Abstract
Background
The COVID-19 pandemic has posed unprecedented challenges to healthcare systems globally, necessitating innovative care models like hospital-at-home and virtual care programs. The Influenzer telemedicine program aims to deliver hospital-led monitoring and treatment to patients at home. Integrating telemedicine technology with domestic visits provides an alternative to traditional hospitalization, with the aim of easing the burden on healthcare facilities without compromising patient safety. To evaluate the effectiveness of the Influenzer program, a randomized controlled trial is proposed. This study aimed to assess the feasibility of the proposed clinical trial design.
Methods
A non-randomized feasibility study was conducted at the Department of Pulmonary and Infectious Diseases at Nordsjaellands Hospital offering a telemedicine-supported early discharge program to patients with lower respiratory tract infections, including COVID-19. The feasibility of trial procedures, including recruitment, adherence, and retention, was analyzed. Also, participants’ characteristics and trajectory during the intervention, including telemedicine and domestic services, were assessed.
Results
Nineteen patients were enrolled from June 2022 to April 2023 and treated at home. Forty patients were not enrolled as 15 (25%) were non-eligible according to study protocol, 15 (25%) refused to participate and 10 (17%) had not been approached. Subjects treated at home had comparable clinical outcomes to those treated in the acute hospital, no major safety incidences occurred and patients were highly satisfied. Participants demonstrated 99% adherence to planned daily monitoring activities. In total, 63% completed all survey assessments at least partially including baseline, at discharge, and 3 months post-discharge, while 89% participated in a follow-up interview. No participants withdrew their consent.
Conclusions
The feasibility study documented that the Influenzer home–hospital program was feasible and well accepted in a Scandinavian setting in terms of no withdrawals and excellent participant adherence to the planned daily monitoring activities. Challenges in the organizational structures including patient recruitment and data collection required resolution prior to our randomized clinical trial. Insights from this feasibility study have led to the improved design of the final Influenzer program evaluation trial.
Trial registration
ClinicalTrials.gov, NCT05087082. Registered on 18 August 2021.
Keywords
Key messages
Recruitment rate: Did identified individuals as potential candidates for the intervention consent? What factors influenced recruitment? Eligibility rate: What proportion of screened patients were identified as potential candidates? What proportion of patients identified as potential candidates for the intervention were eligible? What factors influenced eligibility? Adherence rate: Did participants adhere to the intervention? What factors influenced adherence? Dropout rate: Was the intervention acceptable to the participants? What factors influenced withdrawal? Data collection and retention rate: Were outcome assessments completed? Was retention of the study good? What factors influenced data collection and retention? Safety: Did any major incidences (technical or clinically) challenge the safety of the patients hospitalized at home?
The recruitment rate fell below the target of 80%. The primary reasons cited for declining participation were related to the use of technology and the requirement for self-measurements. Out of 1980 individuals screened for eligibility, 59 (3%) were identified as potentially eligible for participation. The initial exclusion was primarily due to admission diagnoses other than per study protocol. Among 59 potential candidates, 19 individuals (32%) were successfully enrolled in the intervention. Adherence rate to daily scheduled patient activities was 99%, meeting predefined success criteria. There were no dropouts during the study, meeting predefined success criteria. Participants reported the data collection via surveys as challenging. The response rate for all surveys (at least partially) at baseline, end-line, and follow-up was 63%. The interview response rate was 89%.
The revision of the eligibility criteria included the removal of the specific diagnosis, and instead more focused on the planned treatment regimen and whether it could be provided at home. We adjusted the recruitment procedures and material—for example implementing a short cartoon introduction of the activities including technology use and self-measurements in a layman's language. The amendment of the original primary outcome measures and adjustment of the data collection methods together with the implementation of a time study to calculate time use associated with the intervention.
Background
The COVID-19 pandemic has inflicted unprecedented challenges on healthcare systems worldwide, with millions of lives lost and hospital capacities stretched to their limits.1–3 In response to this crisis, innovative care delivery models, including hospital-at-home (HaH) and virtual care programs, have emerged as potential solutions to alleviate the strain on healthcare resources while maintaining quality care.4,5 These initiatives aimed at both COVID-19-positive patients and other patient populations to expand healthcare systems’ capacity and reduce infection risk.4,6–8
Traditional HaH models typically involved domestic visits by clinicians, but their scalability was hindered by the constraints on hospital staff capacity. 9 To address this limitation, innovative approaches such as remote monitoring through telemedicine technology and hybrid models (integrating domestic visits and telemedicine) present a scalable solution to deliver hospital-level services directly to patients’ homes.5,10 These advancements hold significant promise in revolutionizing healthcare delivery, ensuring timely and effective care while easing the burden on healthcare facilities.
