Abstract
Aortic dissection occurring in the infrarenal abdominal aorta is uncommon. We present the case of a patient presenting with an enlarging abdominal aortic aneurysm and concurrent dissection (with associated radiological imaging) and briefly discuss the literature relating to this phenomenon.
Case report
A 66-year-old man was admitted acutely to our institution with a 24-hour history of vomiting and epigastric pain. Clinical history elicited no specific exacerbating or relieving factors and no other family members were unwell. Past medical history included hypertension and a metallic mitral valve replacement for which the patient was maintained on warfarin therapy. On examination he was found to be haemodynamically stable but febrile (temperature 37.8°C) and he demonstrated tenderness in the right upper quadrant and central abdomen.
The patient was known to have an infrarenal abdominal aortic aneurysm (AAA) and a small left-sided popliteal aneurysm and was currently being investigated as an outpatient to assess the options for either open or endovascular (EVAR) repair. He was a non-smoker and had received secondary prevention in the form of statin therapy in addition to his warfarin and anti-hypertensive medication (amlodipine). His last ultrasound scan (Figure 1), performed 3 months prior to admission, had shown a 5.3 cm aneurysm and a computed tomography (CT) scan, undertaken just 1 week previously, had confirmed these findings, with the aneurysm reported to be 6 cm in diameter and suitable for EVAR (Figure 2).
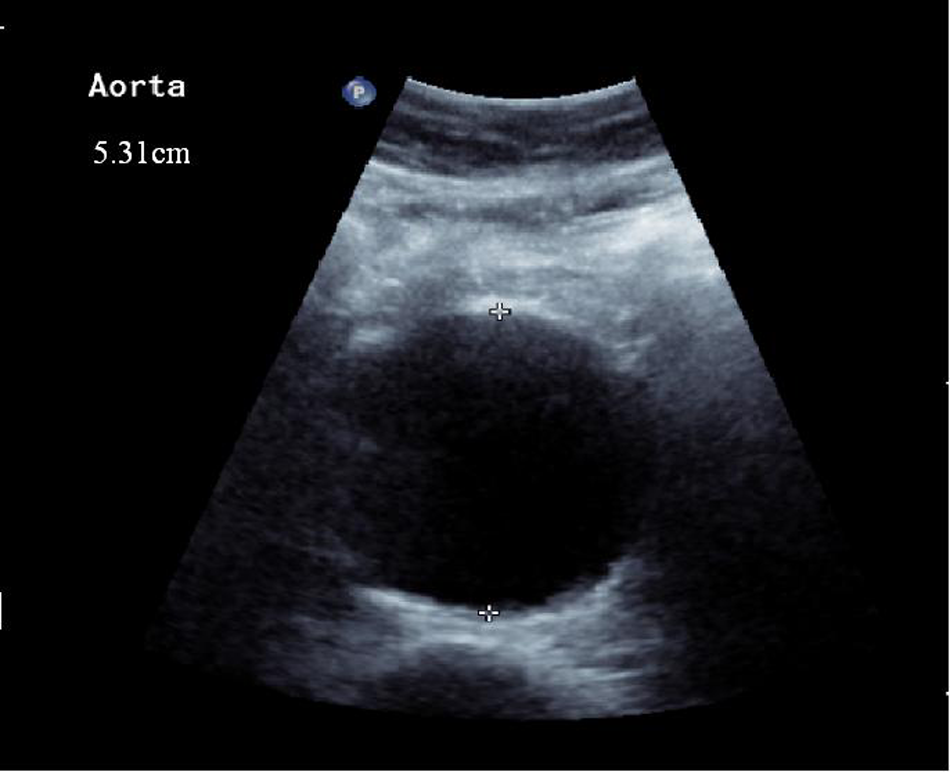
Ultrasound scan of the aorta 3 months prior to admission showing a 5.3 cm AAA.
