Abstract
Background:
Serum neurofilament light (sNfL) chain levels, a sensitive measure of disease activity in multiple sclerosis (MS), are increasingly considered for individual therapy optimization yet without consensus on their use for clinical application.
Objective:
We here propose treatment decision algorithms incorporating sNfL levels to adapt disease-modifying therapies (DMTs).
Methods:
We conducted a modified Delphi study to reach consensus on algorithms using sNfL within typical clinical scenarios. sNfL levels were defined as “high” (>90th percentile) vs “normal” (<80th percentile), based on normative values of control persons. In three rounds, 10 international and 18 Swiss MS experts, and 3 patient consultants rated their agreement on treatment algorithms. Consensus thresholds were defined as moderate (50%–79%), broad (80%–94%), strong (≥95%), and full (100%).
Results:
The Delphi provided 9 escalation algorithms (e.g. initiating treatment based on high sNfL), 11 horizontal switch (e.g. switching natalizumab to another high-efficacy DMT based on high sNfL), and 3 de-escalation (e.g. stopping DMT or extending intervals in B-cell depleting therapies).
Conclusion:
The consensus reached on typical clinical scenarios provides the basis for using sNfL to inform treatment decisions in a randomized pragmatic trial, an important step to gather robust evidence for using sNfL to inform personalized treatment decisions in clinical practice.
Keywords
Introduction
Therapeutic strategies in multiple sclerosis (MS) aim to reduce the relapse rate and to slow disability accumulation. There are more than 20 disease-modifying therapies (DMTs) available, including high-efficacy drugs like natalizumab, 1 alemtuzumab, 2 and B-cell depleting therapies (BCDT: rituximab, ocrelizumab, ofatumumab, and ublituximab),3–5 which provide close to complete suppression of acute inflammatory disease activity. However, such DMTs interfere with physiological immune response functions putting persons with (pw) MS at risk of infections. 6
Several studies have demonstrated the benefits of early therapy initiation or early escalation to high-efficacy DMTs (HET) in relapsing-remitting (RR) MS (“hit hard, hit early” paradigm).6,7 However, controversies persist regarding the pursue of such aggressive strategy due to potential longer-term side effects. 8
Recent smaller studies have also shown that de-escalation could be a safe option in RRMS, decreasing the exposure to MS therapies without recurrence of disease activity.9–11 Conversely, the observation that reduced drug exposure negatively impacts disease progression12,13 suggests that a similar negative effect may occur when de-escalating the dose. Currently, there is no clear consensus on how or when to de-escalate, creating uncertainties and making shared-decision making between physician and patient challenging. Hence, a more personalized or precision medicine approach to MS therapy is urgently needed.14,15
A promising tool is the use of neurofilament light chain (NfL)16,17 as an additive criterion. NfL is a marker of neuroaxonal damage 16 that can be measured in a standardized way in blood making it suitable for use in clinical practice. Serum NfL (sNfL) has been demonstrated as a useful biomarker for both acute inflammatory disease activity and disability accumulation, and treatment response, in real-world settings18–21 and randomized controlled trials.22,23 However, the practical use and added value of this biomarker as part of individual therapeutic decision-making has not been evaluated so far. 24
As part of an ongoing pragmatic randomized trial assessing the added value of informing treatment decisions by 6-monthly sNfL monitoring on patient relevant outcomes (MultiSCRIPT trial—NCT06095271), 25 we conducted this Delphi study. The purpose was to reach a consensus among experts in the field closely involved with the trial and patient consultants on treatment decision algorithms on the inclusion of sNfL information to guide individual patient level treatment decisions for common clinical scenarios encountered in usual care.
Materials and methods
We conducted a modified Delphi study 26 in three rounds (two online and one in-person) over a period of 3 months (26 October 2022 till 27 January 2023). The Delphi methodology aims to gather experts’ opinions until a consensus is reached on a topic—here treatment decision algorithms—through an iterated, structured voting process. The multiple survey rounds allow participants to nuance and reconsider their decisions based on the anonymized votes and comments in the previous online rounds, followed by a moderated discussion during the final in-person round.26,27
Definition: NfL levels
Serum NfL levels are age- and body mass index (BMI)-dependent, thus their normalized values (i.e. adjusted sNfL percentiles or Z scores based on healthy controls) are more meaningful for the clinical interpretation in individual pwMS and to prognosticate future clinical disease activity20,28–30 than the absolute sNfL concentration. Percentiles/Z scores are interchangeable and reflect the deviation of a pwMS’s sNfL from the mean value of age- and BMI-matched healthy individuals (50th percentile; Z score 0). sNfL percentiles/Z scores identify a gradually increased risk of future acute inflammatory events (relapse, lesion formation) as well as worsening of disability. 20 We defined “high” sNfL as >90th percentile. 20 This is based on evidence indicating that pwMS with levels higher than the 90th percentile (Z score 1.28) have about a two-fold higher risk for any sign of clinical or disease activity on magnetic resonance imaging (MRI) in the following year (RR 2.28; 95% confidence interval (CI): 1.11–4.68; p = 0.025). 20 Before applying the treatment decision algorithms based on sNfL values, other possible causes of high sNfL need to be considered such as, for example, head trauma, stroke, relevant sports-related head injury, at least medium severe renal failure (glomerular filtration ratio (GFR) < 60 mL/min/1.73 m2), suboptimal treated diabetes mellitus or any other concomitant disease including incipient neurodegenerative diseases that may lead to relevant neuro-axonal damage.
Serum NfL values <80th percentile are values typically seen in healthy control subjects and were considered as “normal” sNfL in comparison with high sNfL.
Definition: no evidence of disease activity
No evidence of disease activity (NEDA) since last visit was defined as NEDA2 with no relapses and no Expanded Disability Status Scale (EDSS) worsening or as NEDA3 with additionally no MRI activity.
Definition: low, medium and HET
During round 1, participants were asked to classify all DMTs commonly used in Switzerland (i.e. glatiramer acetate, interferon-beta formulations, teriflunomide, dimethyl fumarate, S1P receptor modulators, cladribine, alemtuzumab, BCDT (rituximab, ocrelizumab, ofatumumab), natalizumab) into low, medium, and high efficacy. Based on round 1, the final classification was then constructed and voted on during the second round.
