Abstract
Objectives
This review aimed to present the clinical and health-care outcomes for patients with congenital heart disease (CHD) who use home monitoring technologies.
Methods
Five databases were systematically searched from inception to November 2020 for quantitative studies in this area. Data were extracted using a pre-formatted data-collection table which included information on participants, interventions, outcome measures and results. Risk of bias was determined using the Cochrane Risk of Bias 2 tool for randomised controlled trials (RCTs), the Newcastle–Ottawa Quality Assessment Scale for cohort studies and the Institute of Health Economics quality appraisal checklist for case-series studies.
Conclusion
The use of home monitoring programmes may be beneficial in reducing mortality, enabling earlier and more timely detection and treatment of CHD complication. However, currently, this evidence is limited due to weakness in study designs.
Keywords
Introduction
Congenital heart disease (CHD) 1 is an umbrella term for a broad range of birth defects that affect the function of the heart. Approximately 1% of babies are born with a heart of circulatory condition, which is usually a form of congenital heart disease. The mean prevalence of CHD globally was reported to be 8.224 (SD = 7.817–8.641) per 1000 for the years 1970–2017. 2 It was estimated that in 2017, there were 11,998,283 people living with CHD globally. 3
Many patients with CHD will require lifelong follow-up in order to monitor for signs of deterioration. In light of this, novel strategies for managing these patients may be useful in terms of reducing the burden of repeated health-care attendances, but also by empowering patients to be proactive in the management of their condition.
Telehealth may be a useful tool in the management of cardiac patients. According to the World Health Organization, telehealth is ‘the use of telecommunications and virtual technology to deliver health care outside of traditional health-care facilities’. 4 A systematic review of patients with heart failure reported that telehealth reduced all-cause mortality by 20% (95% confidence interval (CI) 0.68–0.94) and reduced heart failure hospitalisation by 29% (95% CI 0.60–0.83) compared to usual care. It would therefore be of interest to determine any health benefits afforded to patients with CHD through the use of home monitoring, 5 whereby patients would monitor various physiological parameters at home, with the data obtained being reviewed by health-care staff. There has been one paper published to date 6 which has reviewed the use of telehealth for monitoring this patient population. However, this paper was not a systematic review and did not undertake quality appraisal of the included papers. Additionally, this review was published in 2018 and, as such, only included papers published up to 2017, with only the PubMed database searched. This current review will therefore provide a rigorous examination of the literature in this area.
The aim of this systematic review was to provide an overview of the literature on home monitoring technologies used by patients with CHD. The objectives were (a) to present clinical and health-care outcomes for patients who use home monitoring technologies, and (b) to identify areas for research on the use of home monitoring technologies by patients with CHD.
Methods
Search strategy
A computerised search of the following databases from inception to November 2020 was performed: Medline, Embase, Cochrane, CINAHL and Scopus.
Search terms comprising Medical Subject Headings (MeSH), database-specific subject headings and key words were employed: EXP TELEMEDICINE OR telehealth* OR tele-health* OR telemedicine* OR tele-medicine* OR telemonitor* OR tele-monitor* OR telemanagement* OR tele-management* OR teleconsult* OR tele-consult OR telecare* OR tele-care* OR telepharmacy* OR tele-pharmacy* OR telenurs* OR tele-nurs* OR ‘remote monitor*’ OR ‘remote consult*’ OR ‘remote care*’ OR ‘mobile care*’ OR ehealth* or e-health* OR mhealth* OR m-health* OR ‘home monitor*’ OR ‘self monitor*’ OR ‘self manage*’ OR ‘home manage*’ AND EXP HEART DEFECTS, CONGENITAL OR congenital heart disease* OR congenital heart malformation* OR adult congenital heart disease* OR ACHD* OR grown up congenital heart disease* OR GUCH OR (congenital adj3 heart) OR (congenital adj3 cardiac) OR (cardiac adj3 malformation).
Grey literature was sought from the British Library (ETHoS), opengrey.eu, greylit.org, BMJ Best Practice, US Food and Drug Administration (FDA), National Institutes of Health, National Institute for Health and Care Excellence (NICE), Kings Fund, Nuffield Trust and Google Scholar. Hand searching of reference lists of articles was also performed.
