Abstract
Introduction:
The 2024 McDonald criteria for diagnosing multiple sclerosis (MS) include optic nerve involvement as a fifth region for establishing dissemination in space. Optic neuritis (ON) can be detected through optical coherence tomography (OCT) using an inter-eye absolute or percentage difference (IEAD, IEPD) in ganglion cell-inner plexiform layer (GCIPL) thickness.
Objective:
To compare the diagnostic accuracy of GCIPL IEAD/IEPD with GCL and IPL IEAD/IEPD alone for identifying a history of ON.
Methods:
This cross-sectional retrospective study included people with MS (pwMS) who underwent an OCT scan. Diagnostic accuracy was assessed using ROC analysis.
Results:
A total of 241 pwMS (mean age 34.7 years [SD 9.7], 70.1% female) were included. Sixty-eight (28.2%) patients had a documented history of unilateral ON. GCL IEAD (AUC 0.79, cut-off ⩾ 0.06 mm3 or ⩾2µm, 58.9% sensitivity, 85.1% specificity) and IEPD (AUC 0.80, cut-off ⩾ 3%, 48.7% sensitivity, 89.8% specificity) demonstrated excellent diagnostic accuracy for unilateral ON, showing non-inferiority to the established GCIPL IEAD/IEPD.
Conclusion:
GCL IEAD and IEPD provide strong diagnostic accuracy for identifying unilateral ON and can be effectively used as an alternative to GCIPL IEAD/IEPD to facilitate implementation in clinical routine.
Keywords
Introduction
Optic neuritis (ON) is a common early symptom of multiple sclerosis (MS), manifesting as the initial symptom in approximately a quarter of people with MS (pwMS). 1 ON typically causes neuroaxonal damage to the optic nerve and by extension to inner retinal layers, which can be reliably quantified using optical coherence tomography (OCT). 2 Numerous studies have confirmed that interocular asymmetry, expressed as an inter-eye difference (IED) in the peripapillary retinal nerve fiber layer (pRNFL), comprising the unmyelinated axons of the optic nerve, or the macular ganglion cell-inner plexiform layer (GCIPL), a composite measure combining the ganglion cell layer (GCL) and the inner plexiform layer (IPL), are highly sensitive and accurate for detecting neuroretinal degeneration resulting from both clinical and subclinical ON.3–5
The ongoing evolution of diagnostic criteria for MS has led to faster and more accurate diagnosis enabling earlier access to disease-modifying therapies (DMT) for pwMS. Recent evidence suggests that incorporating the optic nerve as a fifth region for demonstrating dissemination in space moderately improves the diagnostic performance by increasing sensitivity without compromising specificity, 6 which is to be reflected in the 2024 revision of the McDonald criteria. With these revisions, OCT is expected to become integral to clinical practice as it shows very good concordance with MRI detection of optic nerve involvement but has some considerable advantages as it is noninvasive, inexpensive, accessible, and produces standardized, reliable quantitative measures. 7 Mean thickness of GCIPL has proven reliable for identifying a history of ON, using either the inter-eye absolute or percentage difference (IEAD, IEPD).5,8–10 However, in clinical routine, other parameters such as GCIPL volume (instead of thickness) or only GCL thickness/volume instead of the composite GCIPL are used, mainly for reasons of the respective default image analysis of different OCT devices.
So far, there are no studies directly comparing the diagnostic accuracy of GCL and IPL IEAD/IEPD to the composite GCIPL IEAD/IEPD in identifying a history of ON in pwMS, which we aimed to provide here.
Methods
Patients and definitions
For this cross-sectional retrospective study, patients who visited the MS outpatient clinic of the Department of Neurology, Medical University of Vienna, between 1 January 2023 and 31 December 2023 were screened and included based on following criteria: (i) relapsing MS diagnosis according to the 2017 version of the McDonald criteria, 11 (ii) age ⩾ 18 years, and (iii) availability of at least one OCT scan at any time of the disease course. 12 For this study, the chronologically first OCT scan was used for the analysis. Data on Expanded Disability Status Scale (EDSS) were obtained at the time of first OCT. 13 PwMS were grouped according to ON status into people with and without a history of unilateral ON. ON was confirmed by experienced neuro-ophthalmologists based on the following criteria: monocular, subacute loss of vision accompanied by (i) orbital pain worsening on eye movements, (ii) reduced contrast and color vision, and/or (iii) relative afferent pupillary defect (RAPD).
Optical coherence tomography
OCT imaging was performed by experienced neuro-ophthalmologists at the Department of Ophthalmology and Optometry using the same spectral-domain OCT (Spectralis OCT, Heidelberg Engineering, Heidelberg, Germany; software Heidelberg eye explorer software version 5.4.8.0) without pupil dilatation in a dark room on both eyes of each patient. For pRNFL measurement, a custom 12° (3.4 mm) ring scan centered on the optic nerve head was used (1536 A-scans, automatic real-time tracking [ART]: 100 averaged frames). 14 For GCIPL measurement, a 20°×20° macular volume scan (512 A-scans, 25 B-scans, vertical alignment, ART: 16 averaged frames) centered on the macula was performed. The mean thickness of the individual retinal layers was computed in an area of 6 mm around the fovea as defined by the Early Treatment Diabetic Retinopathy Study. 15 Image processing was conducted semi-automatically using the built-in proprietary software. All examinations were checked for sufficient quality using OSCAR-IB criteria. 16 OCT examinations that lacked macula scan or did not fulfill OSCAR-IB criteria were excluded from the final analysis.