Amidst this landscape, we have developed the Influenzer telemedicine program, a public–private collaborative effort aimed at providing hospital-led monitoring and treatment for patients with lower respiratory tract infections (LRTI), including COVID-19, in the comfort of their homes. By integrating a standard in-hospital triage algorithm early warning score (EWS) and remote monitoring technology, the program sought to mirror the standards of hospital monitoring while enabling early discharge for eligible patients. 11 We developed a novel telemedicine system including an app for patients and a corresponding web-based dashboard for the hospital site. We conducted a proof-of-concept test with a small group of inpatients, revealing a commendable level of technical safety and usability. The results from this initial test suggested that the telemedicine system can be effectively and safely utilized in clinical settings to remotely monitor hospitalized patients. However, to ascertain the true impact and benefits of the Influenzer program on hospitalized patients’ pathways, a comprehensive large-scale comparative study is required. Such an investigation will shed light on the program's potential to enhance patient outcomes and modernize healthcare delivery in hospital settings.
As a component of preparing for a large-scale clinical trial, feasibility should be assessed comprising key processes including recruitment, adherence, and retention.12,13 Equally important is gaining insights into how research findings can be effectively translated into clinical practice. To achieve this, a comprehensive examination of the implementation process, contextual factors, service delivery, and stakeholder acceptability must be undertaken. 14 By conducting such evaluations, we could optimize trial design and ensure the successful integration of research outcomes into real-world healthcare settings.
We address a gap in the existing evidence on remote monitoring programs implemented worldwide in response to COVID-19. While initial evaluations of these programs showed promise, the lack of comprehensive prospective assessments hinders meta-analysis.15–21 Our clinical research including this feasibility study and subsequent randomized controlled trial (RCT) will fill this gap, contributing valuable evidence to enrich collective knowledge in this area.
Methods
The overall aim of this clinical trial was to evaluate the feasibility of the Influenzer program and provide valuable insights for the design of an RCT. While comprehensive data on all relevant outcomes (e.g. initial program acceptability among stakeholders, and early implementation of the program) were collected during the trial, this report specifically focuses on the investigation of the trial design uncertainties including recruitment, adherence, retention, and data collection. Even though this was a non-randomized feasibility study design, we reported the study following the CONSORT extension to randomized pilot and feasibility trials as many of the principles apply (see Additional file 1). 22
Study design and study settings
This study employed a single-site, non-randomized feasibility design as depicted in the schematic figure of study design (see Additional file 2) and was conducted at the Department of Pulmonary and Infectious Diseases (DPID) within Nordsjaellands Hospital, Capital Region of Denmark. The study design was informed by a feasibility framework suggested by Shanyinde et al. and Bugge et al.23,24
Before commencing this study, DPID's clinical staff received comprehensive information and training on the Influenzer protocol as an integral part of the initial implementation strategy. To ensure the technology's usability and performance quality, a proof-of-concept test was conducted before this feasibility study on a small cohort of inpatients and the DPID clinicians including an iterative process of improvement to the technology from May 2021 to January 2022.
Participants
Patients admitted to the DPID were included in the study at the stage of their medical course when diagnostic procedures were completed, but treatment was still ongoing. All eligibility criteria per protocol are described in detail in Table 1. In general, eligible patients included patients of 18 years or older with LRTI, who were relatively stable in their infection parameters, who didn’t require intensive care treatment, who had no significant care dependency, who had no major communication barriers (primary language) and who possessed adequate digital literacy.
Inclusion and exclusion criteria per study protocol.

Recruitment procedures
Participants were recruited between April 2022 and May 2023 from DPID. Throughout the recruitment period, there were several temporary suspensions of recruitment, totaling three months, due to organizational conditions (see Table 2).
Factors influencing the recruitment process based on the research coordinator log and informal interviews with clinicians and the clinical support group involved in the recruitment.

Abbreviation: DPID, Department of Pulmonary and Infectious Diseases.
Two distinct recruitment procedures were implemented before and after the establishment of the clinical support group of four clinicians (the first and second authors and two project nurses). From April 2022 to August 2022, DPID clinicians were responsible for screening and identifying potential participants, while researchers provided information about the study and obtained consent. The final eligibility assessment, based on all inclusion and exclusion criteria, was conducted by a DPID physician after obtaining consent from potential participants. Patients who did not meet the criteria were either permanently or temporarily excluded, with the possibility of reassessment for inclusion. Eligible patients were educated about intervention-related tasks, such as using mobile medical equipment for self-measurements at home and utilizing the telemedicine app.