The lead review author (R.C.) screened all records identified for inclusion/exclusion criteria. All identified publications were read as either abstracts or full texts. Papers were included if they were full text investigating home monitoring interventions actively used by the patient (or their carer) and published in the English language. Papers were excluded if they were abstracts only, citations, letters, case studies, investigating interventions used for the diagnosis of CHD, technical studies of telehealth equipment and home monitoring that was passively done without any action by the patient. Following the initial screening process, a full text evaluation of 25 articles was performed to determine correlation with the inclusion criteria. A meeting was held between the four members (R.C., C.M.H., S.Mc.F. and J.C.) of the review team to discuss findings, until inclusion was agreed by consensus.
The lead author (R.C.) extracted the necessary data from each included study into a pre-formatted data-collection table.
Quality assessment and risk of bias
Quality assessment and risk of bias was determined for all studies in this review. A meeting was held with all members (R.C., C.M.H., S.Mc.F. and J.C.) of the review team to determine the quality scores and risk of bias for all studies. Agreement on quality scores and risk of bias was gained by consensus. The quality appraisal tools that were used were as follows: the Cochrane Risk of Bias 2 tool 7 for randomised controlled trials (RCTs), the Newcastle–Ottawa Quality Assessment Scale 8 for cohort studies and the Institute of Health Economics (IHE) quality appraisal checklist 9 for case-series studies.
Results
A total of 2025 articles were identified by various literature searches. Twenty-two articles were deemed appropriate for inclusion in this systematic review (Figure 1).
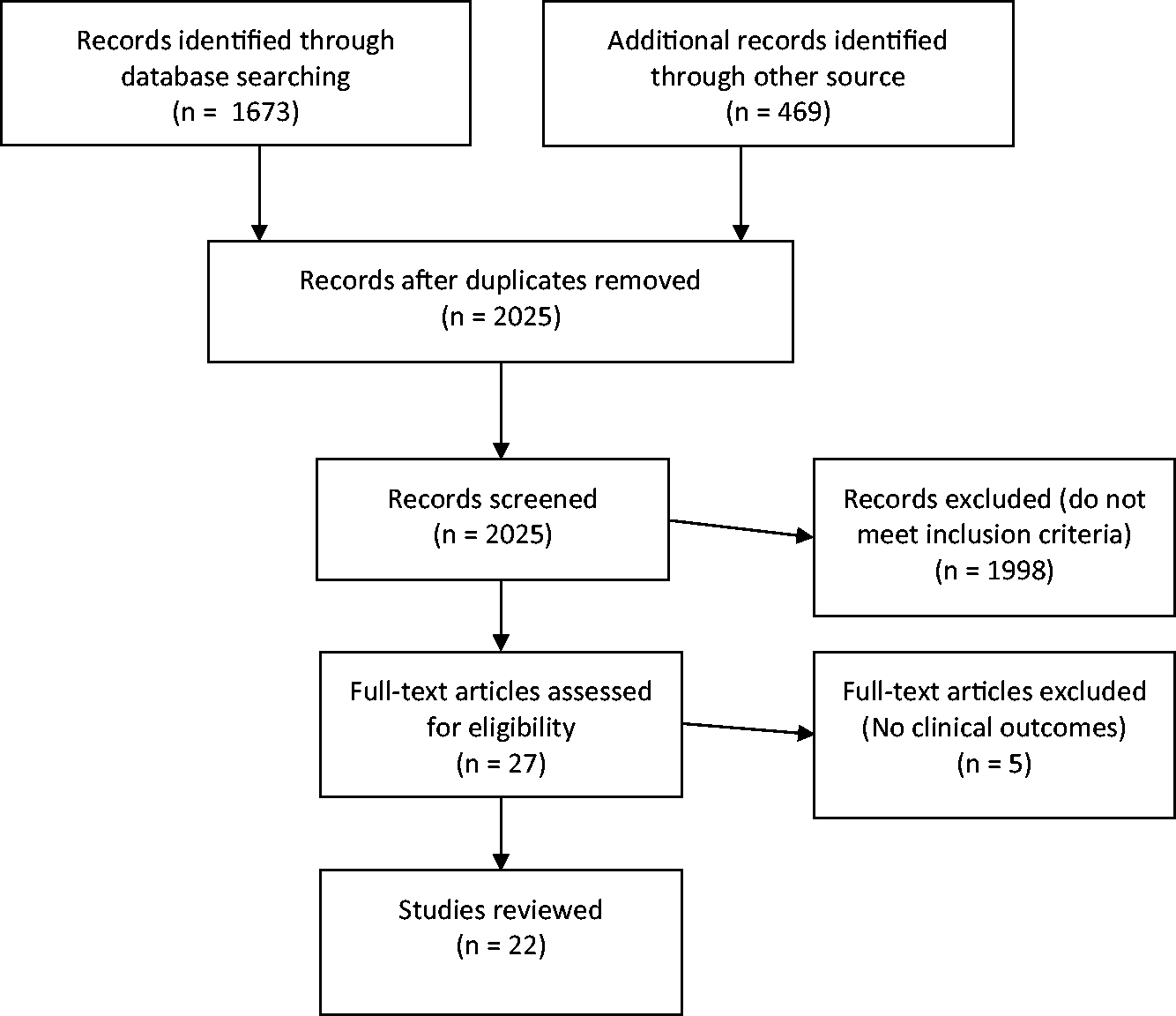
PRISMA flow diagram depicting the study selection process.
