Abstract
Background:
Autologous mesenchymal stem cell neurotrophic factor–secreting cells (NurOwn®) have the potential to modify underlying disease mechanisms in progressive multiple sclerosis (PMS).
Objective:
This open-label phase II study was conducted to evaluate safety/efficacy of three intrathecal cell treatments.
Methods:
Eighteen participants with non-relapsing PMS were treated. The primary endpoint was safety. Secondary endpoints included: cerebrospinal fluid (CSF) biomarkers; timed 25-foot walk speed, nine-hole peg test (9-HPT), low-contrast letter acuity, symbol digit modalities test, and 12-item multiple sclerosis (MS) walking scale. Seventeen participants received all treatments.
Results:
No deaths/adverse events related to worsening of MS, clinical/magnetic resonance imaging (MRI) evidence of disease activation, and clinically significant changes in safety lab results were reported. Two participants developed symptoms of low back and leg pain, consistent with a diagnosis of arachnoiditis, occurring in one of three intrathecal treatments in both participants. Nineteen percent of treated participants achieved pre-specified ⩾ 25% improvements in timed 25-foot walk speed/nine-HPT at 28 weeks compared to baseline, along with consistent efficacy signals for pre-specified response criteria across other secondary efficacy outcomes. CSF neuroprotective factors increased, and inflammatory biomarkers decreased after treatment, consistent with the proposed mechanism of action.
Conclusion:
Based on these encouraging preliminary findings, further confirmation in a randomized study is warranted.
Keywords
Introduction
Progressive multiple sclerosis (PMS) 1 is characterized by the accumulation of central nervous system (CNS) injury-related to inflammation, demyelination, axonal damage, neuronal degeneration, and gliosis in both white and gray matter. 2 Effective reparative therapies to reverse the functional impairments in PMS are lacking.
NurOwn (mesenchymal stem cell neurotrophic factor (MSC-NTF) cells) leverages proprietary technology to isolate, propagate in culture, and differentiate autologous bone marrow–derived mesenchymal stem cells (MSCs) to secrete high levels of neurotrophic factors (NTFs) in addition to their well-documented intrinsic immunomodulatory properties. 3
MSC-NTF cells have been successfully evaluated in animal models relevant to PMS, including experimental autoimmune encephalomyelitis 4 and optic nerve transection. 5 The potential of cell-based therapies to address the unmet biological need of compartmentalized inflammation and deficient neuroprotective mechanisms in PMS has been described. 6 A recent study of intravenous (IV) bone marrow–derived MSCs in participants with MS did not demonstrate efficacy on gadolinium enhancing magnetic resonance imaging (MRI) lesions. Recent studies suggest 7 that the intrathecal route of administration may offer unique advantages, due to direct effects on meningeal inflammation and direct delivery of NTFs. 8 Therefore, the capacity of intrathecally administered MSC-NTF cells to directly modulate inflammation and to promote endogenous neuronal repair makes MSC-NTF cells a promising therapeutic modality in PMS.
In PMS, functional outcomes (timed 25-foot walk test (T25FW), nine-hole peg test (9-HPT), low-contrast letter acuity (LCLA), and symbol digit modalities test (SDMT)) provide additional information in the evaluation of PMS beyond the expanded disability status scale (EDSS).9,10 In addition to the use of validated functional/disability outcomes, neurodegenerative 11 and inflammatory 12 biomarkers provide important biological information in PMS where cerebrospinal fluid (CSF) biomarkers demonstrate residual compartmentalized CNS inflammation 13 that correlates with PMS severity. 14
We report results of the BCT-101 Phase II clinical trial that evaluated the safety, preliminary clinical efficacy, and biomarker outcomes of repeated intrathecal administration of MSC-NTF cells in participants with PMS.
Materials and methods
Study design
The BCT-101 study (NCT03799718) was conducted from March 2019 to March 2021, at four MS academic centers in the United States and in accordance with the International Council for Harmonization Good Clinical Practice Guidelines. The study was designed by the sponsor (Brainstorm Cell Therapeutics, Ltd) in consultation with the site principal investigators and monitored by an independent data and safety monitoring board (Figure 1).
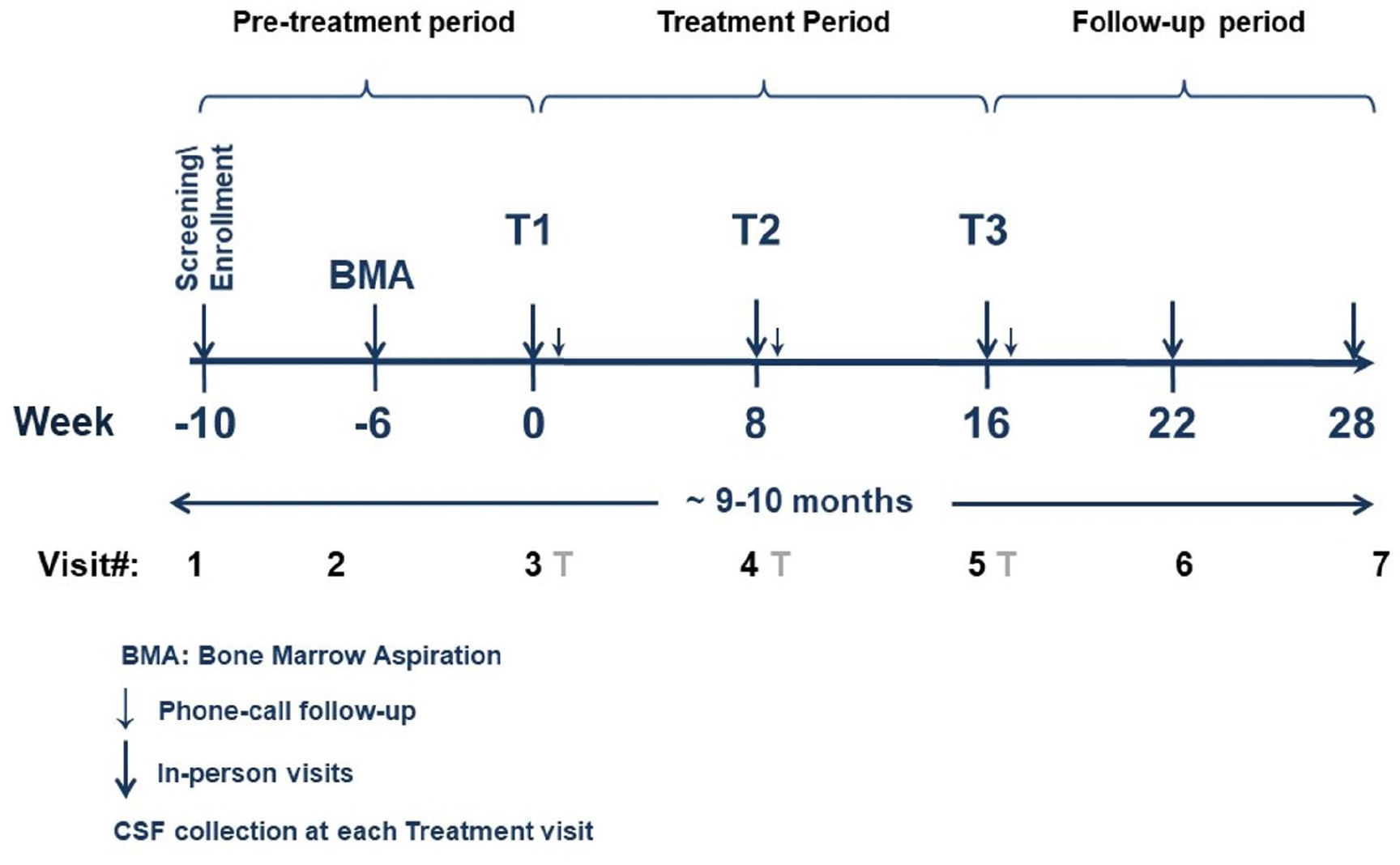
NurOwn progressive MS Phase II trial (BCT-101) design.
