Abstract
Introduction
Telehealth may address healthcare disparities for rural populations. This systematic review assesses the use, effectiveness, and implementation of telehealth-supported provider-to-provider collaboration to improve rural healthcare.
Methods
We searched Ovid MEDLINE®, CINAHL®, EMBASE, and Cochrane CENTRAL from 1 January 2010 to 12 October 2021 for trials and observational studies of rural provider-to-provider telehealth. Abstracts and full text were dual-reviewed. We assessed the risk of bias for individual studies and strength of evidence for studies with similar outcomes.
Results
Seven studies of rural uptake of provider-to-provider telehealth documented increases over time but variability across geographic regions. In 97 effectiveness studies, outcomes were similar with rural provider-to-provider telehealth versus without for inpatient consultations, neonatal care, outpatient depression and diabetes, and emergency care. Better or similar results were reported for changes in rural clinician behavior, knowledge, confidence, and self-efficacy. Evidence was insufficient for other clinical uses and outcomes. Sixty-seven (67) evaluation and qualitative studies identified barriers and facilitators to implementing rural provider-to-provider telehealth. Success was linked to well-functioning technology, sufficient resources, and adequate payment. Barriers included lack of understanding of rural context and resources. Methodologic weaknesses of studies included less rigorous study designs and small samples.
Discussion
Rural provider-to-provider telehealth produces similar or better results versus care without telehealth. Barriers to rural provider-to-provider telehealth implementation are common to practice change but include some specific to rural adaptation and adoption. Evidence gaps are partially due to studies that do not address differences in the groups compared or do not include sufficient sample sizes.
Keywords
Numerous studies have documented health disparities for people living in rural areas of the United States.1–4 Rural-urban differences in access and quality of care contribute to higher mortality4–7 and morbidity from conditions including substance/opioid misuse,8,9 chronic illnesses,10–13 and human immunodeficiency virus/human papillomavirus and other infectious diseases.14,15 Causes of rural-urban differences are varied and related to complex macro and micro sociologic-demographic forces 16 and economic trends;17,18 nevertheless, the need for solutions is urgent. Both the coronavirus disease 2019 (COVID-19) pandemic and acknowledgement of structural racism have focused attention on broad inequities in healthcare in the United States, disproportionate burden on rural populations, and the resulting harms to underserved populations.
Telehealth utilizes technology to provide health care across time and/or distance 19 , encompassing a wide range of interventions, modes, and clinical functions. Telehealth is frequently proposed to address limited access and health disparities20,21 and there is a sizable body of research, including systematic reviews and reviews of reviews,22–29 suggesting telehealth can be effective. However, implementation and spread have been slow,30–32 with steady, but small increases in telehealth use before the COVID-19 pandemic. 33 Rapid increases in the use of telehealth to limit the risk of exposure and transmission during the COVID-19 pandemic may continue to fuel wider adoption and more long-term use.34,35 Growth in telehealth has focused attention on the need to address the digital divide, promote widespread access to telehealth, 36 and assure telehealth promotes equitable healthcare. 37
Rural provider-to-provider telehealth (RPPT) is a subset of telehealth interventions focused on supporting health care providers that treat rural patients and populations through consultations and collaborative patient care as well as mentoring and education.38–40 We conducted this systematic review to identify and synthesize the available research on whether telehealth-supported provider-to-provider communication and collaboration can contribute to improving the health and well-being of rural residents and communities. This review was conducted to inform a National Institutes of Health (NIH) Office of Disease Prevention (ODP) Pathways to Prevention (P2P) workshop, Improving Rural Health through Telehealth-Guided Provider-to-Provider Communication, held on October 12–14, 2021). The panel report and recommendations from this workshop are also published in this journal. 41
Methods
We conducted this review using methods outlined in the Agency for Healthcare Research and Quality (AHRQ) Methods Guide for Effectiveness and Comparative Effectiveness Reviews (hereafter “AHRQ Methods Guide”). 42 Our protocol is available on the AHRQ website (https://effectivehealthcare.ahrq.gov/products/rural-telehealth/protocol) and registered with PROSPERO (registration no. CRD42021233545). Detailed methods and additional information in the full report are available at https://effectivehealthcare.ahrq.gov/products/rural-telehealth/research.
Key questions
This review was designed to answer the following four questions:
What is the uptake of different types of provider-to-provider telehealth in rural areas?
What is the effectiveness of provider-to-provider telehealth for rural patients?
What strategies are effective and what are the barriers and facilitators to implementation and sustainability of provider-to-provider telehealth in rural areas?
What are the methodological weaknesses of the included studies of provider-to-provider telehealth for rural patients and what improvements in study design (e.g. focus on relevant comparisons and outcomes) might increase the impact of future research?