PwMS with a history of bilateral ON or unilateral ON less than 6 months prior to the OCT scan were excluded from the study. Eyes with a history of ON ⩾ 6 months before baseline were eligible for inclusion. Patients with diagnoses of ophthalmologic (i.e. myopia greater than –4 diopters, optic disk drusen, glaucoma, cataract, outer retinal pathologies), neurologic, or drug-related causes of retinal damage not attributable to MS were excluded. 17 All patients were screened for the presence of macular edema and excluded if macular edema occurred. The investigators performing the OCT were blinded to clinical parameters and vice versa. The quantitative OCT study results were reported using the revised Advised Protocol for OCT Study Terminology and Elements (APOSTEL 2.0) recommendations. 18
Standard protocol approvals, registrations, patient consents, and reporting
The study was approved by the ethics committee of the Medical University of Vienna (ethical approval number: 1668/2023). Since this was a retrospective study, the requirement for written informed consent from study participants was waived by the ethics committee. This study adheres to the reporting guidelines outlined within the ‘Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement.
Data availability statement
Data supporting the findings of this study are available from the corresponding author upon reasonable request by a qualified researcher and upon approval by the data-clearing committee of the Medical University Vienna.
Statistics
Statistical analysis was performed using SPSS 29.0 (SPSS Inc, Chicago, IL, USA) and R-Statistical Software (Version 4.0.0). Categorical variables were expressed in frequencies and percentages, continuous variables as mean and standard deviation (SD) or median and range or interquartile range (IQR) as appropriate. Continuous variables were tested for normal distribution by the Kolmogorov–Smirnov test with Lilliefors correction. Univariate comparisons were done by Chi-square-test, Mann–Whitney U test or independent t-test (with Welch’s correction in case of unequal standard deviations between the groups) as appropriate.
The thicknesses of the retinal layers were derived from their respective volumes using the formula: Volume × 1,000/9π. The IEAD was derived by calculating the absolute difference in volume or thickness between the two eyes. The IEPD was then determined by dividing the IEAD by the higher volume or thickness value of the two eyes. To test the correlation between GCIPL IEAD as the reference and GCIPL IEPD, GCL IEAD/IEPD and IPL IEAD/IEPD, the intraclass correlation coefficient (ICC) was calculated based on a mean-rating, absolute-agreement, 2-way mixed-effects models. We performed receiver-operating characteristics (ROC) analyses to compare diagnostic accuracy of GCIPL IEAD/IEPD, GCL IEAD/IEPD, and IPL IEAD/IEPD in distinguishing between pwMS with and without ON. The optimal cut-off values were selected using the Youden index. Areas under the curve (AUC) of the ROC curves were compared using the Delong test. In addition, we conducted a subanalysis in which the inner retinal layer thicknesses were calculated based on the volumes of the inner and outer four quadrants of the macula, explicitly omitting the fovea.
Sensitivity analysis was conducted by excluding patients with treatment initiation prior to the first OCT scan. The significance level was set at a two-sided p-value < 0.05. All multiple analyses were corrected using the Bonferroni method.
Results
In all, 241 pwMS (mean age 34.7 years [SD 9.7], 70.1% female, median disease duration 3.2 years [IQR 0.4–8.5], median EDSS 1.0 [range 0–6.5]) were included. The selection process based on the inclusion and exclusion criteria is shown in Figure 1. Detailed characteristics of the study cohort are given in Table 1.
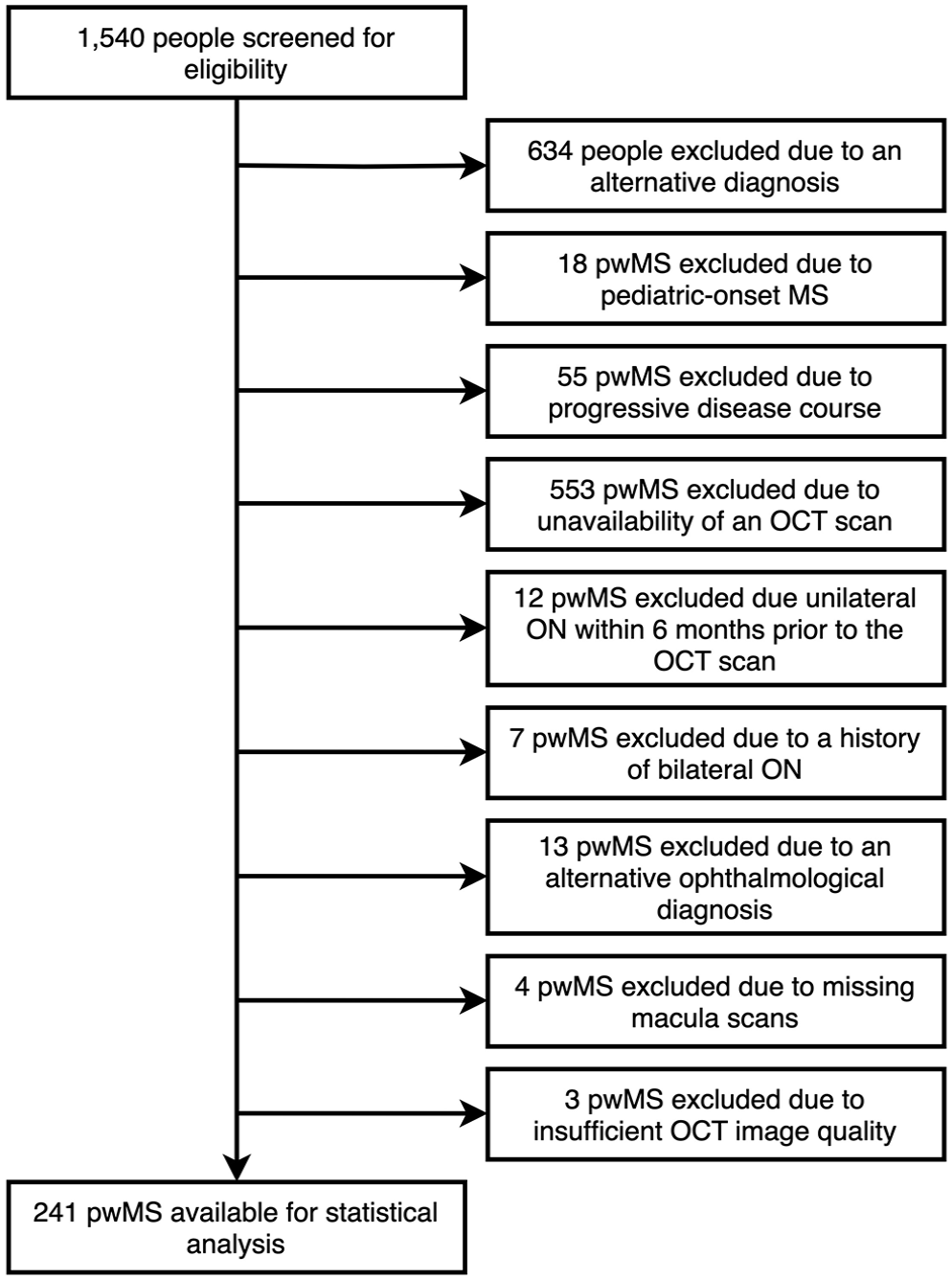
Flow chart of patients’ selection based on the inclusion and exclusion criteria.