Starting in August 2022, the clinical support group was responsible for screening all patients admitted to the DPID. They screened and recruited only on weekdays. If DPID clinicians identified a potential participant during a weekend, a note was made in the electronic patient record (EPR) to notify the group. The amendment to the screening procedure was necessary due to a busy clinical context influencing the recruitment rate.
Intervention
The intervention implemented in this study was a hospital-led early discharge program (Influenzer program), which combined telemedicine and domestic visits for remote monitoring and medical interventions. The duration of patient participation was based on individual medical needs. An overview of all essential services integrated into the program is provided in the supplementary material (see Additional file 3).
Upon completion of the baseline survey assessment at the department, participants were transferred home and remotely monitored by DPID clinicians 24 hours per day via telemedicine. Patients measured vital signs, including blood pressure, rectal temperature, pulse rate, respiratory rate, and oxygen saturation, using provided mobile medical equipment that is described in detail in Additional file 4. These data were manually entered into the telemedicine app. Additionally, patients completed a symptom screening questionnaire in the app that was transferred together with the vital signs. The frequency of data transmission was determined based on an individual risk assessment using EWS triage (see Additional file 5). Data transfer varied from two to six times per day. An algorithm triggered differentiated alerts at DPID based on: (a) predefined clinical ranges; (b) the presence of new symptoms; or (c) non-compliance with the scheduled plan. The synchronized display of symptom screening, vital parameters, and alerts was accessible to clinicians through the web-based dashboard. A virtual ward round was conducted by video once a day. Here, a DPID physician assessed the patient's clinical condition. In addition, patients had the option to request telephone contact via the app or directly call DPID clinicians. For domestic procedures not suitable for telemedicine, a Mobile Hospital-based Care Team (MHCT) integrated into the Emergency Department, or a mobile Clinical Research Unit (CRU) were available. Participants remained enrolled in the program until one of the following conditions was met: (a) they wished to return to the hospital for any reason; (b) they were discharged; (c) in case of clinical deterioration, requiring in-hospital care (escalation of care); or (d) in case of clinically stable condition with no need for hospital-level monitoring, thus, the treatment could continue within other programs until the final discharge (de-escalation of care).
The intervention included survey-based patient assessments conducted at three different time points: baseline, end-line, and follow-up. The assessment results will be reported elsewhere; however, an overview of all questionnaires and the outcome measures of interest is depicted in the supplementary material (see Additional file 6). Baseline surveys were administered at enrollment, end-line surveys were sent at the final discharge and collected within 14 days, and final follow-up surveys were sent 3 months post-discharge and collected within 1 month. Participants were given the option of using either electronic or paper surveys at the baseline assessment, and their chosen modality was maintained for subsequent assessments. A telephone interview was conducted 1 week post-discharge.
Outcomes, data collection, and analysis
The primary focus of this study was to assess the feasibility of the trial design and procedures. Secondary outcomes included capturing the baseline characteristics of participants and describing their treatment trajectory throughout the Influenzer program. Descriptive statistics were employed for data analysis, and the results were presented in descriptive tables. Data were summarized using frequency (%), or median (IQR) due to non-normalized data distribution.
Primary outcomes—feasibility of trial design and procedures
The selection of the feasibility outcomes was guided by a feasibility analytic framework.23,24 For this study, we included the following framework items: recruitment rate; eligibility rate; adherence rate; dropout rate; data collection; retention rate; and whether data could be collected to estimate the intervention costs. The item definition is detailed in Table 3.
Areas of focus for feasibility assessment including research questions, definitions, and findings in the feasibility trial.

Quantitative and qualitative data about recruitment, eligibility, dropout, and retention were collected from research documents such as screening logs and coordinator logs (e.g. the number of individuals screened and approached; the reasons for participation decline; qualitative observations during baseline data collection). Quantitative data about adherence were collected from the telemedicine system and the EPR (e.g. number of planned patient activities, number of completed activities). Quantitative data about intervention costs included costs related to hospital-led care delivery during the intervention and other resources necessary to support the successful execution of the intervention. These data were collected from coordinator logs (e.g. estimated time used for patient recruitment and educational procedures), the telemedicine system (e.g. the number of telemedicine services provided), and the EPR (e.g. the number of domestic care services provided). These data were combined with informal interview data with the clinical support group to get a comprehensive overview of resource use during the intervention and a deeper understanding of what methods could be applied to collect relevant data in the future study.
Data for recruitment, adherence, dropout, and retention rates were analyzed against predefined progression criteria (see Table 3). Also, informal qualitative feedback from DPID clinicians and the clinical support group involved in the trial procedures was notified in the coordinator log and used to gain a comprehensive understanding of the success factors and challenges during the trial. Both analysis of the data against the progression criteria and described experiences were used to inform the discussion of how to proceed with future definitive trial.
