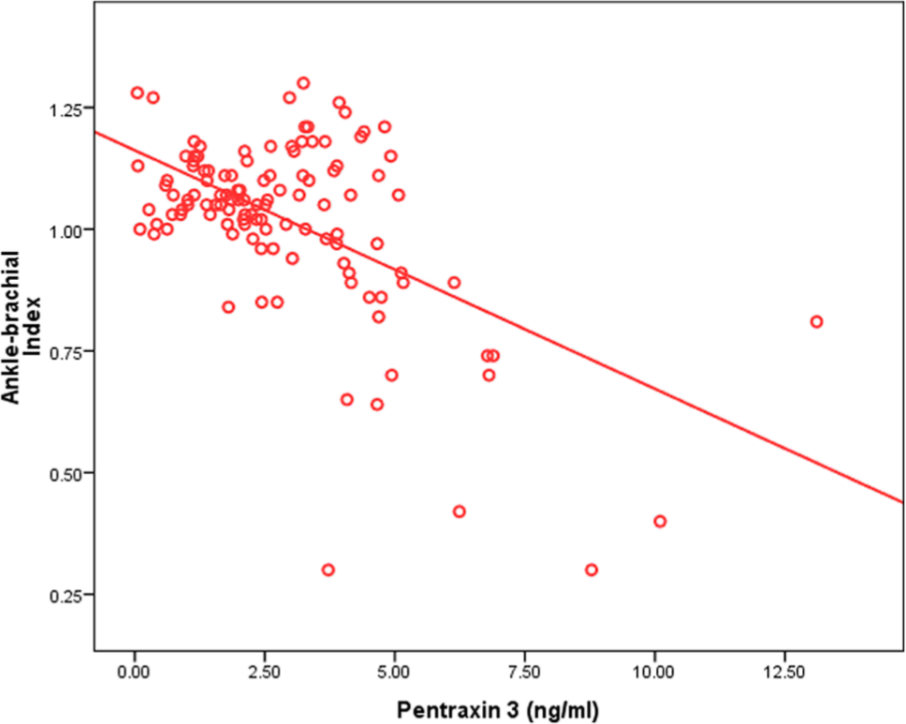
Significant inverse correlation between ankle–brachial index in hemodialysis patients and pentraxin 3 (r = −0.548, p < 0.001).
Correlations between PTX3 and other markers of inflammation
Univariate analysis showed a significant positive correlation between plasma PTX3 levels and hsCRP levels (r = 0.378, p < 0.001; Figure 3). However, there was no significant association between PTX3 and serum albumin or ferritin levels.
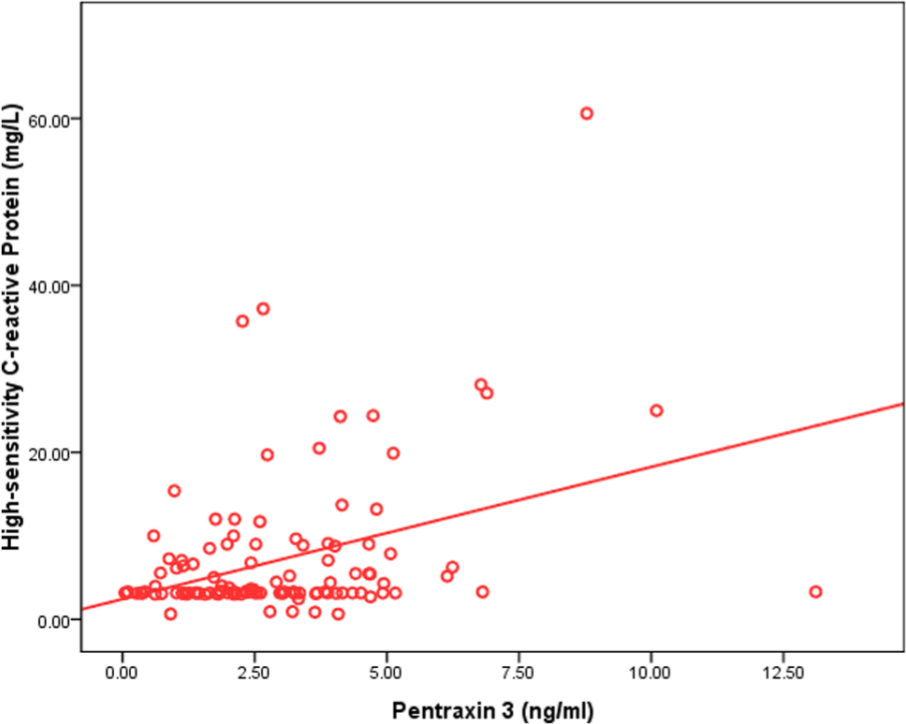
Significant positive correlation between plasma pentraxin 3 levels and high-sensitivity C-reactive protein levels (r = 0.378, p < 0.001).