Algorithms development
The core team (ÖY, PB, CZ, AS, DL, TD, LK, LGH and JK) identified two main settings (1) escalation or horizontal treatment change in case high sNfL and (2) de-escalation in case sNfL <80th percentile. Depending on the currently used DMT (i.e. low, medium or high-efficacy), treatment decision algorithms were further grouped into topics (e.g. escalation from low to medium or HET).
Panel selection
Before the first round of the Delphi study, we invited local investigators (all attending MS neurologists) from the 8 Swiss MS Cohort (SMSC) centers taking part in the MultiSCRIPT trial, international senior MS experts, and patient consultants.
Consensus
For the two online surveys, participants were asked to rate their agreement with the different treatment decision algorithms using a 9-Likert-type scale (1 Strongly disagree to 9 Strongly agree). For the last in-person round, participants were asked to vote by show of hands “agree” or “disagree.” The following agreement thresholds were used: <50% excluded, 50%–79% moderate consensus, 80%–94% broad consensus, 95%–99% strong consensus, and 100% full agreement. More information on the voting rounds in Supplemental Material.
Results
Thirty-two participants were invited and accepted to participate, of which, 18 were experts from the SMSC centers (Basel, Bern, Zurich, Lugano, St Gallen, Aarau, Geneva and Lausanne), 11 international senior MS experts (Germany (n = 4), Spain (n = 3), and United States, Czech Republic, Sweden and Austria (n = 1 each)), and 3 patient consultants. All, except one, participated in both online rounds and 24 participants were present at the hybrid open discussion meeting (14 experts from the SMSC centers, 9 international experts and 1 patient consultant). Of note, all SMSC centers were represented in round 3. Figure 1 provides a detailed overview of the modified Delphi Study. Surveys for rounds 1 and 2 are available as Supplemental Material.
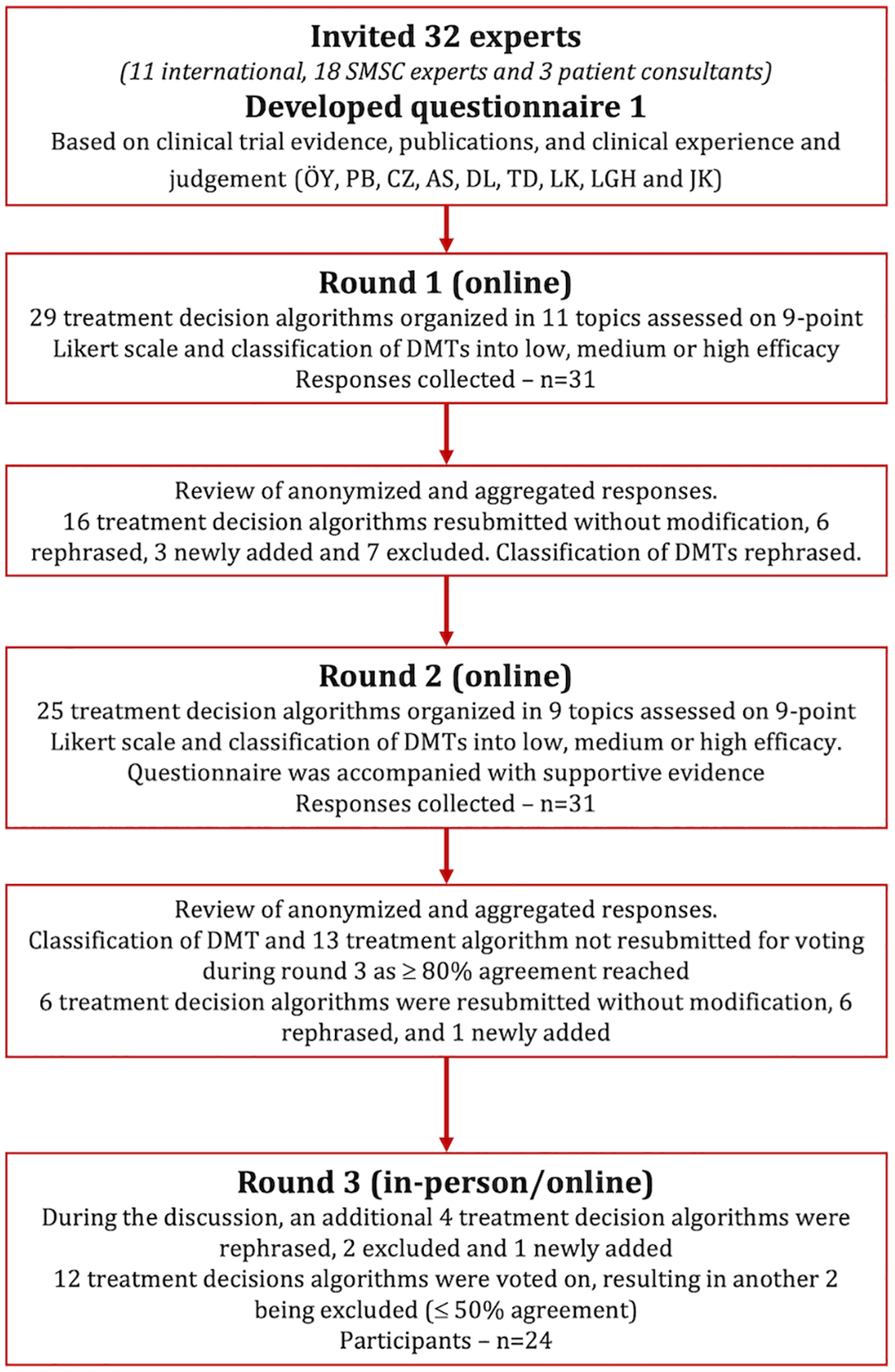
Flow chart of the Delphi process.
Classification of DMTs
A full agreement (100%) was reached for HET to include alemtuzumab, BCDT and natalizumab, a strong consensus (97%) for low efficacy DMTs to include glatiramer acetate, interferon beta and teriflunomide, and a broad consensus (83%) for medium efficacy DMTs to include dimethyl fumarate, S1P receptor modulators and cladribine.
High NfL: escalation algorithms
From untreated to initiating DMT
For pwMS with NEDA3 who are untreated but have high sNfL, we reached broad consensus (80% agreement) to initiate a DMT (Figure 2(a)); agreement was stronger when there was additionally MRI activity (93%–97%).