Data sources and searches
We developed strategies with an expert librarian, had these peer-reviewed by a second librarian, and searched Ovid MEDLINE®, CINAHL®, EMBASE, and Cochrane CENTRAL for studies published between 1 January 2010 and 13 October 2021 (Search strategies included in online Supplement 1). Studies on telehealth have been published before January 2010. However, this start date was selected in consultation with the key informants and NIH ODP stakeholders and corresponded to their request to focus the review on studies using current technology that would be most relevant to future policy and decision-making.
Study selection
Two team members reviewed abstracts and full-text articles to identify studies that met our inclusion criteria (Supplemental Table 1), and any disagreements were resolved through discussion and consensus. To be included, studies had to be of rural patients or populations, or report results separately for rural patients or populations. Trials and observational studies that provided data on use, effectiveness, and facilitators or barriers to implementation of RPPT for inpatient, outpatient, or emergency care and studies of telehealth used for training health care providers who care for rural populations were included. Descriptive articles and studies that reported only data after telehealth implementation (no comparison group) were excluded. Studies had to be conducted in the United States or countries identified as having very high or high human development levels by the United Nations Human Development Report. 43
We defined RPPT as any form of interactive support using telecommunications technology provided to health care professionals who care for rural patients and populations. This included: video, audio, or digital remote consultations across space (e.g. video) or time (e.g. store and forward) as well as remote mentoring or education including rounds or case reviews. We excluded (a) telehealth for patient encounters, (b) remote patient monitoring if the data were transmitted only from a patient to a single provider and were not used as part of provider-to-provider consultations, and (c) communications that were limited to referring a patient to another provider for care and with no other interaction.
To assess use of RPPT we included studies with indicators or measures of uptake such as rates of use as outcomes. For effectiveness we included studies reporting clinical outcomes (e.g. mortality and morbidity) and intermediate outcomes (e.g. treatment, satisfaction, health care services utilization, and economic outcomes) We excluded studies if the outcomes were projections or the results of simulations.
Data extraction and risk of bias assessment
One person extracted data on the study design, setting, patient population, telehealth intervention, participating providers, and outcomes from the included studies. If a study was reported in multiple publications, the study was included once in our counts, but information about the study and data on outcomes were abstracted from all available publications. When extracting data from effectiveness studies, we also extracted methodological weaknesses listed by the study authors. Extracted data were verified for completeness and accuracy by a second person. Two team members independently assessed the risk of bias for individual studies using criteria appropriate for the study design and consistent with the approach in the AHRQ Methods Guide. 42 Studies were rated as low, moderate, or high risk of bias.
Data synthesis and analysis
We summarized surveys and analyses that provided counts or estimates to assess use of RPPT. To address the effectiveness of RPPT, we grouped studies by health care setting, then clinical indication, and summarized findings based on the direction and magnitude of effect. We also assessed the strength of evidence (SOE) for outcomes prioritized by the ODP working group, following the approach described in the AHRQ Methods Guide. 42 Our assessment of implementation summarized the few identified studies that compared approaches, then summarized barriers and facilitators cited in included studies. We classified barriers and facilitators using an existing framework, the Consolidated Framework for Implementation Research (CFIR). 44 We then summarized how frequently barriers and facilitators were reported across all identified studies. Our synthesis of the methodologic weaknesses of the effectiveness studies consisted of summarizing the weaknesses identified by the study authors as well as those identified by the review team.
Results
Our search produced 6329 citations, after triage of abstracts we reviewed 1024 full-text articles and included 166 studies reported in 179 publications (Figure 1). Seven studies met the inclusion criterial for Key Question 1 and provided data on use of provider-to-provider telehealth is rural areas (Key Question 1), 97 studies in 106 publications provided data on effectiveness of RPPT(Key Question 2) and were also used to identify methodological weaknesses in the body of evidence (Key Question 4), and 67 studies in 71 publications were used to summarize barriers and facilitators to implementation of RPPT (Key Question 3). Detailed descriptions of the included studies, as well as the data extracted, are included in the appendix to the full report at https://effectivehealthcare.ahrq.gov/products/rural-telehealth/research.
