Abstract
Background:
Estimating the effect of disease-modifying treatment of MS in observational studies is impaired by bias from unmeasured confounders, in particular indication bias.
Objective:
To show how instrumental variables (IVs) reduce bias.
Methods:
All patients with relapsing onset of MS 1996–2010, identified by the nationwide Danish Multiple Sclerosis Registry, were followed from onset. Exposure was treatment index throughout the first 12 years from onset, defined as a cumulative function of months without and with medium- or high-efficacy treatment, and outcomes were hazard ratios (HRs) per unit treatment index for sustained Expanded Disability Scale Score (EDSS) 4 and 6 adjusted for age at onset and sex, without and with an IV. We used the onset cohort (1996–2000; 2001–2005; 2006–2010) as an IV because treatment index increased across the cohorts.
Results:
We included 6014 patients. With conventional Cox regression, HRs for EDSS 4 and 6 were 1.15 [95% CI: 1.13–1.18] and 1.17 [1.13–1.20] per unit treatment index. Only with IVs, we confirmed a beneficial effect of treatment with HRs of 0.86 [0.81–0.91] and 0.82 [0.74–0.90].
Conclusion:
The use of IVs eliminates indication bias and confirms that treatment is effective in delaying disability. IVs could, under some circumstances, be an alternative to marginal structural models.
Introduction
Several studies have shown that the prognosis of multiple sclerosis (MS) in terms of time to and age at certain disability endpoints defined by the Expanded Disability Status Score (EDSS) 1 and to death has improved,2–6 however, with a noticeable exception. 7 That the natural history of MS appears to be milder in more recent times is probably a result of an interplay between several factors including changes in the diagnostic criteria, the Will Rogers phenomenon, changes in the epidemiology of MS, the impact of early and appropriate disease-modifying treatment and improvement of the general state of health in the population.8,9 A review stated that median time to EDSS 6 (a walking aid is required to walk 100 m) increased from 10 to 31 years in publications from 1984 to 2017. 9 A reason for a better prognosis over time may be disease-modifying treatment (DMT) which became available in most countries from about 1996. With time more of the patients were treated, particularly with earlier start and escalation to high-efficacy drugs.10,11 Studies have compared treated cohorts with historical cohorts before the DMT era and have found a more favourable prognosis in contemporary cohorts in the DMT era than in historical pre-DMT cohorts.12–14 Other studies compared the prognosis in the same population in periods before and after the start of the treatment area and also found a reduction of progression in the period after the start of DMT.15,16 Treated compared with untreated patients from the same populations and cohorts showed advantages of treatment.17,18 Conversely, studies from British Columbia showed weak albeit non-significant positive correlations between treatment and hazards for EDSS 6.7,19,20 These positive correlations may be results of indication bias like in the conventional Cox regression of our study. Contemporary more complete ascertainment of benign cases than earlier may also explain a better prognosis together with a general tendency of increasing incidence. A better prognosis can also be a result of a drift of the natural course of the disease caused by unknown factors or by change in lifestyle, for example, less smoking. 21
In the Danish MS population, we have with the nationwide Danish MS Registry showed decreasing hazards for EDSS 4 and EDSS 6 in the more recent onset cohorts (2001–2005 and 2006–2010) compared with the 1996–2000 onset cohort. 4 This improvement could be caused by a more extensive use of DMT through these decades. In the first onset cohort just after DMT had become available, there was not the same focus on early treatment as with later cohorts, and in these years, some neurologists recommended postponing DMT in patients without a recent relapse and with mild symptoms until the patient had a new relapse or showed new inflammatory lesions on MRI. Conversely, in the years of the later onset cohorts, early treatment was recommended to all patients with relapsing MS, and studies have demonstrated the advantages of early start of treatment.10,22–24 More drugs, particularly second-generation drugs with high efficacy, have become available over the years and have proved their beneficial effect. 11 This could by itself explain the improved prognosis in more recent onset cohorts. However, in daily clinical practice, patients are typically selected individually for initiation or escalation of treatment by their current disease activity at any time through observation. Thus, an element of reverse causation from indication bias may be present. Analyzing the effect of treatment in real-world data (RWD) is particularly difficult for several reasons: One is that – unlike in RCTs – treatment in real-life settings is not a simple binary or three-variate exposure defined at the start. During the observation periods from onset, patients often wait some time from onset to start of DMT, and after start, they may switch treatment to and fro medium- and high-efficacy DMT and no treatment several times. Another major problem is bias because of unknown confounders or known confounders of unknown value.