ROC analysis of PTX3 and HsCRP as predictors of PAD
The ROC curve of PAD showed that the AUC of PTX3 was 0.901 (p < 0.0001). To determine the cut-off value, we generally choose the maximum of the positive likelihood ratio or the Youden’s index. The positive likelihood ratio is equal to the ratio of the true positive rate and the false positive rate. The Youden’s index is equal to the difference between the true positive rate and the false positive rate. With a cut-off value for PTX3 of 4.06 ng/mL, the diagnostic sensitivity and specificity for PAD were 81% and 91.5%, respectively. The ROC curve of PAD showed that the AUC of hsCRP was 0.640 (p < 0.05). With a cut-off value for hsCRP of 3.33 mg/L, the diagnostic sensitivity and specificity for PAD were 57.1% and 56.8%, respectively (Figure 4). For prediction of PAD, the AUC for PTX3 was greater than that for hsCRP. Furthermore, the diagnostic sensitivity and specificity of PTX3 were higher than those of hsCRP (Table 3).
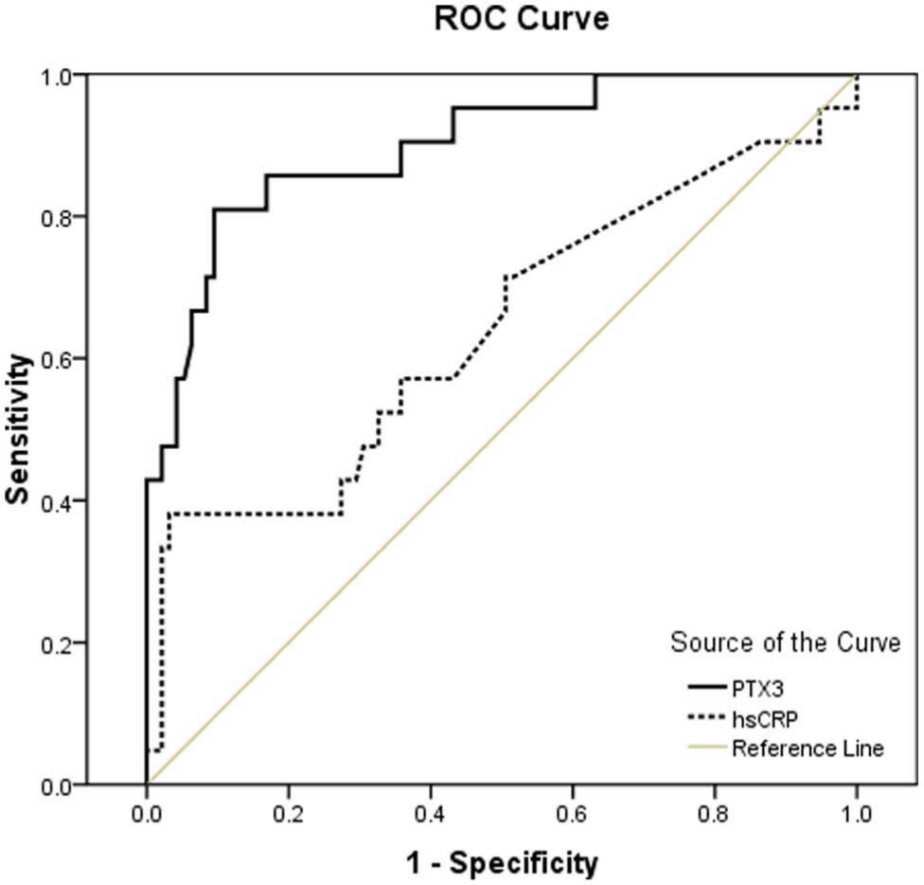
ROC curve for pentraxin 3 and high-sensitivity C-reactive protein to PAD.
