Abstract
Background:
In MS, functional connectivity (FC) dynamism may influence disease evolution.
Objectives:
The objective is to assess time-varying functional connectivity (TVFC) changes over time at 2.5-year follow-up in MS patients according to physical and cognitive worsening.
Methods:
We collected 3T magnetic resonance imaging (MRI) for TVFC assessment (performed using sliding-window analysis of centrality) and clinical evaluations at baseline and 2.5-year follow-up from 28 healthy controls and 129 MS patients. Of these, 79 underwent baseline and follow-up neuropsychological assessment. At 2.5 years, physical/cognitive worsening was defined according to disability/neuropsychological score changes.
Results:
At follow-up, 25/129 (19.3%) MS patients worsened physically and 14/79 (17.7%) worsened cognitively. At baseline, MS patients showed reduced TVFC versus controls. At 2.5-year follow-up, no TVFC changes were detected in controls. Conversely, TVFC decreased over time in parieto-temporal regions in stable MS patients and in default-mode network in worsened MS. In physically worsened MS, basal ganglia TVFC reductions were also found. Reduced TVFC over time in the putamen in physically worsened and reduced TVFC in the precuneus in cognitively worsened were significant versus stable MS.
Discussion:
At 2.5-year follow-up, default-mode network TVFC reductions were found in worsening MS. Moreover, reduced deep gray matter TVFC characterized physically worsened patients, whereas precuneus involvement characterized cognitively worsened MS patients.
Keywords
Introduction
Multiple sclerosis (MS) is characterized by complex pathological substrates, resulting in non-uniform clinical manifestations and variable disease progression. 1 Therefore, determining biomarkers associated with an unfavorable disease course is of paramount importance to optimize patients’ management.
Magnetic resonance imaging (MRI)–derived measures are good candidates for identifying mechanisms associated with disability progression and cognitive deterioration. Conventional MRI often enters in disease monitoring and in evaluating disease-modifying treatment (DMT) efficacy; 2 however, T2-visible MRI abnormalities can partially explain subsequent MS course just at the earliest disease stages.3–5 Conversely, the association between atrophy progression and concomitant worsening of MS disability5–8 and cognitive deficits8,9 is more evident.
Among advanced MRI techniques, resting state (RS) functional magnetic resonance imaging (fMRI) allows to map large-scale functional networks and to establish how MS pathology impairs functional integration. 10 A few longitudinal RS fMRI studies put in relation RS functional connectivity (FC) changes over time with concomitant disability or cognitive worsening.11–13 Faivre et al. 11 described an association between longitudinal RS FC decrease and clinical worsening at 2-year follow-up, suggesting maladaptive mechanisms. Conversely, Koubiyr et al. 13 found an increased coupling among structural and functional networks at 5-year follow-up in patients with clinically isolated syndromes, which was related to cognitive deterioration. Similarly, Huiskamp et al. 12 found a longitudinal increase of ventral attention network centrality in cognitively preserved MS patients transitioning to mild cognitively impaired. Such results suggest a not straightforward relationship between longitudinal RS FC changes and concomitant disease evolution, which may depend from disease stage and specific analysis methods.
Traditionally, RS FC has been quantified using data from the whole RS fMRI examination, making the hypothesis that FC is static across the whole time series. Recently, time-varying functional connectivity (TVFC) 14 has been introduced as a novel technique that aims at measuring variability of RS FC fluctuations during the course of RS fMRI acquisition. Since the human brain is a dynamic system continuously integrating and coordinating external and internal information, TVFC may provide additional information on intrinsic functional brain properties, complementing traditional RS FC data. 15 In MS patients, cross-sectional TVFC analyses showed abnormalities of sensorimotor and cognitive networks,15–18 varying according to disease stage16,18 and correlating with clinical disability 15 and cognitive impairment.12,15–17 Longitudinal TVFC studies in MS are still scanty; however, preliminary evidence showed that cognitive decline at 5-year follow-up was associated with progressive TVFC instability over time. 19 The impact of longitudinal TVFC changes on concomitant clinical disability worsening needs further investigations, since preliminary TVFC studies focused on early MS stages presenting with minimal medium-term disability changes. 18
Here, we hypothesized that TVFC analysis might provide novel insights into the functional mechanisms associated with disease progression in MS; in particular, we supposed that physical and cognitive deterioration might be associated with peculiar TVFC changes in specific brain areas. To address this, we assessed longitudinal TVFC evolution over 2.5 years of follow-up in MS patients, and we investigated specific TVFC changes characterizing MS patients presenting with physical disability progression or cognitive deterioration.
Methods
Ethics committee approval
Approval was received from the institutional ethical standards committee on human experimentation. Written informed consent was obtained from all subjects prior to study participation.
Patients
We retrospectively included from our internal database consecutive MS patients, selected for having two clinical and MRI evaluations separated by a minimum follow-up of 1.5 years. Part (80%) of subjects were included in previous publications, 20 where data were analyzed using a static RS FC approach and follow-up MRI scans were not included. Inclusion/exclusion criteria were: (1) right handedness (five subjects discarded); (2) relapse- and steroid-free, and (3) no DMT changes for at least 3 months before both MRI scans (four subjects discarded); (4) no drug/alcohol abuse history; (5) no MRI contraindications; and (6) no neurological or systemic disorder other than MS (three subjects discarded). Right-handed healthy controls (HCs) with no history of neurological, psychiatric, or systemic disorders, who also underwent two MRI scans separated by a minimum follow-up of 1.5 years, were selected, serving as reference for structural MRI and TVFC analysis.