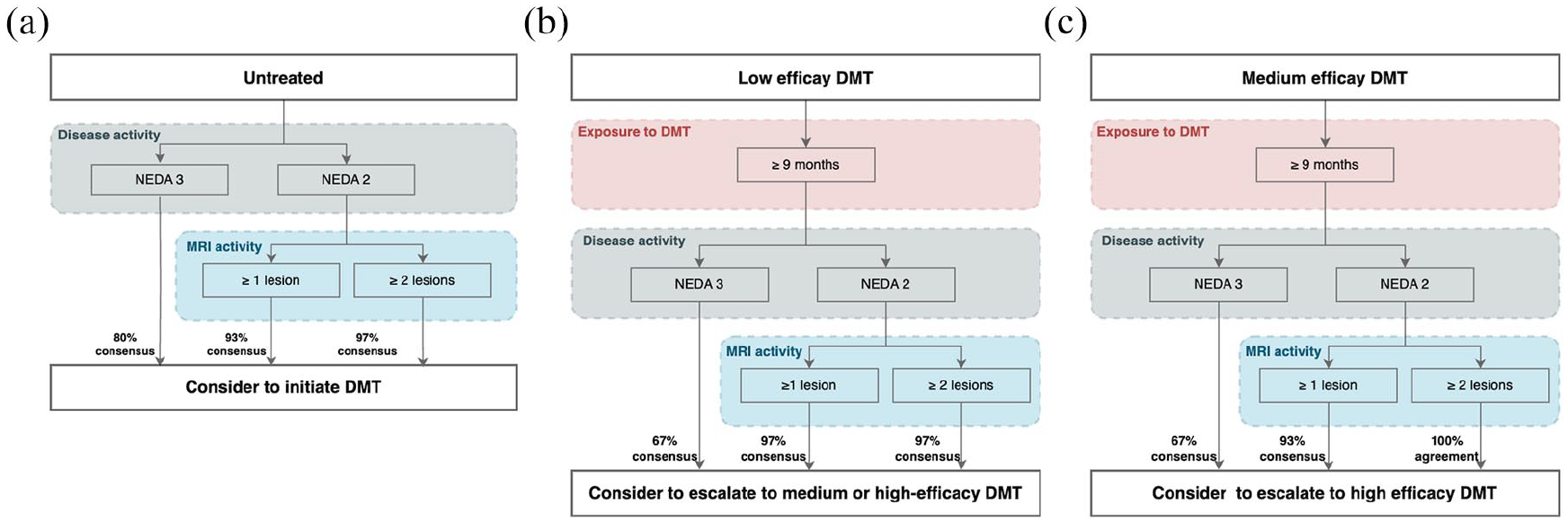
Escalation algorithms in patients with high sNfL (>90th percentile): (a) From untreated to initiating DMT; (b) From low to medium or high efficacy DMT; and (c) From medium to high efficacy DMT.
From low to medium or HET
For pwMS with NEDA3 and high sNfL, moderate consensus was reached (67%) to escalate from low efficacy DMT (Figure 2(b)) to a medium or HET. Experts emphasized that pwMS needed to be on the low efficacy DMT for at least 9 months to exclude high sNfL levels due to the DMT not yet having reached full efficacy; the agreement was stronger when pwMS also showed MRI activity (97%).
From medium to HET
In NEDA3 with high sNfL, moderate consensus was reached (67%) to escalate from a medium to HET (Figure 2(c)). Agreement was again stronger with MRI activity: broad consensus (93%) with one new/enlarging lesion and a full agreement (100%) with at least two new/enlarging lesions.
High NFL: horizontal switch algorithms
Switching from natalizumab
During round 1, consensus on treatment algorithms to shorten natalizumab (Figure 3(a)) intervals from 6 to 4 weeks were divided with 31% disagreeing, 38% undecided, and 31% agreeing to shorten treatment intervals for pwMS with NEDA3 and high sNfL. The algorithms were dropped (Supplemental Material).
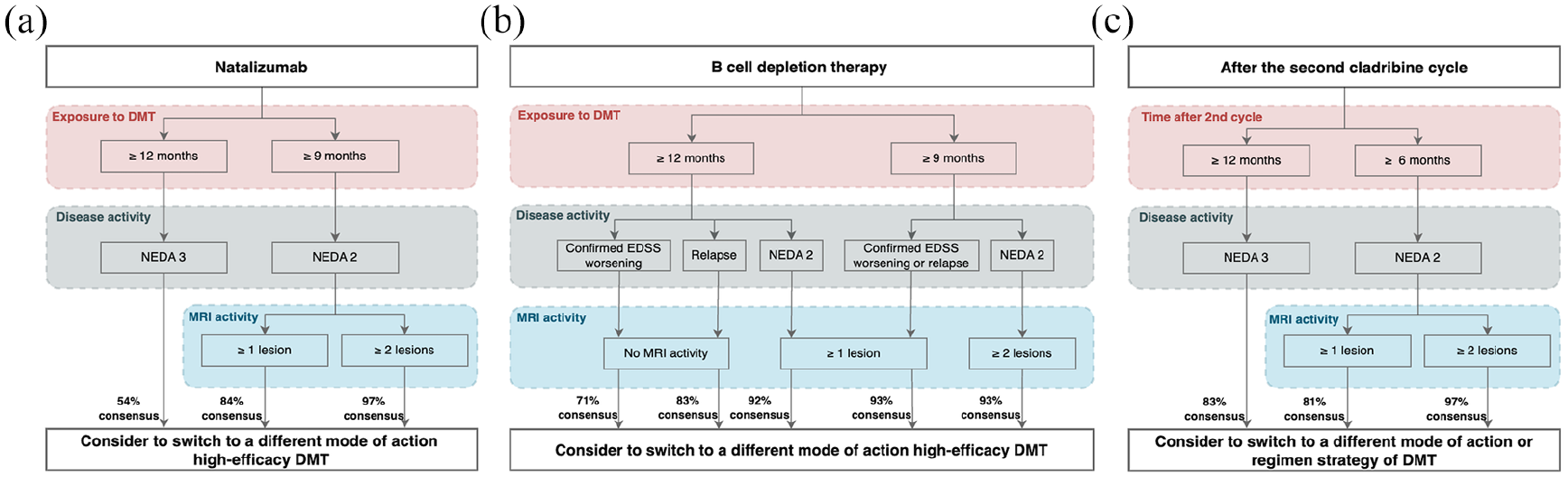
Horizontal switch algorithms in patients with high sNfL (>90th percentile): (a) From natalizumab; (b) From B-cell depletion therapy; and (c) From cladribine to different mode of action of DMT.