Written informed consent was obtained according to the Declaration of Helsinki and was approved by the ethics committee of the institution in which the work was performed.
BCT-101 was an open-label single-arm study. Eligible participants were males and females (18–65 years of age) with primary/secondary progressive multiple sclerosis (PPMS/SPMS), with no relapse for 6 months prior to screening, baseline EDSS scores 3.0–6.5, and ability to walk 25 feet in 60 seconds or less. Participants were allowed to continue use of a stable dose of an approved (nonexcluded) disease-modifying therapy (DMT). After an approximate 10-week pre-treatment period that included an outpatient bone marrow aspiration to obtain mesenchymal cells for manufacturing, participants received three intrathecal administrations of autologous MSC-NTF cells at Weeks 0, 8, and 16, followed by a 12-week post-treatment observation period.
MSC-NTF cells preparation and administration
Approximately, 1–4 weeks after the screening visit, harvested bone marrow was transported to the Connell and O’Reilly Families Cell Manipulation Core Facility at the Dana–Farber Cancer Institute (DFCI) in Boston, MA, USA, where cell manufacturing was completed under Current Good Manufacturing Practice for isolation and expansion of each autologous MSC product before being cryopreserved. The unique NTF secretion profile and micro RNA (miRNA) profiling of MSC-NTF cells has been previously described and involves a medium-based approach that results in overexpression of neuroprotective factors, including NTFs and miRNAs that have shown to be beneficial in several preclinical models of neurodegenerative disease.15,16 The manufacturing process does not include genetic modification of the MSC cells of origin or use of any animal proteins or antibiotics. Fresh autologous MSC-NTF cells were released for transplantation when they fulfilled the cell number, viability, safety (sterility, mycoplasma, and endotoxin), potency (using an enzyme-linked immunosorbent assay (ELISA) for NTF secretion), and identity (CD surface markers) release criteria. Prior to each treatment, the MSC product was thawed, expanded, and induced to differentiate into MSC-NTF cells. MSC-NTF cells were transported back to the clinical site in a validated shipping system at a controlled temperature of 2°C–8°C in a 5 mL syringe containing 100–125 million cells and administered by lumbar puncture at each treatment, as determined in prior experiments.17 –19 The autologous manufacturing process was performed on a per-participant basis with the arrival of fresh bone marrow aspirate to the cleanroom facility at the manufacturing site and was completed once MSC-NTF cells were ready for administration.
Primary and secondary endpoints
The primary endpoint was safety and tolerability of three intrathecal doses of MSC-NTF cells, as assessed based on the incidence of treatment-emergent adverse events as well as clinically relevant changes in vital signs, requirement for concomitant medications, clinical laboratory assessments (hematology, serum chemistry, and urinalysis), and physical/neurological examinations. Brain MRI (fluid-attenuated inversion recovery (FLAIR)) was performed to evaluate T2 lesion status at baseline and at end of study as a safety outcome.
Secondary outcomes also evaluated the clinical efficacy of MSC-NTF cells using clinical outcome measures: T25FW speed (feet/second), 9-HPT (second), EDSS (0–10 scale), LCLA, SDMT (0–120 scale), 12-item MS walking scale (MSWS-12, 0–100 scale), four-component MS functional composite (MSFC-4 score (average z-score)).
Biomarkers (neuroinflammatory, neuroprotective, and neurodegenerative biomarkers) were collected and analyzed from CSF/serum.
Biomarker analyses
CSF samples were collected by lumbar puncture prior to each administration of MSC-NTF cells, for a total of three collections. CSF was immediately centrifuged at 1750 g for 10 minutes and stored at −80°C. Serum samples were collected before and 24 hours after each transplantation, and 1 month after the last treatment, for a total of seven collections. Blood samples were allowed to clot at room temperature for 30 minutes, centrifuged at 1300 × g for 10 minutes to separate serum, and stored at −80 °C. CSF samples were first collected in May 2019, followed by the analyses in March 2021.
Neuroprotective biomarkers (vascular endothelial growth factor A (VEGF-A), hepatocyte growth factor (HGF), neural cell adhesion molecule 1 (NCAM-1), fetuin-A, follistatin, and leukemia inhibitory factor (LIF)), and neuroinflammatory biomarkers (monocyte chemoattractant protein-1 (MCP-1), osteopontin, stromal cell-derived factor 1 (SDF-1), and soluble CD27 (sCD27)) were detected with a highly sensitive, customized ProcartaPlex multiplex immunoassay (Thermo Fisher Scientific, Waltham, MA). Chitotriosidase-1 (CHIT-1) was analyzed by ELISA (MBL International, MA, USA). Neurodegenerative biomarkers (neurofilament light chain (NfL) protein, phosphorylated neurofilament heavy chain (pNFH), and glial fibrillary acidic protein (GFAP)) were analyzed by the Simoa technology assays (Quanterix Corporation, Lexington, MA, USA), performed by VUMC, Amsterdam, NL. The biomarkers were pre-specified and assays were thoroughly validated by matrix evaluation, including spike recovery, parallelism, and sample stability.
MRI analyses
The MRI acquisition/analysis protocol used was developed by Icometrix NV (Leuven, Belgium) and included 2D/3D T1 and 3D FLAIR brain MRI scans, performed at enrollment visit and at 28 weeks. MRI scans were performed following site MRI quality review of dummy runs and MRI data were stored in keeping with Good Clinical Practice guidelines. Central processing of uploaded MRI-digital imaging and communications in medicine (DICOM) format images was performed by Icometrix NV using Icobrain MS software.