Clinical and neuropsychological assessment
Within 2 days from both MRI examinations, MS patients underwent a neurological evaluation, with rating of the Expanded Disability Status Scale (EDSS) score 21 and DMT recording. Besides EDSS score rating, the follow-up neurological assessment also collected information on clinical relapse occurrence and DMT changes (binary coded). At follow-up, patients were considered as physically worsened if they had an EDSS score increase ⩾1.5 when baseline EDSS was 0, ⩾1.0 when baseline EDSS was between 1.0 and 5.5, or ⩾0.5 when baseline EDSS was ⩾6.0. 20
At baseline and follow-up, 79 MS patients underwent a neuropsychological assessment using the Brief Repeatable Battery of Neuropsychological tests
22
(BRB-N, baseline: version A, follow-up: B). Performances were adjusted for age, sex and education and transformed into
MRI acquisition and structural MRI analysis
At baseline and 2.5-year follow-up, all subjects underwent brain MRI using two 3.0T scanners (Scanner 1: Achieva,
TVFC analysis
After RS fMRI pre-processing (Supplementary methods), TVFC was assessed basing on the calculation of degree centrality, 26 a measure quantifying the relative importance of individual gray matter (GM) voxels over the whole-brain networks that was already used in multicenter studies of MS 26 and showed minor scanner-related effects, easy to be controlled using scanner-adjusted statistical models. 27 Using DPABI software (http://rfmri.org/dpabi), we splitted RS fMRI scans into sliding windows of 22 time points (Scanner 1) or 42 time points (Scanner 2), to take into account for different repetition times/acquisition lengths (but at the same time maintaining an equal window length in seconds across scanners), with a shift length of one volume across windows. Degree centrality maps were computed for each window. Then, the coefficient of variation across sliding windows of degree centrality was calculated to produce TVFC maps, taken as a measure of FC dynamism.
Statistical analysis
Analyses of demographic, clinical, and structural MRI variables were performed using SPSS software (IBM, version 26.0, Armonk, NY, USA). Demographic and clinical characteristics of study subjects were compared between HC and MS patients using independent samples
Results
Clinical, neuropsychological, and structural MRI measures
Main study population
Demographic, clinical, and structural MRI data are summarized in Table 1. We initially screened 176 subjects for inclusion; of these, 129 MS patients (103 relapsing-remitting/26 progressive MS) and 28 HCs were finally included. At baseline, age and sex were not different between MS patients and HC, while brain volumetry in all tissue compartments was significantly reduced in MS patients versus HC (Table 1).
Main demographic, clinical, and MRI features of patients with multiple sclerosis (MS) and healthy controls (HCs) at baseline and follow-up evaluations.
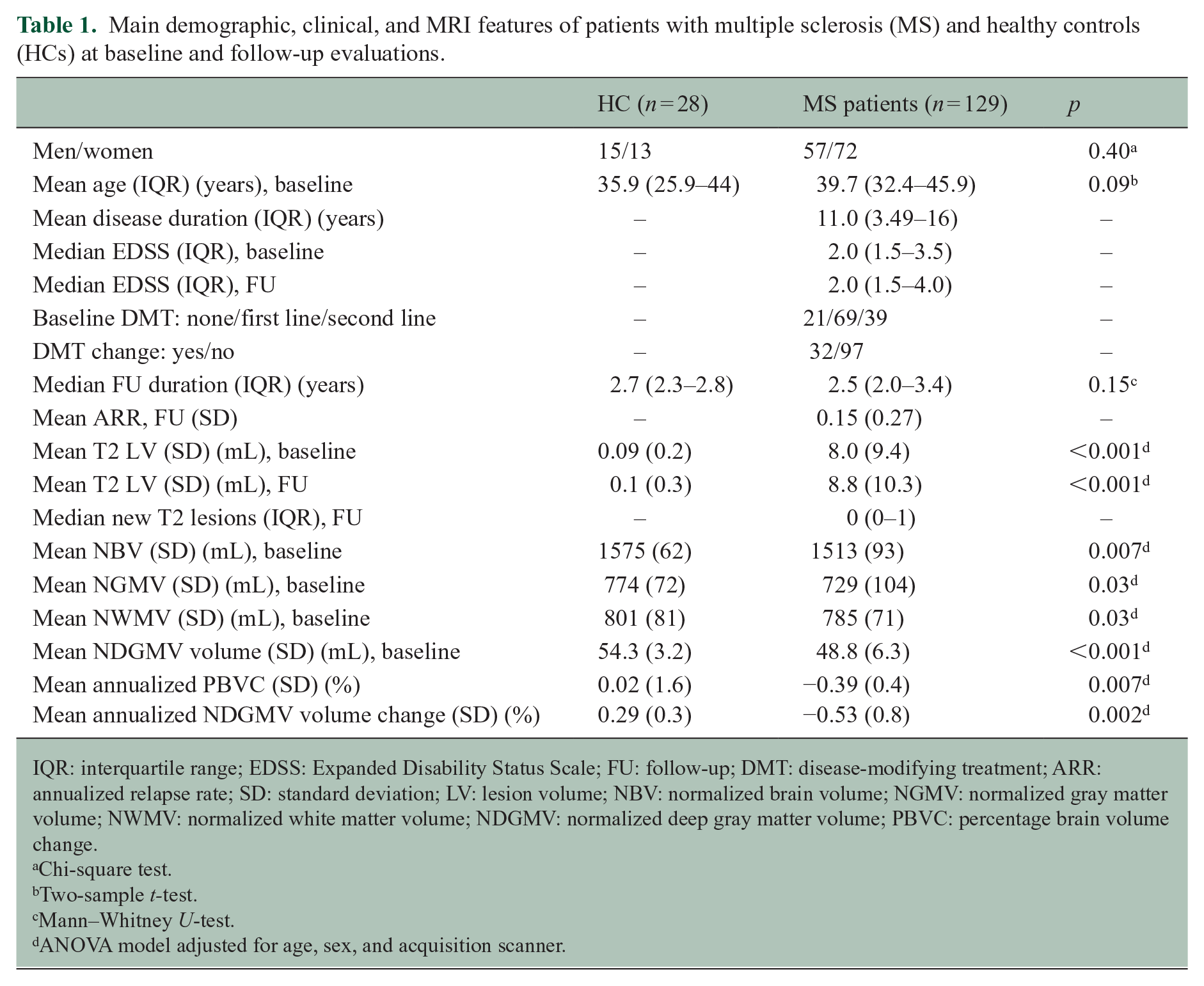
IQR: interquartile range; EDSS: Expanded Disability Status Scale; FU: follow-up; DMT: disease-modifying treatment; ARR: annualized relapse rate; SD: standard deviation; LV: lesion volume; NBV: normalized brain volume; NGMV: normalized gray matter volume; NWMV: normalized white matter volume; NDGMV: normalized deep gray matter volume; PBVC: percentage brain volume change.