Secondary outcomes—baseline characteristics of participants and their program trajectory
Data on participants’ educational background, work situation, civil status, smoking, and alcohol history were collected through the baseline survey. Information regarding age, sex, primary admission diagnosis, and the presence of comorbidities (e.g. heart disease, chronic lung disease, hypertension, diabetes, active oncological disease) was extracted from the EPR.
We collected data on the number and type of received domestic services, the number and type of telemedicine services, and the mode of intervention exit (e.g. discharge, escalation, or de-escalation of care) from the coordinator logs, the telemedicine system, and the EPR. Length of stay data was gathered from the EPR for the following periods: (a) before the intervention; (b) during the intervention; and (c) after the intervention in case of escalation/de-escalation of care. Additionally, we recorded data from the EPR on readmissions for any reason within 30 days post-discharge, as well as deaths occurring within 7 and 30 days post-discharge.
Sample size
For this feasibility study, we aimed to recruit 20 participants. This sample size is in line with previous reports with sample sizes between 10 and 300.25,26
Results
A summary of feasibility results is provided in Table 3 and the enrolment and follow-up are depicted in detail in the CONSORT flow diagram (Figure 1).
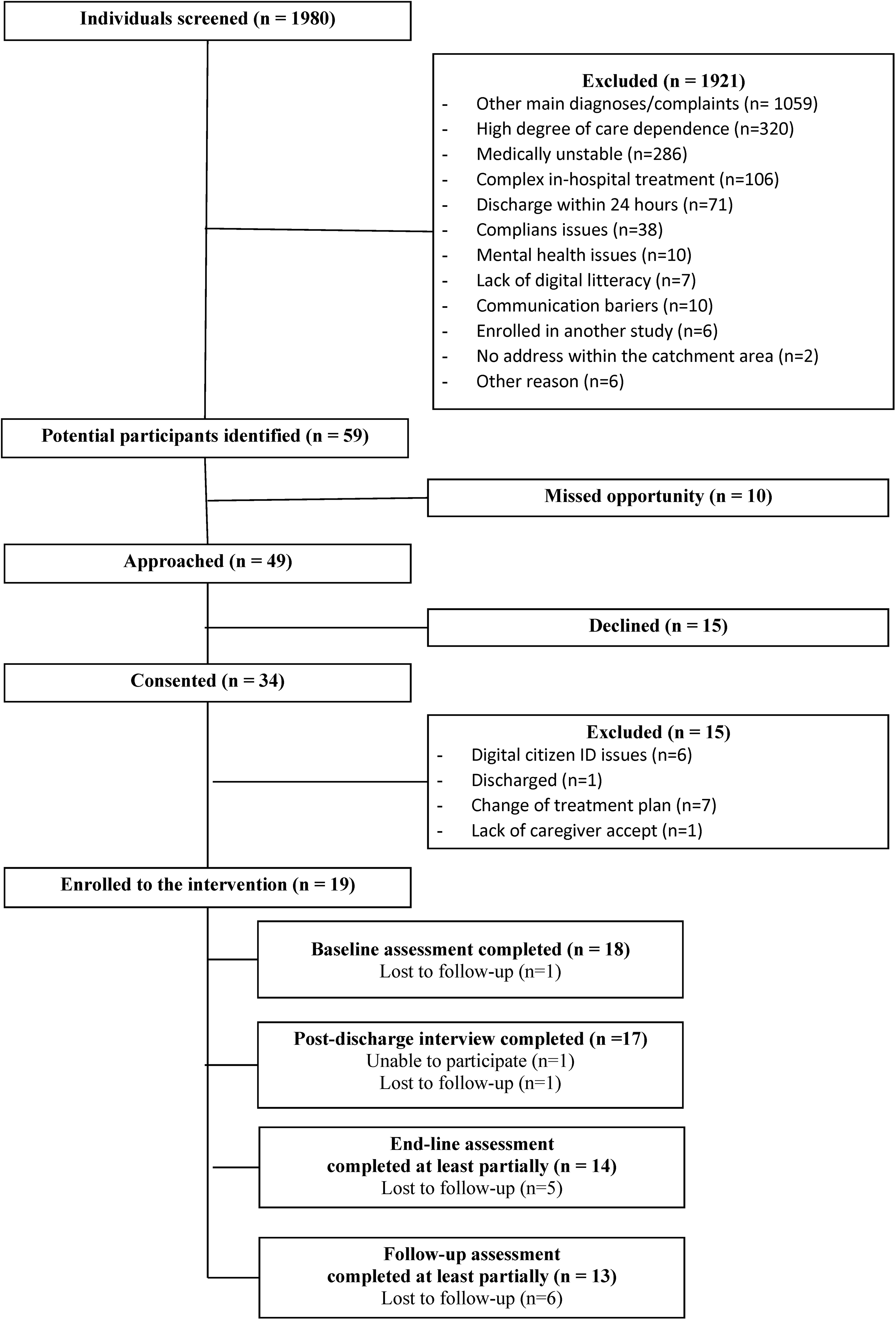
Flow diagram of recruitment and data collection guided by Consort guidelines.