The sensitivity/specificity/PPV/NPV for each of the associated markers (PTX3, CRP, glucose, TG).
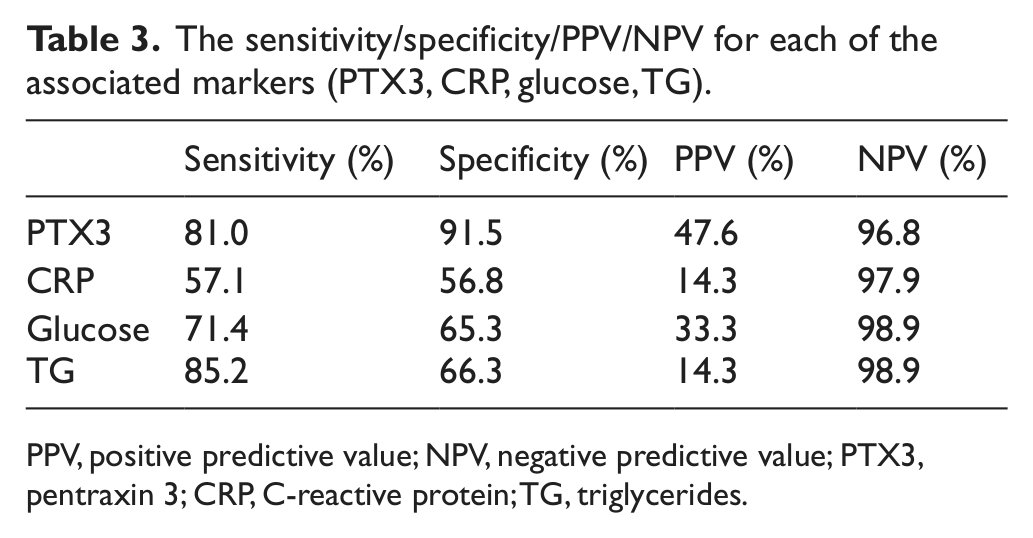
PPV, positive predictive value; NPV, negative predictive value; PTX3, pentraxin 3; CRP, C-reactive protein; TG, triglycerides.
Meanwhile, the C-statistic, which was obtained when using a metric incorporating the traditional risk factors (cholesterol, HDL, systolic blood pressure, age, smoking history, gender), was 0.897 (p < 0.01). PTX3 improved this C-statistic, which was 0.96 (p < 0.01) (Figure 5). CRP did not improve the C-statistic.