Chi-square test.
Two-sample
Mann–Whitney
ANOVA model adjusted for age, sex, and acquisition scanner.
The median follow-up duration was 2.5 years (interquartile range (IQR) = 2.0–3.4 years) in MS patients and 2.7 years (IQR = 2.3–2.8 years) in HC (
Physically worsened MS patients were older (
Cognitive sample
Seventy-nine MS patients (69 relapsing-remitting/10 progressive MS) underwent a complete neuropsychological assessment at baseline and follow-up. The main characteristics of this subpopulation are described in the Supplementary results.
At baseline, 20 MS patients were cognitively impaired and 59 were cognitively preserved. At follow-up, 14/79 (17.7%) patients were classified as cognitively worsened.
TVFC analysis
Baseline TVFC analysis
Results of the comparison of TVFC between MS patients and HC are outlined in Figure 1 and Table 2. At baseline, MS patients showed reduced TVFC compared to HC in the bilateral orbitofrontal cortex (
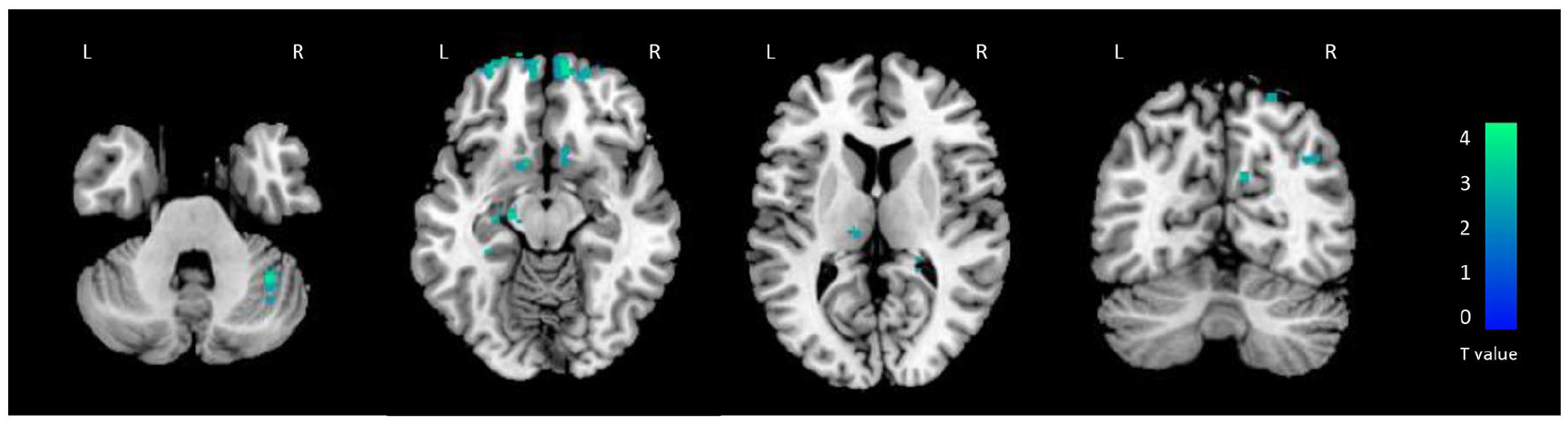
Brain regions showing significant differences of baseline time-varying functional connectivity (TVFC) between healthy controls (HCs) and multiple sclerosis patients (SPM12 two-sample
Brain regions showing significant differences of baseline time-varying functional connectivity (TVFC) between healthy controls (HCs) and multiple sclerosis (MS) patients (SPM12 two-sample
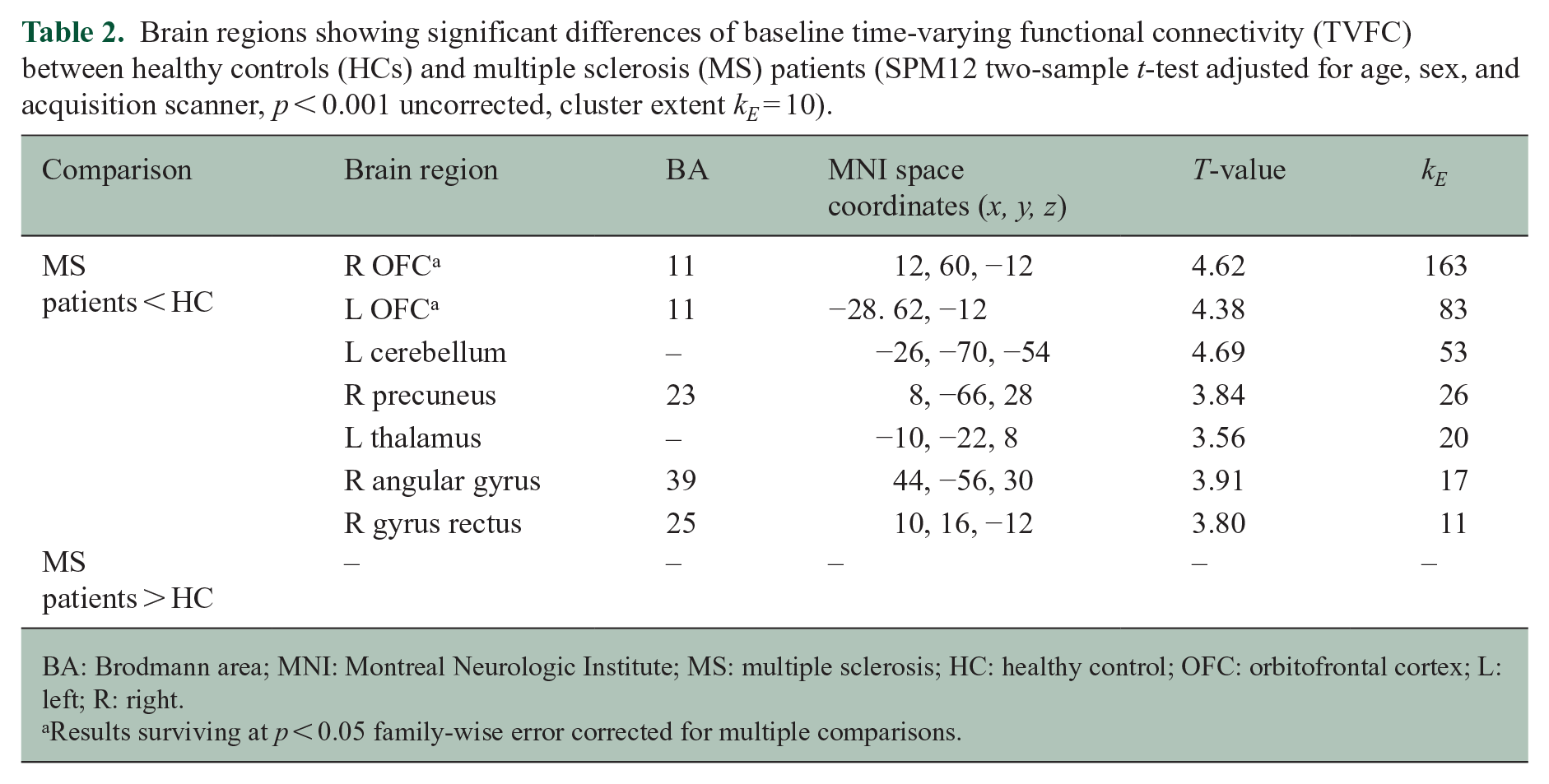
BA: Brodmann area; MNI: Montreal Neurologic Institute; MS: multiple sclerosis; HC: healthy control; OFC: orbitofrontal cortex; L: left; R: right.
Results surviving at
Longitudinal TVFC analysis: physical worsening
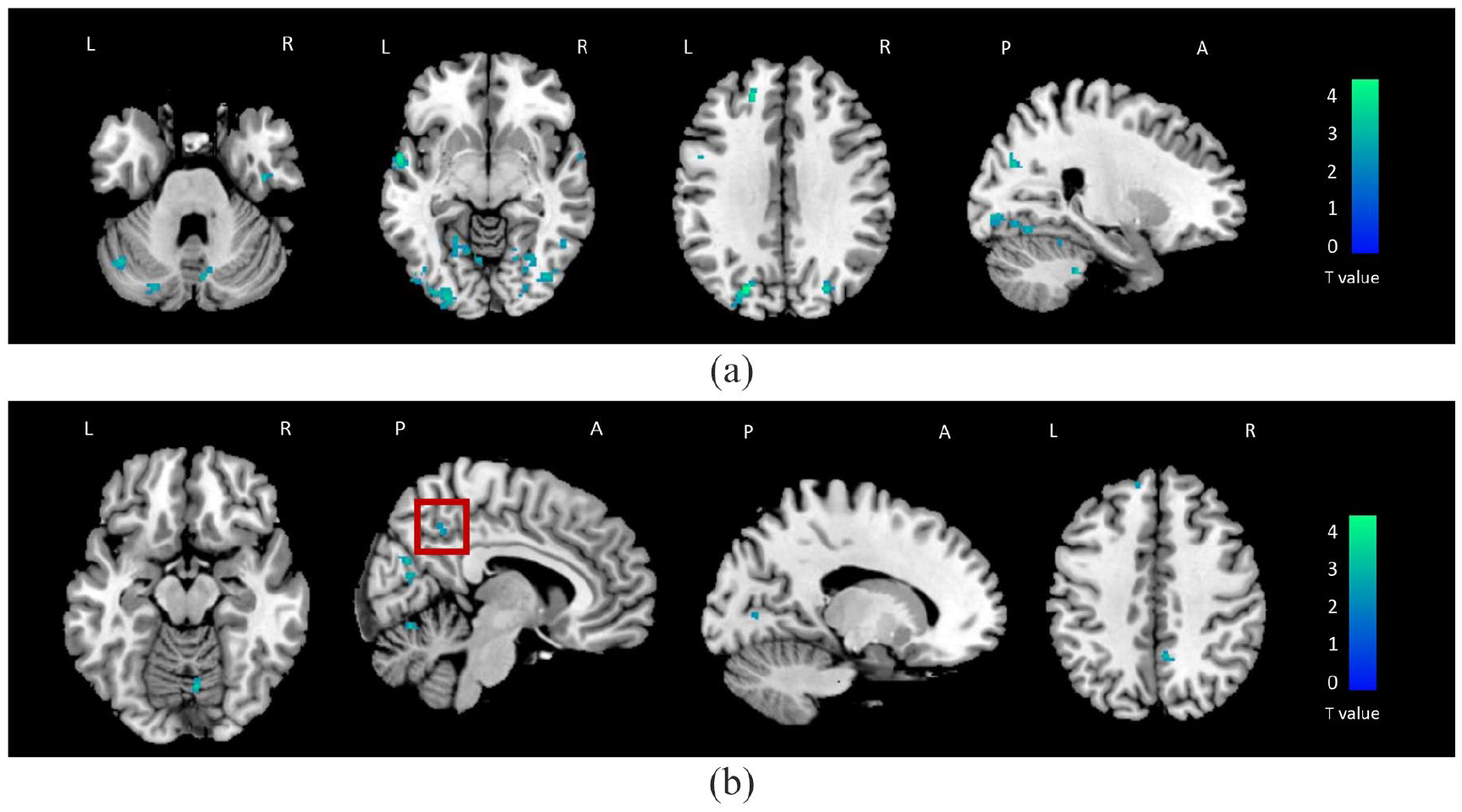
No significant TVFC changes over time were detected within HC. Results of the longitudinal TVFC changes detected within physically stable and worsened MS patients are shown in Figure 2 and Table 3. At 2.5-year follow-up, a widespread reduction of TVFC over time (
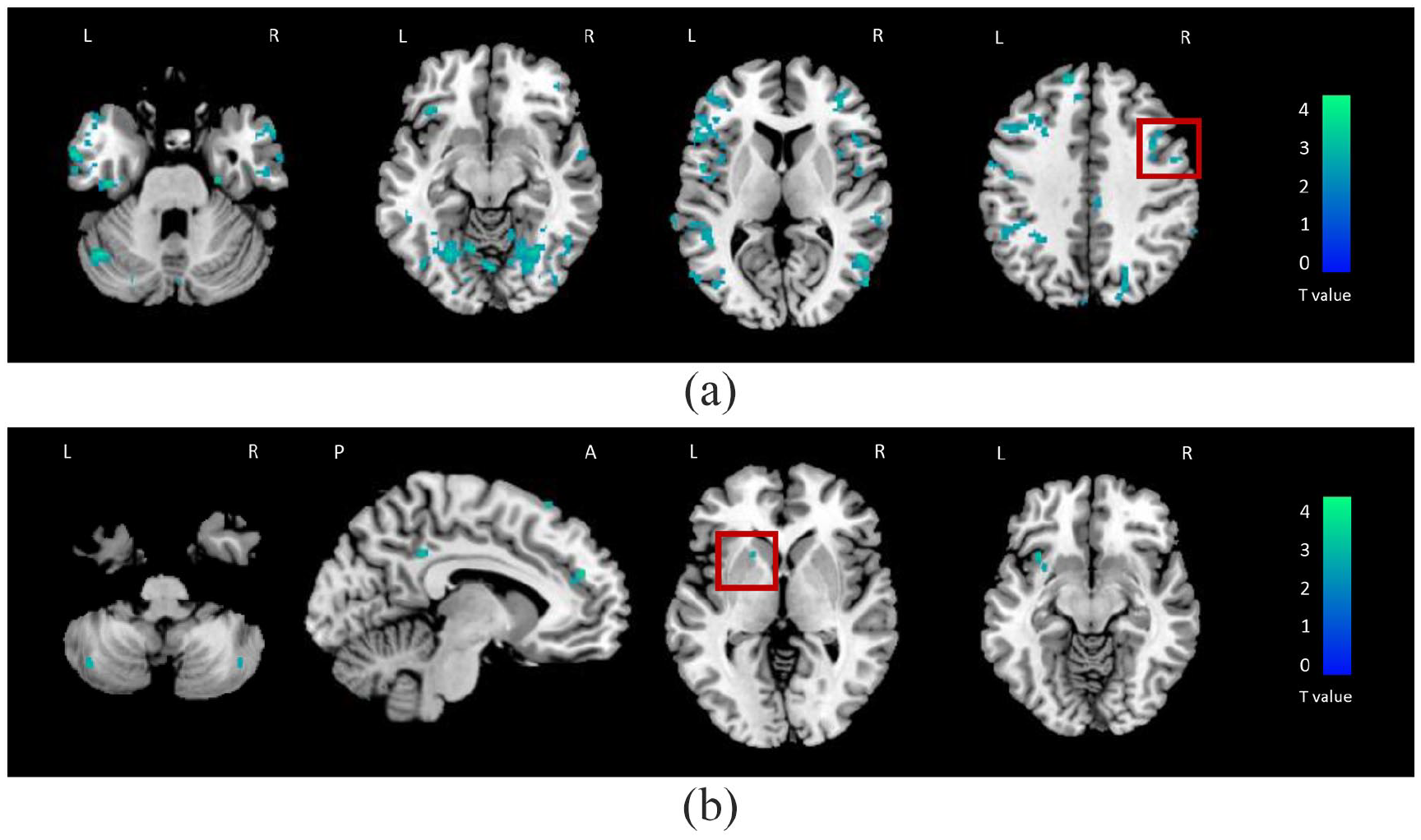
