Abstract
Background:
Uncertainty about disproportionate impact on health care budgets limits implementation of early highly effective treatment (EHT) in multiple sclerosis (MS).
Objective:
To estimate cost-effectiveness of escalation versus EHT disease-modifying treatment (DMT) sequences.
Methods:
Using a health-economic approach, we analysed health benefits (relapse rate reduction, disability prevention), direct/indirect DMT and societal costs of escalation versus EHT DMT sequences. In scenario analyses, we allowed (1) earlier use of alemtuzumab (ALE) and (2) a single retreatment with cladribine (CLA).
Results:
In our model, we showed that the ratio between costs and quality-adjusted life years (QALYs) for the most cost-effective EHT and escalation sequence results into a similar net health benefit with higher costs and also higher QALYs associated with an EHT versus escalation strategy. Earlier use of ALE is more cost-effective than in later lines, even when aggravating the impact of its side-effects tenfold. Retreatment with CLA was more cost-effective in both escalation and EHT sequences.
Conclusions:
Certain EHT sequences are equally cost-effective to escalation sequences and are likely to result in more health at uncertain additional costs. The favourable cost–benefit ratio of CLA and ALE suggests that a wider application of affordable highly effective therapies could promote the cost-effectiveness both EHT and escalation approaches.
Keywords
Introduction
The majority of people with multiple sclerosis (pwMS) will be confronted with therapeutic sequencing of disease-modifying treatments (DMTs). The aim of sequencing is to maximize disease control and thus lifetime quality-adjusted life years (QALYs) and minimize risks based on the mechanism of action, side effects, pharmacokinetic and pharmacodynamic properties of each individual therapy. In practice, there is huge variation in treatment pathways, and the most common treatment approaches are a strategy of escalation and early highly effective treatment (EHT).1,2 Escalation approaches are more risk averse in terms of side-effects, as they start with a first-line low-moderate efficacy DMTs with favourable risk profile. If there are signs of disease breakthrough, they switch to a second-line agent that is considered more effective at the expense of a mostly small to medium increase in risks. If effective, pwMS stay on these safer medications. However, this strategy carries a risk of therapeutic inertia and thus disability accrual with lower lifetime QALYs as observational studies show that early initiation of more effective DMTs prevents neuronal damage later on.3–5 As randomized trials typically assess efficacy of individual drugs rather than treatment strategies, and assessment of cost-effectiveness often erroneously assumes lifetime DMT use, regulators struggle with reimbursement criteria. In European health care systems, access to newer highly effective DMTs is often restricted to later disease stages despite broader regulatory labels. Hence, whether drugs are considered ‘second line’ or ‘highly-effective’ is often based on reimbursement schemes and thus arbitrary. International experts advocate to facilitate early access to highly effective drugs.6,7 The reluctance of regulators is mostly driven by the considerable cost. Several studies have shown that DMTs are the largest cost driver in multiple sclerosis (MS) with striking inflation of drug prices over the past decade.8,9 However, it has not been thoroughly assessed to what extent the reduction in disability seen with EHT translates into societal savings and counterbalances the increased direct costs (i.e. drug costs) that are inherent to the newer agents. Within the EHT strategy, immune reconstitution therapies (IRTs), such as cladribine (CLA) and alemtuzumab (ALE) requiring two treatment cycles rather than continued use are highly effective therapies which at least in theory can be hypothesized to front-load both costs and risk of adverse events (AEs). In this study, we used a health-economic approach to differentiate escalation versus EHT DMT sequences in terms of health benefits (relapse rate reduction, disability prevention), direct DMT costs alongside indirect and societal costs for the care of pwMS. Subsequently, to explore the impact of affordable highly effective IRT within the model, we simulated the impact on cost-effectiveness of using ALE as a third- or second-line option both with and without aggravating the health impact of its autoimmune side-effects and of allowing retreatment with CLA.
Methods
We compared the benefits of escalation and EHT DMT sequences in terms of health outcomes and cost-effectiveness using the ErasmusMC/iMTA MS model.10,11 The model estimates benefits of treatments by combining relative efficacy of DMTs from clinical trial populations with background probabilities from registry data from British Columbia MS Historical Database. While the background probabilities are a heterogeneous sample of MS patients, clinical trials are not. Following this method, our findings are most applicable on the average MS patient for which efficacy of DMTs has been established (i.e. relatively young, predominantly female treatment-naïve relapsing MS). However, the model takes into account natural history events, such as age-associated decrease of annual relapse rate and increased risk of disability accumulation. The principles of modelling treatment sequences in MS and the clinical decision rules underlying this model have been published elsewhere.10–12
DMT sequences
We only included clinically plausible DMT sequences (i.e. excluding switches with the same mode of action). pwMS switch to another DMT when experiencing AE or disease activity (Expanded Disability Status Scale (EDSS)-based progression or relapse) (Supplemental Table S1). Magnetic resonance imaging (MRI) activity was not part of the decision-analytic model as the implications of MRI activity for treatment switches are heterogeneous and detailed MRI data are currently unavailable. In lack of a generally accepted definition, we defined the following treatment strategies reflecting the Dutch setting.
Escalation
We defined escalation strategies as DMT sequences escalating from less to more efficacious drugs based on disease breakthrough activity. Interferon beta (INFB), dimethyl fumarate (DMF) 240 mg, teriflunomide (TER) 14 mg, glatiramer acetate (GLA) 20 mg, ponesimod (PON) 20 mg, ozanimod (OZA)1 mg and fingolimod (FIN) 0.5 mg were included as first-line options as common practice in the Dutch setting. As intolerable side effects occur frequently on first-line DMTs (line 1a), a second first-line DMT was included in the model (line 1b). The probability to switch treatment because of side effects was calculated based on the discontinuation rates in the DMT versus placebo arm of the respective clinical trials. pwMS who experience disease activity on line 1a will have a probability to directly switch to line 2. Second-line options (lines 2 and 3) included PON, OZA, FIN, CLA 3.5 mg/kg, ocrelizumab (OCR) 600 mg, ofatumumab (OFA) 20 mg and natalizumab (NAT) 300 mg. ALE 12 mg was restricted to a last line of treatment (line 4), and retreatment was allowed once if disease activity occurred at least 3 years after the last treatment course. In total, this resulted in 504 escalation strategies.