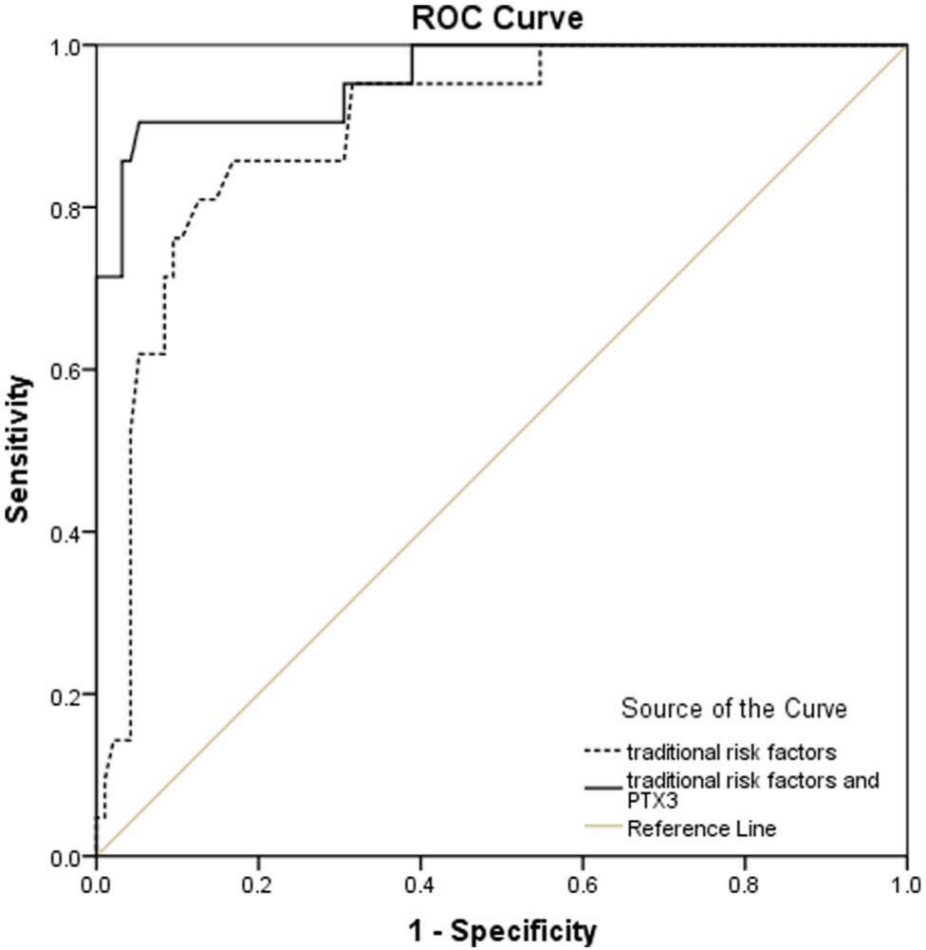
ROC curve for traditional risk factors with and without pentraxin 3 to PAD.
Logistic regression analysis of PTX3 as a predictor of PAD
Markers of inflammation (PTX3, hsCRP, ferritin), age, systolic pressure, serum lipids, blood glucose, serum calcium, serum phosphorus, calcium-phosphorus product, PTH, smoking history, diabetes history, and pre-existing CVD were introduced into the logistic regression equation. We distributed the patients in tertiles of PTX3 by 1 ng/mL, and then compared high versus low levels of PTX3. This showed that an increase in the plasma concentration of PTX3 was associated with an odds ratio (OR) of PAD of 9.755 (95% CI: 2.359–19.354, p = 0.001). Thus, increasing levels of PTX3 were associated with a significant increase in the risk of PAD. In addition to PTX3, smoking history (OR = 5.760, 95% CI: 3.245–9.011, p = 0.004), increasing levels of blood glucose (OR = 10.207, 95% CI: 2.036–16.735, p = 0.004), and increasing levels of TG (OR = 5.406, 95% CI: 1.357–11.80, p = 0.018) were found to be important independent predictors of PAD (Table 4).
Logistic regression analysis of PTX3 as a predictor of PAD.