Recruitment and eligibility
From April 2022 to May 2023, 1980 individuals were screened for eligibility. We identified 59 (3%) individuals as potential participants. Among these, 10 (17%) were not approached, 15 (25%) declined to participate, 15 (25%) were excluded, and 19 (32%) were enrolled in the study. The consent rate was 69% with 34 individuals providing consent out of 49 individuals invited to the study.
The main reasons for initial exclusion were primarily due to admission diagnoses other than per study protocol (55%), high care dependence (17%), or medical instability (15%). Other less frequent reasons included ineligibility for home-based treatment due to complex in-hospital treatment, planned discharge within 24 hours, non-compliance, cognitive impairment, and lack of digital literacy.
Among the 34 individuals who consented, 15 (44%) individuals were later excluded for various reasons. These included changes in treatment (44%); inability to use the telemedicine app due to access issues (because of issues with the personal digital identification that was required for security reasons) (40%); planned discharge (7%); and caregiver refusal (7%).
Overall, 19 (1%) of 1980 screened individuals were successfully enrolled in the study, highlighting the challenges faced during the recruitment and eligibility process. The initial procedure, where DPID clinicians identified potential participants, was challenged by a busy clinic and complex eligibility criteria. After the introduction of the clinical support group, recruitment remained slow due to other factors (e.g. seasonal variation of respiratory infections; logistical limitations; digital literacy issues; and organization resistance to change). Barriers and facilitators of recruitment are detailed in Table 2.
Among 49 invited individuals, 15 (31%) declined participation. Reasons for declining included concerns about performing self-measurements without assistance and using the telemedicine app. Other reasons included medical conditions; previous negative experiences with early discharge programs; and the absence of a cohabiting caregiver for support in emergencies (Table 4).
Baseline characteristics and reasons for declining recruitment among non-participants.
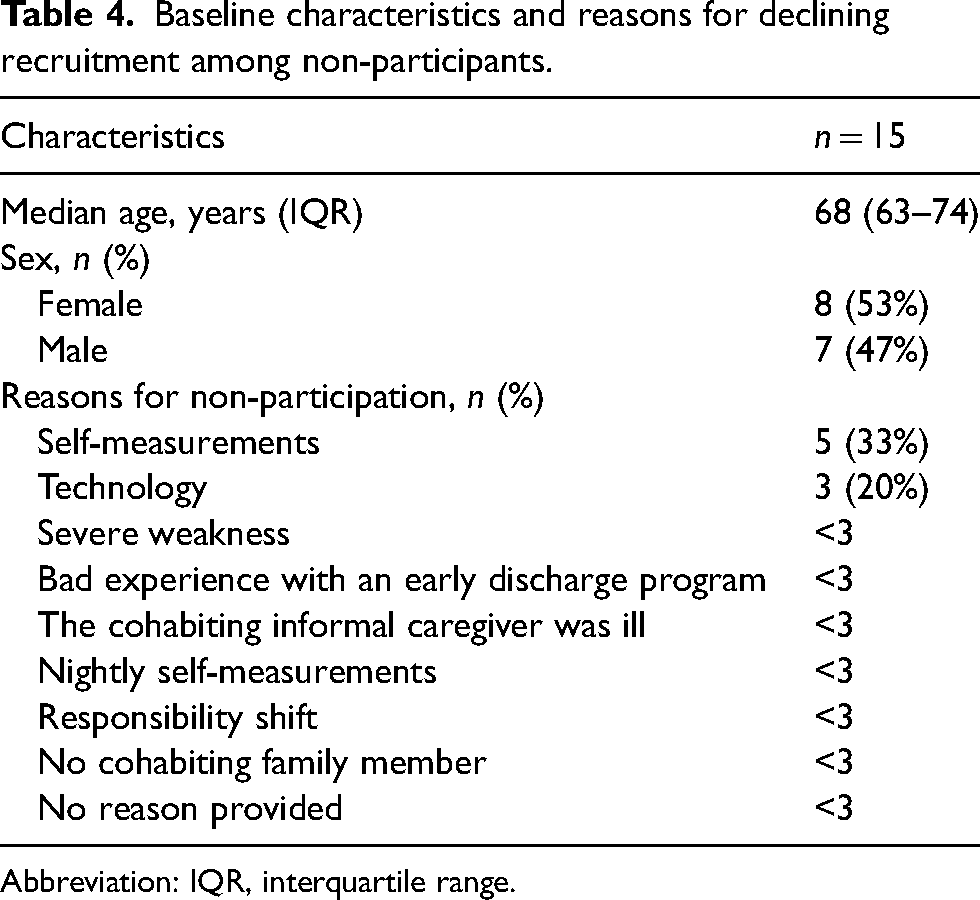
Abbreviation: IQR, interquartile range.