A way to reduce these biases is analyses with instrumental variables (IVs), 25 which is a rather new tool in the MS context. 26 IV is a covariate which is causally associated with exposure but not with outcomes. A recent example of the use of IVs is Mendelian randomization with which the hypothesized causal relationship between migraine and MS was disproved. 27
The aims of this study are to demonstrate how to mitigate the effects of unknown or unmeasured confounders and indication bias with estimations using ‘IVs’ and to show how DMT affects the prognosis of relapsing-onset MS using the nationwide Danish Multiple Sclerosis Registry (DMSR) 28 as a data source.
Methods
Data source
In this study, we have used prospectively collected data from DMSR. The DMSR, 28 formally established in 1956, is a nationwide and population-based registry that collects information on all persons with an approved diagnosis of MS according to the current diagnostic criteria. Since 1996, it has been mandatory to follow all patients under DMT with regular clinical visits and to notify the DMSR about relapses, EDSS, and side effects, enabling high completeness of registration. The registry is clinician-driven. Diagnostic and clinical management of patients with MS and related disorders are carried out by 13 Departments of Neurology/MS clinics in public hospitals, and these are the only units that are authorized to prescribe and dispense disease-modifying drugs. The study candidates are all MS patients fulfilling the current diagnostic criteria with the onset of relapsing MS between 1996 and 2010. Also, patients with clinically isolated syndrome were offered DMT, particularly if they had oligoclonal bands in the cerebro spinal fluid (and thus, they would also have fulfilled the later 2017 McDonald criteria for dissemination in time). 29 A systematic regular recording of EDSS was in the majority of patients, particularly in earlier onset cohorts, undertaken only after their start of DMT. Consequently, EDSS values have been missing in the interval from the onset of MS until the start of the patient’s first DMT treatment. To avoid problems with left truncation, we replaced these missing values with expected EDSS values for each year, calculated by linear interpolation from EDSS = 0.0 the day before the onset of MS to the recorded value at the start of the first DMT. We compared the three onset cohorts: 1996–2000, 2001–2005 and 2006–2010, and we limited the observation of each patient to the first 12 years from onset in all three cohorts to make them comparable. EDSS values assessed within 90 days from the onset of a relapse were disregarded. We used two EDSS endpoints: sustained EDSS 4 (significant disability but able to walk without aid or rest for 500 m), and sustained EDSS 6 (walking aid required to walk 100 m). An EDSS endpoint was sustained when and if EDSS never subsequently got below the value, and near the end of observation it should be confirmed at least at one subsequent visit.
Study design
We conducted a prospective cohort study with treatment index as IVs and time to EDSS 4 and EDSS 6 as dependent variables.
Treatment
At any moment through follow-up, DMT was classified into three categories: 0 for no treatment; 1 for treatment with moderate-efficacy DMT (meDMT); and 2 for treatment with high-efficacy DMT (heDMT). The group of meDMTs included Interferon beta-1a, interferon beta-1b, peginterferon beta-1a, glatiramer acetate, teriflunomide and dimethylfumarate, and the group of heDMTs included natalizumab, fingolimod, ofatumumab, alemtuzumab, cladribine, mitoxantrone, ocrelizumab and rituximab. For the analyses, we used a ‘treatment index’ which we calculated as 12 × (yHE + yME × 0.5)/yObs), where yHE is the years under heDMT; yME is the years under meDMT; and yObs is the number of years observed within the 12-year follow-up period. In the analyses, we used treatment index as exposure variables.
Statistical analyses
In the first part, we analyze the association between treatment index and time to the endpoints EDSS 4 and EDSS 6 using conventional Cox regression adjusted for age at onset and sex for all cohorts together, and for each of the three onset cohorts separately. All hazard ratios (HRs) are calculated per unit treatment index. To validate the direction of the HRs, we also correlated the progression rate (EDSS increase/observation time) with the treatment index for each onset cohort and altogether.