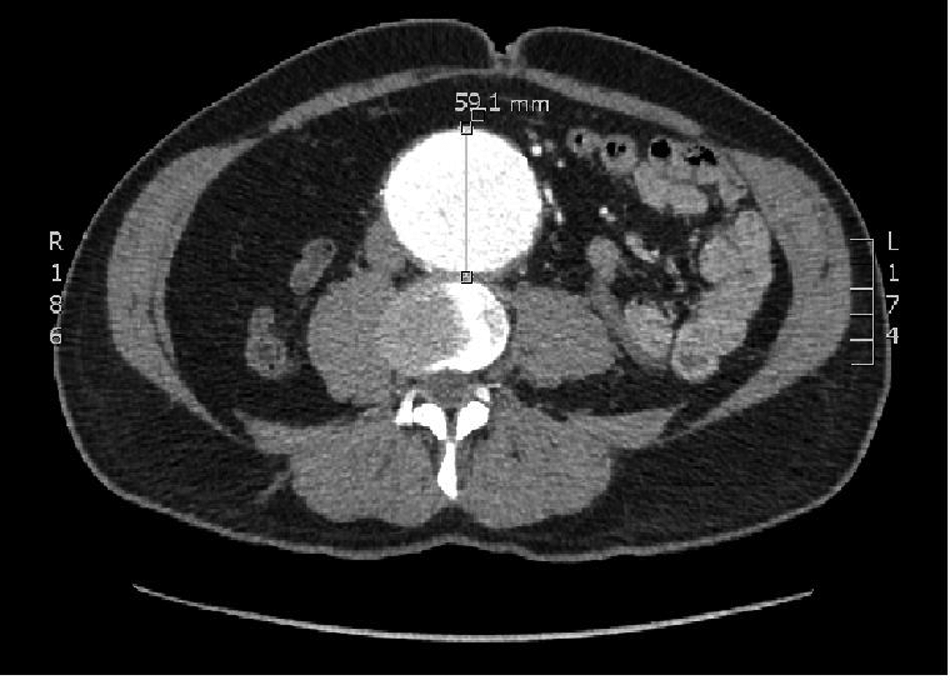
A CT scan undertaken 1 week prior to admission showing a 6 cm AAA suitable for endovascular repair.
In light of this history, his clinical signs and his normal blood chemistry, the patient underwent a CT scan which revealed a 6.4 cm AAA with an associated, localized aortic dissection (Figure 3) arising from the superior origin of the aneurysm at the L2 level and completely confined to the aneurysm sac (Figure 4). There was normal arterial filling of the flap and all distal vessels despite the inferior mesenteric artery arising from the false lumen anteriorly. The CT scan also showed stranding of the surrounding mesenteric fat, a feature indicating possible impending rupture.
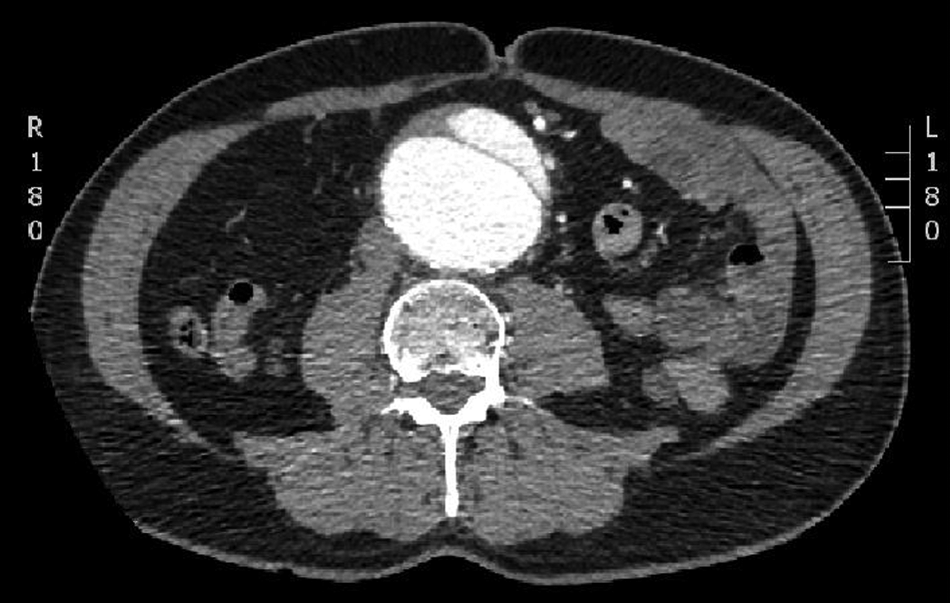
CT scan demonstrating aneurysm expansion and aortic dissection with disruption of the surrounding fat.