Brain regions showing significant changes of time-varying functional connectivity (TVFC) over time in physically stable (a) and in physically worsened (b) multiple sclerosis patients (SPM12 repeated-measure full-factorial model, adjusted for age, sex, and acquisition scanner,
Brain regions showing significant changes of time-varying functional connectivity (TVFC) over time within physically stable and worsened multiple sclerosis (MS) patients at 2.5-year follow-up (SPM12 repeated-measures full-factorial model, adjusted for age, sex, and acquisition scanner,
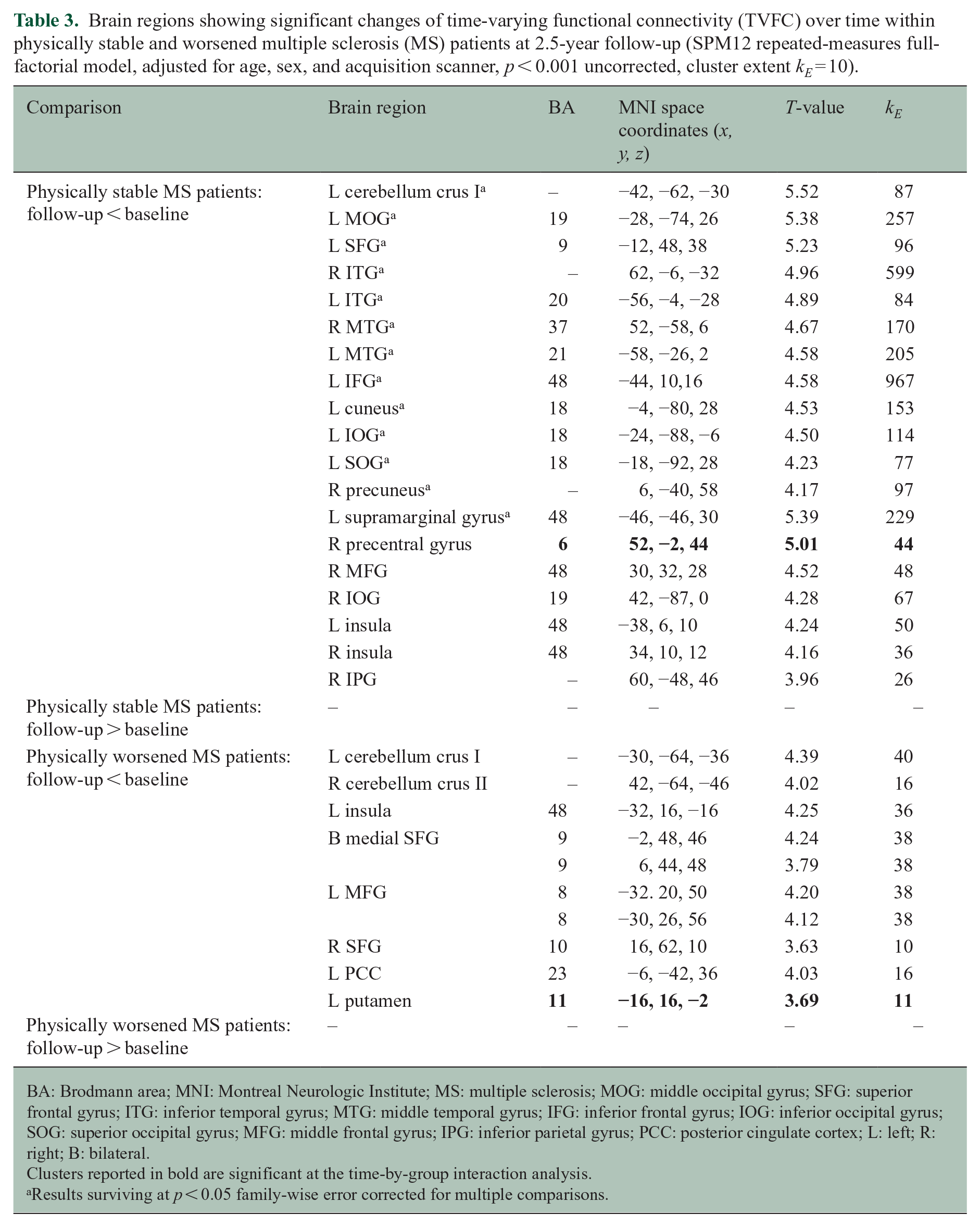
BA: Brodmann area; MNI: Montreal Neurologic Institute; MS: multiple sclerosis; MOG: middle occipital gyrus; SFG: superior frontal gyrus; ITG: inferior temporal gyrus; MTG: middle temporal gyrus; IFG: inferior frontal gyrus; IOG: inferior occipital gyrus; SOG: superior occipital gyrus; MFG: middle frontal gyrus; IPG: inferior parietal gyrus; PCC: posterior cingulate cortex; L: left; R: right; B: bilateral.
Clusters reported in bold are significant at the time-by-group interaction analysis.
Results surviving at
Longitudinal TVFC analysis: cognitive worsening
Results of the longitudinal changes of TVFC within cognitively stable and worsened MS patients are shown in Figure 3 and Table 4. At follow-up, a reduction of TVFC versus baseline was found within cognitively stable MS patients in the bilateral superior occipital gyrus, left lingual cortex, right fusiform gyrus, and left superior temporal gyrus (
Brain regions showing significant changes of time-varying functional connectivity (TVFC) over time in cognitively stable (a) and in cognitively worsened (b) multiple sclerosis patients (SPM12 repeated-measure full-factorial model, adjusted for age, sex, and acquisition scanner,
Brain regions showing significant changes of time-varying functional connectivity (TVFC) over time in cognitively stable and worsened multiple sclerosis (MS) patients at 2.5-year follow-up (SPM12 repeated-measures full-factorial model, adjusted for age, sex and acquisition scanner,