Early HET
In an EHT strategy, pwMS start with CLA, OCR, OFA or NAT in line 1, followed by other high-efficacy DMTs (NAT, CLA, OCR, OFA and ALE) in lines 2–4. To guarantee comparability with escalation strategies, ALE is also restricted as a last-line therapy allowing retreatment once. In total, this resulted in 12 DMT sequences.
Scenario analyses
The base case scenario analyses cost-effectiveness of an escalation versus EHT treatment approach. In the first-scenario analysis, we allowed earlier treatment with ALE (i.e. in line 2 and 3). This resulted in 2,808 escalation and 36 EHT sequences. In a sensitivity analysis of this ‘early ALE’ scenario, the quality-of-life loss due to autoimmune thyroid events was increased tenfold (i.e. from −0.05 to −0.5 during the first year of treatment). This increase corresponds to the quality-of-life decline from EDSS 0 to 7–8. In the subsequent years, we decreased the base case parameter from −0.02 to −0.2 which compares to quality-of-life decline from EDSS 0 to EDSS 2–3. In the second-scenario analysis, we arbitrarily allowed one single retreatment with CLA when disease activity occurred at least 3 years after the last treatment course.
Input data
Efficacy, in terms of annualized relapse rates and 24-week confirmed disability progression (CDP), of DMTs was based on network meta-analyses (NMA) 12 and assumed constant irrespective of first- or second-line position in the DMT sequence. The NMA included data of 77 trials in relapsing MS comprising a total of 26,096 participants followed for 22,159.1 person-years. Detailed methods of this NMA have been included in the Supplemental material of our previous work. 12 Analyses were performed from a societal perspective and included costs to cover the DMTs, other health care use (i.e. DMT administration, monitoring, inpatient care, day admissions, tests, other drugs, relapses and DMT switches), productivity loss using the friction cost method and informal care of pwMS. Drug acquisition costs of DMTs were based on the most recent (2022) publicly available list prices in the Netherlands (Table 1) and can be found in the Supplementary data of our previous work. 12 In accordance with the Dutch guideline for conducting economic evaluations in health care, costs and QALYs were discounted with 4% and 1.5% accounting for the diminishing value of future costs and benefits, respectively. 13
Comparison of costs and effects of first-line therapies in the most cost-effective escalation sequence. The most cost-effective treatment based on NHB is indicated in bold, and differences in costs and QALYs for other sequences are indicated relative to this sequence. As a preference for line 1b is difficult to assert, we did not specify line 1b treatment.
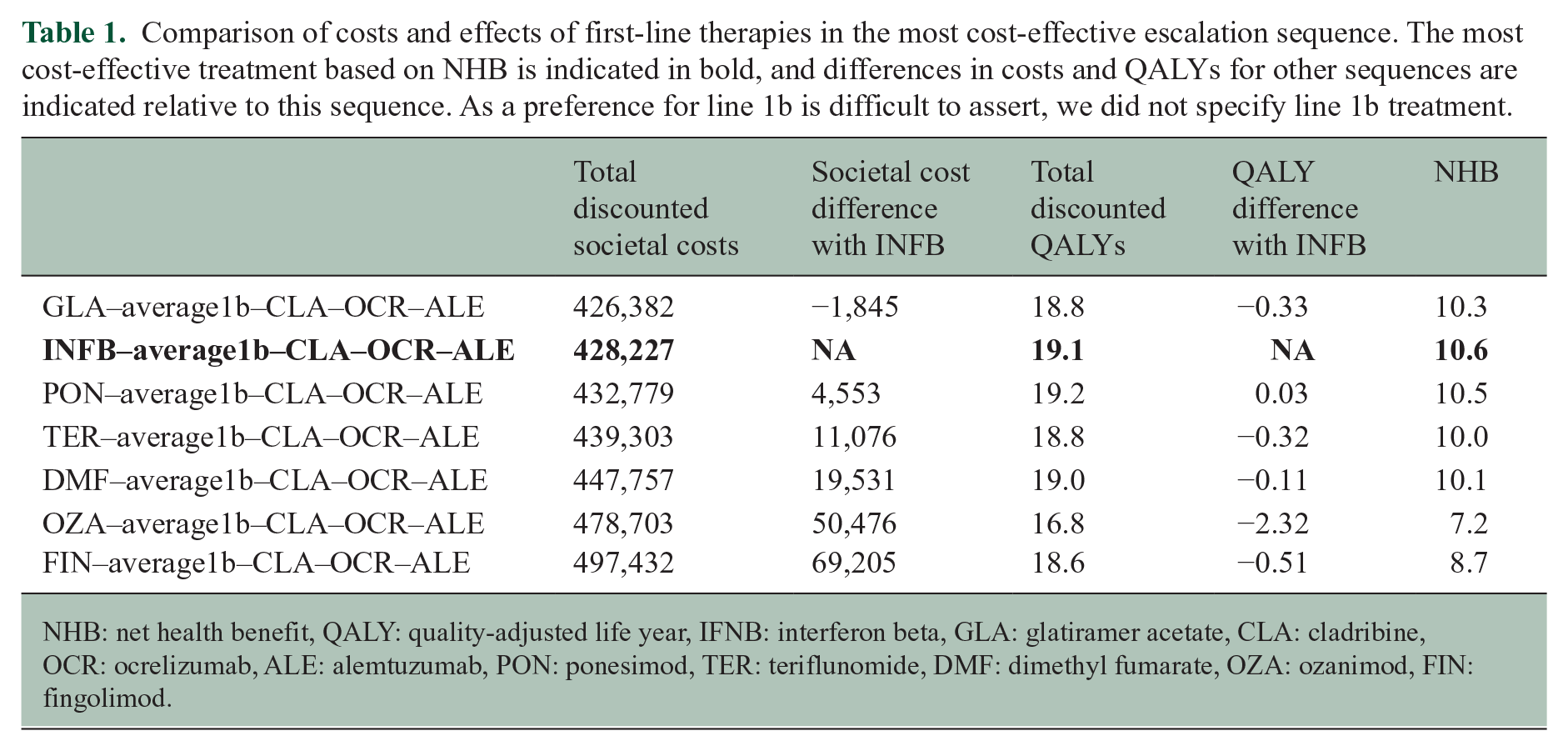
NHB: net health benefit, QALY: quality-adjusted life year, IFNB: interferon beta, GLA: glatiramer acetate, CLA: cladribine, OCR: ocrelizumab, ALE: alemtuzumab, PON: ponesimod, TER: teriflunomide, DMF: dimethyl fumarate, OZA: ozanimod, FIN: fingolimod.