Study characteristics
The main study characteristics are presented in Table 1. These studies were published between 2001 and 2020. The main types of study were RCT, cohort and case series. In total, they included 2809 participants, with the number in individual studies ranging between 14 and 494 participants.
Summary of study characteristics.
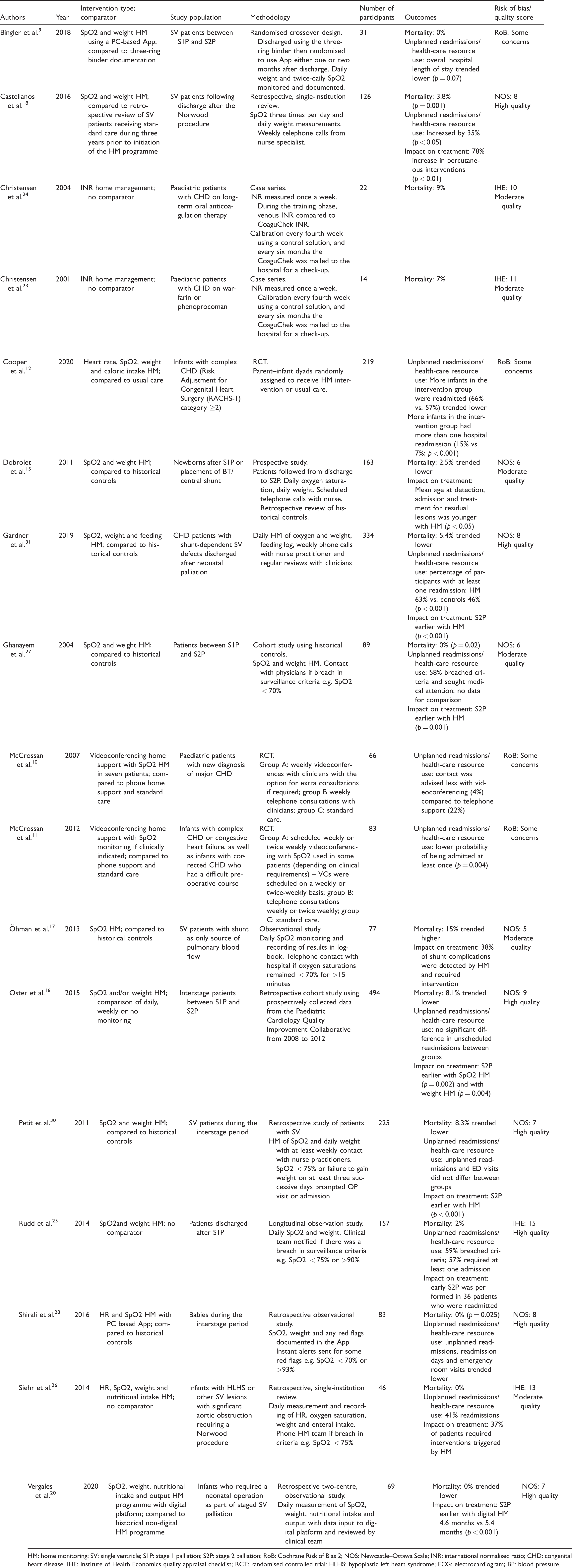
HM: home monitoring; SV: single ventricle; S1P: stage 1 palliation; S2P: stage 2 palliation; RoB: Cochrane Risk of Bias 2; NOS: Newcastle–Ottawa Scale; INR: international normalised ratio; CHD: congenital heart disease; IHE: Institute of Health Economics quality appraisal checklist; RCT: randomised controlled trial: HLHS: hypoplastic left heart syndrome; ECG: electrocardiogram; BP: blood pressure.