Experts were reluctant (37% agreement) to switch natalizumab treatment after 9 months to another HET in pwMS with NEDA3 and only high sNfL. However, with a minimum treatment duration to 12 months, 54% agreed on switching from natalizumab to another HET with a different mode of action.
Agreement increased if there were additional signs of disease, for example, MRI activity; most experts would then switch even if the treatment duration was only 9 months with 84%–97% agreement depending on the number of new/enlarging lesions (one or two and more lesions).
Switching from BCDT
Experts were reluctant (20% in agreement, 30% undecided and 50% in disagreement) to switch from BCDT (Figure 3(b)) in pwMS with NEDA3 and high sNfL. The treatment decision algorithms were dropped following round 1 (Supplemental Material).
However, if pwMS on BCDT with high sNfL showed clinical and/or MRI activity, most experts would switch to another HET with a different mode of action. Consensus ranged from moderate (71%) to broad (93%) depending on the additional sign of disease activity and depending on the duration (i.e. ⩾9 or 12 months) of the BCDT.
Switching from cladribine
There was a broad consensus (83%) to switch from cladribine (Figure 3(c)) to another DMT if pwMS with NEDA3 showed high sNfL. However, experts recommended to switch only if high sNfL occurs 12 months after the second cycle, allowing the medication to exert its full effect. If pwMS on cladribine have both high sNfL and MRI activities, agreements ranged from 81% to 97% depending on the number of new/enlarging lesions to switch cladribine to a drug with a different mode of action even 6 months after the second cycle.
sNfL < 80th percentile: De-escalation algorithm
From HET
During round 1, de-escalation algorithms for extending natalizumab treatment intervals from 4 to 6 weeks reached 72% agreement. However, it was dropped as it is already according to standards. Most experts were not in favor of stopping (10% agreement) or de-escalating (21%) BCDT to a lower efficacy DMT pwMS with NEDA3 who have sNfL <80th percentile. Those algorithms were dropped after round 1 (Supplemental Material).
In the first two rounds, de-escalation based on B-cell counts (i.e. extending treatment interval as long as B-cells are fully depleted) was introduced but dropped in round 3 due to lack of evidence on safety and efficacy of such an approach. 31
De-escalation was recommended for pwMS on BCDT when they are NEDA3 for at least 2 years and have sNfL <80th percentile (Figure 4(a)), with a moderate consensus (74%) to extend the treatment interval up to 12 months for ocrelizumab and rituximab, or 8 weeks for ofatumumab.
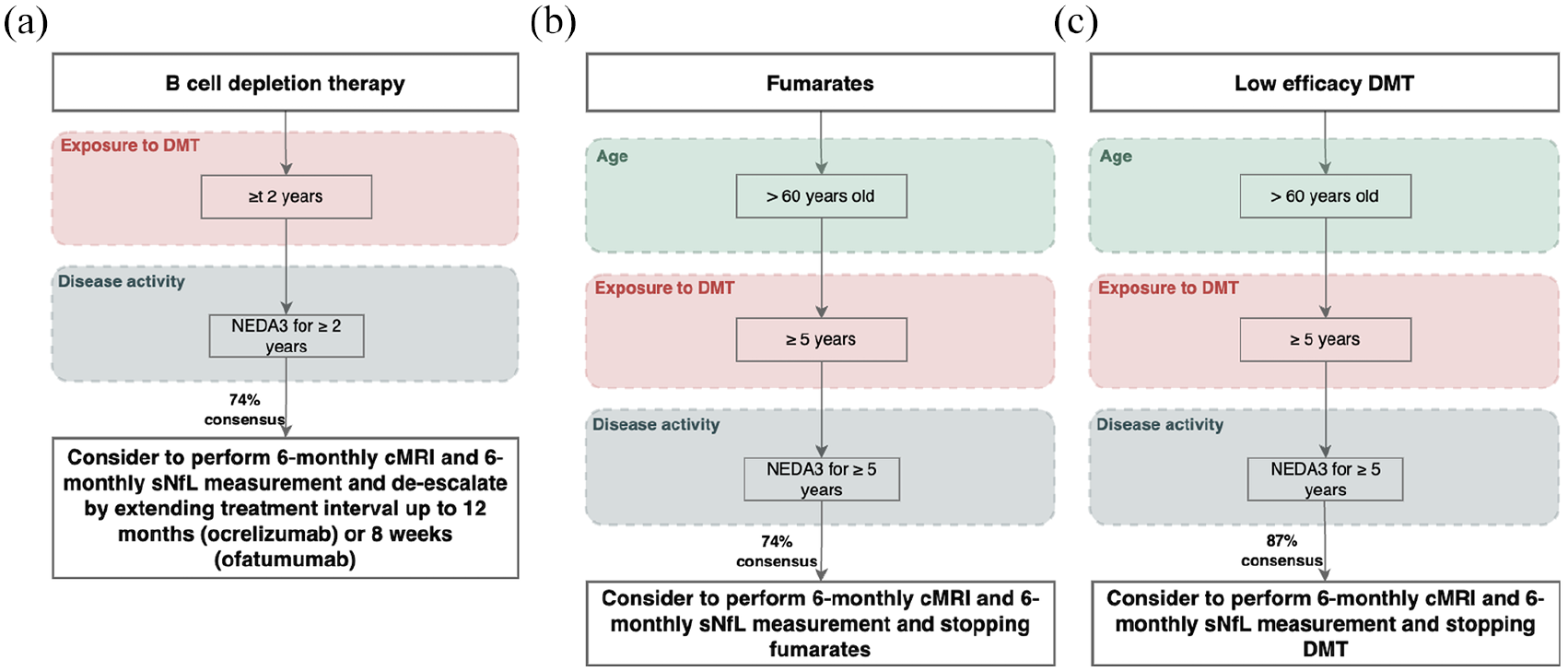
De-escalation algorithms in patients with “normal” sNfL (<80th percentile): (a) From B-cell depletion therapy to extending treatment interval; (b) From fumarates to stopping fumarates; and (c) From low efficacy DMT to stopping DMT treatment.