Statistical analysis
Safety analyses included a summary of discontinuations and associated reasons, along with adverse events summarized by system organ class and preferred term of Medical Dictionary for Regulatory reporting. Adverse events were further summarized by severity, and relationship to study intervention. Laboratory data and vital signs were summarized by changes from baseline to Week 28 and incidence of abnormalities.
Efficacy analyses were based on observed data with no imputation for missing data. Continuous variables were assessed by absolute and percent change from baseline to each postbaseline assessment and were summarized along with the number of participants with available data (n), means, and standard deviation (SD). For categorical data, the number/percentage of participants was summarized. Denominator for percentages was set to the number of participants with observed data at that timepoint. Efficacy analyses were conducted by evaluating the number and percentage of responders based on pre-defined response thresholds. Since there was no concurrent control, no hypothesis testing was performed.
To provide relevant clinical context, similar pre-specified analyses were conducted on a matched cohort from the Comprehensive Longitudinal Investigation of MS at the Brigham & Women’s Hospital (CLIMB) registry from Tanuja Chitnis, MD, Brigham & Women’s Hospital, Harvard Medical School. Forty-eight select participants out of 500 total eligible participants matched to BCT-101 inclusion criteria (males/females, ages 18–65 at screening visit with clinical diagnosis of PMS based on the 2017 revised MacDonald Criteria and confirmation by the Investigator that the disease has entered the progressive stage for at least 6 months prior to enrollment, and disability status at screening with an EDSS of 3.0–6.5). CLIMB data were collected for each participant at two timepoints, 1–2 years apart. CLIMB efficacy assessment results were obtained using a linear approximation to interpolate changes through Week 28. The matched CLIMB participants were pre-specified and completed at the time of BCT-101 study initiation.
All biomarker data were log-transformed and percent changes were calculated by taking the antilog enabling graphs to be presented in the original units. Geometric means and the first (Q1) and the third (Q3) quartiles were presented. For HGF, one patient had an extreme negative value, so that, the value was excluded to estimate Q1, but remained in the analysis of the geometric mean.
Data availability
The authors confirm that the core of the data supporting the findings of this study is available within the article.
Results
Participants
The CONSORT diagram is presented in Figure 2. A total of 23 participants with primary/secondary PMS were screened. Of these, 20 underwent bone marrow aspiration. In two participants, autologous bone marrow culture failed to yield adequate number of MSC/MSC-NTF cells and they did not receive treatment. In total, 18 participants (ten females and eight males) with a mean ± SD EDSS score of 5.4 ± 1.3, and a mean age of 47.4 ± 9.6 years were treated. The mean disease duration was 17.7 ± 7.9 years since first MS symptoms. Demographics/baseline characteristics of study participants are detailed in Table 1. The majority had a diagnosis of SPMS (14/18; 78%) and most (13/18; 72%) were receiving anti-CD20 therapies (ocrelizumab/ rituximab). In the cohort of matched CLIMB patients, 26/48 (54%) were receiving anti-CD20 therapies.
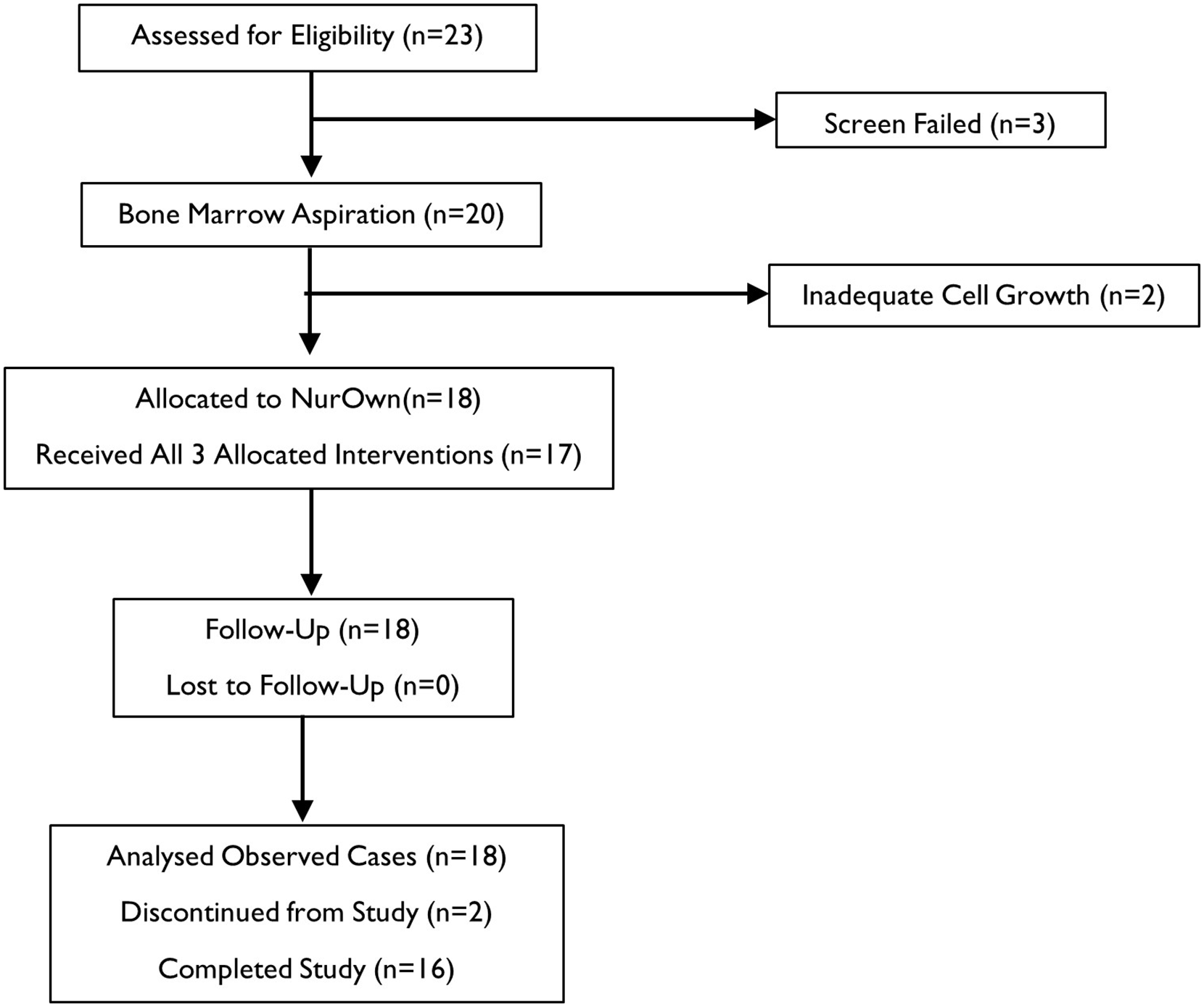
CONSORT flow diagram.