Inherent biases with this type of analyses are caused by unknown or unmeasured cofounders and by indication because disease activity during observation also prompts decisions about treatment. To avoid these biases, we used a novel epidemiological method ‘IV’. An IV is an external covariate which is causally associated with exposure and only indirectly with the outcome (via exposure). Estimations with IV are statistical techniques used to estimate the causal relationship between an intervention and an outcome variable while controlling for unknown or unmeasured confounders. Figure 1 shows a diagram of causal pathways. The IV technique has been extended to be used in survival statistics with Cox regression.30–32 As shown in Figure 2, the MS onset period is causally associated with treatment pattern, and the onset period thereby fulfils the conditions of being an IV. Not being available in all commercial statistical software, IV-time-to-event analyses have now become accessible with the package ‘ivtools’ under the statistical language R. 33 We used ‘two-stage estimation’ and ‘G-estimation’. In ‘two-stage estimation’, we first regress the treatment index on the IV with the covariates sex and age at onset. Next, we enter this fitted value for each case instead of exposure into a regression on the outcome to get valid estimates of the treatment effect. ‘Two-stage’ estimation may sometimes lead to bias outside the linear modelling framework, and in these cases, G-estimation is more robust. The G-technique constructs prediction of the counterfactual outcome for each subject if treatment was given versus if treatment was withheld. Results for both techniques will be shown.
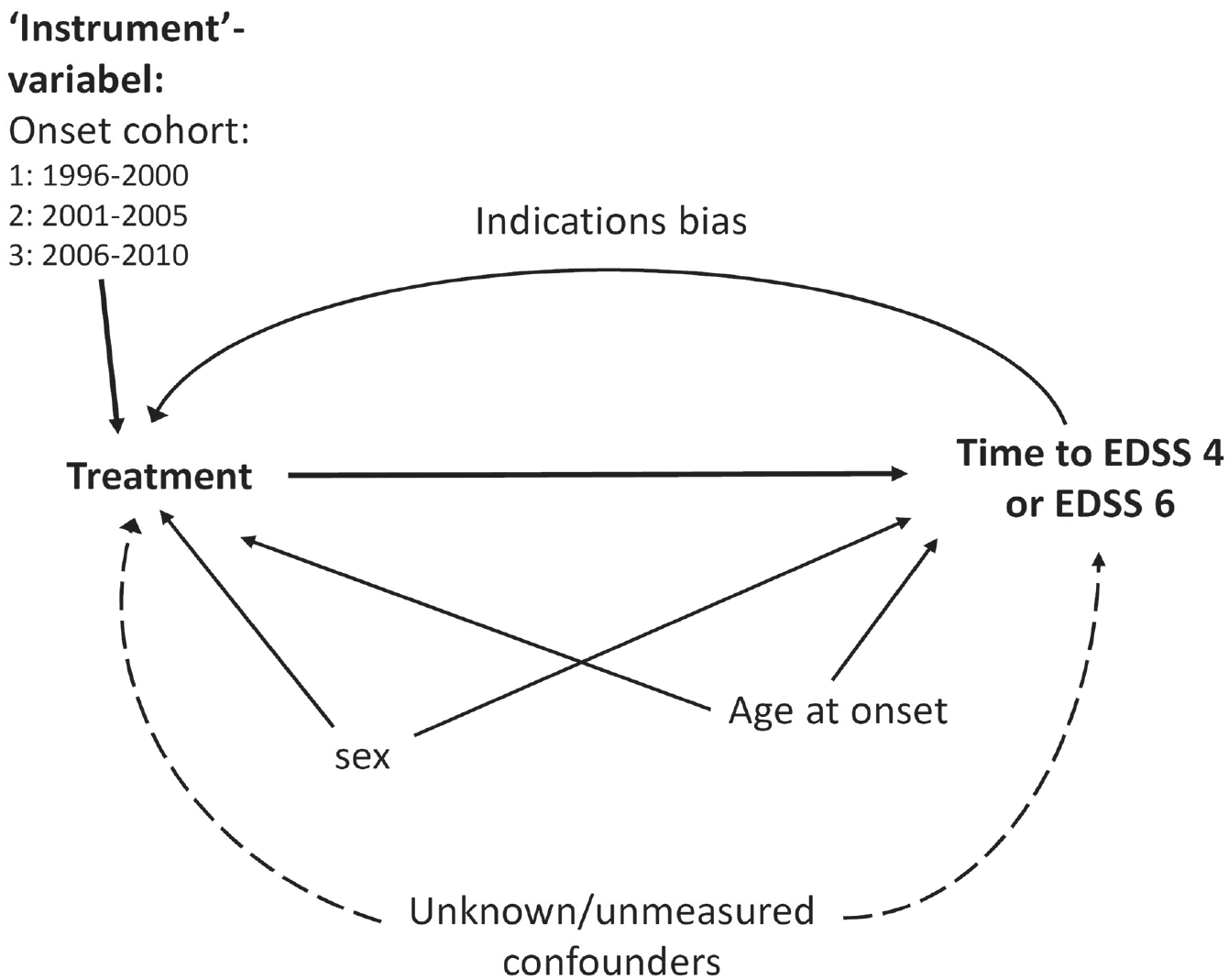
Causal pathways.