Risk of bias
The Cochrane Risk of Bias 2 tool was used for all RCTs (N = 4; Figure 2). All four studies9–12 were assessed as having some concerns, with all indicating a moderate risk of bias. The most common deficit in methodological quality was in the domains for randomisation and selection of the reported result. No study reported whether allocation sequence was concealed until participants were enrolled and assigned to the intervention. No study reported whether the data were analysed in accordance with a pre-specified analysis plan or whether the outcome assessors were blinded to the intervention.
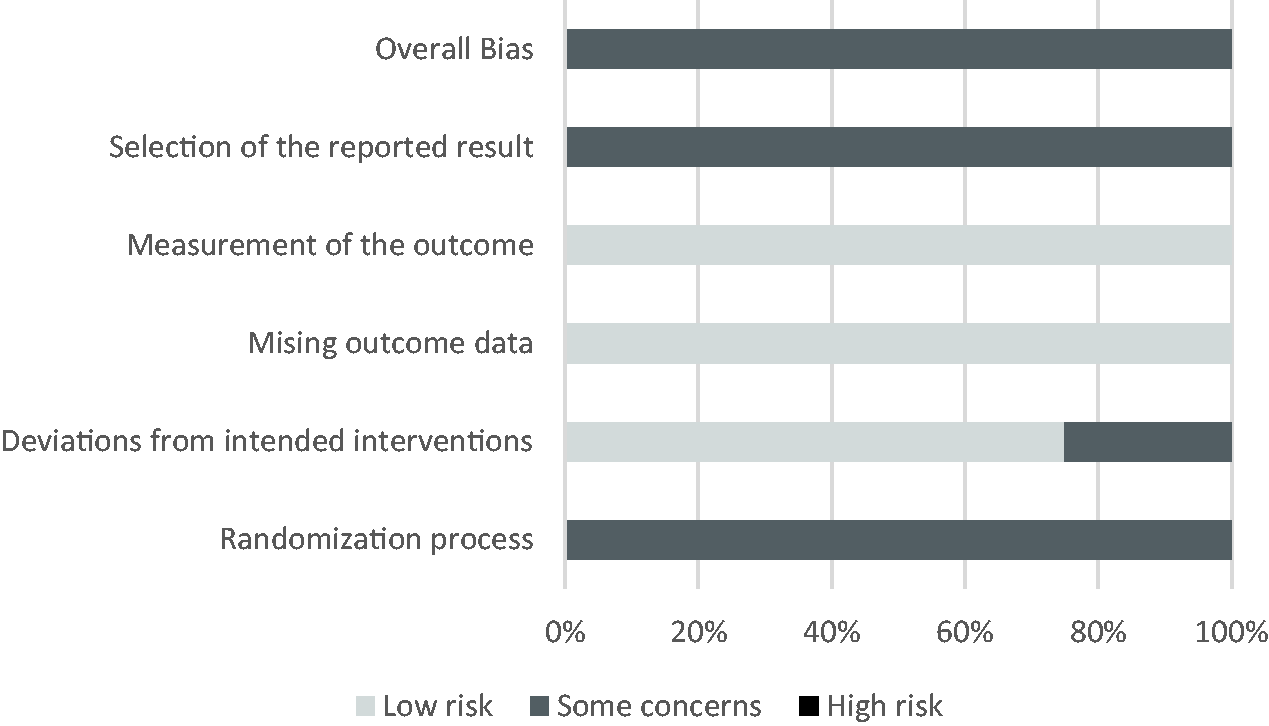
Risk of bias in studies.
Newcastle–Ottawa Quality Assessment Scale
The Newcastle–Ottawa Quality Assessment Scale was used for the quality appraisal of all cohort studies (N = 12; Figure 3). The most common shortfalls in methodological quality related to the domains of comparability and outcome. Five studies scored zero in the domain of comparability.13–17 These studies did not provide information on whether they controlled for factors that could affect the outcome measured. Only four studies 16 ,18–20 reported that the outcomes measured were controlled for multiple variables. One study 13 was rated as low quality for the outcome domain. This study did not provide details of how the outcome was assessed or whether all subjects were accounted for.