Clinical characteristics of the study cohort.
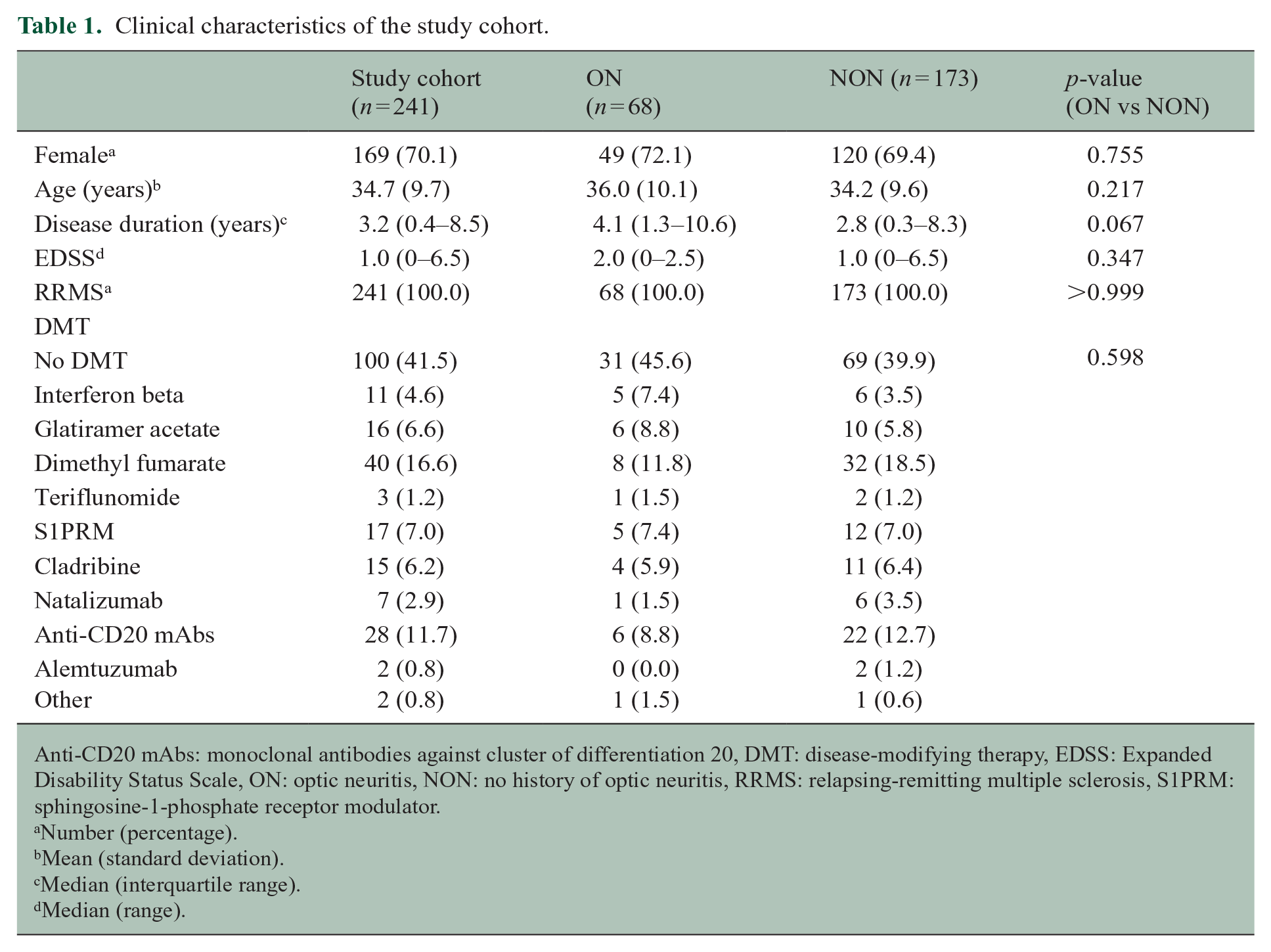
Anti-CD20 mAbs: monoclonal antibodies against cluster of differentiation 20, DMT: disease-modifying therapy, EDSS: Expanded Disability Status Scale, ON: optic neuritis, NON: no history of optic neuritis, RRMS: relapsing-remitting multiple sclerosis, S1PRM: sphingosine-1-phosphate receptor modulator.
Number (percentage).
Mean (standard deviation).
Median (interquartile range).
Median (range).
Sixty-eight (28.2%) patients had a documented history of unilateral ON. Characteristics of the study cohort are given in Table 1. As expected, mean pRNFL and GCIPL thicknesses were significantly lower in the ON group. Similarly, median IEAD and IEPD were significantly higher in all layers in the ON group (Table 2).
OCT characteristics of the study cohort according to the history of ON.