Adherence and dropout
Participants demonstrated adherence to daily activities, completing 179 out of 180 planned self-measurements (99%). No participants withdrew their consent during the intervention.
Data collection and retention
Data collection and retention faced challenges—especially survey completion at discharge and 3 months post-discharge (end-line and follow-up). Out of 19 participants, 18 (95%) completed the baseline assessment, 14 (74%) completed the end-line assessment at least partially, and 13 (68%) completed the follow-up assessment at least partially. Only 12 (63%) participants completed all three survey assessments at least partially, while 17 (89%) participated in the follow-up interview.
Participants faced challenges during baseline surveys due to lengthy questionnaires, energy limitations, cognitive impairment, and font size issues when filling out on a smartphone. Some preferred paper-based surveys due to limited electronic mail access. Non-responders cited time constraints and lack of motivation upon a reminder call from the researchers.
Intervention costs
An overview of identified resources used during the program and additional resources required to initiate the program are depicted in Additional file 7. It was feasible to collect the most relevant cost data including the number of video or telephone assessments; domestic visits; and assessment of incoming alerts. Participants received on average 23 telemedicine assessments by a physician and 25 domestic visits. The clinicians assessed on average 95 self-measurements, 14 severe alerts, and 19 non-severe alerts per patient. Medical services and resources utilized, which rely on hospital-based clinical resources were on average $550.
Participant characteristics
Most participants were male (58%), with a median age of 65 years (IQR 59-72). The primary admission diagnosis for most participants was unspecified pneumonia (47%), and most of them lived with a partner (68%). Detailed characteristics of the participants are presented in Table 5.
Baseline characteristics of participants.
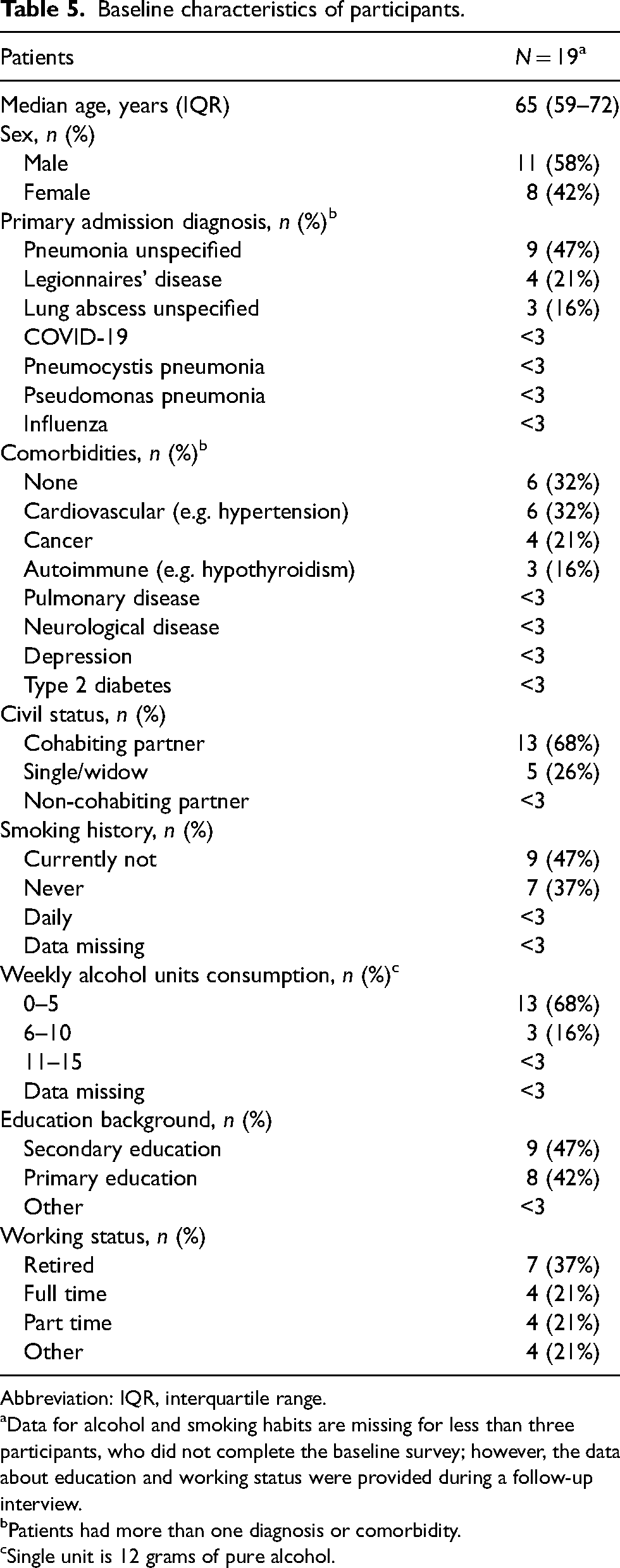
Abbreviation: IQR, interquartile range.
