Abstract
Lymphopenia is a known adverse effect in patients with relapsing multiple sclerosis (RMS) treated with fumaric acids. We present a case series of four patients diagnosed with RMS with prolonged lymphocyte stability on dimethyl fumarate for over 1 year who developed significant lymphopenia after transitioning to diroximel fumarate. This case series highlights the need for further research to elucidate the risk of lymphopenia in patients switching between fumaric acids.
Keywords
Introduction
Dimethyl fumarate (DMF), diroximel fumarate (DRF), and monomethyl fumarate (MMF) are disease-modifying therapies (DMTs) approved for the treatment of relapsing forms of multiple sclerosis (RMS).
DMF and DRF are fumaric acid prodrugs of the active metabolite MMF. Fumarates are thought to exert therapeutic effect by activating the nuclear factor (erythroid-derived 2)-like 2 (Nrf2) pathway involved in cellular response and oxidative stress.1,2 The EVOLVE-MS-1 trial demonstrated that most patients on DRF experienced a 30% drop in absolute lymphocyte count (ALC) over the first year which stabilized to a level above the lower limit of normal. About 10% of patients experienced an ALC less than 0.5 × 109/L (grade 3 lymphopenia). 3 Patients may transition between fumarates for many reasons including tolerability, insurance coverage, and financial barriers. This case series highlights four patients on DMF with prolonged ALC stability who developed significant lymphopenia after switching to DRF leading to its discontinuation.
Methods