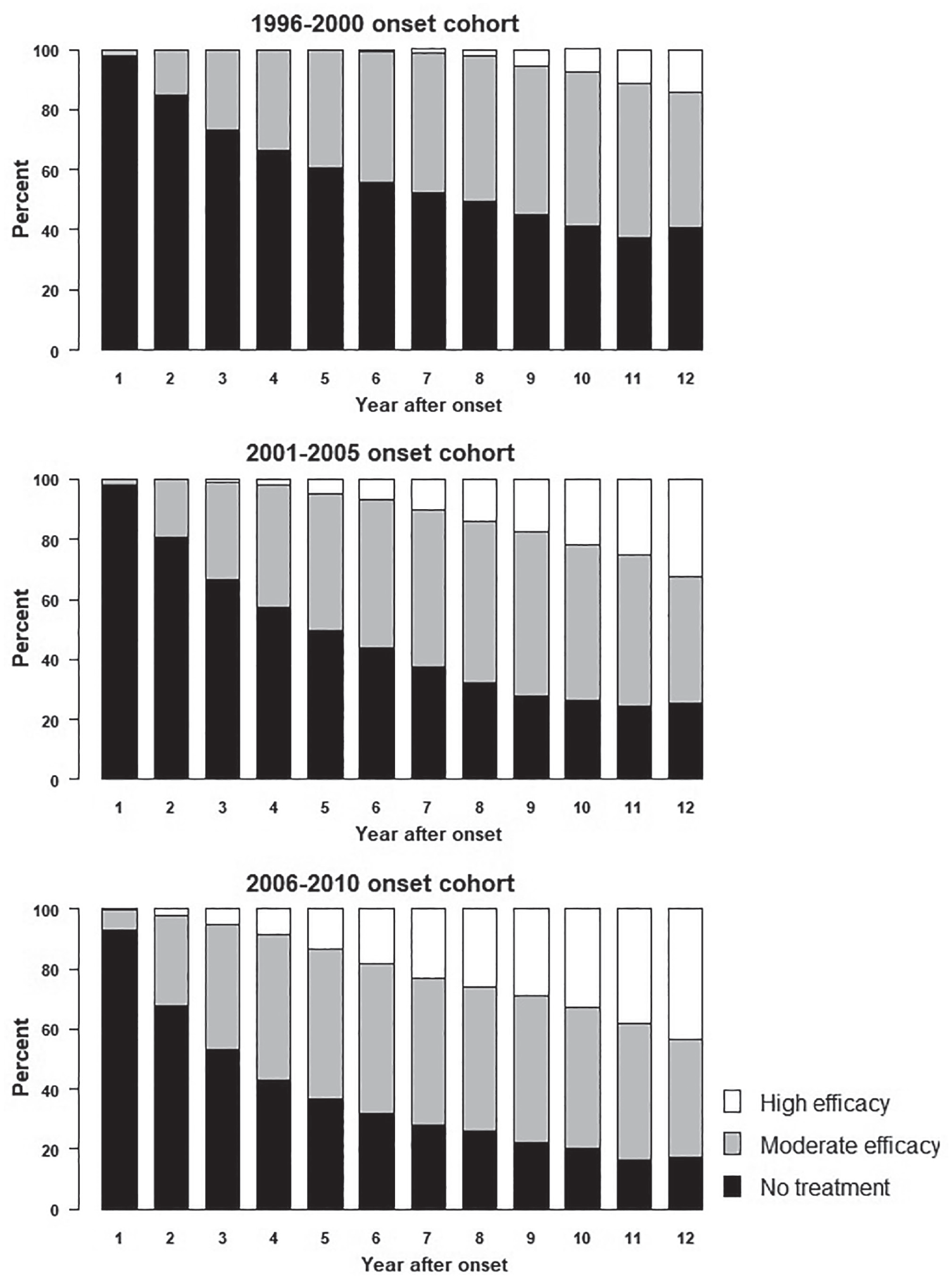
Treatment pattern within the first 12 years after onset in the three onset cohorts.