Health-economic analyses
The model simulated 10,000 pwMS per DMT sequence and the outcomes represent the average costs and QALYs of this virtual population. We ranked escalation and/or EHT sequences according to net health benefit (NHB) which is a rewritten form of the more common incremental cost-effectiveness ratio (ICER). However, as it is not a ratio, it is better suited to rank treatments. NHB reflects the cost-effectiveness of a treatment and expresses the net benefit of a treatment in terms of QALYs, adjusted for the cost given the value of a QALY (NHB = QALYs – (costs/cost-per-QALY threshold)). The appropriate cost-per-QALY threshold for pwMS in the Netherlands is €50,000.
For both scenario analyses, we estimated the uncertainty of our findings. The large number of escalation strategies prohibits meaningful probabilistic sensitivity analysis (PSA) over all sequences. Therefore, uncertainty around the most cost-effective escalation sequences is illustrated graphically by visualizing DMTs that rank in the top 10% of cost-effectiveness (i.e. NHB). For comparisons across EHT sequences, all model parameters (such as the efficacy of DMTs and cost estimates of health care utilization) were varied simultaneously by sampling from their distributions in a PSA. The PSA was performed for the most cost-effective EHT sequences starting with CLA, OCR, OFA or NAT and for the most cost-effective escalation versus EHT sequences. We conducted the PSA with 500 sampling iterations while sampling 1000 pwMS per DMT sequence.
Results
Cost-effectiveness of escalation strategies
Figure 1 shows that the top 10% of most cost-effective escalation sequences start with IFNB, PON or GLA. No particular preference can be found for line 1b, although FIN and OZA are rarely present. CLA appears most often in the second line. OCR, OFA and NAT appear often in line 3 and are therefore treatments of similar NHB. Disregarding uncertainty, the most cost-effective escalation sequence starts with an IFNB, followed by PON when experiencing side effects on IFNB, with pwMS switching subsequently to CLA, OCR and ALE in case of disease activity or side effects on the prior line of treatment. This sequence yields 19.2 QALYs, and € 427,702 total societal costs (of which 203,693 drug costs) and results in 4.6 lifetime relapses and a time to EDSS 6 of 23.8 years (Table 1). The