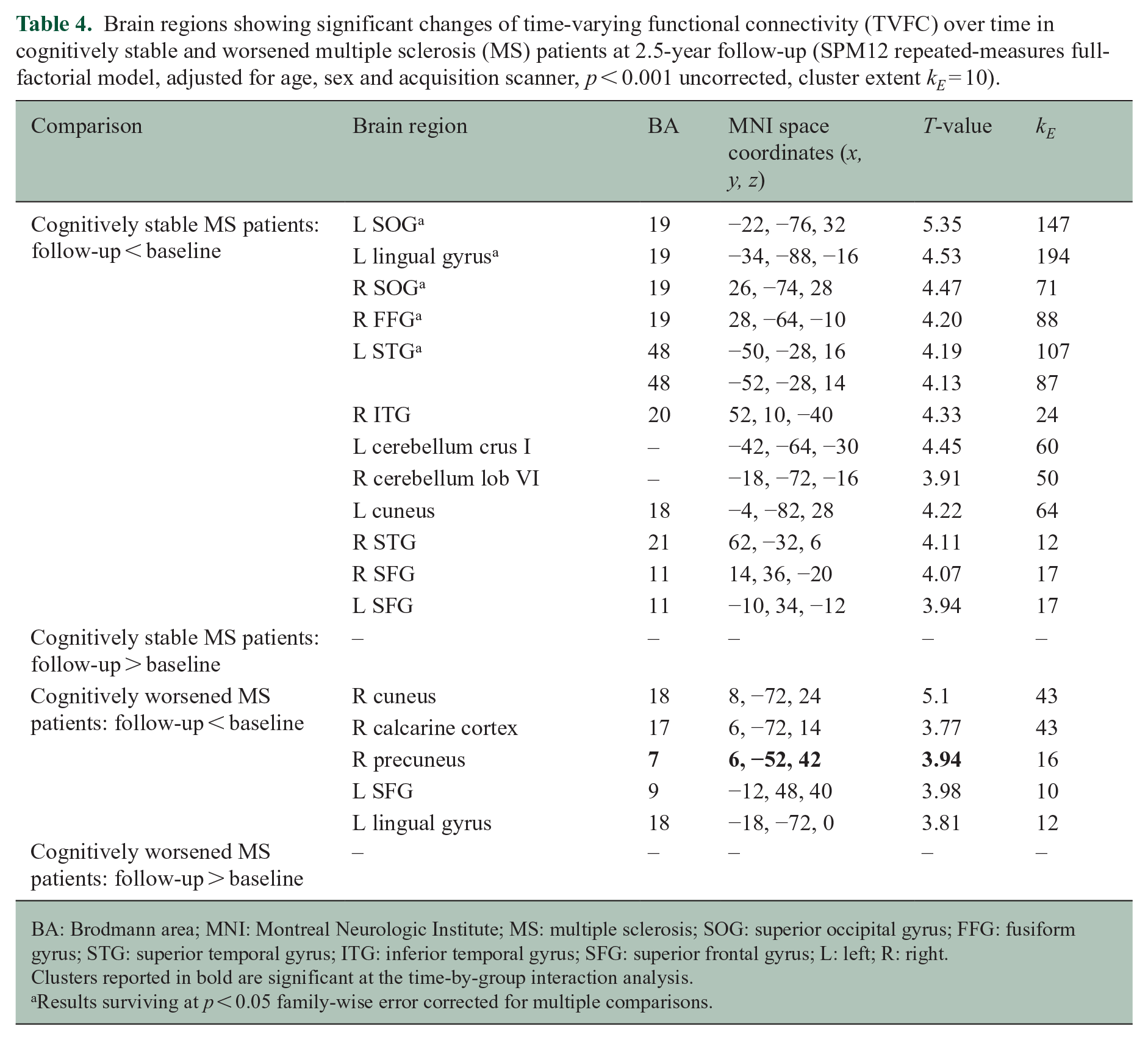
BA: Brodmann area; MNI: Montreal Neurologic Institute; MS: multiple sclerosis; SOG: superior occipital gyrus; FFG: fusiform gyrus; STG: superior temporal gyrus; ITG: inferior temporal gyrus; SFG: superior frontal gyrus; L: left; R: right.
Clusters reported in bold are significant at the time-by-group interaction analysis.
Results surviving at
Conversely, TVFC decrease over time was found within cognitively worsened MS patients not only in the right cuneus but also in right calcarine cortex, right precuneus, left superior frontal cortex, and left lingual cortex (
Validation analyses
To better validate our data, three sensitivity analyses were performed, to test: (1) effects of “pure” physical and cognitive worsening; (2) exclusion of patients deteriorating just in one cognitive domain; and (3) scanner effects. Results from sensitivity analyses are reported in the Supplementary results and confirm the main study findings. In particular, the analysis of “pure” effects of disability and cognitive worsening confirmed a peculiar TVFC reduction over time in the right precuneus within the “pure” cognitively worsening group, and a peculiar TVFC reduction over time in the left putamen within the “pure” physically worsening group, even if these findings were observed at a more liberal threshold compared to that used in the main analysis.
Discussion
Here, we explored changes over time of network connectivity dynamism in MS patients and their association with concomitant worsening of physical and cognitive deterioration. In general, MS patients were characterized by decreased TVFC over 2.5-year follow-up. However, while stable MS patients presented TVFC reductions predominantly in sensorimotor and associative cortices, physically and cognitively worsening MS patients exhibited reduced TVFC over time in default-mode network regions. In addition, the time-by-group interaction analysis indicated a specific link of physical disability worsening with decreased TVFC of the left putamen, while the same analysis indicated a link of cognitive deterioration with decreased TVFC of the right precuneus.