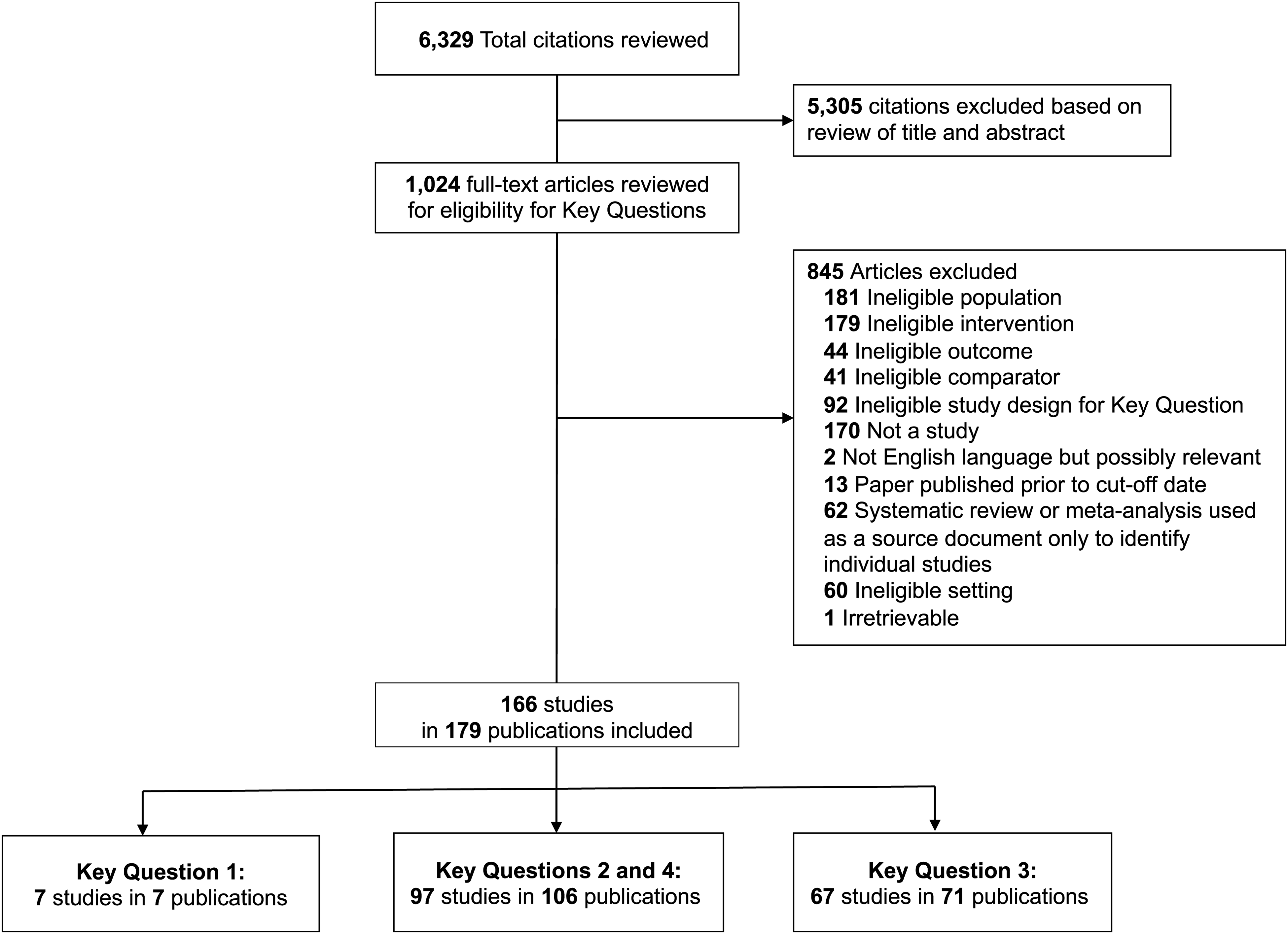
Literature flow diagram. Key Question 1: What is the uptake of different types of provider-to-provider telehealth in rural areas? Key Question 2: What is the effectiveness of provider-to-provider telehealth for rural patients? How does provider-to-provider telehealth affect outcomes for patients and populations? How does provider-to-provider telehealth affect outcomes for healthcare providers? How does provider-to-provider telehealth affect outcomes for private and public payers? Key Question 3: What strategies are effective and what are the barriers and facilitators to implementation and sustainability of provider-to-provider telehealth in rural areas? Key Question 4: What are the methodological weaknesses of the included studies of provider-to-provider telehealth for rural patients and what improvements in study design (e.g. focus on relevant comparisons and outcomes) might increase the impact of future research
Use of provider-to-provider telehealth for rural populations
We did not identify comprehensive data on the use of RPPT in the United States. However, we identified seven studies that reported on national or regional use of provider-to-provider telehealth for emergency or inpatient care and included data specific to rural areas.45–51 These studies were published since 2015 but included data collected prior to the COVID-19 pandemic.