We identified 82 patients at Vanderbilt University Medical Center Comprehensive Multiple Sclerosis Clinic who switched from DMF to DRF (n = 69) or MMF (n = 13) between January 2019 and July 2022. Baseline demographics and clinical information pertinent to the development and management of lymphopenia were collected in an electronic database. Our clinic obtains a complete blood count with differential at baseline, 6 months, and every 6–12 months thereafter (per recommended guidelines). Additional lab draws are left up to the prescribing physician. All ALC values are included on the graph and pertinent labs noted in the table (Table 1). We retrospectively identified four patients on DMF for at least a year with a stable lymphocyte count and no history of grade 3 lymphopenia. Affordability and access issues prompted a change to DRF; however, patients subsequently developed grade 3 lymphopenia resulting in discontinuation.
Patient demographics.
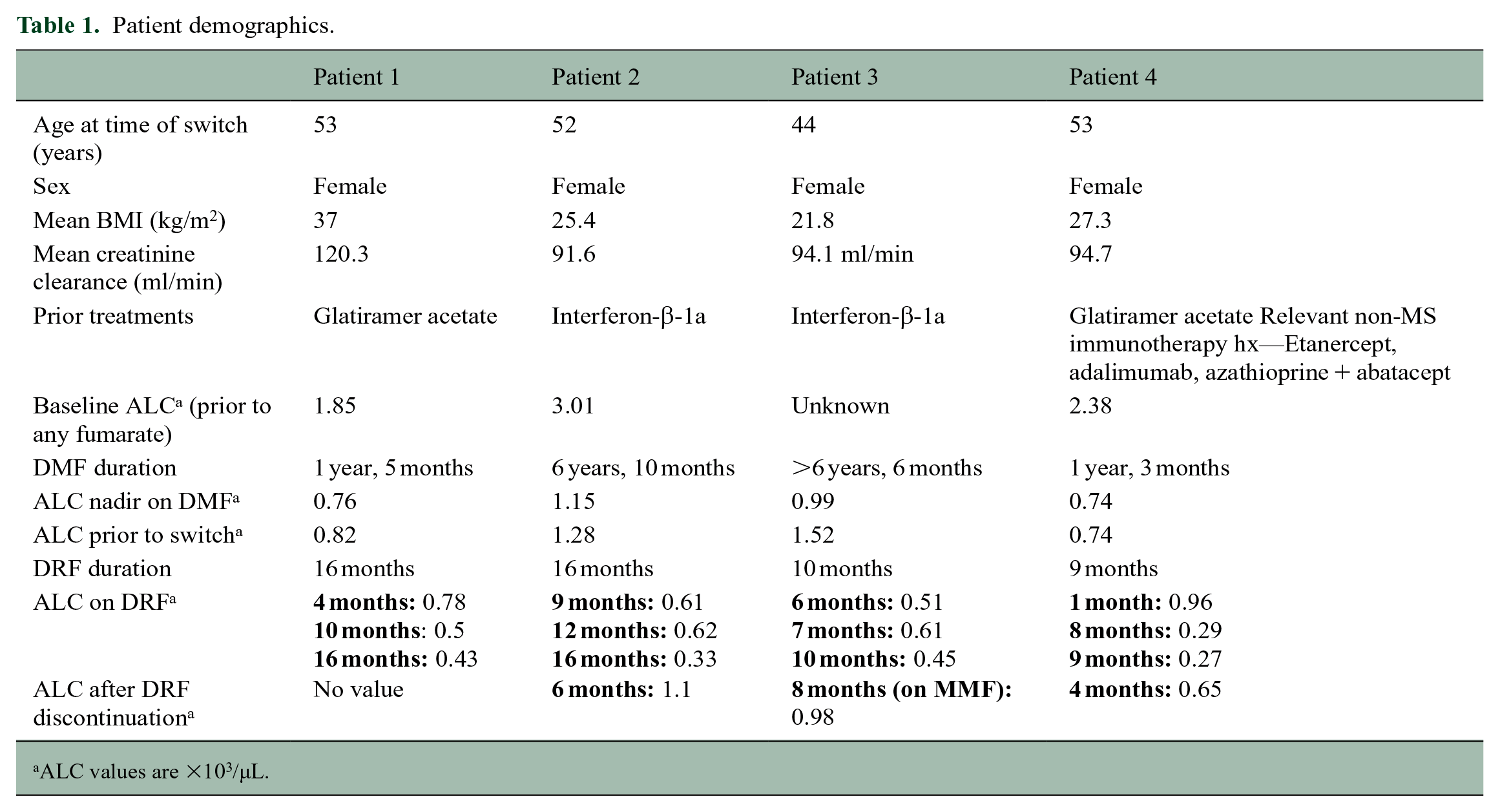
ALC values are ×103/µL.
Cases
Four patients developed grade 3 lymphopenia after transitioning from DMF to DRF. ALC remained stable over a prolonged period then dropped after the switch (Figure 1), absent other identifiable causes.
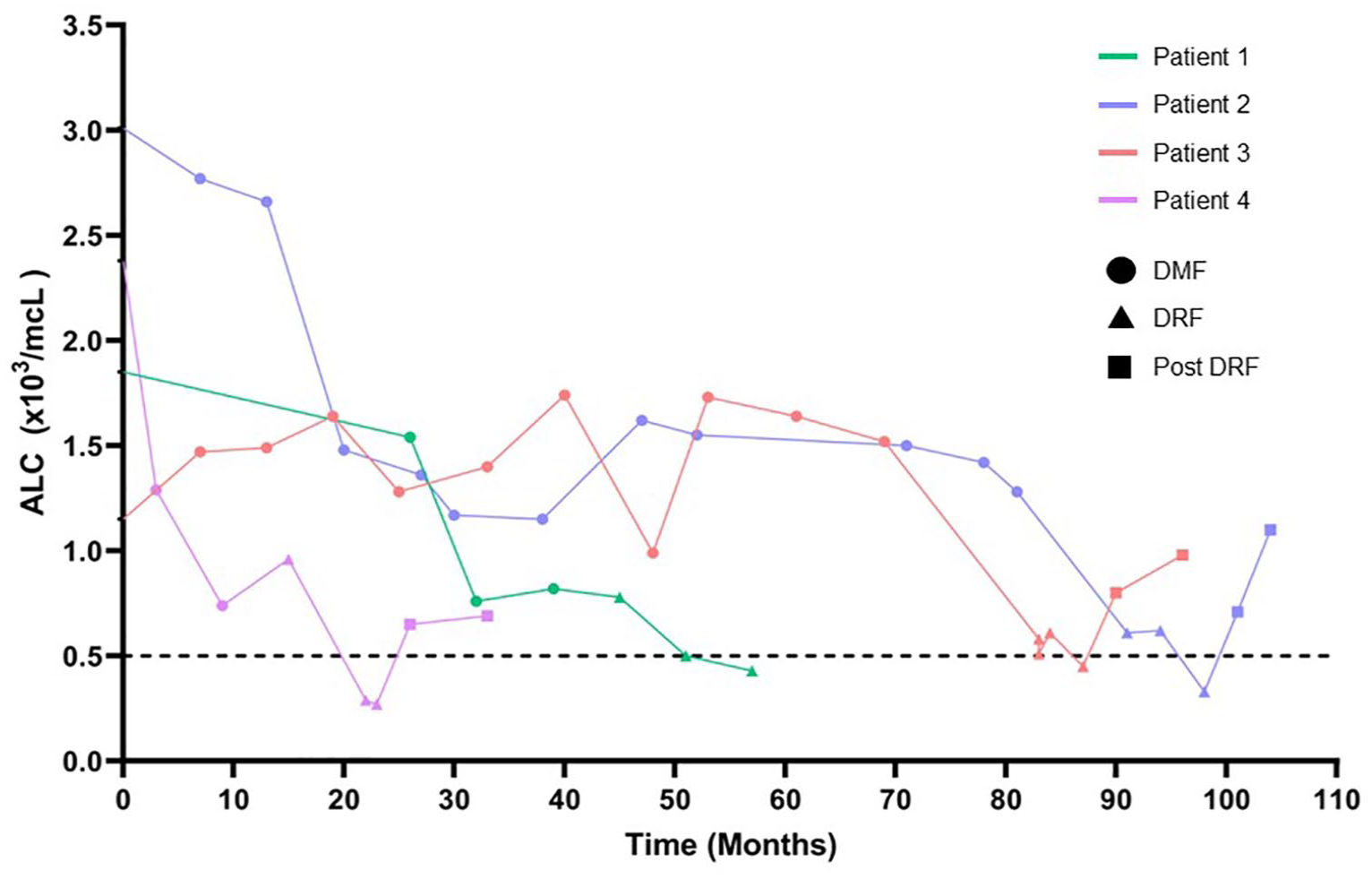
ALC values plotted as a function over time in months for four individual patients. Circles represent ALC while on DMF, diamonds represent ALC on DRF, and squares represent ALC post-DRF.
Case 1