Demographics and baseline characteristics: BCT-101 versus CLIMB.
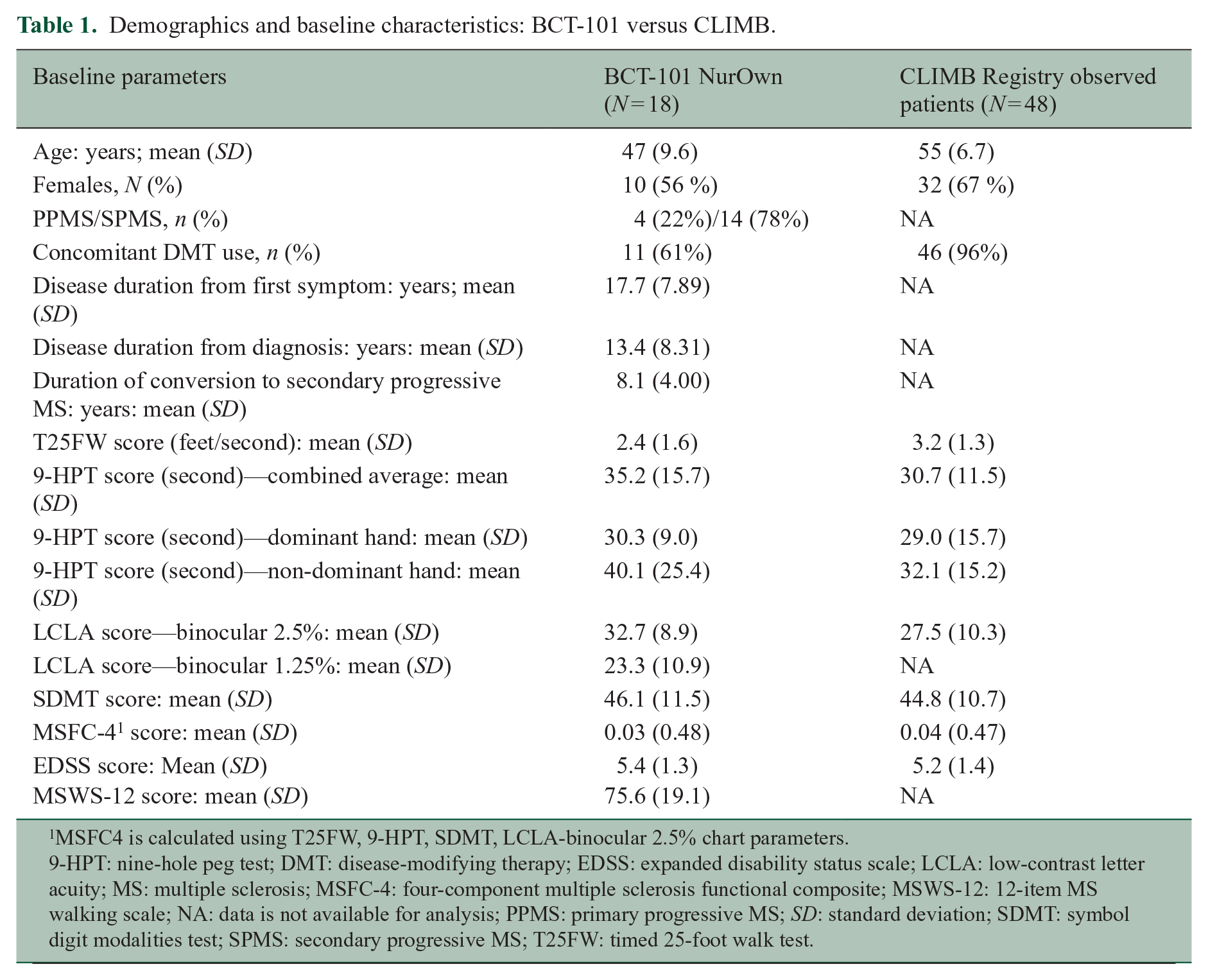
MSFC4 is calculated using T25FW, 9-HPT, SDMT, LCLA-binocular 2.5% chart parameters.
9-HPT: nine-hole peg test; DMT: disease-modifying therapy; EDSS: expanded disability status scale; LCLA: low-contrast letter acuity; MS: multiple sclerosis; MSFC-4: four-component multiple sclerosis functional composite; MSWS-12: 12-item MS walking scale; NA: data is not available for analysis; PPMS: primary progressive MS; SD: standard deviation; SDMT: symbol digit modalities test; SPMS: secondary progressive MS; T25FW: timed 25-foot walk test.
Primary endpoint
Safety
Of the 20 participants enrolled, 18 were treated, 17 received all three treatments, and one received two treatments. Two participants discontinued due to procedure-related adverse events, including feeling cold, muscle weakness, and pyrexia in one participant and arachnoiditis in another. There were no study deaths or adverse events related to MS relapses. Two serious treatment-emergent adverse events occurred during the study resulting in participant hospitalization (Table 2). Two treated participants developed symptoms of low back and leg pain, consistent with a diagnosis of arachnoiditis, occurring in one of three intrathecal treatments in both participants. Lumbar MRI in both cases showed characteristic clumping of lumbar roots. Both participants were treated with epidural cortisone injections and analgesics, and the symptoms completely resolved in one participant, who subsequently completed the third intrathecal treatment without the adverse event recurrence. In the second case, the symptoms occurred only after the third intrathecal treatment and did not fully resolve.
Overall summary of treatment-emergent adverse events (TEAEs).