NHB of this sequence is 10.64. However, numeric differences in NHB between the top 10% sequences are small (10.64 vs 10.11 for the first vs 50th highest ranking). The full ranking is available in the Supplemental Material. Alternatives to IFNB in the first line are more expensive except for GLA. Among the S1PR modulators, PON is the only treatment that is more costly (€ 4,553) and also yields more QALYs (0.03) compared to IFNB. Oral DMTs (TER and DMF) are € 11,076 and € 19,531 more costly without yielding more QALYs, respectively.
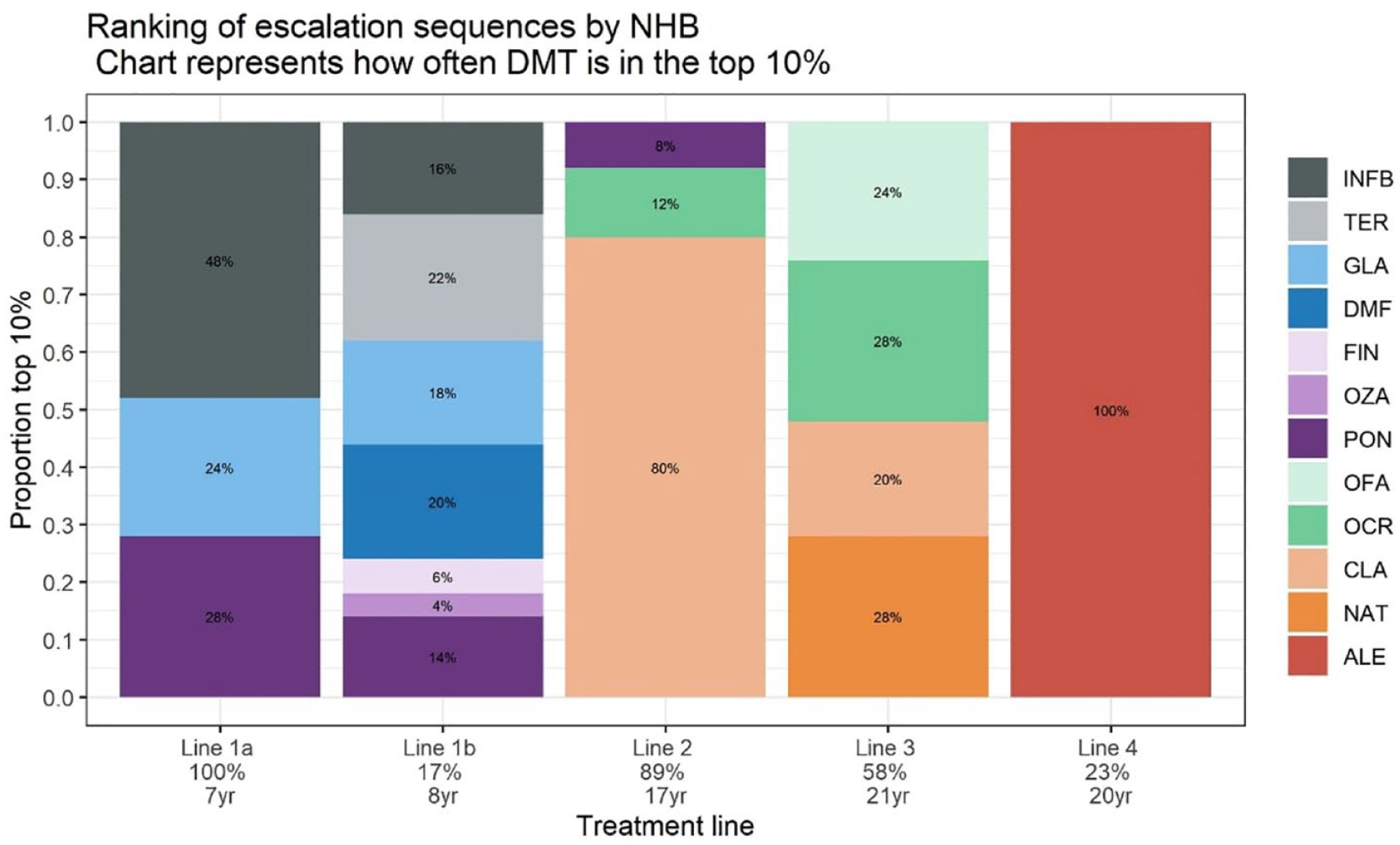
Visualization of the top 10% DMT escalation sequences ranked by cost-effectiveness. The model simulated 10,000 pwMS per DMT sequence with 504 DMT sequences representing escalation treatment approaches. The percentages in the vertical bars represent the proportion of sequences containing that specific DMT in a certain line in the top 10% DMT sequences ranked based on cost-effectiveness based (i.e. NHB). The percentage below the line numbers on the x-axis represents the proportion of the total cohort that receives the specific line. The duration in years below the line numbers represents the average duration of treatment in the specific line.
Cost-effectiveness of early HET strategies
The most cost-effective EHT strategy starts with OCR, with pwMS subsequently switching to CLA, NAT and ALE. This sequence yields 20.4 QALYs, and € 494,703 total societal costs (of which € 290,094 drug costs) and results in 3.9 life-time relapses and a time to EDSS 6 of 26.6 years. The NHB of this sequence is 10.55 (Supplemental Material). Numeric differences between all 12 sequences are not large ranging from 9.42 to 10.55. The cost-effectiveness plane in Figure 2 shows that there is considerable uncertainty about the most cost-effective first-line DMT in an EHT treatment approach without a clear preference for CLA, NAT, OCR or OFA. However, they all have in common that the first- or second-line treatment is CLA indicating that including CLA early in an EHT sequence is cost-effective.
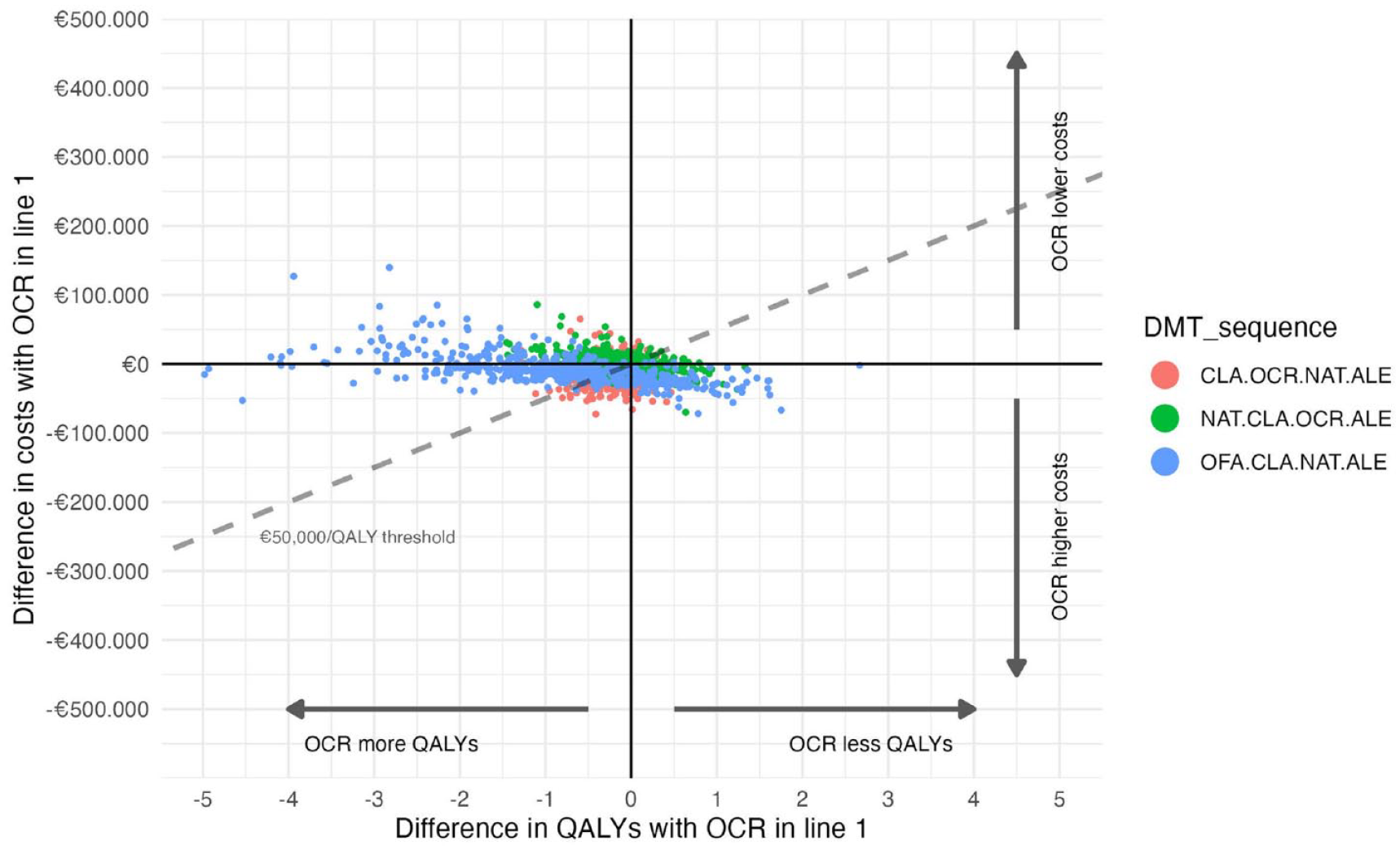
Cost-effectiveness plane of the most cost-effective early highly effective treatment sequence. DMT sequences starting with CLA, NAT or OFA compared to the most cost-effective DMT sequence with an early highly effective treatment approach (i.e. OCR–CLA–NAT–ALE).