Baseline TVFC analysis showed that MS patients had decreased TVFC versus HC in bilateral orbitofrontal, parietal, cerebellar, and thalamic regions. This is not the first study detecting a decreased connectivity dynamism in MS compared to HC, especially in frontal16,17,29 and parietal16,17 lobes, as well as in the cerebellum and in subcortical nuclei. 16 This finding is likely to indicate a disease-induced loss of flexibility in functional brain interactions, leading to impaired efficiency in information exchange between distant brain areas, especially those related to sensorimotor and associative functions. Of note, cross-sectional TVFC abnormalities detected by previous studies were generally characterized by concomitant TVFC increases and reductions,15,16,18,30 which was not the case of our study. The variety of approaches used to assess TVFC 15 and the investigation of different disease phases are both likely to have a role in explaining such heterogeneity of findings.16,18,29
The most novel part of our study was the longitudinal TVFC assessment, which was the topic of only a few previous investigations18,19 putting such changes in relationship with physical and cognitive deterioration. The first result we would like to highlight is that TVFC tended to decrease at 2.5-year follow-up both within stable and worsened MS patients, although brain regions involved by such a decrease differed. While stable patients mainly showed TVFC decrease over time in sensorimotor and associative regions, physically and cognitively worsening patients exhibited TVFC decrease in regions of the default-mode network and deep GM. Taken together, these results suggest that in our entire cohort RS FC is going toward a more static, less flexible configuration, suggesting that information exchange within and