Data management was performed using SPSS v 25 and Excel Visual Basic, and Cox regression with and without IV was performed using R 4.2.2.
Results
All 6033 patients with relapsing onset of MS (ROMS) from 1996 to 2010 were targeted. Only 19 were excluded because they dropped out of treatment and had been observed with less than two control visits or less than 1 year, leaving 6014 cases to be included. Table 1 shows the distribution of clinical and demographical variables by onset cohort. The greatest differences between the tree onset cohorts were the shortening of time from onset of MS to initiation of treatment the treatment indexes which increased from mean values of 3.32 to 5.09. The different distributions on the three treatment levels for each of the first 12 years for the three onset cohorts are shown in Figure 2.
Descriptives by onset cohort.
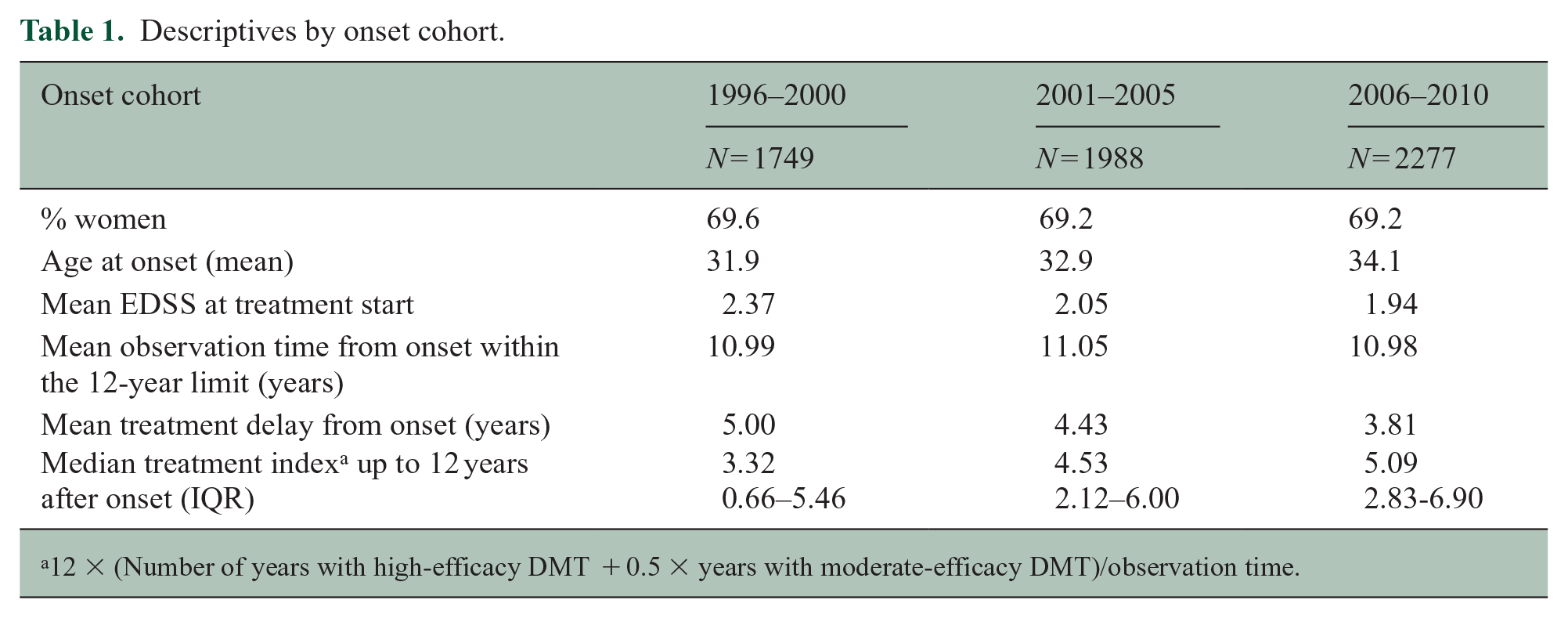
12 × (Number of years with high-efficacy DMT + 0.5 × years with moderate-efficacy DMT)/observation time.