Data for alcohol and smoking habits are missing for less than three participants, who did not complete the baseline survey; however, the data about education and working status were provided during a follow-up interview.
Patients had more than one diagnosis or comorbidity.
Single unit is 12 grams of pure alcohol.
Participant's program trajectory
During the program, 19 participants received a total of 65 days of home-based care, with a median length of stay of 2 days (IQR 1·5-5) (Table 6). Among the participants, 11 (58%) were discharged after the program, while 3 (16%) had their care de-escalated to other home-based programs. Five (26%) participants were required to return to the in-hospital settings (escalation of care), but none of them needed ventilator support or Intensive Care Unit admission, and there were no deaths upon the escalation. Reasons for returning to the hospital included insecurity and confusion about self-measurements or technology use; in-hospital diagnostic and therapeutic intervention requirements; and subjective worsening with elevated EWS scores. No deaths or severe adverse events occurred during the intervention.
Characteristics of the hospital-led early discharge program (Influenzer) and services delivered to the participants during total program enrollment length.

Abbreviation: IQR, interquartile range.
Non-severe alerts defined as Early Warning Score between 3 and 6.
Severe alerts defined as Early Warning Score > 6.
Patients can request telephone contact from clinicians using the telemedicine app.
In case of escalation (hospital-based stay) or de-escalation (home-based stay) of care before the final discharge.
During home-based care, only a few participants required domestic isolation due to COVID-19, while numerous received intravenous antibiotics (47%), and used supplemental oxygen therapy (21%). Hospital-based nursing staff provided domestic visits for intravenous medication and blood analysis to 12 (63%) participants counting a total of 39 visits, while 6 (32%) individuals received domestic visits from laboratory technologists for blood analysis counting a total of 9 visits.
Regarding telemedicine application during home-based care, 18 (95%) participants received video or telephone assessments by a physician counting in total 47 individual assessments, with video modality offered to 16 (84%) participants. Six (32%) participants used the app for urgent or non-urgent contact requests in total generating 15 requests. No major incidents occurred during the study period related to the technical solution or the patients clinical situation at home, challenging the safety of the patient.
During the total of 65 days of home-based care, participants received daily video or telephone assessments by a physician 47 times (72%); daily domestic visits for intravenous treatment including blood analysis 39 times (60%); assessment of non-severe alerts defined as EWS score between 3 and 6 counted 37 days (57%); assessments of severe alerts defined as EWS score > 6 counted 27 days (42%); assessment of contact requests counted 15 days (23%); and domestic visits only for blood analysis counted 9 days (14%).
Discussion
Our study introduces the Influenzer telemedicine program, designed to deliver hospital-led monitoring and treatment for LRTI, including COVID-19, at patients’ homes. The primary aim was to assess the feasibility of the proposed clinical trial and provide insights for a future RCT.
In this study, the trajectory of participants demonstrated successful program implementation, facilitating early discharge, and providing remote monitoring supported by domestic visits. No deaths occurred during the program or upon escalation to in-hospital settings, providing some indication of clinical safety. However, the program's safety and appropriateness for managing LRTI at home need to be rigorously evaluated in a future large comparative trial.
Participants who readily joined the intervention tended to be younger than 60 years of age and more accustomed to using technology daily. This indicates that more attention is needed to ensure that older patients with limited digital literacy get adequate introduction and training to the program. Otherwise, older patients may prefer a conventional hospital-based hospitalization albeit they might benefit from a HaH program as suggested by Norman et al. 21 Overall, participants’ high adherence and no dropouts indicated a favorable acceptance of the Influenzer intervention.