A 53-year-old female with RMS developed grade 3 lymphopenia 16 months after switching from DMF to DRF. Her medical history was unremarkable. She started DMF in December 2017, stopped, and then restarted in November 2019 after radiologic progression was noted. Baseline ALC was normal and decreased by 51% after 6 months on DMF. She transitioned to DRF in April 2021 because of prohibitive cost and sporadic gastrointestinal (GI) upset. Ten months after switching, her ALC remained at or below 0.5 × 103/µL, and DRF was discontinued.
Case 2
A 52-year-old female diagnosed with RMS whose ALC was stable on DMF developed grade 3 lymphopenia 16 months after transitioning to DRF. No other contributing factors were identified. Her baseline ALC was normal and remained above normal limits while on DMF for almost 7 years. Insurance coverage prompted a change to DRF in February 2021. ALC began to decrease and DRF was discontinued in June 2022 because of persistent lymphopenia. The patient’s ALC recovered to normal limits 6 months after DRF was stopped.
Case 3
A 44-year-old female diagnosed with RMS developed grade 3 lymphopenia 9 months after transitioning from DMF to DRF, absent other contributing factors. She presented to our clinic in August 2014 and remained on DMF until February 2021. Her ALC remained at or above 0.99 × 103/µL. In February 2021, insurance prompted a change from DMF to DRF. Her ALC significantly decreased, and she developed a severe sinus infection in August 2021. In December 2021, she developed grade 3 lymphopenia prompting a switch from DRF to MMF. Shortly thereafter, her ALC began to rebound.