Base case scenario: cost-effectiveness of escalation versus early HET strategies
The most cost-effective escalation (INFB–PON–CLA–OCR–ALE) and EHT (OCR–CLA–NAT–ALE) sequence have a similar NHB, 10.64 and 10.55, respectively. Differences between the two are uncertain as shown in the PSA (Figure 3). Although the EHT sequence yields more QALYs, it is uncertain if this DMT sequence is less or more expensive than the escalation sequence. The probability that the EHT sequence is cost-effective compared to the escalation sequence at a cost per QALY threshold of € 50,000 is 46.2% (i.e. percentage of all dots below the € 50,000/QALY threshold line in Figure 3). This is only a small difference with 50%, which would suggest absolute indifference between the two DMT sequences.
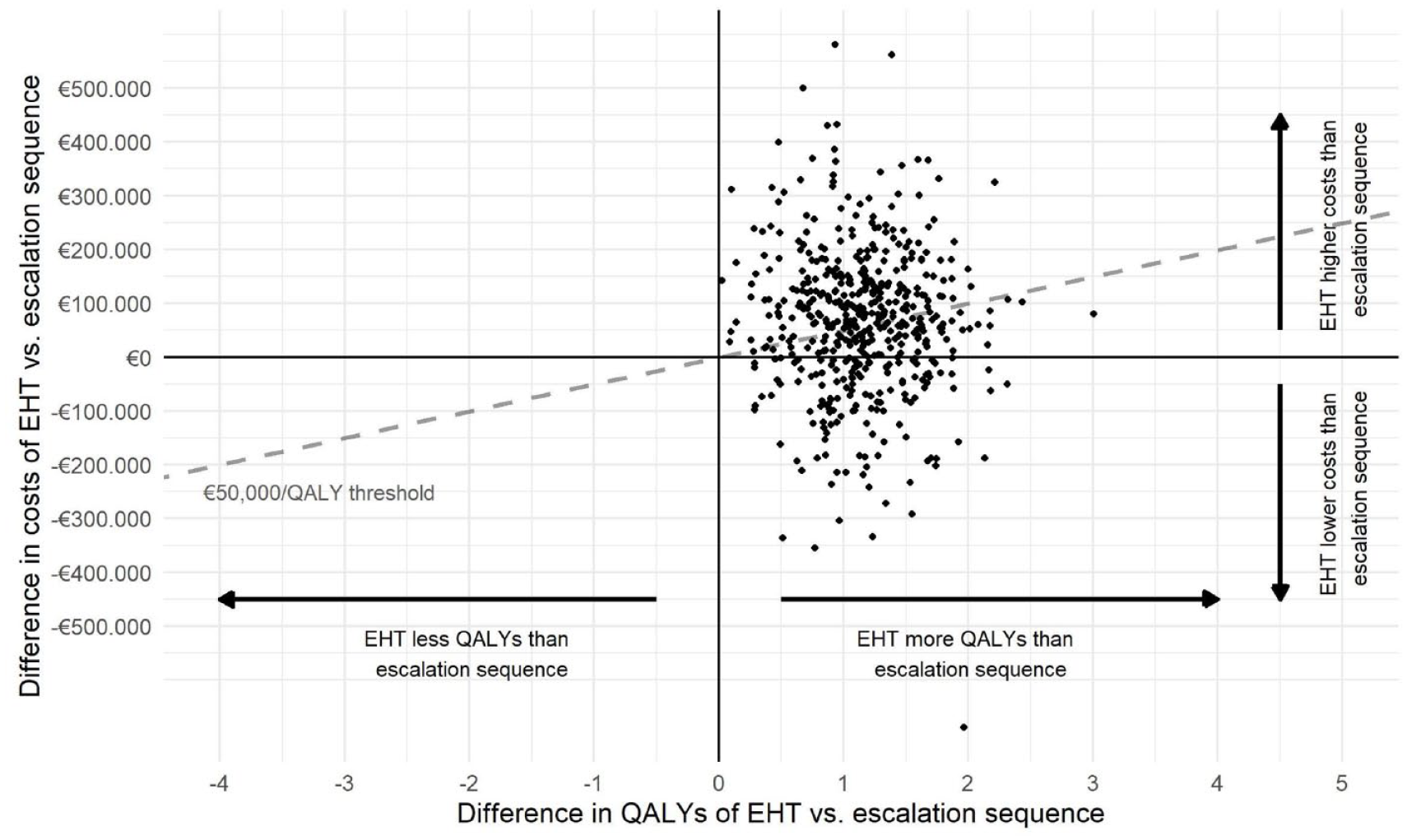
Cost-effectiveness plane of the most cost-effective escalation versus early highly effective treatment sequence. INFB–PON–CLA–OCR–ALE is the most cost-effective escalation sequence and OCR–CLA–NAT–ALE is the most cost-effective EHT sequence.