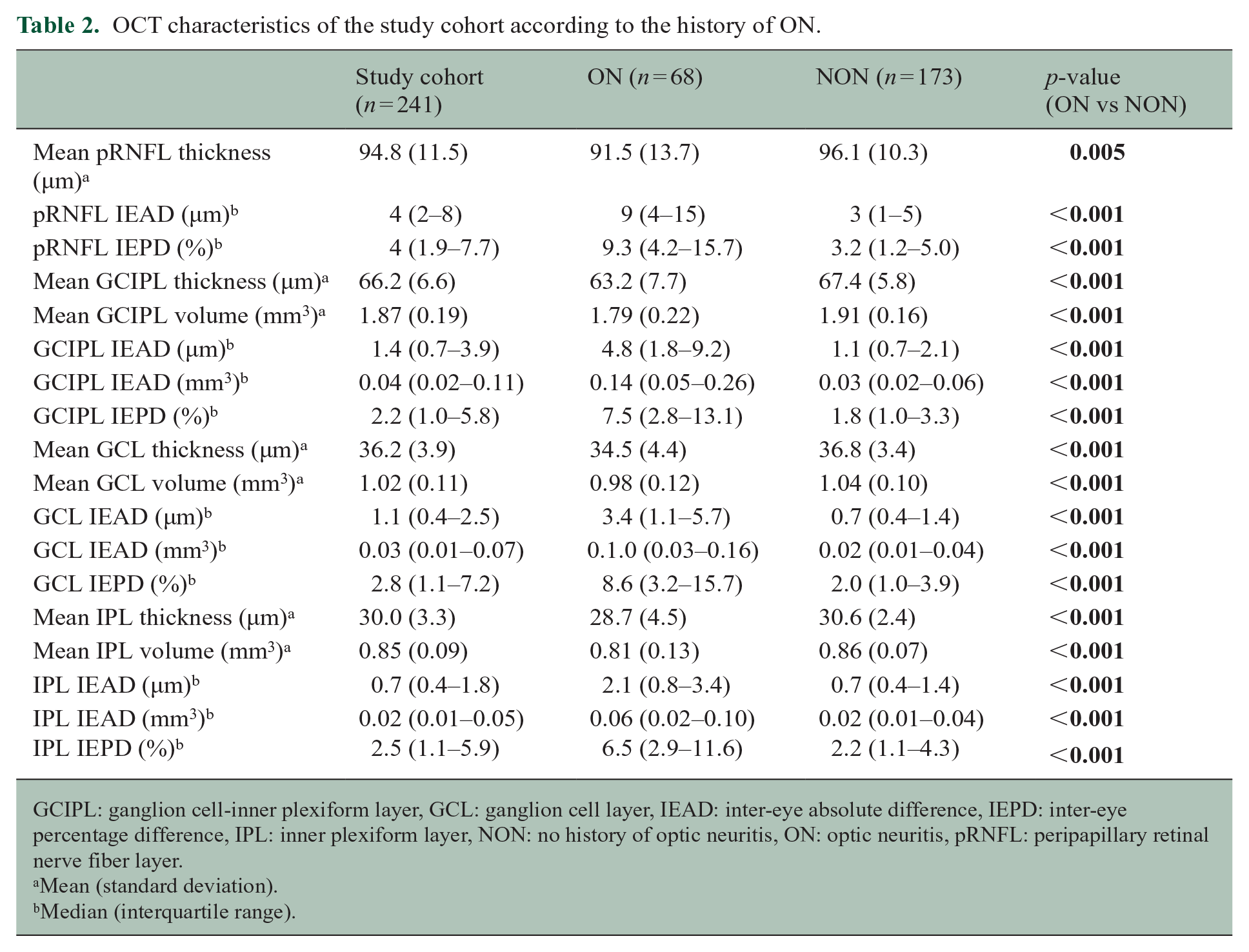
GCIPL: ganglion cell-inner plexiform layer, GCL: ganglion cell layer, IEAD: inter-eye absolute difference, IEPD: inter-eye percentage difference, IPL: inner plexiform layer, NON: no history of optic neuritis, ON: optic neuritis, pRNFL: peripapillary retinal nerve fiber layer.
Mean (standard deviation).
Median (interquartile range).
GCIPL IEAD correlated strongly with GCL IEAD (ICC 0.92, 95% CI 0.79–0.96, p < 0.001) but only moderately with IPL IEAD (ICC 0.68, 95% CI 0.39–0.81, p < 0.001). Moreover, GCIPL IEPD correlated even more strongly with GCL IEPD (ICC 0.97, 95% CI 0.95–0.98, p < 0.001) and IPL IEPD (ICC 0.94, 95% CI 0.92–0.95, p < 0.001).
Inter-eye absolute difference
The AUC were 0.76 (cut-off ⩾ 5µm, 48.5% sensitivity, 87.0% specificity, PPV 73.5%, NPV 69.4%) for pRNFL IEAD and 0.81 (cut-off ⩾ 4µm, 66.1% sensitivity, 84.1% specificity, PPV 57.4%, NPV 88.4%) for GCIPL IEAD (Figure 2), indicating GCIPL IEAD as the superior model. Among patients with ON, 18 (26.5%) and 29 (42.6%) did not meet the cut-off values for pRNFL and GCIPL IEAD, respectively (Figure 3). For GCL and IPL IEAD, the corresponding AUC were 0.79 (cut-off ⩾ 0.06 mm3 or ⩾2µm, 58.9% sensitivity, 85.1% specificity, PPV 63.2%, NPV 82.7%) and 0.76 (cut-off ⩾ 0.04 mm3 or ⩾1.2µm, 51.1% sensitivity, 85.4% specificity, PPV 67.6%, NPV 74.6%), respectively, demonstrating superior diagnostic performance of GCL IEAD by improving sensitivity without compromising specificity (Figure 2). Among patients with ON, 25 (36.8%) and 22 (32.4%) did not meet the cut-off values for GCL and IPL IEAD, respectively (Figure 3). Both GCL or GCIPL IEAD seemed equally effective in detecting a history of ON (Table 3).