Conventional analyses
In the first part of the analyses with conventional Cox regression adjusted for sex and age at onset for each onset cohort, DMT unexpectedly appeared to be harmful as the HRs per unit treatment index were greater than 1.0 for all three onset cohorts. These results are expanded in Table 2 with stratification by onset cohort and 95% confidence interval (CI). The HR estimates for both endpoints were closer to unity in the latest 2006–2010 onset cohort, where it did not attain statistical significance, than in the preceding cohorts. This reflects that confounding by indication became less pronounced with time.
Conventional Cox regression results a per unit treatment index by onset cohort.
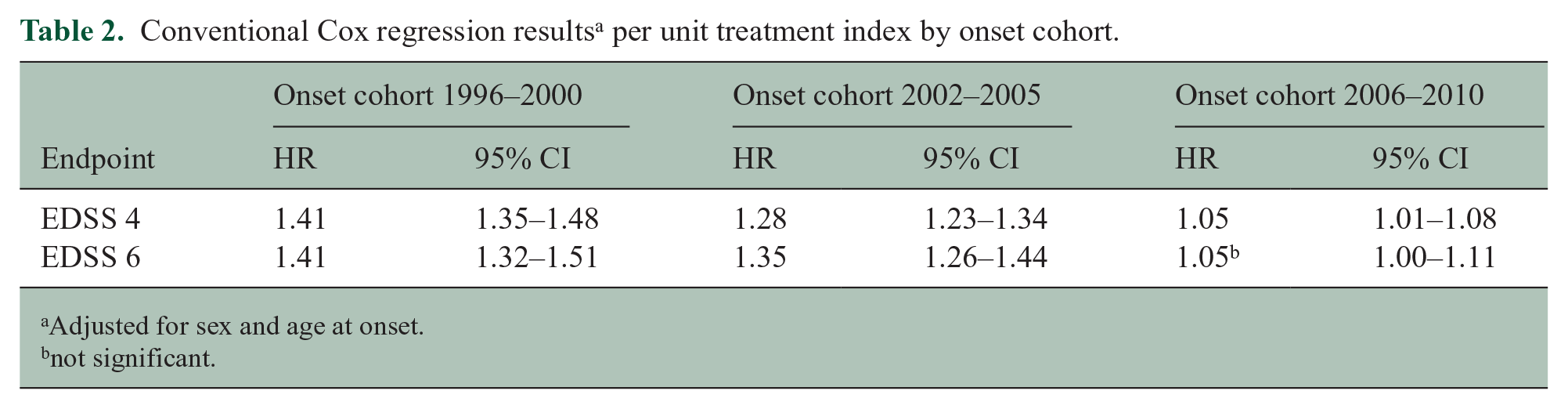
Adjusted for sex and age at onset.
not significant.
To validate these results, we also correlated progression rate with treatment index in non-parametric analysis. Again, the correlation was positive with Spearman ϱ = 0.287; 95% CI by bootstrapping with 1000 samples: 0.263–0.313; p < 0.0001. This correlation diminished with time: For the 1996–2000 cohort, Spearman ϱ was 0.513 (95% CI 0.472–0.557); for the 2001–2005 cohort, it was 0.357 (0.312–0.400), and for the 2006–2010 cohort, it was 0.078 (0.039–0.117).