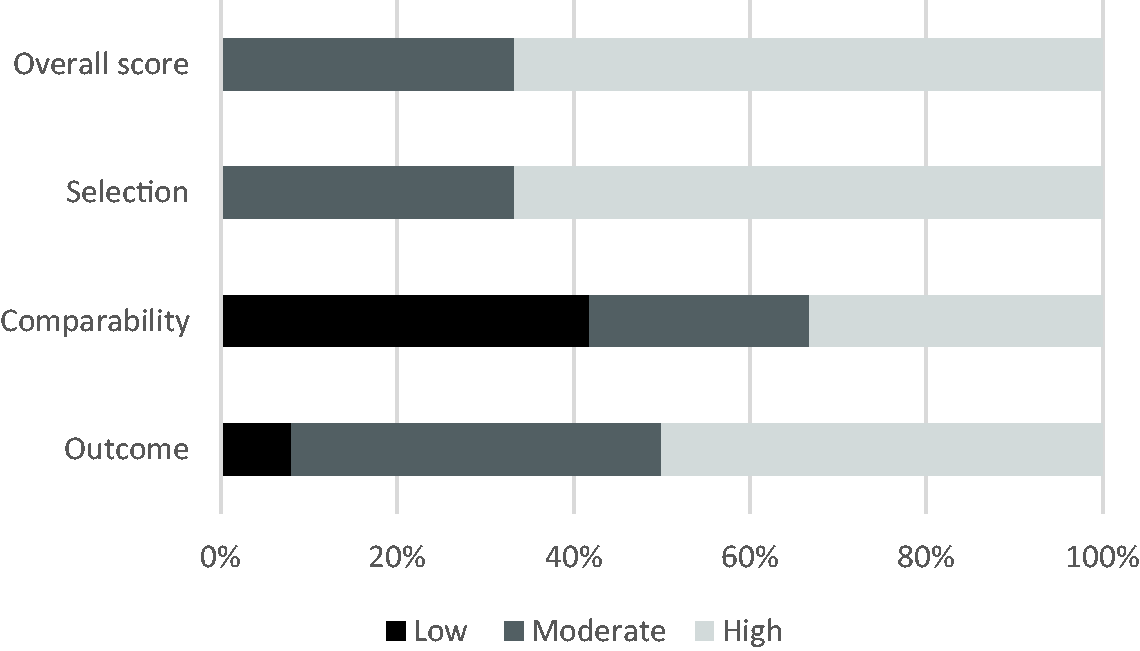
Newcastle–Ottawa Quality Assessment Scale.
IHE quality appraisal checklist
The IHE quality appraisal checklist for case-series studies was used to assess the quality of the remaining studies which used a case-series design methodology (N = 6; Figure 4). The most common deficiencies in methodological quality were for the domains of competing interests and sources of support, statistical analysis and study design. Two studies 21 , 22 provided details on competing interests and sources of support, with the remaining four studies23–26 not providing any information in relation to this. The domain for statistical analysis also identified three studies23–25 which were judged to have a low level of quality based on the IHE quality appraisal checklist.
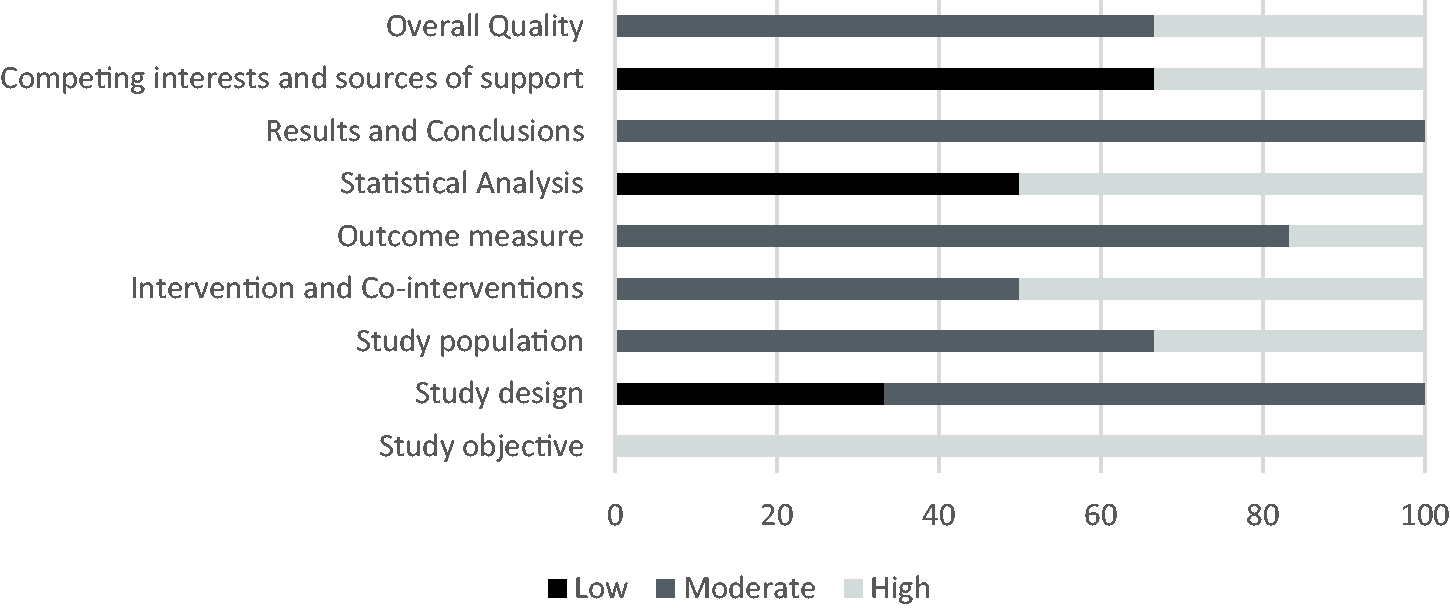
Institute of Health Economics quality appraisal checklist.