across different functionally specialized brain systems is progressively hampered over time. However, not all TVFC decreases are necessarily detrimental: only those occurring in critical networks (such as the default-mode network) are likely to have an impact on MS deterioration. Indeed, the default-mode network is one of the most important brain networks, being consistently altered in MS patients. 10 From a dynamic point of view, such a network is thought to have a central role in linking new external information to previously acquired data and in facilitating information exchange with other high-order networks, such as the fronto-parietal and the central executive networks. 10 It is therefore conceivable that a TVFC decrease localized in the default-mode network has a major impact on MS disease worsening. This is also supported by findings from another recent longitudinal study, which used static RS FC and found default-mode network dysfunction in intact MS converting to cognitively impaired MS patients. 12
With this regard, it is interesting to note that our findings related to cognitive worsening indicate a peculiar TVFC reduction at time-by-group interaction analysis in the right precuneus, which is once again a central node of the default-mode network. The precuneus has a major role in a wide spectrum of integrated tasks (e.g. visuo-spatial imagery, episodic memory retrieval, self-processing, and consciousness). 31 Previous RS FC studies consistently showed that the default-mode network gets stuck in a more central26,32 and less dynamic state12,17 in cognitively impaired MS patients. In addition, abnormal TVFC of the precuneus was previously shown to have a role in explaining cognitive performance of patients with neuromyelitis optica spectrum disorder. 33 This is probably explained by the high-energy consumption of the posterior parietal cortex, creating a hypoxic environment which in turn might promote neurodegeneration and worsening of cognitive performances.