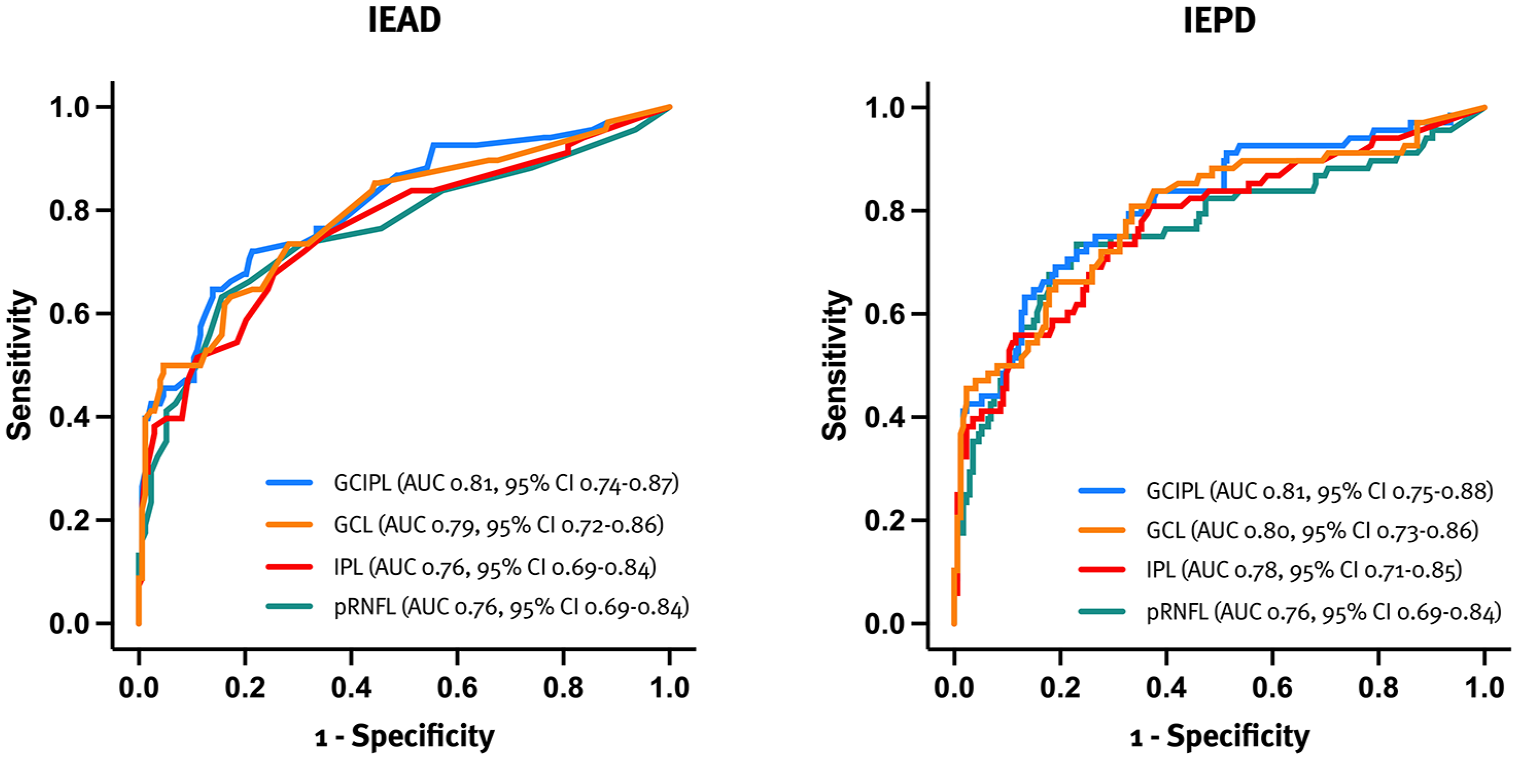
ROC curve analysis identifying patients with a history of ON.
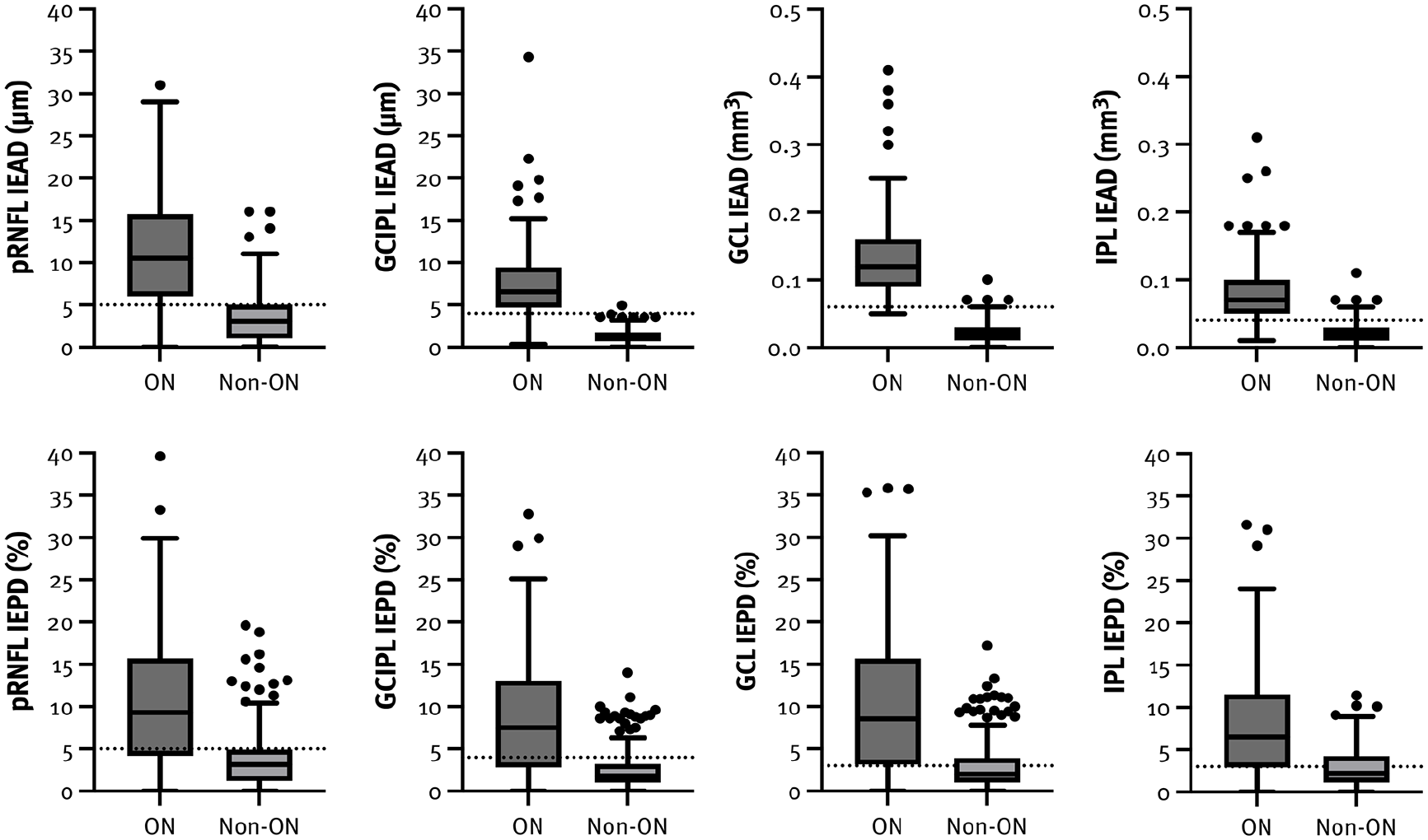
Box plots illustrating the IED in patients with and without a history of ON, with dotted lines indicating the optimal cut-off values.