From medium efficacy DMTs
Panelists highlighted the need to differentiate between S1P modulators and dimethyl fumarates due to concerns regarding the potential rebound of severe disease activity after stopping S1P modulators, 32 which is not known to occur with dimethyl fumarate (Figure 4(b)).
De-escalation to a lower efficacy DMT from S1P receptor modulators or dimethyl fumarates were dropped due to insufficient agreement (below 50%; 26% and 48% respectively). A moderate consensus (74%) was reached to stop dimethyl fumarates in pwMS with sNfL <80th percentile and NEDA3 for the past 5 years when introducing an age limit and a minimum treatment duration: above 60 years of age and receiving fumarates for at least 5 years.
From low efficacy DMT
Similarly, a broad consensus (87% t) was reached to stop low efficacy DMT in pwMS over 60 years old, treated for at least 5 years, and with NEDA3 for the past 5 years and sNfL <80th percentile.
For monitoring and safety reasons, for all de-escalation algorithms, it was advised to perform 6-monthly cMRI and sNfL measurements, and to re-escalate if re-occurrence of disease activity (clinically, by MRI or high sNfL).
Discussion
This Delphi study resulted in a consensus on the classification of DMTs as low, medium and high-efficacy, 9 escalation treatment decision algorithms (7 ranging from broad (≥80% agreement) to full agreement (100%) and 2 with a moderate consensus (50% to 79% agreement)), 11 horizontal treatment changes decision algorithms (9 broad consensus (≥80% agreement) and 2 moderate consensus (50% to 79% agreement)), and 3 de-escalation treatment decision algorithms (1 broad consensus (≥80% agreement) and 2 moderate consensus (50% to 79% agreement)).
The rationale to escalate DMT based on sNfL values is derived from studies showing that higher efficacy drugs lead to lower sNfL values compared to pwMS without treatment or on lower efficacy drugs.20,22,23 It also has been shown that sNfL is increased in a proportion of pwMS with NEDA3, and significantly prognosticating disease activity in the following years illustrating that sNfL and MRI provide only partially overlapping information on the future disease course. Although consensus increases with additional imaging disease activity, sNfL does capture disease activity beyond conventional clinical and imaging measures 20 and potentially capturing it earlier. 33 MRI lacks sensitivity for lesions below detectability by imaging and spinal cord MRI are less frequently done in routine—sNfL could complement those gaps in detection. Importantly, data from the OPERA studies have shown that persons with RRMS on ocrelizumab with high sNfL at week 48 after treatment start had a significantly higher chance for future disability accumulation up to 9 years after starting the DMT. 34
Patients’ preference was frequently highlighted along with the concern in the de-escalation questions, on the risk of changing a treatment that works and has achieved NEDA3. Although patients who de-escalate cannot do better in terms of relapse rate or EDSS worsening, de-escalation might increase the quality of life in these patients due to fewer infusions, less risk for side effects/risks (also long-term) and reduce treatment burden and costs.
The long-term effects of immunosuppressive treatments like BCDT are not fully known. The immune system is essential for combating infections and providing tumor cell surveillance, making it challenging to balance better-defined shorter-term outcomes against the less understood long-term (10–50 years) benefits and harms.
Another drawback with de-escalation pertains to progression independent of relapse activity (PIRA). De-escalation may lead to subsequent disease activity as subtle inflammation might be missed by NfL measurements, clinical assessment, or MRI at the time of the decision.
Our understanding of the extent to which DMTs contributes to the pure neurodegenerative aspect of the disease remains limited. For example, ocrelizumab has been showed to reduce disability accumulation in primary progressive (PP)MS (ORATORIO trial) suggesting that its mode of action exerts this by suppressing brain diffuse inflammation that remains below detection threshold by standard measures. By de-escalating DMT, we may expose patients to a risk for developing more disability accumulation in the longer term. On the contrary, the effect size of ocrelizumab on disability progression was at best moderate (24%) in the ORATORIO trial, 35 however, which still may be relevant for pwMS. If we believe that anti-inflammatory type DMTs might have a relevant effect on disability accumulation, de-escalation cannot be recommended as a standard unless their effect on this hidden inflammation can be quantitated by novel biomarkers.
Experts were reluctant to switch horizontally within HET with NEDA3 and high sNfL, likely because HET is often the “final” treatment option. However, consensus increased when additional signs of clinical or imaging disease activity where included in the treatment decision algorithms—as was the case for all algorithms. sNfL application in usual care is in its infancy which may explain the hesitancy from experts to base decisions solely on sNfL. The MultiSCRIPT trial will provide essential evidence to further understand the role of sNfL in treatment decisions and to conduct exploratory analyses on its impact on patient outcomes.
With the Delphi study, we aimed to provide a minimal set of treatment decision algorithms that experts have agreed upon for the most frequent clinical scenarios to be applied within the MultiSCRIPT trial 25 in patients with RRMS. The herein proposed treatment decision algorithms may not apply to other clinical context. Caution is needed when using them more broadly, especially when elevated sNfL may be due to other potential causes (e.g. head trauma, stroke, relevant sports-related head injury, at least medium severe renal failure (GFR < 60 mL/min/1.73 m2), suboptimal treated diabetes mellitus or potential other incipient neurodegenerative diseases). This is particularly relevant in the absence of clinical or imaging evidence of disease activity. If uncertainty exists regarding the cause of high sNfL, repeat measurements may be warranted. Furthermore, factors such as comorbidities, neutralizing antibodies, among others, which may lead to suboptimal treatment effect, need to be factored in. The suggested algorithms assume that such special conditions do not apply if they do, decisions should follow best clinical practices, which may include maintaining the current treatment. They are non-binding, and patients and treating physician can always overrule the recommendations made here in the Delphi process.
Our findings illustrate the interest in using sNfL as a biomarker for personalized treatment decisions. sNfL monitoring offers additional information that may support earlier escalation of DMTs and safer de-escalation but is not intended to replace clinical and neuroimaging assessments. The added value of sNfL in informing treatment decision needs yet to be established; the MultiSCRIPT trial aims to provide such needed evidence.
The Delphi process has several limitations.
First, anonymity was lost in the final in-person meeting, which may have impacted willingness to express deviating views. However, the open discussion between experts allowed clarifications and to efficiently fine tune some of the algorithms.