Overall, the risk of bias results indicate that the RCTs were all rated as having some concerns; cohort studies were rated as being of moderate to high quality, with 64% bring rated as high quality; and case series were all rated as moderate to high quality, with 67% of these studies receiving a rating of high quality. Whilst many of the case studies and cohort studies were judged to be high quality, it must be borne in mind that the studies themselves were less robust by virtue of their design methodology compared to the randomised cohort studies.
Synthesis of results
Thematic analysis of outcome measures was undertaken by the lead author (R.C.) who systematically examined the pre-formatted data-extraction table to identify outcome measures recorded by each of the papers. Themes were discussed and agreed with the co-authors. There were three common outcome measures: mortality, unplanned readmissions/health-care resource use and impact on treatment.
Mortality
There were 17 studies that included mortality as an outcome measure (Table 2). Five studies found that home monitoring programmes were associated with a significant improvement in mortality. 13 , 14 , 18 , 27 , 28 Mortality was reduced by between 10.2% and 17%. The study with the smallest reduction in mortality reported 12.4% mortality for historical controls compared to 2.2% for the study group. 29 The largest reduction in mortality reported 17% mortality for historical controls compared to 0% in the study group. 28
Summary of mortality rates.

Five studies found that mortality rates in the home monitoring group trended lower, with mortality reduced by between 3.8% and 7.6%. 15 , 16 , 20 , 30 , 31 The study with the biggest reduction in mortality reported a mortality rate for historical controls at 13% compared to 5.4% in the home monitoring group (p = 0.2). 31 The study with the smallest reduction in mortality reported that mortality was reduced from 12.1% to 8.3% (p = 0.924) with home monitoring. 30 While there was a trend for lower mortality in these studies, the results were not significant. One study reported 0% mortality in both groups. 9
There were four studies that reported mortality rates. However, there was no comparator.23–26 Mortality rates in these studies ranged from 0% to 9%.
Two studies found that mortality trended higher for the home monitoring groups. 17 , 19 One study reported that mortality in the home monitoring group was 15% versus 10% in the historical controls. 17 In the other study, mortality in the home monitoring group was 5.4% versus 2.4% in the historical controls (p = 0.71). 19
Unplanned readmissions/health-care resource use
Unplanned readmissions/health-care resource use was reported in 14 studies (Table 3). Four studies23–26 reported data for the home monitoring cohort, but there were no data for comparison. Three studies reported data on breaches in surveillance criteria (e.g. a SpO2 < 70%). 13 , 25 , 27 Between 31% and 59% of patients breached surveillance criteria, with up to 57% of patients requiring observation and being admitted to hospital. Readmissions were reported in two studies. 21 , 26 One study 26 reported that 41% of patients had 27 readmissions, and another study 32 reported that there were two (3.6%) presentations to ED and 1 (1.8%) hospitalisation.
Summary of unplanned readmissions/health-care resource use.

Summary of impact on treatment.