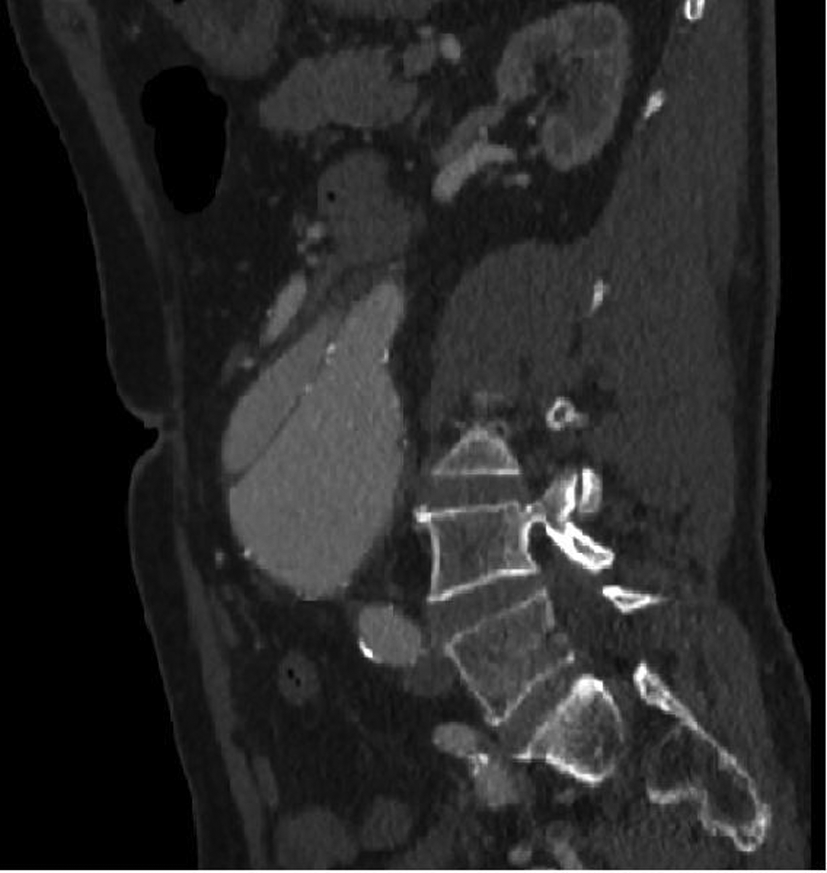
CT scan demonstrating the extent of the dissection, localized within the aneurysm sac.
The diagnosis was explained to the patient and attending relatives and consent was obtained to undertake a stent-graft procedure to treat the aneurysm and concomitant dissection. The patient’s warfarin level was found to be high (international normalized ratio (INR) of 3.1) and hence Beriplex® (prothrombin complex concentrate, CSL Behring Ltd, Haywards Heath, UK) and vitamin K were given to reverse activity prior to surgery. Bilateral groin ‘cut downs’ were performed and an aorto-bifemoral Endurant® device (Medtronic, Watford, UK) was inserted under radiological control. There was minor difficulty in deploying the stent-graft and angiography revealed kinking of the left limb (due in part to atherosclerotic disease within the vessel), which was treated with 15 mm balloon angioplasty and a Bard LUMINEXX™ 14 × 2 cm stent (Angiomed, Karlsruhe, Germany). No endoleak was seen post stent-graft insertion (Figure 5) and the patient made an excellent recovery with no immediate postoperative complications. He was discharged home on day 5 after stabilization of his INR and check CTA to confirm exclusion of the false lumen (Figure 6).