Second, the first-round survey was drafted based on the core team expertise without formal systematic review of the literature. Personalization of treatment strategies in MS is only at its infancy and the evidence regarding escalation and de-escalation strategies are currently limited. Still, it is unlikely that the core team was not aware of the research and trials done in the field.
Third but related, the panel was small with a majority Swiss MS experts. Performing the Delphi was motivated by the MultiSCRIPT trial 25 conducted within the SMSC assessing the added value of informing treatment decisions by 6-monthly sNfL monitoring on patient relevant outcomes including NEDA3 and quality of life that started in February 2024. 36 It was important to gather consensus among the physicians also taking part in the trial based on their expertise and knowledge of the evidence as in routine care. The treatment algorithms developed herein may be prone to changes as we learn from the evidence generated.
Fourth, there are no generally accepted threshold to define a consensus in a Delphi study. We chose to include all treatment decision algorithms that had reached ≥50% consensus acknowledging different thresholds of consensus (i.e. moderate < broad < strong < full agreement). The added value of sNfL to monitor disease activity in clinical practice remains to be determined. Those treatment decision algorithms are intended to help physicians and patients in the application of NfL in their decision-making within a clinical trial and to provide insight on what can be considered. They do not intend to be clinical guidelines nor to supplant patients’ and physicians’ preferences.
Finally, not all the experts in the panel could attend the hybrid meeting. One cannot exclude that the results reached in the three rounds are biased toward the experts present during the meeting. However, the majority was present (77%) and the bias was minimized as all members of the panel and core team are co-authors of the current manuscript.
Conclusion
The treatment decision algorithms we have developed and agreed upon are steppingstones to implementing sNfL into clinical routine care toward a more personalized care for people with MS. The algorithms have recently been applied in a pragmatic randomized clinical trial embedded in the Swiss MS Cohort assessing the superiority of 6-monthly sNfL monitoring compared with usual care. 25 The evidence generated during the trial will allow to adjust, if necessary, the treatment decision algorithms before being implemented in routine care.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251335466 – Supplemental material for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study
Supplemental material, sj-docx-1-msj-10.1177_13524585251335466 for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study by Özgür Yaldizli, Pascal Benkert, Lutz Achtnichts, Amit Bar-Or, Viviane Bohner-Lang, Claire Bridel, Manuel Comabella, Oliver Findling, Giulio Disanto, Sebastian Finkener, Claudio Gobbi, Cristina Granziera, Marina Herwerth, Robert Hoepner, Dana Horakova, Nicole Kamber, Michael Khalil, Philipp Kunz, Patrice Lalive, Ralf Linker, Johannes Lorscheider, Stefanie Müller, Johanna Oechtering, Victoria Pettypool, Fredrik Piehl, Caroline Pot, Patrick Roth, Marie Théaudin, Mar Tintore, Carmen Tur, Denis Uffer, Marjolaine Uginet, Jochen Vehoff, Heinz Wiendl, Tjalf Ziemssen, Chiara Zecca, Anke Salmen, David Leppert, Tobias Derfuss, Ludwig Kappos, Lars G Hemkens, Perrine Janiaud and Jens Kuhle in Multiple Sclerosis Journal
Supplemental Material
sj-docx-2-msj-10.1177_13524585251335466 – Supplemental material for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study
Supplemental material, sj-docx-2-msj-10.1177_13524585251335466 for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study by Özgür Yaldizli, Pascal Benkert, Lutz Achtnichts, Amit Bar-Or, Viviane Bohner-Lang, Claire Bridel, Manuel Comabella, Oliver Findling, Giulio Disanto, Sebastian Finkener, Claudio Gobbi, Cristina Granziera, Marina Herwerth, Robert Hoepner, Dana Horakova, Nicole Kamber, Michael Khalil, Philipp Kunz, Patrice Lalive, Ralf Linker, Johannes Lorscheider, Stefanie Müller, Johanna Oechtering, Victoria Pettypool, Fredrik Piehl, Caroline Pot, Patrick Roth, Marie Théaudin, Mar Tintore, Carmen Tur, Denis Uffer, Marjolaine Uginet, Jochen Vehoff, Heinz Wiendl, Tjalf Ziemssen, Chiara Zecca, Anke Salmen, David Leppert, Tobias Derfuss, Ludwig Kappos, Lars G Hemkens, Perrine Janiaud and Jens Kuhle in Multiple Sclerosis Journal
Supplemental Material
sj-docx-3-msj-10.1177_13524585251335466 – Supplemental material for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study
Supplemental material, sj-docx-3-msj-10.1177_13524585251335466 for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study by Özgür Yaldizli, Pascal Benkert, Lutz Achtnichts, Amit Bar-Or, Viviane Bohner-Lang, Claire Bridel, Manuel Comabella, Oliver Findling, Giulio Disanto, Sebastian Finkener, Claudio Gobbi, Cristina Granziera, Marina Herwerth, Robert Hoepner, Dana Horakova, Nicole Kamber, Michael Khalil, Philipp Kunz, Patrice Lalive, Ralf Linker, Johannes Lorscheider, Stefanie Müller, Johanna Oechtering, Victoria Pettypool, Fredrik Piehl, Caroline Pot, Patrick Roth, Marie Théaudin, Mar Tintore, Carmen Tur, Denis Uffer, Marjolaine Uginet, Jochen Vehoff, Heinz Wiendl, Tjalf Ziemssen, Chiara Zecca, Anke Salmen, David Leppert, Tobias Derfuss, Ludwig Kappos, Lars G Hemkens, Perrine Janiaud and Jens Kuhle in Multiple Sclerosis Journal
Supplemental Material
sj-pdf-4-msj-10.1177_13524585251335466 – Supplemental material for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study