The remaining nine studies included data for a control group. There was a significant increase in unplanned readmissions, 12 , 18 , 31 with rates up to 35% higher. 18 However, the duration of the readmissions was shorter for the home monitoring group compared to controls, with a median of three days (interquartile range (IQR) 2–7 days) versus four days (IQR 2–10 days; p = 0.002). 31 Conversely unplanned readmissions, readmission days and emergency room visits trended lower with the use of the home monitoring with an App, but this did not reach statistical significance. 28
Two studies found no difference in rates of unplanned readmissions/health-care resource use. 16 , 30 There was no significant difference in unscheduled readmissions (any cause) between weekly versus daily monitoring, or between no monitoring versus daily monitoring. 16
Contact with health service professionals was advised 18% less often with videoconferencing compared to phone support (p < 0.01). 10 The probability of being admitted at least once to hospital was 37% lower with videoconferencing support compared to standard care (p = 0.004). 11 Home monitoring using the app was associated with significantly shorter unplanned length of stay in the intensive care unit (1 (IQR 1–2) vs. 6 (IQR 1–16); p = 0.03). 9
Impact on treatment
The impact on treatment was reported in 15 studies (Table 4). Seven studies reported that age at stage 2 palliation (S2P) was younger in the home monitoring group, 14 , 15 , 20 , 26 , 27 , 30 , 31 and age at S2P was 22 days earlier with home monitoring (p < 0.002). 31 S2P in the home monitoring groups was earlier (p = 0.016 and p = 0.001). 14 , 27 One study found that the age of S2P for the home monitoring group was 150 ± 52 days versus 120 ± 114 days for the historical controls (p < 0.001). 30 A study comparing weight and/or SpO2 monitoring against no monitoring reported that S2P was 26 days earlier for those with SpO2 monitoring versus no SpO2 monitoring (p = 0.002) and 21 days earlier for those with weight monitoring compared to no weight monitoring (p = 0.004). 16 It was also reported that S2P was carried out early in 36 patients (23% of participants) who were readmitted due to home monitoring events. 25
Five studies reported on interventions and treatments that were undertaken due to breaches in home monitoring criteria. 13 ,17–19, 26 Two studies included historical controls and found that there were more interventions/procedures undertaken in the home monitoring group, 18 , 19 with a 78% increase in percutaneous interventions (p < 0.01)18 and more major post-Norwood procedures (p = 0.02)19 in the home monitoring groups. Three studies did not have a comparator and reported that there were between 17% and 57% of patients who breached surveillance criteria and required intervention or diagnostic catheterisation. 13 , 17 , 26
One study found that the mean age at detection, admission and treatment of residual lesions after shunt placements was younger in the home monitoring group (p < 0.005). 15 It was reported in one study that in 25% of patients with a previous diagnosis of arrhythmia, a recurrence of the arrhythmia was confirmed and treatment initiated. 22 In the final study, it was reported that medication changes were made in 11% of patients based on home monitoring. 21
Discussion
The period between S1P and S2P is known to be a precarious time for CHD patients, with haemodynamic instability. 32 Just over half (56%) of the studies found a significant reduction or a trend for lower mortality with home monitoring.
Interstage mortality (i.e. mortality that occurs in patients between S1P and S2P) is reported in the literature to be between 11% and 19%.33–35 Reported mortality rates compare favourably to the literature, with 10 demonstrating lower mortality.13–16, 18 , 20 , 27 , 28 , 30 , 31 A 15% mortality was noted by one study, 17 while the remaining studies were <11% when home monitoring was used. This would imply that the use of home monitoring may have beneficial effects, which lead to a lower mortality in this patient population.
No minor or major clinical complications were reported for patients who were home monitoring their international normalised ration (INR) using a prothrombin time (PT)-INR point-of-care device, 23 , 24 which suggests that mortality was unrelated to the INR levels.
Studies that reported outcomes for mortality included RCT (n = 1), 9 cohort studies (n = 12)13–20,27,28,30,31 and case series (n = 4).23–26 Whilst 59% of studies reported either a significant reduction or a trend for lower mortality, these were all cohort studies, with 9/10 studies using historical controls. Furthermore, 3/10 studies were rated as moderate quality. The two studies that reported a trend for higher mortality also used historical controls which is problematic and introduces bias into the research. Medical and surgical treatments may have improved between the two time periods, data may be recorded differently and so on. Therefore, the evidence is weak for this outcome.
Between 1.8% and 63% of patients had unplanned readmissions. In the context of the literature available, these results compare favourably. A study in 2016 reported that 65.5% of patients had at least one unplanned readmission in the interstage period. 36 Similarly, a more recent study in 2018 found that 75% patients had unplanned readmissions. 37 In 62.5% of the studies that reported on unplanned readmissions, rates were lower than reported in the literature for interstage patients. 36 , 37 An important finding from this review is that no studies reported unplanned readmissions using home monitoring significantly more than expected from the wider literature.