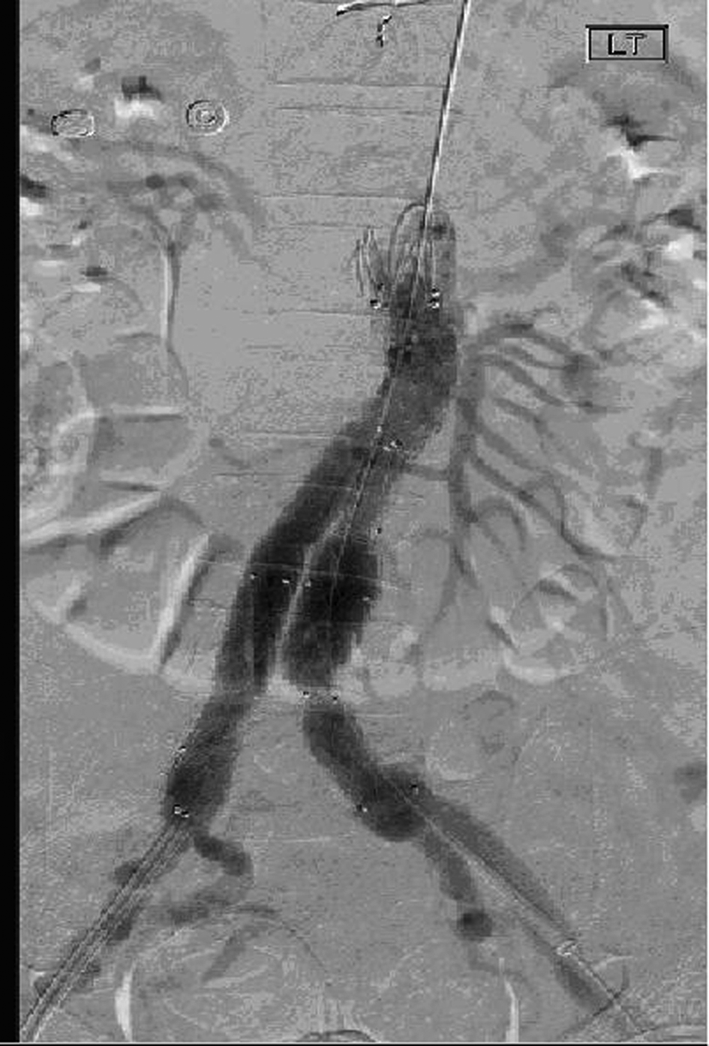
Angiogram showing post-stent appearance with no evident endoleak.