First scenario: earlier treatment with ALE in early highly effective and escalation sequences
The average cost-effectiveness of the EHT and escalation sequences with ALE in line 4 (base case analysis) or earlier in lines 2 and 3 are shown in Tables 2 and 3, respectively. Interestingly, allowing ALE in line 2 is more cost-effective (i.e. higher NHB) in both treatment strategies compared to the base case analyses. We conducted a sensitivity analysis in which the quality-of-life reduction of an autoimmune thyroid event is set at an extremely high level. In that analysis, the NHB of earlier ALE use in line 2 for escalation (IFNB–GLA–ALE–CLA–OFA) was 12.18 and 11.77 for EHT (OCR–ALE–CLA–NAT). This suggests that for both the escalation and EHT strategies, earlier ALE use is more cost-effective than later line ALE even with aggravated side effects for ALE, as the NHB of the scenario is higher than that of the base-case with later line ALE use.
Cost-effectiveness of first-scenario analysis allowing earlier treatment with alemtuzumab in early highly effective treatment strategy. Average cost-effectiveness outcomes of all possible DMT sequences in which alemtuzumab is used in line 2, 3 or 4 (base-case analysis) in an early highly effective setting.
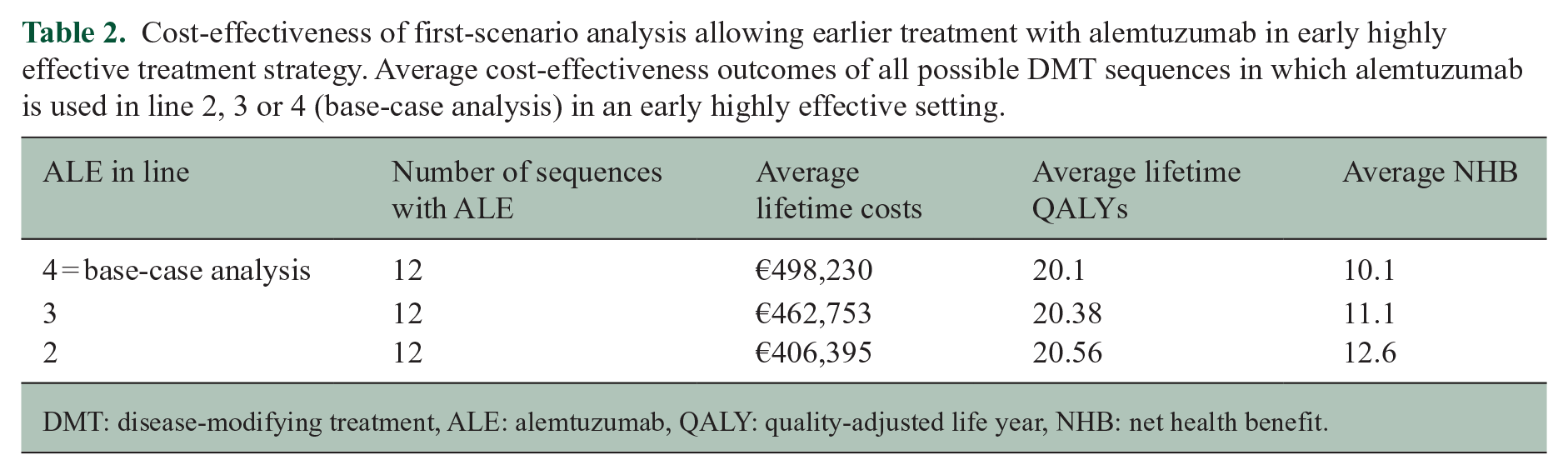
DMT: disease-modifying treatment, ALE: alemtuzumab, QALY: quality-adjusted life year, NHB: net health benefit.
Cost-effectiveness of first-scenario analysis allowing earlier treatment with alemtuzumab in escalation treatment strategy. Average cost-effectiveness outcomes of all possible DMT sequences in which alemtuzumab is used in line 2, 3 or 4 in an escalation setting.