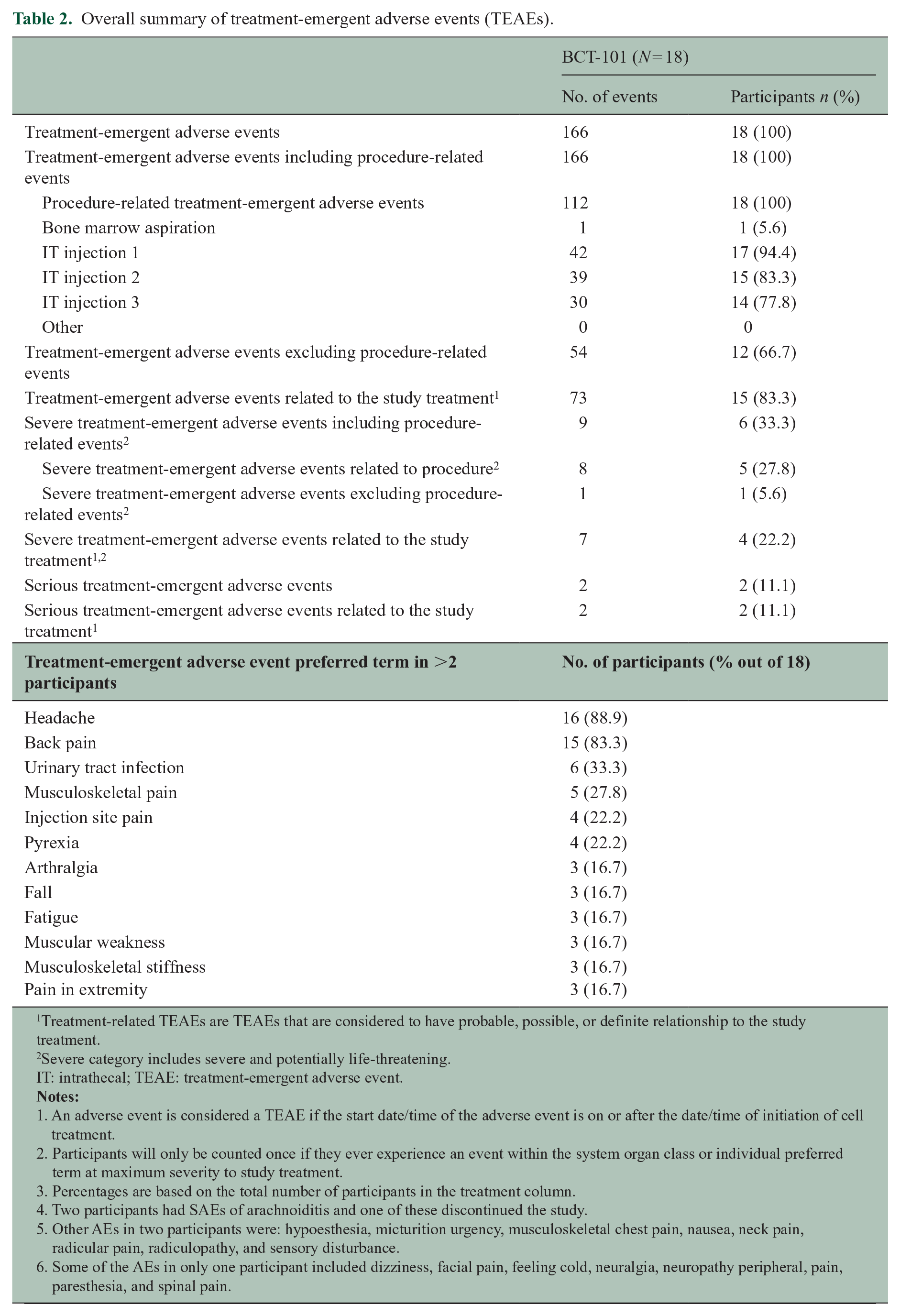
Treatment-related TEAEs are TEAEs that are considered to have probable, possible, or definite relationship to the study treatment.
Severe category includes severe and potentially life-threatening.
IT: intrathecal; TEAE: treatment-emergent adverse event.
1. An adverse event is considered a TEAE if the start date/time of the adverse event is on or after the date/time of initiation of cell treatment.
2. Participants will only be counted once if they ever experience an event within the system organ class or individual preferred term at maximum severity to study treatment.
3. Percentages are based on the total number of participants in the treatment column.
4. Two participants had SAEs of arachnoiditis and one of these discontinued the study.
5. Other AEs in two participants were: hypoesthesia, micturition urgency, musculoskeletal chest pain, nausea, neck pain, radicular pain, radiculopathy, and sensory disturbance.
6. Some of the AEs in only one participant included dizziness, facial pain, feeling cold, neuralgia, neuropathy peripheral, pain, paresthesia, and spinal pain.
There were no clinically significant changes following dosing in safety lab results (complete blood count, coagulation, chemistry, and urinalysis) or vital signs (heart rate, respiratory rate, or blood pressure) in any subject.
No changes were observed on mean brain MRI-FLAIR lesion volume or count in BCT-101 or matched CLIMB patients estimated over 28 weeks and they were comparable at baseline.
Secondary and exploratory efficacy endpoints
Summary of Responder Analysis—Clinical Efficacy Endpoints Over 28 Weeks
Using a pre-specified threshold for a clinical response of 25% or greater improvement in T25FW speed or 9-HPT (combined dominant and non-dominant hands), 19% (3/16) of treated participants were classified as responders at Week 28 (Table 3). Looking at each of these two endpoints individually, 14% (2/14) and 13% (2/15) of MSC-NTF cell-treated participants were classified as responders, respectively. In contrast, ⩽ 5% of matched CLIMB patients achieved any of these pre-specified outcomes. Thirty-eight percent (6/16) of treated participants showed at least a 10-point improvement in the MSWS12. Sixty-seven percent (10/15) showed at least a 3-point improvement in the SDMT. Forty-seven percent (7/15) of treated participants showed at least an eight-letter improvement in LCLA at the 1.25% contrast threshold. Twenty-seven percent (4/15) showed at least an eight-letter improvement in LCLA 2.5% contrast threshold. None of the participants with baseline EDSS ⩽ 5.5 showed improvement of ⩾ 1.0, while 30% (3/10) of participants with baseline EDSS > 5.5 showed improvement of ⩾ 0.5.
Responder analysis —efficacy endpoints over 28 weeks in BCT-101.