Analyses with IV
However, by introducing IV analyses, the HR estimates turned around as the indication bias disappeared, and it disclosed a strong beneficial effect of treatment: The HRs for the EDSS 4 and EDSS 6 endpoints with G-estimates were reduced by 14% and 18% per unit treatment index. Details are displayed in Table 3 both without IV and with IV with ‘two-stage’- and with ‘G-estimation’.
Cox regression results a per unit treatment index, corrected with IV – methods.
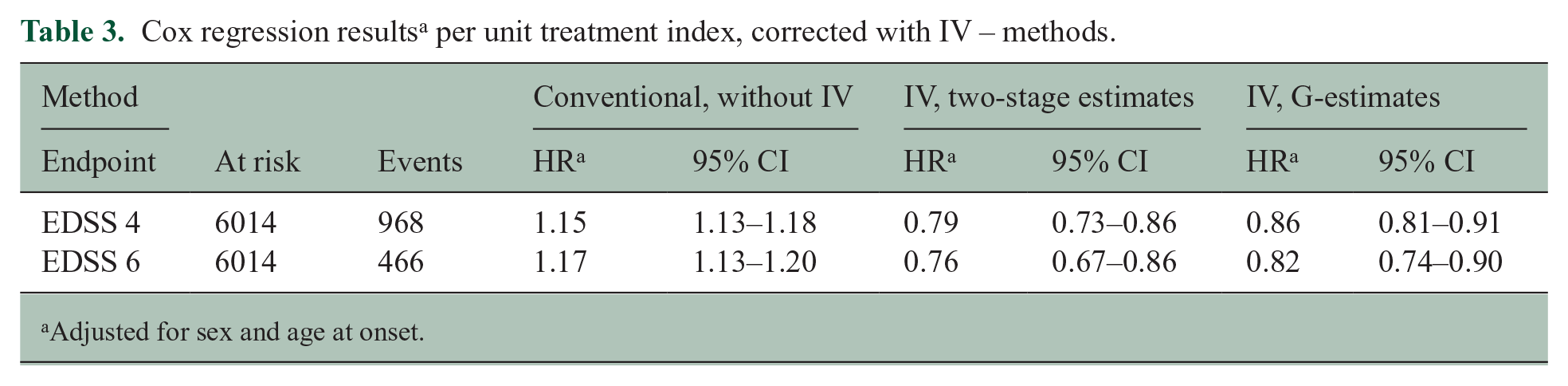
Adjusted for sex and age at onset.
Discussion
This observational study of 6014 MS patients with ROMS from the DMSR showed that within 12 years from onset, the risk for sustained EDSS 4 and 6 at first seemed to increase with increasing treatment index, thus suggesting a harmful effect of DMT. These results are obviously incorrect. However, after abating the effects of reverse causality, indication bias, and unmeasured confounders by Cox regression estimated with IV, we found that DMT has a strong effect in reducing the risk of reaching the endpoints of irreversible EDSS 4 or EDSS 6. Likewise important is that with IV estimations it is possible to correct for otherwise incorrigible bias, and that onset cohort in the present context is a proper IV. To be an IV, the covariate (in case cohort) must be associated with exposure (treatment) and have no direct effect on the outcome except through exposure. 31 One of the studies showing the beneficial effect of early treatment start had also regressed for covariates causally associated with exposure but not with outcome. 24 Along with time to EDSS endpoints from the onset, age at the endpoints has also shown to be higher in recent onset cohorts attributable to changes in treatment patterns. This has been shown by Capra et al. 16 and Magyari et al. 4 We do not believe that change in diagnostic criteria is a significant unmeasured confounder in our study because the onset period for the cases in the study only spans from 1996 to 2010 which period, we only used the Poser Criteria and the McD1 criteria, and their sensitivity was not much different in contrast to subsequent changes with the later generations of the McD criteria.