While this study had successful aspects as mentioned above, other challenges needed to be addressed. The original study design wasn’t feasible and needed adjustments before RCT initiation. Recruitment faced difficulties due to organizational factors, complex eligibility criteria, patient preferences for hospital-based care, and their limited digital literacy. Adherence to daily patient activities in the intervention was high, but survey completion at discharge and follow-up was suboptimal, questioning the suitability of the Short Form-36 questionnaire that was originally proposed to capture one of the primary outcome measures (quality of life). It was only possible to estimate theoretical time use for several tasks during the program delivery (e.g. time use for telephone calls to participants, and documentation in EPR by clinicians) because no automatic or manual data registry was in place. Thus, measuring exact data on clinicians’ time use needed to be applied in the RCT design to assess the intervention costs including time used for program activities.
Similar recruitment issues and attrition problems were reported in other remote monitoring and home-based care studies.27–29 Studies reported participation barriers including technical competence, threats to identity, and disruptions to services, and recommended adaptations to clinical workflows and eligibility criteria.28,29 Other reported patient age, education level, eHealth literacy, and resistance to change as important factors for telemedicine implementation. 30
Addressing the identified challenges in recruitment and data collection was crucial for obtaining robust and meaningful data for program effectiveness evaluation. We present an overview of our recommendations for the RCT design based on the identified challenges in Table 7. Based on this feasibility study, we implemented the insights in the design of the RCT before its initiation. The final trial design improvements included revision and simplification of the eligibility criteria; design of a multicenter trial; supplemental cartoon video illustrating the intervention in simple terms to avoid information overload; revision of the number and type of surveys; and revision of the primary outcome measures based on the logic model developed during the feasibility trial.
Recommendations for future Influenzer program trial design based on feasibility study data.

To our knowledge, this study is the first to report on a telemedicine-supported early discharge intervention in a Danish sample of individuals with LRTI. This feasibility study focuses solely on methodological issues, avoiding unintended outcome exploration. Progression criteria were established for three key factors (recruitment, dropout, and patient adherence) a priori (ClinicalTrials.gov Identifier: NCT05087082). The largest limitation of this study is the non-randomized design that prohibited the assessment of the randomization process; thus, its feasibility remains uncertain for the final RCT trial.
Conclusion
In conclusion, this feasibility study documented that the Influenzer home–hospital program was feasible and well accepted in a Scandinavian setting in terms of no withdrawals and excellent participant adherence to the planned daily monitoring activities. Further, no major safety incidences were present during the intervention indicating an acceptable patient safety. Challenges in the organizational structures including patient recruitment and data collection required resolution prior to our randomized clinical trial. Insights from this feasibility study have led to the improved design of the final Influenzer program evaluation trial. Our results also shed light on the potential of the Influenzer program for hospital-led monitoring and treatment at home. Based on the identified methodological challenges, we were able to implement design improvements timely to initiate the final RCT in June 2023 (ClinicalTrials.gov Identifier NCT05920304). The RCT findings will subsequently advance evidence-based medical practices, and potentially enhance healthcare delivery for patients admitted due to acute infections.
Supplemental Material
sj-doc-1-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-doc-1-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Supplemental Material
sj-docx-2-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-docx-2-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Supplemental Material
sj-docx-3-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-docx-3-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Supplemental Material
sj-docx-4-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-docx-4-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Supplemental Material
sj-docx-5-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-docx-5-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Supplemental Material
sj-docx-6-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-docx-6-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Supplemental Material
sj-docx-7-jtt-10.1177_1357633X241254572 - Supplemental material for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study
Supplemental material, sj-docx-7-jtt-10.1177_1357633X241254572 for Transforming health care: Investigating Influenzer, a novel telemedicine-supported early discharge program for patients with lower respiratory tract infection: A non-randomized feasibility study by Tatjana Sandreva, Maria Normand Larsen, Maja Kjær Rasmussen, Thyge Lynghøj Nielsen, Charlotte von Sydow, Thomas Andersen Schmidt and Thea K Fischer in Journal of Telemedicine and Telecare
Footnotes
Abbreviations
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study has ethics approval number 98381 provided by the Regional Committee of Human Health Research Ethics, Capital Region of Denmark.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Influenzer project is partly funded by Innovation Fund Denmark (grant number 0176-00017B). The funding organization was not involved in the study design, data collection, data analysis, data interpretation, or manuscript writing. The research was conducted without any influence from the funder.
Data availability statement
Data collected for this study will not be made publicly available to preserve individuals’ privacy under the European General Data Protection Regulation. The datasets supporting the conclusions of this article will be shared on request for academic purposes.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