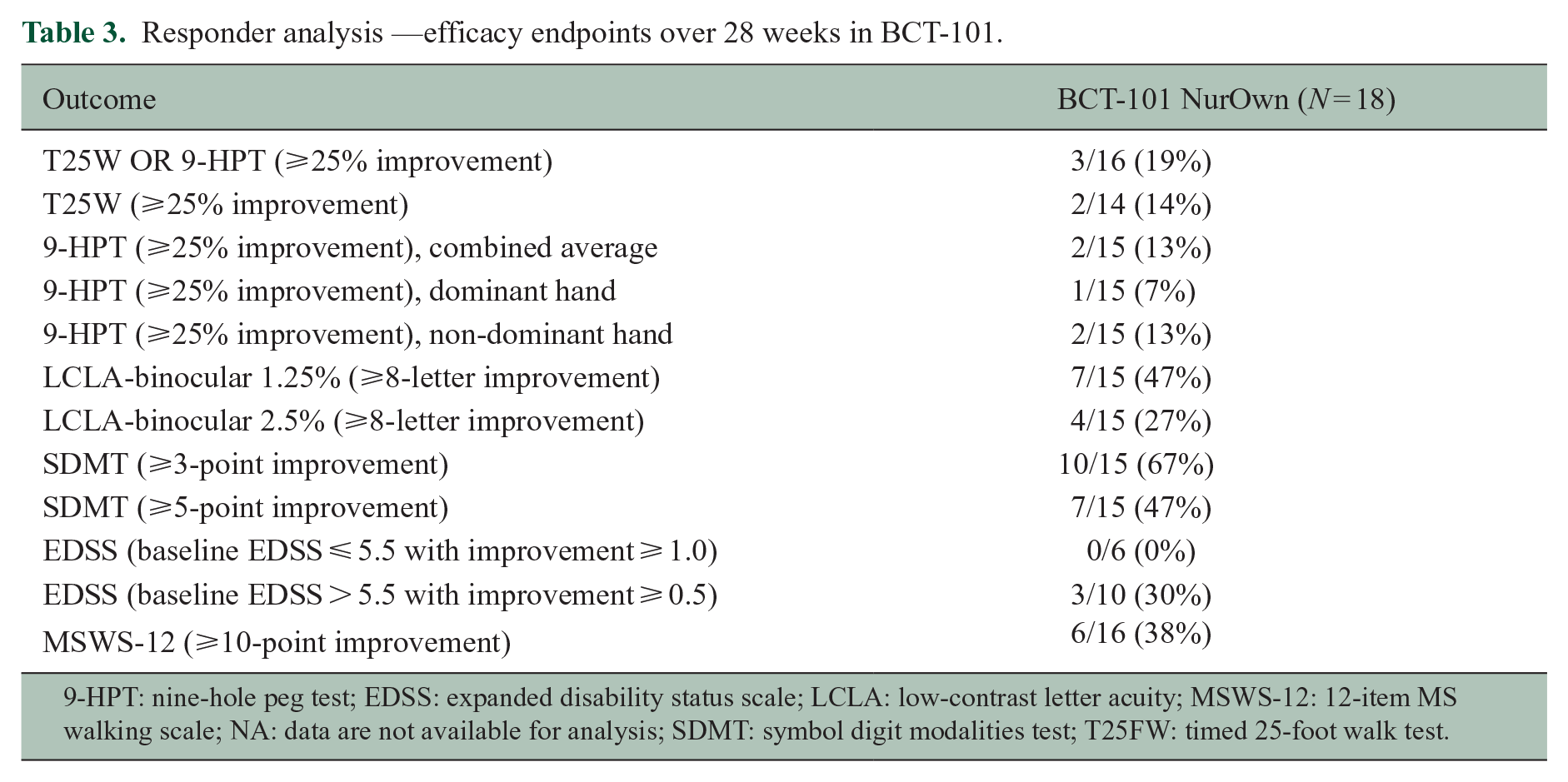
9-HPT: nine-hole peg test; EDSS: expanded disability status scale; LCLA: low-contrast letter acuity; MSWS-12: 12-item MS walking scale; NA: data are not available for analysis; SDMT: symbol digit modalities test; T25FW: timed 25-foot walk test.
Median Change from Baseline—Clinical Efficacy Endpoints Over 28 Weeks
The outcomes across key efficacy endpoints are highlighted in Figure 3 and Table 4. MSC-NTF cell-treated participants showed a median change from baseline to Week 28 of −0.05 feet/second in T25FW. MSC-NTF cell-treated participants showed a median improvement from baseline of −0.8 second on 9-HPT (combined both hands).
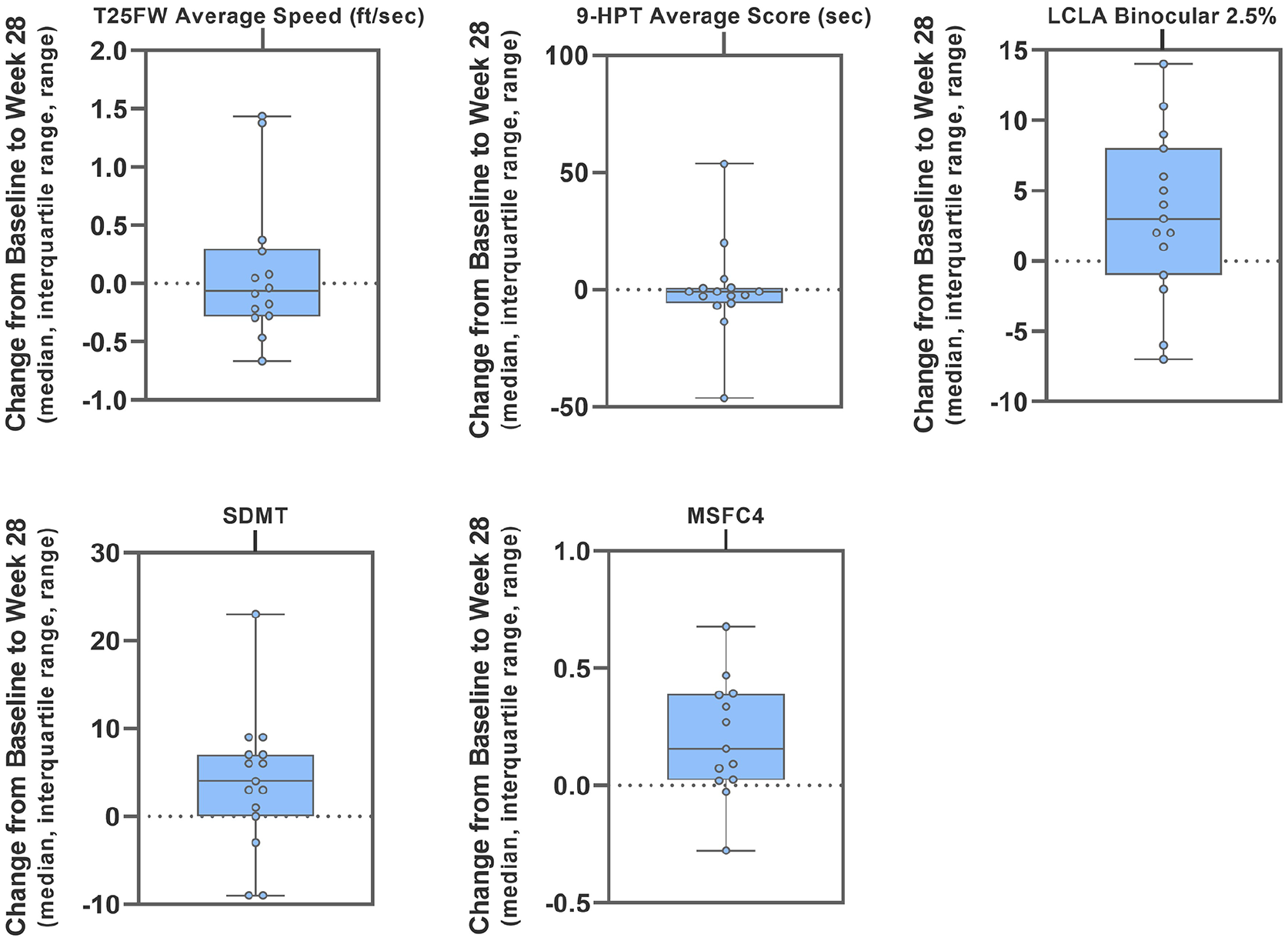
Efficacy endpoints—median change from baseline at 28 weeks.