Parameters of diagnostic accuracy of IEAD and IEPD of pRNFL, GCIPL as well as GCL and IPL alone for identifying a history of ON.
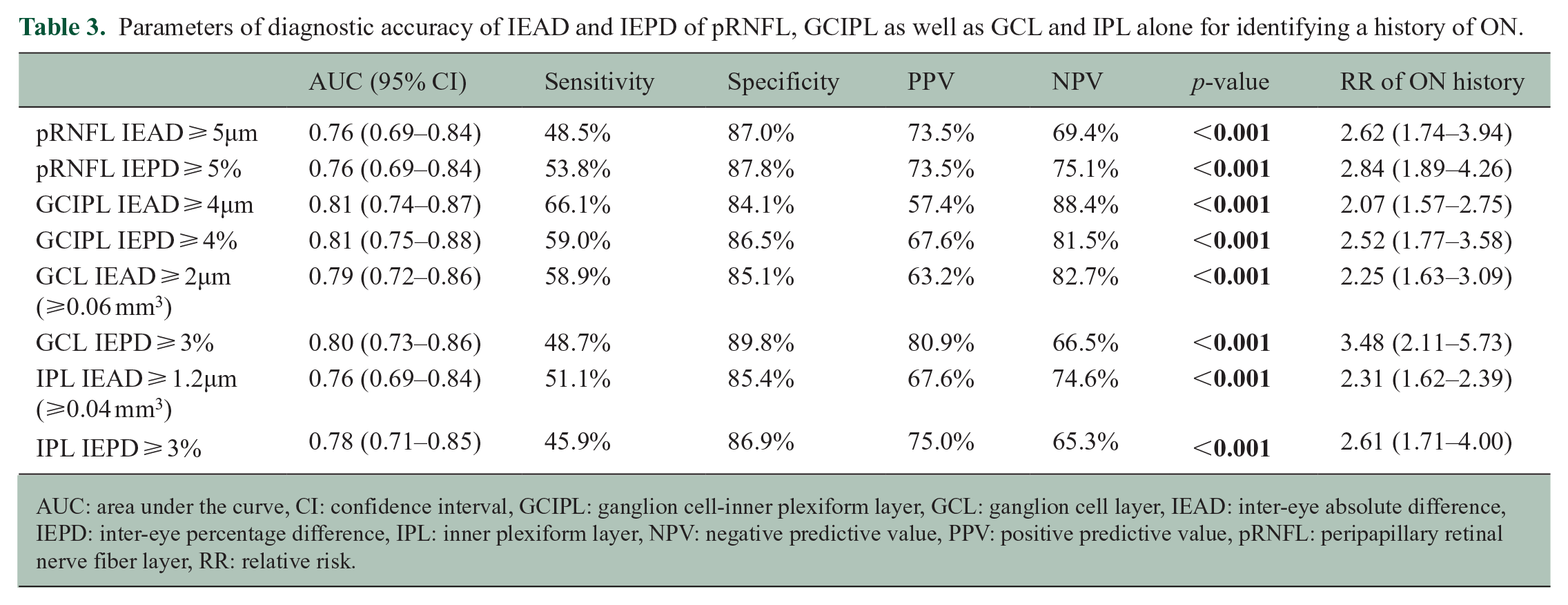
AUC: area under the curve, CI: confidence interval, GCIPL: ganglion cell-inner plexiform layer, GCL: ganglion cell layer, IEAD: inter-eye absolute difference, IEPD: inter-eye percentage difference, IPL: inner plexiform layer, NPV: negative predictive value, PPV: positive predictive value, pRNFL: peripapillary retinal nerve fiber layer, RR: relative risk.
Inter-eye relative difference
The AUC were 0.76 (cut-off ⩾ 5%, 53.8% sensitivity, 87.8% specificity, PPV 73.5%, NPV 75.1%) for pRNFL IEPD and 0.81 (cut-off ⩾ 4%, 59.0% sensitivity, 86.5% specificity, PPV 67.6%, NPV 81.5%) for GCIPL IEPD (Figure 2), indicating GCIPL IEPD as the superior model. Among patients with ON, 18 (26.5%) and 22 (32.4%) did not meet the cut-off values for pRNFL and GCIPL IEPD, respectively (Figure 3). For GCL and IPL IEPD, the corresponding AUC were 0.80 (cut-off ⩾ 3%, 48.7% sensitivity, 89.8% specificity, PPV 80.9%, NPV 66.5%) and 0.78 (cut-off ⩾ 3%, 45.9% sensitivity, 86.9% specificity, PPV 75.0%, NPV 65.3%), respectively, demonstrating superior diagnostic performance of GCL IEAD by improving both sensitivity and specificity (Figure 2). Among patients with ON, 13 (19.1%) and 17 (25.0%) did not meet the cut-off values for GCL and IPL IEPD, respectively (Figure 3). Both GCL or GCIPL IEPD seemed equally effective in detecting a history of ON (Table 3). Detailed diagnostic accuracy metrics are provided in Table 3.