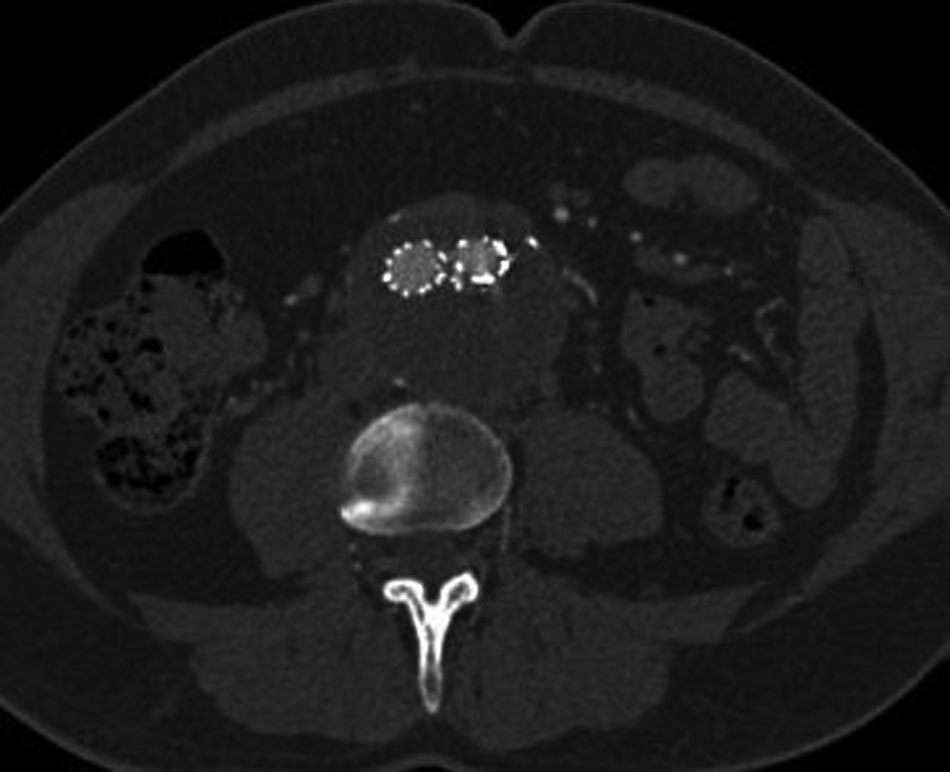
CT angiogram showing aneurysm repair (stent), resolution of the aortic dissection and obliteration of the false lumen.
Clinically, the patient has recovered well and investigation has revealed no evidence of either infection or connective tissue disorder. Post-op radiological follow-up did reveal a small type 2 endoleak, arising from the lumbar vessels; however, subsequent scans have shown evidence of sac shrinkage with a current sac size measurement of 5.5 cm (from 6.4 cm). After much multidisciplinary team (MDT) discussion, a strategy of close surveillance has been adopted to monitor progression and/or need for intervention, although the patient is aware that any increase in sac size may put him at higher than normal rupture risk.
Discussion
Aortic dissection occurring in the infrarenal abdominal aorta (IAAD) is uncommon,1,2 and on a background of aneurysmal disease is rare, with Cambria et al. finding that only 5% of all acute dissections ‘occur in the aorta of patients with coexistent or previously operated degenerative aneurysmal disease’. 3 In most cases, the location of the intimal flap is infrarenal (90% of cases reported by Farber et al. 4 ) and dissection may extend into the iliacs or remain localized to the aneurysm sac, as in our case.
Our patient presented with abdominal pain and fever, as did 30% of the patients with IAAD identified by Farber et al., 4 and this may represent an important clinical feature in the diagnosis of this condition. Indeed, there are reports in the literature of aortic dissection presenting as pyrexia of unknown origin 5 and Turner and Pusey describe a ‘post-dissection syndrome’ of systemic illness, fever and weight loss. 6 Other presentations of abdominal aorta dissection include back pain, end-organ ischaemia, peripheral embolization and even loin pain and urinary retention. 7 Generally, however, dissecting AAAs are more clinically evident than their non-dissecting counterparts because of their rapid evolution, although the presenting symptoms may initially be heterogeneous and non-specific. 8
Although the natural history and management of this combined pathology is poorly understood, the presence of both aneurysmal disease and aortic dissection is associated with a significant risk of vessel rupture due to loss of integrity of the aortic wall and increased wall tension. Although debate exists about when these patients should be offered intervention, a review of the literature appears to favour early surgical repair 9 and probably at an aneurysm size of just 3 cm, considerably below the usual ‘cut-off’ of 5.5 cm. 4 Endovascular repair appears to be feasible, 10 although some authors report difficulties in stent deployment in the presence of a large intimal dissection flap and Lai and Coleman advocate the use of an intravascular ultrasound in such complex cases. 11
This case clearly demonstrates how aortic disease can evolve rapidly and reports an important example of the successful use of endovascular repair for a patient with dual aortic pathology and significant bleeding tendency.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