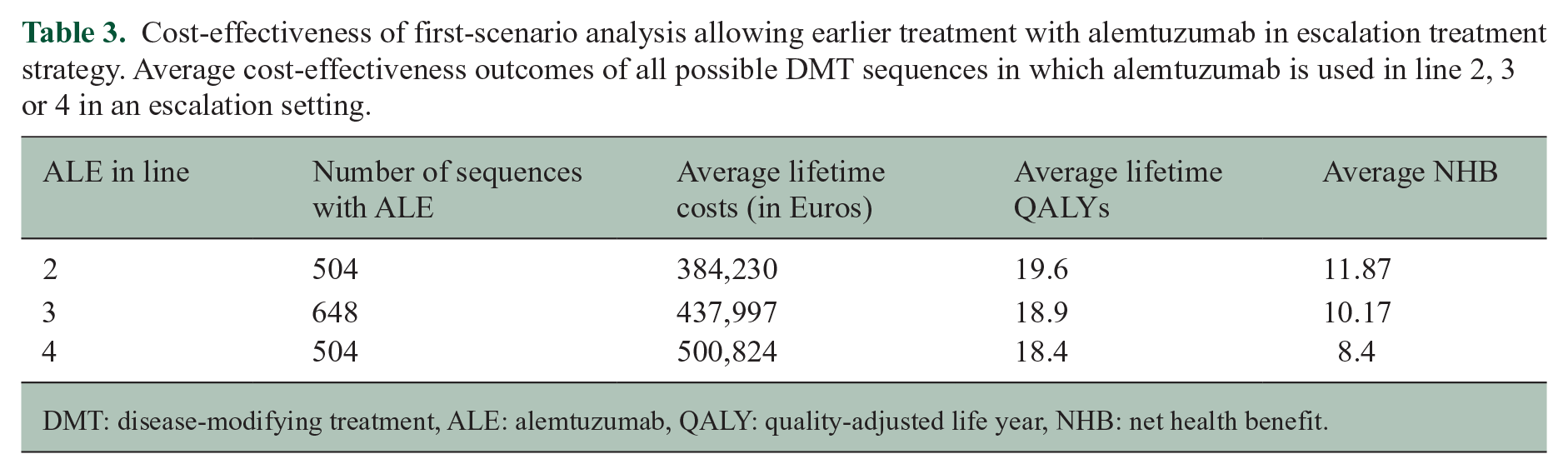
DMT: disease-modifying treatment, ALE: alemtuzumab, QALY: quality-adjusted life year, NHB: net health benefit.
Second scenario: retreatment with CLA
In both escalation and EHT sequences, allowing retreatment with CLA was more cost-effective (i.e. higher NHB) than the base-case scenario without retreatment with CLA (assuming equal efficacy) (Table 4). Allowing retreatment with CLA did result in less lifetime QALYs compared to the base-case as retreatment delays the switch to more effective and also more costly DMTs. However, this reduction in QALYs was offset by a reduction in costs resulting in a higher NHB of sequences with CLA retreatment.
Cost-effectiveness of second-scenario analysis allowing retreatment with cladribine. Average difference in cost-effectiveness of DMT sequences with cladribine retreatment compared to base-case scenario without cladribine retreatment.
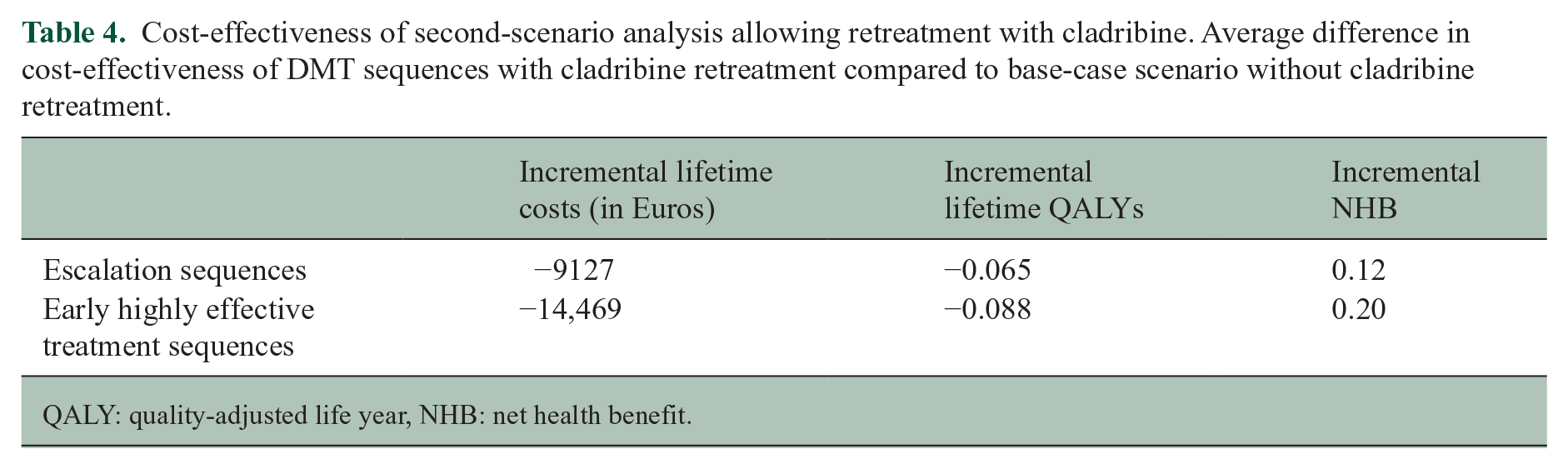
QALY: quality-adjusted life year, NHB: net health benefit.