Most studies reported lower use in rural than in urban areas, but studies that examined changes over time reported increases in both rural and urban hospitals. A study conducted in 2018 found that urban hospitals were twice as likely as rural hospitals to use telehealth for intensive care unit (ICU) and stroke care. 46 A study of telehealth use for heart attack and stroke assessment reported that rural hospital use increased from 6% in 2012 to 16% in 2017, though this was still lower than urban hospitals. 45 One study that reported higher use in rural than in urban areas focused on rates of telestroke use (8.6 vs. 2.3 per 1000), using Medicare fee-for-service claims to compare rates at the patient, rather than hospital, level. 51 Two studies of telehealth use for any reason by emergency departments (EDs) in New England in 2014 50 and nationwide in 2016 49 reported use by 49% and 54% of rural EDs, though this differed by region, with less use in the southern United States. Additionally, studies of telepsychiatry use in rural EDs in 2016 47 and by rural mental health facilities from 2010 to 2107 48 reported less than 30% of those surveyed used telehealth.
Effectiveness of provider-to-provider telehealth for rural patients
We assessed effectiveness and SOE after grouping studies by setting and clinical topic. Table 1 presents outcomes for patients, providers, and payers. Findings are summarized in the text below by setting and in terms of SOE by outcome in Supplemental Table 2.
Effectiveness in inpatient, outpatient and emergency care.
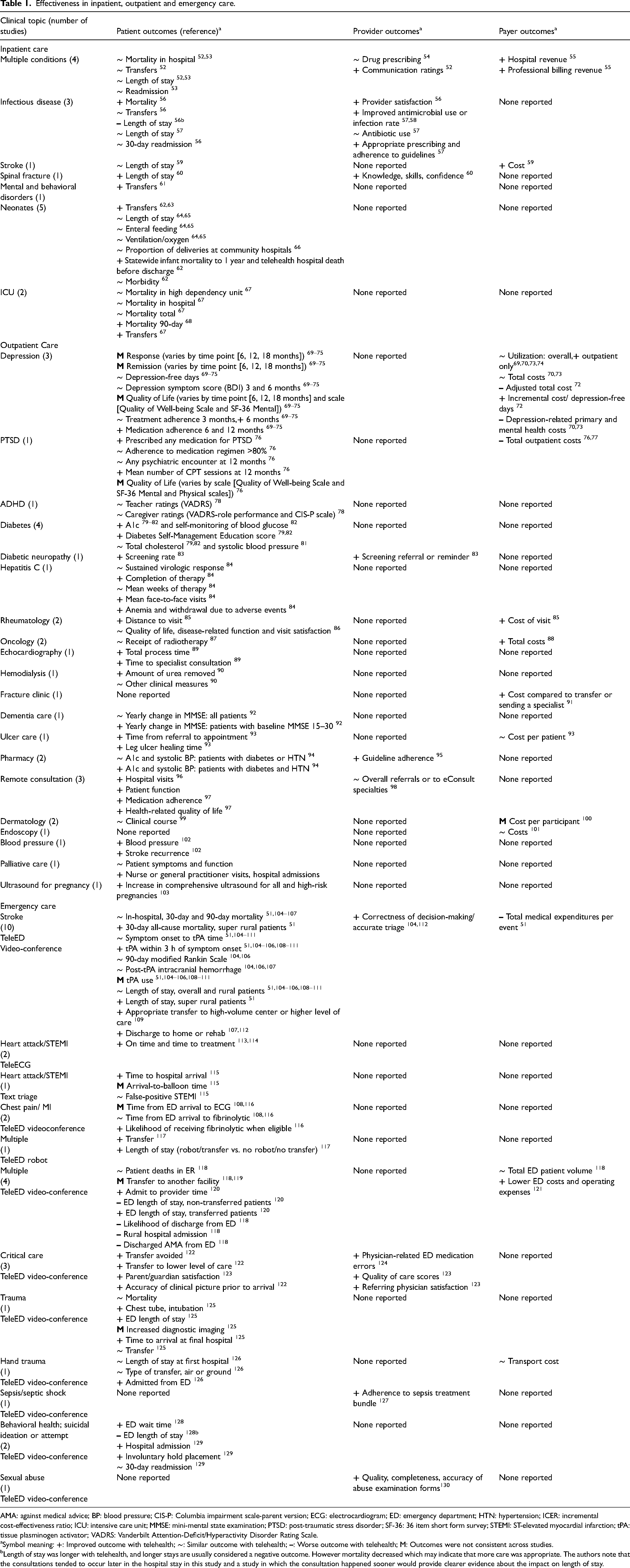
AMA: against medical advice; BP: blood pressure; CIS-P: Columbia impairment scale-parent version; ECG: electrocardiogram; ED: emergency department; HTN: hypertension; ICER: incremental cost-effectiveness ratio; ICU: intensive care unit; MMSE: mini-mental state examination; PTSD: post-traumatic stress disorder; SF-36: 36 item short form survey; STEMI: ST-elevated myocardial infarction; tPA: tissue plasminogen activator; VADRS: Vanderbilt Attention-Deficit/Hyperactivity Disorder Rating Scale.