Our results seem to be partly in contradiction with one recent report, 19 which found a more unstable network organization in cognitively declining MS patients at 5-year follow-up. Notably, such a study had a longer follow-up duration than our study, and it used a different methodological TVFC approach. More specifically, the network flexibility parameter found to be increased at follow-up versus baseline was cohesion, that is, the number of mutual switches between subnetworks, while the number of independent switches was not increased. 19 This seems to indicate that subnetwork configuration was more “fragmented” at follow-up versus baseline, but groups of nodes had a more “fixed” membership in the same subnetworks, in turn suggesting a not straightforward increase of RS FC dynamism. Another possible explanation of such discrepancies might be the intrinsically limited temporal resolution of RS fMRI scans, which probably hampers precise investigations of brain connectivity dynamism. 34 Future studies complementing RS fMRI with electrophysiological data may allow more reliable inspections of TVFC reorganization.
With regard to physical worsening, the time-by-group interaction analysis highlighted a specific effect of decreased TVFC of the putamen. This deep GM nucleus is primarily engaged in motor control, but it has also some role in motor learning, executive functions, behavior, and emotion. 35 Deep GM damage is known to be severe and extensive in MS patients; in particular, deep GM atrophy was found to be increasing especially in patients presenting with clinical worsening; 5 moreover, atrophy rate of the putamen was found to be the highest among the other nuclei. 5 In light of this, we can suppose that structural and functional changes of deep GM are particularly relevant for disability accumulation and that they represent useful metrics for monitoring clinical disease progression.
We do acknowledge that this study is not without limitations. First, MRI was performed using two different 3.0T scanners. However, the same scanner was used at baseline and follow-up for the same subject, and all MRI statistical analyses were adjusted for scanner. Second, TVFC abnormalities localized in the orbitofrontal cortex should be interpreted with caution, since RS fMRI acquisitions are characterized by distortions in this region. Third, the sample of cognitively and physically worsened MS patients was relatively small, probably because of the short follow-up duration; a lack of statistical power might explain why we observed limited within-group TVFC changes in worsening MS patients. A larger sample size (obtained, e.g., in future multicenter studies) and longer follow-up would be needed to increase the statistical power and confirm or expand our results. Fourth, the definition of cognitive worsening over time was chosen in agreement with the recent work of Dong et al., 25 basing on a cohort having similar follow-up duration. However, there is still no general consensus on methods for assessing cognitive decline in MS studies, and it is possible that different thresholds and approaches may lead to bigger/smaller numbers of cognitively worsening patients. Also, despite we tried to mitigate learning effects using two different versions of the BRB-N battery, we cannot exclude that practice effects are still present in our data, since a cognitive evaluation for HC was not available. Finally, larger and more comprehensive studies including additional functional and microstructural MRI metrics, as well as genetic, environmental, and biological data, would be necessary.
To conclude, we observed reduced TVFC of the default-mode network in physically and cognitively worsened patients at 2.5-year follow-up, while stable MS patients presented decreased TVFC especially in sensorimotor and associative areas of the occipital, parietal and temporal lobes. Reduced connectivity dynamism in deep GM characterized physically worsened patients, while precuneus involvement was the peculiar finding in cognitively worsened MS patients. Centrality dynamics reductions in deep GM and precuneal regions may represent useful metrics to predict and monitor cognitive and clinical disability accumulation in this condition.
Supplemental Material
sj-docx-1-msj-10.1177_13524585241231155 – Supplemental material for 2.5-Year changes of connectivity dynamism are relevant for physical and cognitive deterioration in multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585241231155 for 2.5-Year changes of connectivity dynamism are relevant for physical and cognitive deterioration in multiple sclerosis by Maria A Rocca, Giulia D’Amore, Paola Valsasina, Nicolò Tedone, Alessandro Meani and Massimo Filippi in Multiple Sclerosis Journal
Footnotes
Data Availability Statement
The anonymised data set used and analyzed during this study is available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.A.R. received consulting fees from Biogen, Bristol-Myers Squibb, Eli Lilly, Janssen, and Roche and speaker honoraria from AstraZeneca, Biogen, Bristol-Myers Squibb, Bromatech, Celgene, Genzyme, Horizon Therapeutics Italy, Merck Serono SpA, Novartis, Roche, Sanofi, and Teva. She receives research support from the MS Society of Canada, the Italian Ministry of Health, the Italian Ministry of University and Research, and Fondazione Italiana Sclerosi Multipla. She is Associate Editor for Multiple Sclerosis and Related Disorders. G.D.A., A.M., and N.T. have nothing to disclose. P.V. received speakers’ honoraria from Biogen Idec. M.F. is Editor-in-Chief of the Journal of Neurology, Associate Editor of Human Brain Mapping, Neurological Sciences, and Radiology; received compensation for consulting services from Alexion, Almirall, Biogen, Merck, Novartis, Roche, Sanofi; speaking activities from Bayer, Biogen, Celgene, Chiesi Italia SpA, Eli Lilly, Genzyme, Janssen, Merck Serono, Neopharmed Gentili, Novartis, Novo Nordisk, Roche, Sanofi, Takeda, and TEVA; participation in Advisory Boards for Alexion, Biogen, Bristol-Myers Squibb, Merck, Novartis, Roche, Sanofi, Sanofi-Aventis, Sanofi-Genzyme, Takeda; scientific direction of educational events for Biogen, Merck, Roche, Celgene, Bristol-Myers Squibb, Lilly, Novartis, Sanofi-Genzyme; and he receives research support from Biogen Idec, Merck Serono, Novartis, Roche, Italian Ministry of Health, the Italian Ministry of University and Research, and Fondazione Italiana Sclerosi Multipla.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