Discussion
In this health-economic modelling study, we looked at the most cost-effective DMT sequences when starting escalation versus EHT strategies in pwMS. We showed that the ratio between costs and QALYs for the most cost-effective EHT and escalation sequence results into a similar NHB with higher costs and also higher QALYs associated with an EHT versus escalation strategy. This implies that certain EHT sequences have an equal likelihood of being cost-effective as certain escalation sequences. Against expectations, cost-effectiveness is thus not a defining criterion between both treatment strategies. Importantly, the QALY gains seen with EHT are considerable. The most cost-effective EHT strategy gains a little over 1 QALY versus its escalation counterpart. Although UK health benefits are discounted at a higher rate compared to the Netherlands, an analysis of 129 drugs approved on the National Health Service found 0.27 median incremental QALYs of new drugs relative to the best available alternative. 14 Nonetheless, EHT strategies carry a risk of overspending health care budgets as shown in the probabilistic analysis. There is a 53.8% chance that the EHT strategy produces more QALYs but at unacceptable costs. Hence, prescribing EHT requires accepting this risk of overspending. For both EHT and escalation strategies, the NHB improves when CLA is used in earlier treatment lines, suggesting this is an important strategy to improve cost-effectiveness. Alternatively, cheaper drug prices for other HETs would significantly improve cost-effectiveness while maximizing health gain at the same time in the model. The currently upcoming generic first-line drugs and biosimilars of highly effective drugs have an uncertain societal economic impact in the Dutch health care system because it is possible that savings realized at the hospital level are not translated to health care insurance companies at a societal level. Regardless of this uncertainty, the obvious candidate to tip this balance in favour of EHT is rituximab. The drug has recently been incorporated in the World Health Organization (WHO) 15 essential medicine list for treatment of MS and has a list price that is about one fifth of OCR. In a previous study, we have shown that treatment with rituximab would already be cost-effective when its efficacy on disability progression would match the effect of first-line therapies. 16 In addition, the advent of biosimilars might significantly alter the equipoise between escalation and EHT. Robust evidence of effectiveness and equivalence has already been demonstrated for NAT and we are currently awaiting its official list price. 17
The disconnect between drug costs and long-term societal savings is an important barrier to prioritize cost-effective DMT strategies in practice. Pharmacoeconomic considerations of traditional payers (i.e. health care department, insurance companies) and risk assessment of regulating bodies lead to discrepancies between the regulatory label and clinical use of DMTs. Our most cost-effective EHT sequence starting with OCR, which is equally cost-effective as the most cost-effective escalation strategy, is not reimbursed in many European countries. 18 On the other hand, the European Medicine Agency restricted the use of ALE in 2019 based on the occurrence of rare cardiovascular side effects whereas high efficacy of this drug early in the disease has been clearly shown.19–21 In addition, the use of ALE has further declined because of reduced tolerance for its side effects by clinicians and pwMS with the advent of alternative HETs. However, qualitative research convincingly shows that autoimmune thyroid events do not offset the overall gain in quality-of-life. 22 To accommodate individual variability in risk trade-offs, we demonstrated that the health benefits of ALE in terms of preventing disability accrual outweigh even in the most pessimistic scenario the burden from its autoimmune side effects. Admittedly, the impact of cardiovascular side effects was not assessed. However, given the rarity of these events 21 and the very extreme increase in quality of life we modelled secondary to a frequent AE, these are unlikely to impact on the conclusions.
Cost-effectiveness of DMT sequencing is hampered by lack of trial and real-world data on how to deal with disease breakthrough after IRTs.23,24 Among the EHT strategies in our model, CLA, which is the quintessential example of an IRT, was overrepresented in first or second line suggesting that it offered the best balance between costs and health benefits. This is not unexpected as its use is limited to two treatment cycles with a low monitoring burden after the second cycle. 25 Cost-effectiveness studies with a more limited scope focussing on CLA versus ALE/NAT or versus FIN in pwMS with high disease activity drew the same conclusion.26,27 In the scenario analyses, cost-effectiveness in both escalation and EHT strategies improved when conservatively allowing retreatment with CLA once in case of disease breakthrough at least 3 years after the second cycle. This is in line with recent expert opinion on the matter. However, remaining knowledge gaps on cumulative dose and safety will need to be filled before widespread adoption in practice will take place.23,24 Importantly, also for anti-CD20 there is preliminary evidence that it could be used as an IRT.28,29
We acknowledge that our model simulates current practice of DMT sequencing beyond a time frame for which real-world data exist. In the top 10% most cost-effective escalation sequences, the model predicts that pwMS would take between 45 and 53 years to cycle through treatment lines 1, 2 and 3. If there is disease activity after this period, 23% of the population would move to ALE (line 4) in the base-case. While it may seem unlikely, it is not possible to verify our simulation as EMA approval for the first DMT (IFNB) was in 1995 (i.e. 28 years ago). Also, as all fourth-line treatments were ALE and as this is equal for all sequences in the base-case, it does not affect the relative ranking between sequences in the base-case. Furthermore, with the expansion of the DMT armamentarium both neurologists and pwMS have developed reduced tolerance for side effects of first-line injectables that might not be reflected by the decision rules applied in the model. Nonetheless, if pwMS sequence faster to a more cost-effective second line, such as CLA, the NHB would only increase keeping first-line injectables as a favourable first option. Finally, this study’s results for the Dutch setting may not be transferable to other countries due to country-specific DMT list prices and switching practices. In addition, DMT switches because of insurance coverage and formulary changes have not been taken into account. Nevertheless, the principles of weighing benefits of treatments against their costs are not and are of increasing relevance with the continuously rising costs of MS treatment.
In conclusion, the expanding DMT landscape translates into ample variation in DMT sequences with absence of prospective comparative studies guiding the optimal long-term treatment strategy, especially after IRT. Although pharmacoeconomic considerations and risk assessments facilitate escalation strategies in clinical practice, our model taking into account costs and benefits shows there is currently no certain difference between the most cost-effective escalation and EHT strategies. Importantly, this would be significantly altered by cheaper highly effective drugs, such as off-label CD20 monoclonals or biosimilars.
Supplemental Material
sj-docx-1-msj-10.1177_13524585241258692 – Supplemental material for Benefits of early highly effective versus escalation treatment strategies in relapsing multiple sclerosis estimated using a treatment-sequence model
Supplemental material, sj-docx-1-msj-10.1177_13524585241258692 for Benefits of early highly effective versus escalation treatment strategies in relapsing multiple sclerosis estimated using a treatment-sequence model by Ide Smets, Matthijs Versteegh, Simone Huygens, Beatrijs Wokke and Joost Smolders in Multiple Sclerosis Journal
Footnotes
Data availability statement
Data are available at the corresponding author upon reasoneable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: B.H.A.W. declares no conflict of interest. S.A.H. and M.M.V. are shareholders of Huygens & Versteegh, which conducts research for government organizations and pharmaceutical companies, including research in MS. I.S. has received honoraria from Merck, Biogen Idec and Sanofi. J.S. received lecture and/or consultancy fee from Biogen, Merck, Novartis, Roche, and Sanofi Genzyme.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.A.H. and M.M.V. have received funding from Merck for MS-related research.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