A subanalysis, conducted by calculating the inner retinal layer thicknesses using the volumes of the inner and outer quadrants of the macula while explicitly excluding the fovea, demonstrated no significant change in the model’s accuracy (Suppl. Figure 1). Similarly, a sensitivity analysis excluding pwMS with treatment initiation before their first OCT scan did not significantly alter the overall results or the impact of individual variables.
Discussion
While OCT based IED in pRNFL and GCIPL thickness is a well-established marker of ON, in clinical routine, other OCT parameters are frequently used as a substitute for GCIPL, although data on comparative accuracy are scarce. Our findings demonstrate that GCL is a sensitive and specific alternative to GCIPL for identifying a history of ON, offering a viable option with minimal loss of diagnostic accuracy to facilitate implementation in clinical practice.
Historically, the optic nerve was not recognized as a region confirming dissemination in space in MS, which may have significantly delayed its diagnosis and treatment initiation, potentially leading to worse long-term visual and neurological outcomes. 19 However, several studies have provided sufficient evidence for the inclusion of the optic nerve in the 2024 revision of the McDonald criteria to address this gap.6,20,21 Due to the low contrast difference between GCL and IPL on OCT scans, these layers are often combined into a composite measure in most devices, GCIPL. 18 Yet, advances in image acquisition and automatic segmentation of retinal layers now allow for accurate separate quantification of GCL and IPL, eliminating the need for this composite approach. 22 Moreover, from a neuroanatomical perspective, GCL, housing the cell bodies of the third-order neurons in the visual pathway, is critical for assessing neuroaxonal loss following ON. The IPL, however, contains synapses of ganglion cells and various types of interneurons, including horizontal, bipolar, amacrine, Müller, and interplexiform cells. This complex network allows for the assessment of synaptic loss, as an early indicator of circuit dysfunction in MS that appears to precede disability progression. It has been shown to correlate strongly with serum biomarkers like glial fibrillary acidic protein (GFAP), highlighting the role of astrocytes and microglia in disease progression. 23
From a methodological perspective, focusing solely on the GCL offers practical advantages, particularly in light of the challenges posed by various OCT devices and segmentation algorithms. Some OCT devices come with rigid default settings that are challenging to adjust, making the manual summation of both the GCL and IPL layers both time-consuming and labor-intensive. This is especially crucial in clinical routine where efficiency is paramount. Simplifying the process by focusing solely on GCL analysis could streamline OCT assessments, improving clinical efficiency and enhancing patient care. Conversely, it would be advantageous for OCT manufacturers to provide automated GCIPL measurement through updated software, further streamlining the procedure.
Our findings indicate that both GCL and GCIPL can be reliably used in clinical practice to distinguish between patients with and without previous ON, thus establishing affection of the optic nerve. Conversely, IPL demonstrated slightly lower diagnostic accuracy compared to GCIPL, potentially increasing the risk of misdiagnosis in people with symptoms not suggestive of typical demyelination or non-specific MRI findings, especially in the presence of comorbidities, e.g. migraine or hypertension.24,25 Given the strong correlation between GCL and GCIPL, and the fact that GCL offers a good balance between sensitivity and specificity, GCL could be used instead of GCIPL where deemed necessary for implementation in routine clinical use.
Moreover, IEPD appeared to be more accurate in identifying ON, which is particularly important due to several key advantages. As a dimensionsless value, IEPD addresses the challenge of integrating data from different OCT devices and segmentation algorithms, which often vary across centers. 26 It also accounts for inter-eye interactions, simplifying statistical analyses by reducing the need for complex models. 18 In addition, IEPD may help overcome exclusions related to ophthalmological comorbidities, ethnic differences, and physiological variability, thereby enhancing its clinical applicability. These findings could aid in earlier diagnosis, which is critical for timely initiation of DMT and ultimately improving the long-term prognosis. In addition, we propose that a cut-off value of ⩾3% can be applied to GCL IEPD, offering comparable sensitivity with a slight trade-off in specificity to streamline its integration into clinical practice.
Although the underlying mechanism of inner retinal layer thinning in MS is not fully understood, it likely involves both direct damage from ON and retrograde transsynaptic axonal degeneration, which occurs secondary to axonotmesis within pathways directly or indirectly associated with the visual cortex. 27 Abnormal interocular asymmetry, however, is not specific for MS and may occur in other conditions, e.g. glaucomatous, ischemic or compressive optic neuropathy. Therefore, it is essential to rule out any plausible alternative causes by a comprehensive ophthalmological examination. Also, using abnormal interocular asymmetry on OCT to assess optic nerve involvement is not applicable in cases of bilateral ON, 28 which were excluded from the study. Its diagnostic accuracy can also be compromised if the time-interval between ON and OCT is too short, as neuroretinal loss is typically detected with a delay of 3–6 months after ON. However, in clinical practice, patients often report transient visual symptoms suggestive of previous ON. In such cases, the validated IED threshold can be utilized by OCT to objectively identify previous episodes of ON.
It is important to note that relying solely on OCT to detect optic nerve involvement in suspected MS cases can present significant challenges, particularly in patients without clinically confirmed ON or those with ophthalmological and neurological comorbidities. In addition, caution must be exercised when interpreting OCT findings in older adults and nonwhite populations, as these groups have a higher prevalence of other inflammatory (e.g. neuromyelitis optica spectrum disorder and neurosarcoidosis) and non-inflammatory conditions (e.g. ischemic optic neuropathy). This emphasizes the necessity of applying diagnostic criteria within the appropriate clinical context to avoid potential misdiagnosis.