Case 4
A 53-year-old female diagnosed with RMS developed grade 3 lymphopenia 8 months after transitioning from DMF to DRF. ALC decreased by 69% after 9 months on DMF and remained stable. Insurance coverage prompted a change from DMF to DRF in January 2021. Eight months after switching, she developed grade 3 lymphopenia. The level was repeated 1 month later, and DRF was discontinued. Her ALC rebounded 3 months after stopping DRF.
Discussion
Lymphopenia is a well-established adverse effect of fumaric acids when used for treatment in MS. ALC reduction is typically noted in the first year then plateaus over time. 4 We identified four patients who switched from DMF to DRF and developed grade 3 lymphopenia despite having a prolonged stable ALC on DMF. Per the US Prescribing Information, the terminal half-life of DRF is approximately 1 hour, and accumulation does not occur after multiple doses. Despite similarities between fumaric acids, there are known pharmacokinetic and pharmacodynamic differences. Unlike DMF, DRF is metabolized into an inactive metabolite, 2-hydroxyethyl succinimide, which is eliminated renally. Metabolite accumulation limits the use in moderate to severe renal impairment. 5 None of our patients had renal or hepatic impairment.
DRF and MMF have demonstrated improvement in GI tolerability compared to DMF which may lead to better adherence and consequential effects on lymphocyte counts.3,6 With our patients, affordability was the primary driver for transitioning from DMF to DRF, and significant GI upset was not identified as a main reason for switching. In addition, a reduction in lymphocyte count was not noted in the 13 patients who switched to MMF, and ALC rebounded after patient 3 switched from DRF to MMF. This suggests differences in pharmacodynamic properties between fumaric acids. 7
Studies have identified potential risk factors for developing lymphopenia on fumaric acids. These include a lower baseline ALC, older age (56 years and older), and longer disease duration.7,8 One paper demonstrated a lower risk of lymphopenia in patients with a body mass index (BMI) ⩾30 kg/m2 while another showed an increased risk when BMI falls between 25 and 30 kg/m2.8–10 Our patients’ BMI ranged from 21.8 to 37, and all were <56 years old. Longbrake et al. demonstrated an increased risk of developing grade 3 lymphopenia in patients older than 55. While the mechanism of DMF-induced lymphopenia is not fully understood, it is thought to be related to T-cell apoptosis. In addition, lymphopoiesis is known to decline over time due to thymic involution. 11 While most of our patients were older in age, we do not believe immunosenescence significantly influenced our results given their historical stability on DMF and the short duration on DRF to nadir ALC (mean = 13 months, range = 9–16 months). In addition, all patients had similar prior DMT exposure.
We note several limitations. Our case series is a single-center retrospective analysis of switches from DMF to DRF or MMF. Our sample size is relatively small so the true incidence of grade 3 lymphopenia in this population may differ from what we have reported. In addition, we could not obtain prescription fill data to calculate proportion of days covered to assess potential adherence differences between DMF and DRF. However, insurance coverage was the primary driver of each switch, and there was no indication any patient was switched because of adherence issues on DMF.
To our knowledge, this is the first case series identifying patients with prolonged ALC stability on DMF who developed grade 3 lymphopenia after transitioning to DRF. Further studies are warranted to elucidate the risk of ALC reduction in patients switching between fumaric acids.
Footnotes
Data Availability Statement
Data sharing not applicable to this article as no data sets were generated or analyzed during this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.S. has no conflicts of interest to disclose. J.K. has received personal compensation through consulting fees from Biogen, Genentech, TG Therapeutics, Sanofi, and EMD Serono, and he has a leadership role with the CMSC-Physician Assistant Special Interest Group. A.B. has received speaking honoraria and personal compensation for authoring chapters on MS for ACCP PSAP 2021 and ACSAP 2022, and she has also received personal compensation for her involvement in the ASHP Biosimilar Steering Committee (resource center, webinar, podcast). H.M. has received personal compensation through consulting fees from Biogen, ER Squibb and Sons, and Genentech, he has also received speaking honoraria for participating as a speaker for Biogen, ER Squibb and Sons, Alexion, TG Therapeutics, and EMD Serono, and he has received personal compensation as an expert witness on behalf of the US government for the Vaccine Injury Program for the Department of Justice.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