Median changes from baseline over 28 2eeks in BCT-101.

9-HPT: nine-hole peg test; EDSS: expanded disability status scale; LCLA: low-contrast letter acuity; MSFC-4: four-component multiple sclerosis functional composite; MSWS-12: 12-item MS walking scale; NA: data are not available for analysis; SDMT: symbol digit modalities test; T25FW: timed 25-foot walk test.
MSFC4 is calculated using T25FW, 9-HPT, SDMT, LCLA-binocular 2.5% chart parameters.
Worsening.
The composite MSFC-4 (normalized T25FW, 9-HPT, LCLA, and SDMT) median change showed an improvement from baseline of 0.10. MSWS-12 and EDSS showed no median change from baseline at Week 28.
CSF Biomarkers
Treatment resulted in consistent trends for increases in the percent change from baseline to Week 16 in CSF neuroprotective factors (VEGF-A, HGF, NCAM1, follistatin, LIF, and fetuin-A) and a reduction in percent change from baseline in most CSF inflammatory biomarkers (MCP-1, SDF-1, osteopontin, and CD27) (Table 5 and Figure 4). CSF neurodegenerative biomarkers (NfL, pNFH, and GFAP) did not show consistent changes following treatment. Due to small the sample size, no statistical inferences were made, while focusing on trends.
CSF biomarkers: mean change from baseline at Week 16.
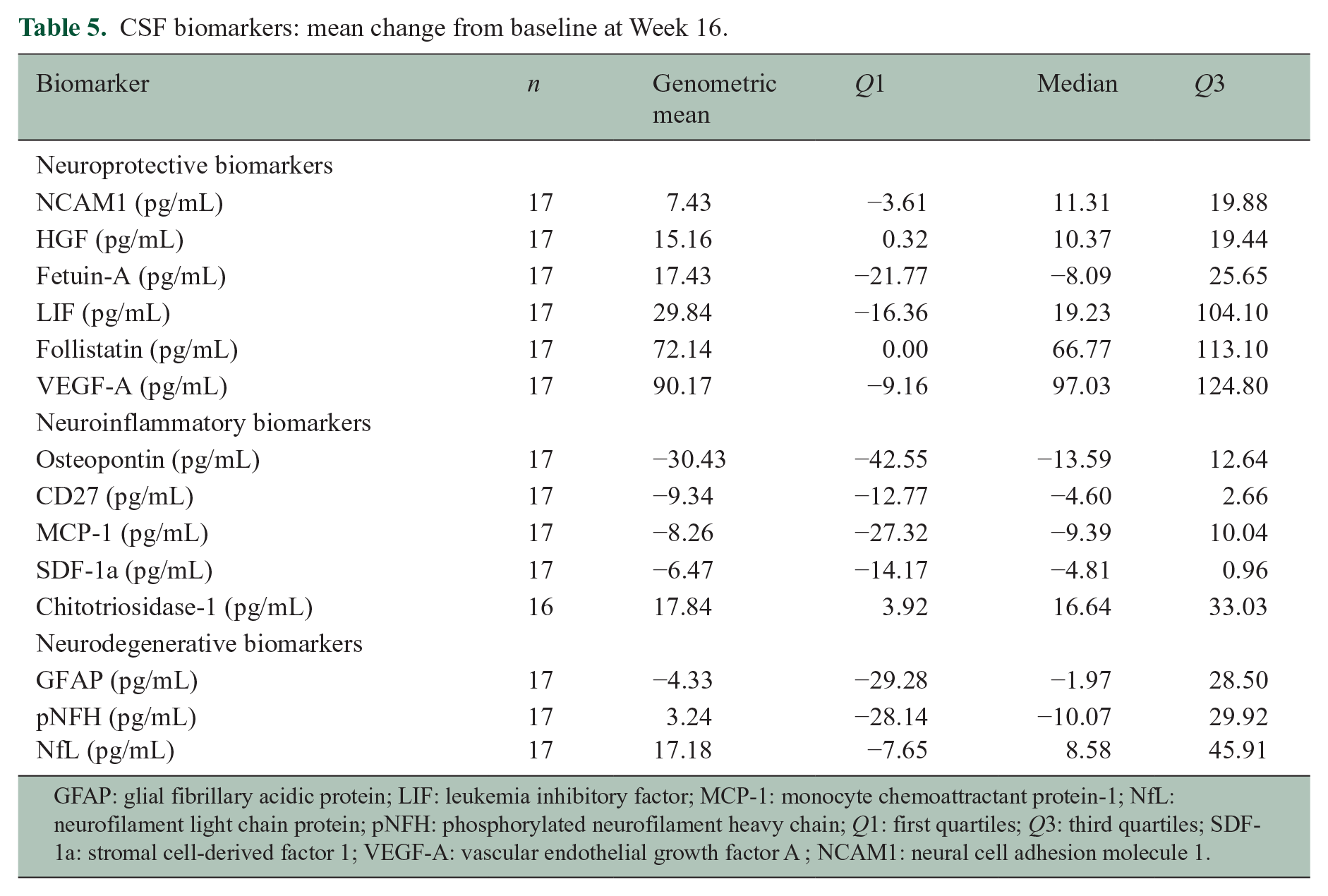
GFAP: glial fibrillary acidic protein; LIF: leukemia inhibitory factor; MCP-1: monocyte chemoattractant protein-1; NfL: neurofilament light chain protein; pNFH: phosphorylated neurofilament heavy chain; Q1: first quartiles; Q3: third quartiles; SDF-1a: stromal cell-derived factor 1; VEGF-A: vascular endothelial growth factor A ; NCAM1: neural cell adhesion molecule 1.