Supplemental material, sj-pdf-4-msj-10.1177_13524585251335466 for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study by Özgür Yaldizli, Pascal Benkert, Lutz Achtnichts, Amit Bar-Or, Viviane Bohner-Lang, Claire Bridel, Manuel Comabella, Oliver Findling, Giulio Disanto, Sebastian Finkener, Claudio Gobbi, Cristina Granziera, Marina Herwerth, Robert Hoepner, Dana Horakova, Nicole Kamber, Michael Khalil, Philipp Kunz, Patrice Lalive, Ralf Linker, Johannes Lorscheider, Stefanie Müller, Johanna Oechtering, Victoria Pettypool, Fredrik Piehl, Caroline Pot, Patrick Roth, Marie Théaudin, Mar Tintore, Carmen Tur, Denis Uffer, Marjolaine Uginet, Jochen Vehoff, Heinz Wiendl, Tjalf Ziemssen, Chiara Zecca, Anke Salmen, David Leppert, Tobias Derfuss, Ludwig Kappos, Lars G Hemkens, Perrine Janiaud and Jens Kuhle in Multiple Sclerosis Journal
Supplemental Material
sj-pdf-5-msj-10.1177_13524585251335466 – Supplemental material for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study
Supplemental material, sj-pdf-5-msj-10.1177_13524585251335466 for Personalized treatment decision algorithms for the clinical application of serum neurofilament light chain in multiple sclerosis: A modified Delphi Study by Özgür Yaldizli, Pascal Benkert, Lutz Achtnichts, Amit Bar-Or, Viviane Bohner-Lang, Claire Bridel, Manuel Comabella, Oliver Findling, Giulio Disanto, Sebastian Finkener, Claudio Gobbi, Cristina Granziera, Marina Herwerth, Robert Hoepner, Dana Horakova, Nicole Kamber, Michael Khalil, Philipp Kunz, Patrice Lalive, Ralf Linker, Johannes Lorscheider, Stefanie Müller, Johanna Oechtering, Victoria Pettypool, Fredrik Piehl, Caroline Pot, Patrick Roth, Marie Théaudin, Mar Tintore, Carmen Tur, Denis Uffer, Marjolaine Uginet, Jochen Vehoff, Heinz Wiendl, Tjalf Ziemssen, Chiara Zecca, Anke Salmen, David Leppert, Tobias Derfuss, Ludwig Kappos, Lars G Hemkens, Perrine Janiaud and Jens Kuhle in Multiple Sclerosis Journal
Footnotes
Authors Contributions
Ö.Y., P.B., A.S., C.Z., T.D., L.K., D.L., L.G.H., P.J., and J.K contributed to the conception and design of the study. L.A., A.B-O., V.B-L., C.B., M.C., G.D., C. Go., C.Gr., M.H., R.H., D.H., M.K., P.K., P.L., R.L., J.L., S.M., J.O., V.P., F.P., C.P., P.R., M.Th., M.Ti., C.T., D.U., J.V., H.W., and T.Z. contributed to the acquisition and analysis of data. All authors contributed to drafting the text or preparing the figures.
Data availability statement
Additional data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PB, AB-O, VB-H, CB, GD, OF, SF, MH, NK, PK, VP, CP, DU, MU, JV, DL, and PJ have nothing to declare; OY received grants from ECTRIMS/MAGNIMS, University of Basel, Pro Patient Stiftung, University of Basel, Free Academy Basel, Swiss Multiple Sclerosis Society, Swiss National Science Foundation and advisory board/lecture and consultancy fees from Roche, Sanofi-Genzyme, Almirall, Biogen and Novartis; LA served on scientific advisory boards for Celgene, Novartis Pharmaceuticals, Merck, Biogen, Sanofi-Genzyme, Roche, and Bayer; received funding for travel and/or speaker honoraria from Celgene, Biogen, Sanofi-Genzyme, Novartis, Merck Serono, Roche, Teva, and the Swiss MS Society; and research support from Biogen, Sanofi, Genzyme, and Novartis; MC received compensation for consulting services and speaking honoraria from Bayer Schering Pharma, Merk Serono, Biogen-Idec, Teva Pharmaceuticals, Sanofi-Aventis, Genzyme, Bristol-Myers Squibb, and Novartis; Ente Ospedaliero Cantonale (employer) received compensation for C. Go.’s speaking activities, consulting fees, or grants from AbbVie, Alexion, Almirall, Biogen, Bristol Meyer Squibb, Eisai, Lilly, Lundbeck, Merck, Merz, Novartis, Organon, Pfizer, Sandoz, Sanofi, Teva Pharma, Roche; The University Hospital Basel (USB) and the Research Center for Clinical neuroimmunology and Neuroscience (RC2NB), as the employers of CGr, have received the following fees which were used exclusively for (research support from Siemens, GeNeuro, Genzyme-Sanofi, Biogen, Roche. They also have received advisory board and consultancy fees from Actelion, Genzyme-Sanofi, Novartis, GeNeuro, Merck, Biogen and Roche; as well as speaker fees from Genzyme-Sanofi, Novartis, GeNeuro, Merck, biogen and Roche; RH received speaker/advisor honorary from Merck, Novartis, Roche, Biogen, Alexion, Sanofi, Janssen, Bristol-Myers Squibb, Teva/Mepha and Almirall. He received research support within the last 5 years from Roche, Merck, Sanofi, Biogen, Chiesi, and Bristol-Myers Squibb. He also received research grants from the Swiss MS Society, the SITEM Insel Support Fund and is a member of the Advisory Board of the Swiss and International MS Society. He also serves as deputy editor in chief for Journal of Central Nervous System disease and is part of the ECTRIMS Young Investigator Committee; DH was supported by the Charles University: Cooperatio Program in Neuroscience, by the project National Institute for Neurological Research (Program EXCELES, ID Project No. 174 LX22NPO5107)—Funded by the European Union—Next Generation EU, and by General University Hospital in Prague project MH CZ-DRO-VFN64165. She also received compensation for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck, Bayer, Sanofi-Genzyme, Roche, and Teva, as well as support for research activities from Biogen Idec; MK received funding for travel and speaker honoraria from Bayer, Novartis, Merck, Biogen Idec and Teva Pharmaceutical Industries Ltd. and serves on scientific advisory boards for Biogen Idec, Merck Serono, Roche, Novartis and Gilead; PL reports that the Geneva University Hospital received honoraria for speaking from Biogen, Merck, Roche; consulting fees from Biogen, Merck, Novartis, Roche; research grants from Biogen, Merck, Novartis; RL received compensation for activites with Biogen, Celgene/BMS, Janssen Cilag, Pfizer, Merck, Novartis, Sanofi and Roche as well as research support from Biogen and Novartis; JL’s institution has received research grants from Novartis, Biogen, Innosuisse—Swiss Innovation Agency, and the MSBase Foundation, and honoraria for advisory boards and/or speaking fees from Novartis, Roche and Teva. He received conference travel support