The apparent and spurious strong detrimental effect of treatment in the conventional analysis may be attributed to the fact that the clinical parameters used for the selection of patients for DMT, particularly in the first periods of the DMT era, are also strong predictors of disability, and this effect overshadowed the beneficial effect of DMT in the conventional analyses. In the years defied by the first onset cohorts, the medical authorities felt responsible for reserving this at that time unprecedentedly expensive treatments to patients with more disease activity who at that time was thought to benefit the most from it. This selection lessened when it later was shown that early treatment results in a better prognosis.
In the two studies from British Columbia that failed to show significant associations between β-interferon exposure and the hazard for disability,7,19 they even found a positive albeit non-significant association between treatment and EDSS 6. These positive correlations are probably results of indication bias like in the conventional Cox regression of our study. The same may be true for another study from British Columbia which failed to show a beneficial effect of beta-interferon on the time to conversion to secondary progressive MS. 20
In the more recent study by Kalincik et al., 18 they pre-empted reverse causality and indication bias by using a marginal structural models which is a counterfactual framework like that of the G-estimation, used in our study, and they found that that treatment reduced the hazards of EDSS 6 with about 60% which is comparable with the protective effect of 18% per unit treatment index in our study.
There are some limitations in our study. The initial course of EDSS from onset up to initiation of DMT was not recorded, but for each year in that period, it was imputed by linear interpolation between a value of null just before onset and the recorded value at the day of treatment start. With this, patients have been followed since onset, and this reduces immortal time bias caused by left truncation at the expense of precision of early time estimates. A general limitation of a study from different centres is the interrater variability and the semi-narrative character of the Kurtzke EDSS- and their underlying Functional System-scales with adjectives like ‘mild’, ‘moderate’ and ‘severe’, and that EDSS assessments sometimes are performed by different physicians within the different centres. However, all patients in our study were treated in dedicated MS clinics, and the definitions of EDSS and FS were always at hand, in the first 12 years of registration on the rear side of the score sheet and later as a drop-down menu.
The strength of our study is the completeness and validity of the registration and substantial statistical power.
In conclusion, this large study confirms reassuringly that DMT is beneficial to people with MS with relapsing onset of MS, as it is highly effective in delaying disability. Moreover, it shows that bias caused by unmeasured confounders, not least indication bias and reversed causality, can be eliminated by using available IVs. Observational studies in real-life settings are an important supplement to clinical trials and the evolving statistical methods improves the validity and reliability of the findings. Whenever IVs are at hand, they should be used to correct for unknown and unmeasured bias in cohort studies of the effects of DMT.
Footnotes
Acknowledgements
We thank all our colleagues in the Danish Multiple Sclerosis Group from the 14 Danish MS Centres who systematically and currently notified the DMSR with data.
Author Contributions
Concept and design: N.K.-H., L.C.T., M.M.
Acquisition of data: N.K.-H., M.M.
Interpretation of data: N.K.-H., L.C.T., M.M., P.S.S.
Drafting the manuscript: N.K.-H., L.C.T.
Critical revision of the manuscript for the important intellectual concept: N.K.-H., L.C.T., M.M., P.S.S.
Statistical analysis: N.K.-H., L.C.T.
Administrative, technical, or material support: M.M.
Data Sharing Information
The data underlying this article cannot be shared publicly due to data protection regulation. Data are accessible to authorized researchers only for the purpose of checking the present study and after application to the Danish Health Data Authority and the board of the Danish Multiple Sclerosis Registry.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N.K.-H. did not declare any competing interests. L.C.T did not declare any competing interests. M.M. has served in scientific advisory board for Sanofi, Novartis, and Merck and has received honoraria for lecturing from Biogen, Merck, Novartis, Roche, Genzyme, and Bristol Myers Squibb. P.S.S. has received personal compensation for serving on advisory boards for Biogen, Merck, Novartis, and Teva; was on steering committees or independent data monitoring boards in trials sponsored by Merck and Novartis; and has received speaker honoraria from Biogen, Merck, Teva, BMS/Celgene, and Novartis. No other disclosures were reported.
Ethical Approval
Involvement of ethical committees was not required because this was an observational register study without study interventions. Before the data were delivered from the register for analysis, the patients’ identities were removed. The Danish Data Protection Agency granted permission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The Danish Multiple Sclerosis Society funds the Danish Multiple Sclerosis Registry.