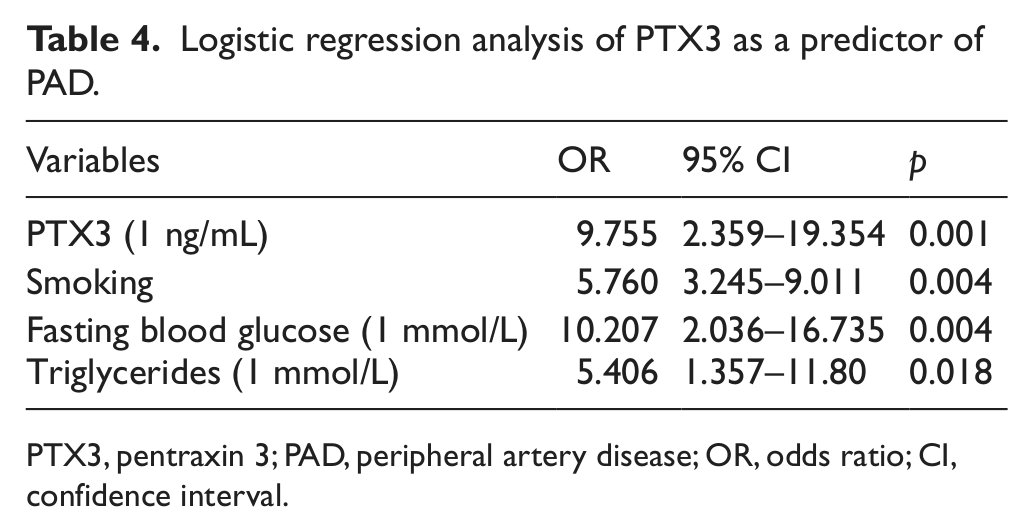
PTX3, pentraxin 3; PAD, peripheral artery disease; OR, odds ratio; CI, confidence interval.
Discussion
To our knowledge, this is the first reported study investigating PTX3 in relation to PAD in a group of HD patients. Salient observations of this study are that plasma levels of PTX3 may be a better predictor of PAD in HD patients than hsCRP. Moreover, we confirmed earlier reports of a PAD prevalence in maintenance HD Asian patients of around 18%. Because of the study design, only correlative associations have been presented and, thus, this study does not provide mechanistic explanations.
Our study showed that the incidence of PAD in HD patients was 18%. Epidemiological studies have reported the prevalence of PAD in the general population to be 1–5%.14,15 The Chronic Renal Insufficiency Cohort study data showed PAD in 7% of adult CKD non-dialysis (ND) patients. 16 In HD patients, the incidence of PAD is higher than that of the general population. Based on the United States Renal Data System (USRDS) 2008 database, the prevalence of PAD was 13.9–14.7% in chronic HD patients. In Japan, Ono et al. showed that the incidence of PAD was 16.5% in 1010 chronic HD patients using the ABI method. 17 In Taiwan, Chen et al. reported that the incidence was 15.6% in 125 HD patients, using the same method. 18 The incidence of PAD was 18% in our study, close to that in Japan and Taiwan.
Traditional risk factors alone do not explain the unacceptably high prevalence and incidence of CVD in this population. 4 The search for non-traditional risk factors that may be involved in the pathogenesis of CVD in ESRD patients, especially for chronic inflammation, has been an area of intense investigation. Moreover, it is increasingly recognized that CKD is also associated with a state of persistent microinflammation. 5 Indeed, several inflammatory biomarkers, such as hsCRP and interleukin (IL)-6, are robust predictors of outcome in this patient population.
PTX3 is a recently discovered inflammatory biomarker; it is a secreted glycoprotein belonging to the pentraxin family. PTX3 is a long pentraxin, structurally related to, but distinct from, classic short pentraxins, such as CRP and serum amyloid P. 6 In healthy subjects, PTX3 levels were reported to average between 1.34 ± 0.9 and 2.28 ± 1.33 ng/mL.6,7 Thus, the levels in control subjects observed in the present study (1.70 ± 0.85 ng/mL) were consistent with previous studies. The plasma PTX3 levels in HD patients as well as the non-PAD group were higher than those of the control group. PTX3 levels were markedly elevated in HD patients. This result is consistent with the recent study of Boehme et al. 11 Our study also demonstrated that ESRD was associated with a state of persistent microinflammation.