Between 31% and 61% of patients required observation and/or evaluation as a result of data from home monitoring. Two studies 10 , 11 found that contact with health service professionals was advised less often. This suggests that home monitoring is a valuable resource for detecting clinical deterioration in this patient population.
The increases in unplanned readmissions and health-care resource use may be explained by the fact that subtle deteriorations in clinical status could be picked up more frequently due to the home monitoring protocols. These increases should be viewed alongside mortality rates. The two studies that found a significant increase in unplanned readmissions also reported either a significant reduction or a trend for lower mortality rates with the use of home monitoring.
Studies that reported data for this outcome included RCTs (n = 4),9–12 cohort studies (n = 7)13,16,18,27,28,30,31 and case series (n = 3). 21 , 25 , 26 Three of the four studies which reported lower unplanned readmissions/health-care resource use were RCTs, which were all judged to have some concerns regarding bias. The other study was a cohort study using historical controls and was rated as high quality. Two of the three studies that reported an increase in relation to this outcome were cohort studies, using historical controls, with one study being an RCT. The remaining studies reported either no differences between groups or had no controls. Based on the design methodologies and quality appraisal scores, there is a stronger evidence that home monitoring reduces unplanned readmissions/health-care resource use. However, the evidence remains limited for this outcome.
Of the 15 studies13–19, 20 – 22 ,25–27, 30 , 31 that reported on the impact that home monitoring had on treatment, 47% found that S2P was performed at a younger age in the home monitoring groups. 13 , 14 , 20 , 25 , 27 , 31 The timing of S2P varies among centres and is a clinically led decision. Literature on the optimal timing of S2P reports that timing differs depending on whether patients are low, intermediate or high risk. A study in 2018 reported S2P being performed after three months of age was associated with maximal 2 year survival in low/intermediate risk infants. 38 Additionally, another study reported that median age at S2P for the study cohort was 155 days (IQR 109–214 days). 39 The earlier timing of S2P may be a reflection of earlier detection of clinical deterioration or clinicians being providing with more data on the patient, enabling them to make a more informed decision on the timing of S2P.
In studies without a control group, the intervention rates were reported to be between 37% and 38% of patients. The rates of re-interventions in these studies compare favourably to the literature, with a retrospective analysis of 1157 interstage patients reporting that 50% of patients required reintervention. 40
Studies for this outcome included cohort studies (n = 11)13–20,27,30,31 and case series (n = 4), 21 – 22 ,25–26 with all but one being of high quality. There were 9/15 (60%) studies that found that either treatment was undertaken significantly earlier with home monitoring or significantly more interventions were undertaken in the home monitoring groups. 14 , 16 ,18–20, 25 , 27 , 30 , 31 Eight of these were cohort studies using historical controls. 14 , 16 ,18–20, 27 , 30 , 31 Of the remaining studies with either no data for comparison or no control group, only one was rated as high quality, and this was a case series design, with the rest rated as moderate quality. However, whilst most studies reported an either earlier treatment or increased interventions, given that these were cohort studies which primarily used historical controls, the methodological shortfalls in the studies mean that the evidence is limited for this outcome.
Strengths and limitations of the review
This review restricted the search to English language articles and excluded passive forms of home monitoring, which may have caused some relevant research to be excluded. Additionally, this review only considered clinical and health-care outcomes, and as such, qualitative outcomes such as anxiety levels, patient perceptions and so on have not been considered.
There have been no systematic reviews published on this topic to date, and this review systematically examined the full scope of the literature on home monitoring by patients with CHD. As such, this review provides a comprehensive review and interpretation of the data available.
Areas for further research
This systematic review included patients with CHD at any age who were undergoing home monitoring of their CHD. Only 2/20 (9.1%) of these studies were undertaken in adults with CHD. Improvements in survival rates of patients with CHD have led to increasing numbers of children and adults living with complex CHD. Approximately 90% of babies born with cardiovascular abnormalities are expected to reach adulthood, 41 and as surgical techniques continue to improve, this could increase further in the future. In light of this, further research should focus on home monitoring interventions for adults with CHD, as home monitoring of this patient population has the potential to relieve pressure in the health service.
Conclusion
This systematic review examining the clinical and health-care outcomes for CHD patients when using home monitoring programmes identified that home monitoring may be beneficial in reducing mortality, enabling earlier and more timely detection and treatment of CHD complications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