from Novartis and Bristol-Myers Squibb; SM received honoraria for travel, honoraria for lectures/consulting and/or grants for studies from Almirall, Alexion, Bayer, Biogen, Bristol-Myers Squibb SA/Celgene, Genzyme, Merck-Serono, Teva, Novartis and Roche; JO received research support by the Swiss MS Society and served on advisory boards for Roche and Merck; FP received research grants from Janssen, Merck KGaA and UCB, and fees for serving on DMCs in clinical trials with Chugai, Lundbeck and Roche, and preparation of expert witness statement for Novartis; PR received honoraria for lectures or advisory board participation from Alexion, Bristol-Myers Squibb, Boehringer Ingelheim, Debiopharm, Galapagos, Merck Sharp and Dohme, Laminar, Midatech Pharma, Novocure, QED, Roche, and Sanofi and research support from Merck Sharp and Dohme and Novocure; The CHUV has received for MTh grants for advisory boards, funding for participation in medical meetings from Alexion, Merck, Sanofi, Roche, Biogen, and Novartis; MTi received compensation for consulting services, speaking honoraria and research support from Almirall, Bayer Schering Pharma, Biogen-Idec, Genzyme, Janssen, Merck-Serono, Novartis, Roche, Sanofi-Aventis, Viela Bio and Teva Pharmaceuticals. Data Safety Monitoring Board for Parexel and UCB Biopharma; CT is currently being funded by a Junior Leader La Caixa Fellowship (fellowship code is LCF/BQ/PI20/11760008), awarded by “la Caixa” Foundation (ID 100010434). She has also received the 2021 Merck’s Award for the Investigation in MS, awarded by Fundación Merck Salud (Spain) and a grant awarded by the Instituto de Salud Carlos III, Ministerio de Ciencia e Innovación de España (PI21/01860) and she has also received honoraria from Roche and Novartis and is a steering committee member of the O’HAND trial and of the Consensus group on Follow-on DMTs; HW declares scientific advisory boards/consultant fee for Biogen, Evgen, Genzyme, MedDay Pharmaceuticals, Merck Serono, AbbVie, Actelion, Johnson & Johnson, Novartis, Roche Pharma AG, Sanofi-Aventis and Swiss Multiple Sclerosis Society, speaker honoraria and travel support from Alexion, Biogen, Cognomed, F. Hoffmann-La Roche Ltd., Gemeinnützige Hertie-Stiftung, Merck Serono, Novartis, Roche Pharma AG, Genzyme, Teva, and WebMD Global and his research is funded by the German Ministry for Education and Research, Deutsche Forschungsgemeinschaft, Else Kröner Fresenius Foundation, Fresenius Foundation, the European Union, Hertie Foundation, NRW Ministry of Education and Research, Interdisciplinary Center for Clinical Studies Muenster and RE Children’s Foundation, Biogen, GlaxoSmithKline GmbH, Roche Pharma AG, and Sanofi-Genzyme; TZ declares advisory boards fees from Biogen, BMS, Merck, Novartis, Roche, Sanofi and Teva; speaker fees from Almirall, Alexion, Biogen, BMS, Hexal, Novartis, Roche, Sanofi, Teva and Viatris; research support from Biogen, BMS, Novartis, Roche, Sanofi and Teva; Ente Ospedaliero Cantonale (employer) received compensation for C.Z.’s speaking activities, consulting fees, or grants from AbbVie, Alexion, Almirall, Biogen, Bristol Meyer Squibb, Eisai, Lilly, Lundbeck, Merck, Merz, Novartis, Organon, Pfizer, Sandoz, Sanofi, Teva Pharma, Roche. CZ is recipient of a grant for senior reseachers provided by AFRI (Area Formazione accademica, Ricerca e Innovazione), EOC; AS received speaker honoraria for activities with Bristol-Myers Squibb, CSL Behring, Merck, Neuraxpharm, Novartis, and research support by the Baasch Medicus Foundation, the Medical Faculty of the University of Bern, the Swiss MS Society and the regional association of North Rhine-Westphalia of the German MS Society (DMSG Landesverband NRW); TD has served on scientific advisory boards, steering committees, and data safety monitoring boards for Alexion, Actelion, Biogen, Celgene, Genzyme, GeNeuro, Merck, Mitsubishi Pharma, Novartis, Roche, Octapharma, and MedDay; has received travel and/or speaker honoraria from Biogen, Genzyme, Merck, Novartis, Roche, and Merck-Serono; has received research support from Alexion, Biogen, Novartis, Roche, the Swiss MS Society, the European Union and the Swiss National Foundation. LK has received no personal compensation. His institutions (University Hospital Basel/Stiftung Neuroimmunology and Neuroscience Basel) have received and used exclusively for research support payments for steering committee and advisory board participation, consultancy services, and participation in educational activities from: Actelion, Bayer, BMS, df-mp Molnia & Pohlmann, Celgene, Eli Lilly, EMD Serono, Genentech, Glaxo Smith Kline, Janssen, Japan Tobacco, Merck, MH Consulting, Minoryx, Novartis, F. Hoffmann-La Roche Ltd, Senda Biosciences Inc., Sanofi, Santhera, Shionogi BV, TG Therapeutics, and Wellmera, and license fees for Neurostatus-UHB products; grants from Novartis, Innosuisse, and Roche. LGH employer RC2NB is supported by Foundation Clinical Neuroimmunology and Neuroscience Basel. RC2NB has a contract with Roche for a steering committee participation of LGH. JK received speaker fees, research support, travel support, and/or served on advisory boards by Swiss MS Society, Swiss National Research Foundation (320030_212534/1), University of Basel, Progressive MS Alliance, Alnylam, Bayer, Biogen, Bristol-Myers Squibb, Celgene, Immunic, Merck, Neurogenesis, Novartis, Octave Bioscience, Quanterix, Roche, Sanofi, and Stata DX.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is part of the MultiSCRIPT trial supported by the Swiss National Science Foundation as part of the Investigator Initiated Clinical Trial program (grant no. 33IC30_205806/1). The funder had no role in the design and conduct of the study, nor in the preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethical considerations
Not applicable, all participants to the Delphi Study are co-authors.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