The main observation of the present study was the significant relationship between plasma PTX3 and PAD in the HD patients. Even after adjusting for several confounding factors such as age, gender, BMI, fasting blood glucose, and hsCRP, plasma PTX3 concentration was still significantly associated with ABI. In a multiple logistic model, plasma PTX3 independently predicted the presence of PAD. These observations suggest a close link may exist between microinflammation and PAD. These results are consistent with those of Boehme et al. 11 The mechanism of action of PTX3 is that PTX3 binds to the C1q component of the complement cascade and participates in the clearance of apoptotic cells. This is similar to the mechanism of hsCRP. A recent study reported that oxLDL (especially lysophosphatidic acid (LPA)) upregulated PTX3 in human endothelial cells via LPA-receptors and nuclear factor kappa-B activation. PTX3 could also contribute to the accumulation of monocytes in atherosclerotic lesions. 19 Another study reported that PTX3-positive neutrophils infiltrated into atherosclerotic plaques, suggesting that PTX3 derived from neutrophils, as well as macrophages, plays an important role in atherogenesis. 10 Thus, PTX3 may play a role in the atherosclerotic process of PAD in HD patients apart from CRP.
PTX3 is a recently described inflammatory molecule that belongs to the same family as CRP. We found an association between PTX3 and hsCRP in HD patients. Suliman et al. also found a strong association between PTX3 and CRP in HD patients. 12 Moreover, they reported the same result in CKD patients in another study. 20 Consistently, the relationship between PTX3 and CRP in our study could be explained by the fact that ESRD patients are characterized by a state of systemic low-grade inflammation and the increase in PTX3 in such patients could be the result of exposure of various tissues to inflammatory stimuli induced by the unphysiological uremic milieu. 12 Thus, in ESRD patients, PTX3 may be a reliable inflammation marker, in addition to CRP.
Our ROC analysis showed that not only the diagnostic sensitivity but also the specificity of PTX3 was higher than those of hsCRP. The AUC of PTX3 was 0.901. The diagnostic sensitivity and specificity in PAD were 81% and 91.5%, respectively. However, the AUC of hsCRP was 0.640. The diagnostic sensitivity and specificity in PAD were only 57.1% and 56.8%, respectively. This suggests that PTX3 may be a better inflammation marker than hsCRP. Logistic regression analysis in our study also corroborated this. The result of the logistic regression analysis was that increasing levels of PTX3 were associated with a significantly increased risk of PAD, but this was not true for hsCRP. These findings were consistent with those of Boehme et al. 11
We acknowledge several limitations of this study. Because of the cross-sectional nature of the study, the analysis leaves unanswered the question of whether higher levels of plasma PTX3 can predict future PAD in HD patients. An additional limitation of our study is the relatively small number of patients, particularly the relatively small number of patients with PAD. Thus, caution should be exercised in the interpretation of our results. The patient population was also limited to those on hemodialysis and may not be generalizable to the broader population with PAD.
In summary, we showed that high levels of PTX3 were found in HD patients with PAD. Furthermore, we showed that PTX3 may be a better predictor of PAD in HD patients than hsCRP. Because this is the first study to examine PTX3 in relation to the presence of PAD in HD patients, the present findings should be confirmed in future studies.
Footnotes
Conflict of interest
The authors report no conflicts of interest.
Funding
This work was supported, in part, by the National Basic Research Program of China 973, Program No. 2012CB517600 (No. 2012CB517602). The Project was also sponsored by SRF for ROCS, SEM, and the National Society Foundation (81102700). The work was also sponsored by grant 09dZ1973600 and 10JC1410100 from the Science and Technology Commission of Shanghai Municipality, China, and 2010L063A from the Shanghai Health Bureau.