Symbol meaning:
Length of stay was longer with telehealth, and longer stays are usually considered a negative outcome. However mortality decreased which may indicate that more care was appropriate. The authors note that the consultations tended to occur later in the hospital stay in this study and a study in which the consultation happened sooner would provide clearer evidence about the impact on length of stay.
Effectiveness for inpatient care
Seventeen studies, reported in 18 articles, evaluated RPPT for inpatient care (Table 1). The majority (n = 12) were studies that compared outcomes before and after the implementation of telehealth,52,54–62,66,67 two (reported in three articles) were prospective cohort studies,63,64,131 and three were retrospective cohort studies.53,65,68 Eleven were conducted in the United States,52,53,55,56,58,62–66,68 five in Australia,54,57,60,61,67 and one in Scotland. 59 Risk of bias was rated as high for six studies52,57,58,60,62,66 and medium for 11 studies.53–56,59,61,63–65,67,68
Summary of evidence of effectiveness for provider education and mentoring.

A1c: glycated hemoglobin; CBT: cognitive-behavioral therapy; ECHO: Extension for Community Healthcare Outcomes.44,154
Symbol meaning:
Summarizing these studies, we found evidence that telehealth consultations in rural hospitals resulted in no difference in length of hospital stay (six studies; low SOE) or transfers (three studies; low SOE) compared with usual care, which was in-person or phone consultations. Additionally, telehealth-supported care for neonates at rural hospitals resulted in no difference in clinical outcomes when compared to transfer and care at a hospital with a Level 4 neonatal intensive care unit (two studies; Low SOE). Also, mortality rates were not different when patients were treated in rural hospitals with remote ICUs rather than transported to more distant hospitals (two studies; low SOE).
Effectiveness for outpatient care
Thirty-two studies (in 35 publications) evaluated the use of RPPT interventions to support outpatient care.69–74,76–82,84–103,155 Sixteen studies (reported in 17 publications) were randomized controlled trials (RCTs),69–73,76–79,81,86,89,95,98–100,102 six were prospective cohort studies,74,83,85,92,96,155 six were retrospective cohort studies,80,84,87,88,91,101 four were pre-post study designs (same group measured before and after implementation),82,90,94,97 and two were before-after studies (different groups/systems measured before and after implementation).93,103 Eleven of the studies were conducted in the United States;69–71,76,78,82,84,85,94,95,99,100,103 the remainder were conducted in Canada,86,90,98 Australia,83,88,91,155 Korea,79,92,97 China, 102 Denmark, 80 New Zealand, 87 Spain, 96 United Kingdom,93,101 Chile, 74 Taiwan, 81 and Sweden. 89 Risk of bias was rated as high for eight studies,80,82,89,91,93,97,101,103 medium for 21 studies,71,74,76,78,79,81,83–88,90,92,94–96,99,100,155,156 and low for two studies.98,102
RPPT for outpatient care varied widely across interventions and outcomes (Table 1). Outpatient telehealth consultations with specialists in various clinical specialties (e.g. diabetes, depression, outpatient medication management) resulted in improvements in clinical outcomes compared with care without specialist involvement. For patients with diabetes, RPPT resulted in improvements in A1c and self-management in patients with diabetes but had no effect on blood pressure or cholesterol levels (4 studies; Low SOE). In patients with depression, RPPT was associated with some improvement in treatment response, medication adherence, and satisfaction (three studies; Low SOE), and higher utilization and corresponding costs for outpatient consultations due to increased access, resulting in overall benefit based on cost-effectiveness analyses (two studies; Low SOE). Studies found that outpatient telepharmacy consultations improved guideline adherence and patient outcomes but only for patients with both diabetes and hypertension (two studies; Low SOE).
Effectiveness for emergency care
We included 28 studies of RPPT for emergency care, either by emergency medical services (EMS) or EDs.51,104–130 Two of these studies were RCTs,104,112 10 were prospective cohort studies,106,108,110,111,113–117,127 eight were retrospective cohort studies,51,121–125,129,130 and eight were studies that compared outcomes before and after telehealth initiation.105,107,109,118–120,126,128 Eighteen of these studies were conducted in the United States,51,104,108,111,116,118–130 two were conducted in Italy,113,114 two in Australia,105,107 and one each in Canada, 117 Spain, 110 Finland, 106 Turkey, 115 and Japan. 109 Risk of bias was rated as high for one study, 108 medium for 26 studies,51,105–107,109–130 and low for one RCT. 104
Telehealth consultations for emergency assessment and initial care of stroke, heart attack, or chest pain at rural hospitals resulted in similar rates of mortality (five studies; Low SOE) and similar time to treatment when patients were treated locally as opposed to transferred, suggesting telehealth did not cause delays (eight studies; Low SOE). Telehealth consultations by specialists for critical care and trauma patients in rural EDs had a generally positive impact on transfers, with results reporting fewer unnecessary transfers, more appropriate transfers, or similar rates compared with care without RPPT (five studies; Low SOE).