This study has some limitations that should be acknowledged. The retrospective study design introduces a number of potential biases, though these were partly mitigated by the detailed and standardized patient characterization. Still, replication of our data in an independent cohort is needed for confirmation. An additional limitation of our study is the potential floor effect due to the absence of minimal inner retinal layer thickness as part of the exclusion criteria. To mitigate this, we calculated IEPD rather than IEAD, as the former is less sensitive to the floor effect. We emphasize that the study cohort almost exclusively consists of patients of Caucasian origin, limiting applicability to other ethnicities. A notable limitation of our study is the inherent difficulty in precisely segmenting the GCL and IPL, a challenge that has led previous research teams to adopt a composite measure. While the layers were automatically segmented without manual error correction, this approach enhanced the generalizability of our results and minimized potential bias from the rater’s experience. Furthermore, our results are not directly applicable to other OCT devices, although previous findings suggest that retinal layer thickness thresholds might be independent of the OCT manufacturer. Importantly, it appears that the separate reporting of GCL and IPL thicknesses is a feature unique to the Spectralis platform. In contrast, other platforms, such as Cirrus, provide a combined GCIPL thickness in their reports, thereby addressing a key limitation highlighted in this study. Moreover, our study did not evaluate the model’s performance in patients with subclinical ON, thereby restricting the generalizability of our findings in this clinically highly relevant population. Furthermore, our study did not include other visual function assessments, such as low-contrast letter acuity and visual evoked potentials (VEP). Finally, our findings depend on rigorous quality control of OCT scans and careful exclusion of confounding ophthalmologic pathologies such as severe myopia and optic disk drusen. These potential sources of error may be more pronounced in real-world settings where OCT devices and protocols may vary.
Conclusion
In conclusion, GCL IEAD ⩾ 2µm (0.06 mm3) and, more notably, IEPD ⩾ 3% are reliable cut-off values for identifying unilateral ON, demonstrating non-inferiority to the combined GCIPL IEAD/IEPD in identifying a history of ON. With the inclusion of the optic nerve as a fifth region in the revised McDonald criteria 2024 to assess dissemination in space, our study provides robust evidence that GCL IEAD/IEPD can be used to identify a history of ON to facilitate implementation in clinical routine.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251332895 – Supplemental material for Diagnostic accuracy of inter-eye difference of ganglion cell layer alone in identifying optic neuritis in multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585251332895 for Diagnostic accuracy of inter-eye difference of ganglion cell layer alone in identifying optic neuritis in multiple sclerosis by Nik Krajnc, Fabian Föttinger, Markus Ponleitner, Barbara Kornek, Fritz Leutmezer, Stefan Macher, Paulus Rommer, Christiane Schmied, Karin Zebenholzer, Gudrun Zulehner, Tobias Zrzavy, Thomas Berger, Berthold Pemp and Gabriel Bsteh in Multiple Sclerosis Journal
Footnotes
Data availability
Data supporting the findings of this study are available from the corresponding author upon reasonable request by a qualified researcher and upon approval by the data-clearing committee of the Medical University Vienna.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NK has participated in meetings sponsored by, received speaker honoraria or travel funding from Alexion, BMS/Celgene, Janssen-Cilag, Merck, Novartis, Roche, and Sanofi-Genzyme and held a grant for a Multiple Sclerosis Clinical Training Fellowship Program from the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS). FF has participated in meetings sponsored by, received speaker honoraria or travel funding from Novartis. MP has participated in meetings sponsored by, received speaker or consulting honoraria or travel funding from Amicus, Merck, Novartis, and Sanofi-Genzyme. BK has received honoraria for speaking and for consulting from Biogen, BMS-Celgene, Johnson & Johnson, Merck, Novartis, Roche, Teva, and Sanofi-Genzyme outside of the submitted work. No conflict of interest with respect to the present study. FL has participated in meetings sponsored by, received speaker honoraria or travel funding from Actelion, Almirall, Biogen, Celgene, Johnson & Johnson, MedDay, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva, and received honoraria for consulting Biogen, Celgene, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. SM declares no conflict of interest relevant to this study. PR has received honoraria for consultancy/speaking from Alexion/Astra Zeneca, Allmiral, Amgen/Horizon, Amicus, Biogen, Merck, Novartis, Roche, Sandoz, Sanofi has received research grants from Amicus, Biogen, Merck, Roche. CS declares no conflict of interest relevant to this study. KZ received speaking honoraria or travel grants from Biogen, Celgene/BMS, Novartis, and Sanofi-Genzyme. GZ has participated in meetings sponsored by or received travel funding from Biogen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. TZ has participated in meetings sponsored by or received travel funding from Biogen, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. TB has participated in meetings sponsored by and received honoraria (lectures, advisory boards, consultations) from pharmaceutical companies marketing treatments for MS Allergan, Bayer, Biogen, Bionorica, BMS/Celgene, Genesis, GSK, GW/Jazz Pharma, Horizon, Janssen-Cilag, MedDay, Merck, Novartis, Octapharma, Roche, Sandoz, Sanofi-Genzyme, Teva, and UCB. His institution has received financial support in the past 12 months by unrestricted research grants (Biogen, Bayer, BMS/Celgene, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva and for participation in clinical trials in multiple sclerosis sponsored by Alexion, Bayer, Biogen, Merck, Novartis, Octapharma, Roche, Sanofi-Genzyme, and Teva. BP has received honoraria for consulting from Novartis, has received honoraria for advisory boards/consulting from Chiesi and GenSight, and has received speaker honoraria from Novartis, Chiesi and Santen. GB has participated in meetings sponsored by, received speaker honoraria or travel funding from Biogen, Celgene/BMS, Lilly, MedWhizz, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva, and received honoraria for consulting Biogen, Celgene/BMS, Merck, Novartis, Roche, Sanofi-Genzyme, and Teva. He has received unrestricted research grants from Celgene/BMS and Novartis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Austrian MS Research Society.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