Effectiveness for education and mentoring
Twenty-three studies evaluated RPPT for education and mentoring (Table 2),132–153,157 including three RCTs144,151,153 and 20 observational studies. Observational study designs included pre-post,132–136,138,139,141–143,146,148,150,152 prospective cohort,137,149 and retrospective cohort studies.140,156 All studies were conducted across multiple clinical sites or health care organizations. Sixteen studies were performed in the United States,132,134–141,143–146,149,150,157 four in Australia,147,148,151,152 one in Canada, 142 and one in Vietnam. 153 One study was rated low risk of bias, 142 11 were rated medium risk of bias,132,133,137,138,140,144,145,149,151,153,157 and 11 were rated high risk of bias.134–136,139,141,143,146–148,150,152
Extension for Community Healthcare Outcomes (ECHO) programs is a model that uses video for remote instruction and case-based learning and was designed to promote best practices in rural healthcare158,159 and has been use to expand access to specialty care. 160 ECHO programs were associated with better or equivalent patient outcomes (two studies; Low SOE), including reduction in A1c in patients of trainees after ECHO compared with before participation (one study), and similar Hepatitis C viral response and serious adverse events rates at the “spoke” site with ECHO participation to those at an academic medical center (one study). ECHO and non-ECHO video training programs (a) resulted in desired changes in provider behavior (e.g. increased appropriate prescribing practices, screening, and patient counseling) (eight studies; Low SOE) and (b) were associated with increased provider confidence, efficacy, and scores on knowledge tests (ten studies; Low SOE).
Implementation of provider-to-provider telehealth for rural patients
Sixty-seven studies in 71 publications addressed the implementation of RPPT.49,59,118,161–227 Most of these studies were program evaluations that combined data from several sources, such as site visits, observations, surveys, and interviews. A smaller number were qualitative research studies that analyzed interviews, focus groups, or documents and then categorized or cataloged specific barriers and facilitators to initial implementation, ongoing operations, longer-term sustainment, or spread of the use of telehealth.
Information on the barriers and facilitators was first recorded as described by the study authors. Then, because studies used different terms, we mapped these using CFIR constructs (mapping tables are provided in the full report) to standardize our description and allow us to summarize and compare these across studies. Identified barriers and facilitators mapped to 19 of the 39 constructs that make up CFIR.
Figure 2 illustrates how the 219 facilitators and 192 barriers we identified map to the CFIR constructs. Each construct and the number of times it occurred in our data is included in the label, and the width of the line represents the relative frequency. Short definitions of each construct are included as notes to the figure; comprehensive descriptions are available in CFIR articles and tools.44,154 The figure illustrates the point that most constructs can either facilitate or impede implementation, depending both on context and their presence or absence. The two most frequently cited constructs in our analysis were Available Resources and Access to Knowledge & Information. Available Resources is a broad concept including the wide range of investments an organization dedicates to implementing or sustaining an innovation or program. This was cited 60 times, and almost equally split between mention as a facilitator and as a barrier. Access to Knowledge & Information was cited 57 times, most frequently, though not exclusively, as a facilitator. We repeated the analysis by health care setting (available in the full report). Barriers and facilitators were similar across different settings. Assessed and summarized using CFIR constructs, the included implementation studies and program evaluations provided information about what is needed to translate knowledge about effectiveness of RPPT into actual practice.
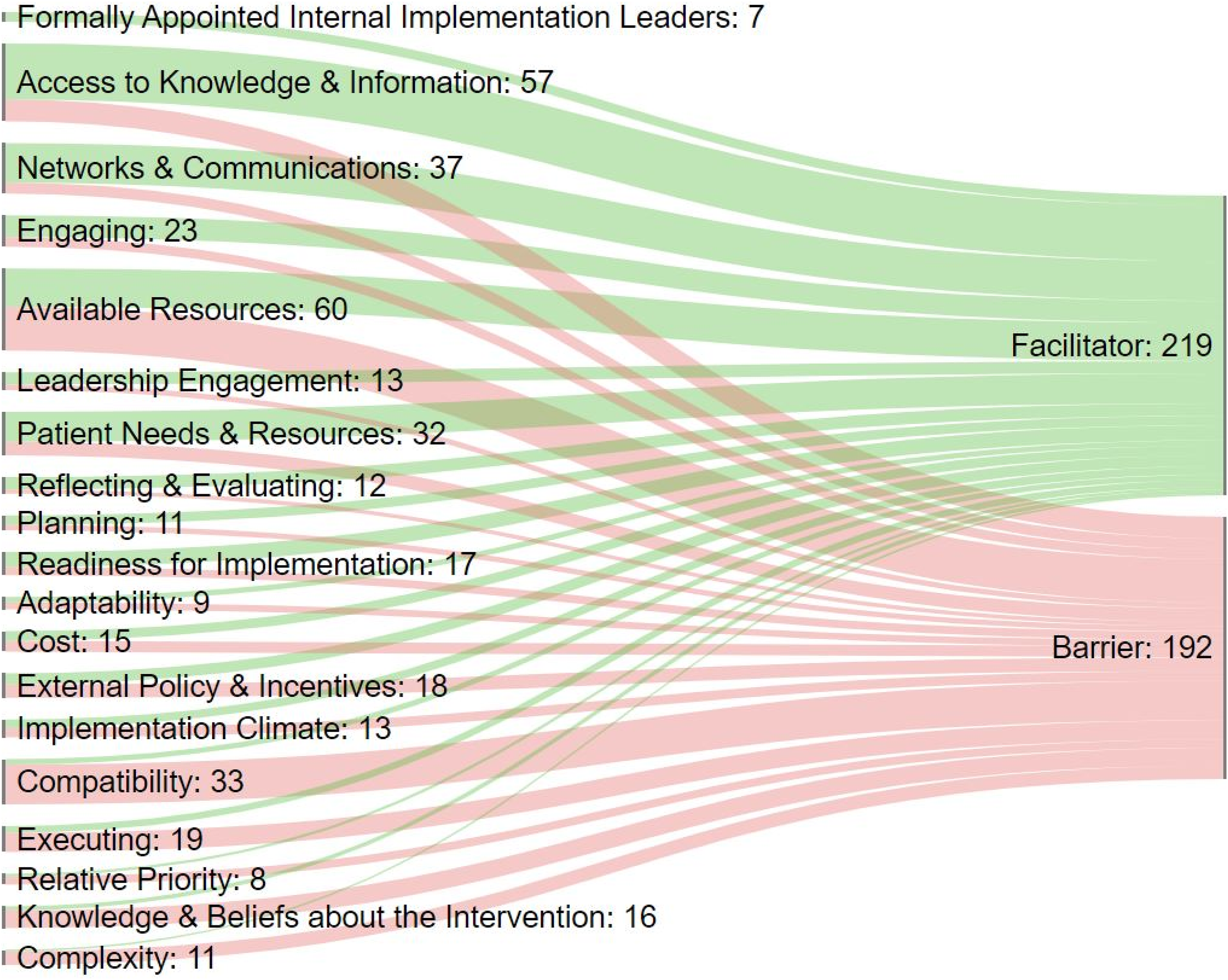
Facilitators and barriers to implementation*.
Many facilitators and barriers were not specific to rural settings. The studies highlight that telehealth needs to alleviate burden on providers, the technology needs to work, staff resources and reimbursement need to be allocated to provide both start-up infrastructure and ongoing support, and training of both support staff and clinicians is needed. Engagement from a range of stakeholders from patients to health system leadership and governments was cited in studies as essential. Some issues were raised in identified studies that are specific to rural programs. In the United States, insufficient internet in rural areas remains a persistent barrier. Lack of understanding of the rural environment by urban-based consultants and educators risks reducing the utility of teleconsultations and remote training programs. Remote consultants for the care of rural patients can be used for either frequent events or serve as a resource for rare events in rural healthcare, but needed technology and procedures are different in these two cases and technology and operations needed to be tailored to frequency of use. Most RPPT programs are local or in a single health system, yet sustainment requires long-term commitment and resources, on a scale that may not be feasible for smaller rural organizations.
Methodologic challenges in studying provider-to-provider telehealth for rural patients
We abstracted limitations cited by the authors and combined these with our risk of bias and applicability assessments to identify and categorize the methodological weaknesses of the available evidence. Studies did not routinely control for confounders related to patients, providers, facilities, and differences in telehealth implementation across study sites, and have been hampered by data limitations, such as missing data or inaccuracies in data collected for reasons other than research. In our assessment, it is often difficult to attribute impact to telehealth because less rigorous study designs, rather than RCTs or prospective, well-designed observational studies, were used in more than two-thirds of the included studies (Table 3).
Study designs and number of sites by setting for effectiveness studies.

ED: emergency department; EMS: emergency medical services; RCT: randomized controlled trial.
Same patients or providers evaluated pre and post-intervention.
Do not have the same patients or providers in both time points.
Discussion
The research on RPPT for collaboration in the delivery of rural health care provided an evidence base that addressed questions about use, effectiveness, implementation, and methodological weaknesses, but the evidence was uneven and could not support definitive, universally applicable conclusions.
To answer Key Question 1, we identified seven studies that reported national or regional trends of increasing telehealth use, with differences across specialties and geographic locations. Increasing use was evident even before the onset of the COVID-19 pandemic. For effectiveness (Key Question 2), we identified and synthesized studies that assessed the impact of RPPT and found evidence that outcomes were better or similar with RPPT for applications in inpatient, outpatient, and emergency care. We also summarized studies demonstrating that telehealth for rural provider education and mentoring, including ECHO programs, may improve patient outcomes, change provider behavior, and increase provider knowledge and confidence in treating specific conditions. To address Key Question 3, we categorized barriers and facilitators, finding most were common to practice change initiatives, though some were specific to the rural context. We identified important methodological weaknesses (Key Question 4) in the RPPT effectiveness studies, including less rigorous study designs and small sample sizes.
Our synthesis of the available evidence was qualitative because the modes, functions, and outcomes studied and how they are measured were heterogeneous. We grouped studies of similar topics that assessed similar outcomes allowing these conclusions. However, there were several instances where only one study was identified, or two or more studies reported conflicting results leading to an assessment of “unclear effect” or “insufficient evidence.” Classification of barriers and facilitators to implementation in 67 studies confirmed that common barriers for change in practice, including inadequate provider time, technology, and other resources, are limiting the spread of RPPT. However, there are also specific barriers, such as incomplete understanding of rural context and lack of long-term commitments to maintaining the infrastructure and staffing needed. Our assessment of the methodological weaknesses of the effectiveness studies found these frequently employed less rigorous designs, had small sample sizes, and often did not minimize possible bias through design or analytic approach.
Our review had limitations associated with our methods. We only included studies published in English about research conducted in developed countries. Searching for studies of RPPT was challenging as telehealth is a broad term and studies do not consistently include “rural” in titles or abstracts. We were unable to conduct quantitative synthesis (i.e. meta-analyses) and our qualitative synthesis combined studies with similar, but not identical, outcomes.
There were also limitations due to the nature of the evidence base. Research on telehealth in general is often not based on a clear model of how telehealth is expected to affect outcomes and whether telehealth needs to produce outcomes that are better than standard care or if equivalence with in-person alternatives is sufficient. It is often unclear if the goal of telehealth is to provide care that is as good as care provided without telehealth or if the investment in telehealth requires outcomes to be better. While telehealth should increase patient and provider satisfaction, there is no agreement on how to prioritize clinical outcomes, resource use, costs, and potential harms.
Our assessment of the methodological weaknesses of the included studies suggests directions for future research. Additional research is needed to measure outcomes at multiple time points or over longer periods of time, as short-term outcomes may differ from longer-term outcomes (e.g. provider retention of knowledge acquired through remote education and mentoring). Additional trials would strengthen the evidence base, as would observational studies that include contemporaneous comparison groups and multiple sites. The former would allow the impact of telehealth to be separated from historical change or the potentially unique characteristics of specific sites or providers/consultants. More complete descriptions are needed of both telehealth interventions and comparators. Clear statements of the intended impact of telehealth would help inform assessments of fit and help clarify whether telehealth was designed to replace in-person services or add additional services.
The existing evidence base was insufficient to allow us to unequivocally endorse all potential uses of RPPT as tools for improving health care for rural patients. Nevertheless, the studies we identified and summarized do not report harm or negative consequences. More importantly, they suggest it is likely that the application of telehealth can improve patient outcomes such as access to and quality of care, provider outcomes such as knowledge and self-efficacy, and payer outcomes such as reduced costs or maintenance of payments to rural providers.
Supplemental Material
sj-docx-1-jtt-10.1177_1357633X221139892 - Supplemental material for Telehealth-guided provider-to-provider communication to improve rural health: A systematic review
Supplemental material, sj-docx-1-jtt-10.1177_1357633X221139892 for Telehealth-guided provider-to-provider communication to improve rural health: A systematic review by Annette M Totten, Dana M Womack, Jessica C Griffin, Marian S McDonagh, Cynthia Davis-O'Reilly, Ian Blazina, Sara Grusing, and Nancy Elder in Journal of Telemedicine and Telecare
Footnotes
Acknowledgements
AHRQ staff, an NIH ODP working group, an NIH-convened content area expert group, and a technical expert panel helped refine the project scope. The draft report was presented at an NIH ODP P2P workshop. Experts in the field, AHRQ and NIH partners, and the public reviewed earlier drafts of the full report. The investigators are solely responsible for the contents of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This project was funded by the National Institutes of Health (NIH) through an interagency agreement with the Agency for Healthcare Research and Quality (AHRQ), U.S. Department of Health and Human Services (HHS) under Contract No. 75Q80120D00006/Task Order 75Q80120F32001. The findings and conclusions in this document are those of the authors, who are responsible for its contents; the findings and conclusions do not necessarily represent the views of AHRQ or NIH. Therefore, no statement in this report should be construed as an official position of AHRQ, NIH, or HHS.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