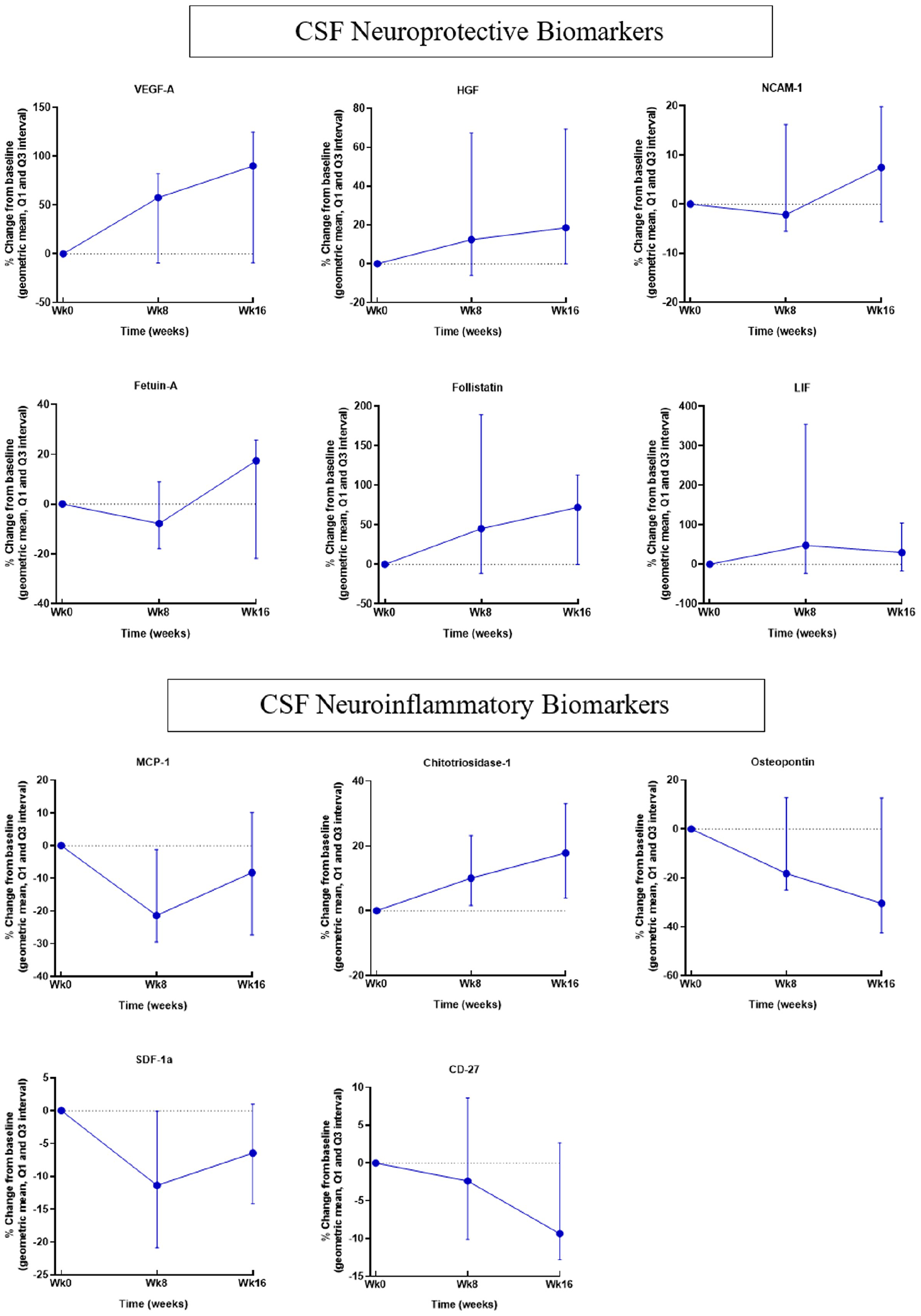
CSF neuroinflammatory and neuroprotective biomarkers.
Discussion
This open-label, single-arm phase II study of MSC-NTF cells in participants with PMS demonstrated good overall safety/tolerability. Arachnoiditis was observed in two participants, each occurring in one of three intrathecal treatments. This adverse event can be seen following routine lumbar puncture, epidural steroid injection, intrathecal treatment in the context of lumbar degenerative disk disease, 20 and following intrathecal administration of adipose-derived MSCs in amyotrophic lateral sclerosis (ALS). 21 Arachnoiditis may be confirmed by MRI as clumping of lumbar nerve roots as was observed, although the MRI features lack specificity and may not always be accompanied by symptoms. 22
Recent studies in ALS, 21 multiple system atrophy, 23 and spinal cord injury 24 have confirmed that intrathecal MSC can be safely administered in doses up to 100 × 10. 6 In the current study, two participants discontinued due to treatment-emergent adverse events (arachnoiditis and nonspecific symptom). No other significant safety signals were detected. There was no change in brain T2 lesion volume or count in brain FLAIR MRI measures over 28 weeks to suggest disease activation. The safety of intrathecal administration of MSC-NTF cells has been demonstrated in Phase II/III randomized clinical trials in ALS.18,19 The majority of the adverse events in that study were related to the intrathecal administration procedure, which were generally short-lived and mild/moderate in severity.
Based on pre-specified thresholds, encouraging responses were observed in T25FW, 9-HPT, SDMT, and LCLA tests. These functional endpoints add important outcome information in PMS. 25 The observed efficacy outcomes were greater than that observed in matched CLIMB patients, however, in the absence of a randomized control group, these observations require cautious interpretation. We observed positive changes in MSWS-12, a validated patient-reported measure of walking function. 26 EDSS was unchanged following MSC-NTF cell treatment. In PMS, T25FW may record more worsening events per unit time compared to EDSS or 9HPT and may precede and predict EDSS worsening. 27
We enrolled stable PMS participants who were relapse-free for 6 months at screening and maintained on a stable dose of their previously prescribed DMTs, with a majority (13/18) receiving anti-CD20 therapy. MSC-NTF cells do not express CD20 mRNA or protein (data on file Brainstorm Cell Therapeutics). We did not observe changes in MRI FLAIR lesion count or volume, suggesting the absence of measurable disease activity during the clinical trial. Therefore, it is not possible to extrapolate the observations in this study to PMS patients experiencing clinical or MRI disease activity.
CSF biomarker analyses demonstrated reductions across most inflammatory biomarkers, including MCP-1, sCD27, SDF-1, and osteopontin. Both MCP-1 and SDF-1 play a role in the recruitment of inflammatory cells into the CNS.28,29 CSF soluble CD27 (sCD27) may be an important marker of meningeal inflammation/intrathecal T-cell activation in MS. It shows comparable changes in SPMS/PPMS patients decrease following treatment. Osteopontin may be an early indicator of intrathecal inflammation in PMS. 30 Osteopontin haplotypes may be associated with MS disease progression. 31
We also observed consistent increases across CSF neuroprotective biomarkers, including VEGF-A, HGF, NCAM1, follistatin, LIF, and fetuin‑A. CSF-VEGF-A, 32 HGF, 33 and NCAM1 34 levels are reported to be decreased in PMS. We did not observe consistent changes in neurodegenerative biomarkers following treatment; CSF-NfL, for example, has not shown a clear relationship with measures of disability in PMS. 35 While Petrou et al. 8 have shown a reduction of CSF neurofilament following in PMS patients with active disease, other studies have shown no change in CSF-NfL after intrathecal MSC therapy 36 or serum NfL after IV MSCs. 37 MSC-NTF cells have been observed to decrease NfL and pNfH in ALS participants across 28 weeks. 19 Further studies are needed to determine the utility of CS- NfL and other neurodegenerative biomarkers as treatment outcome measures in PMS.
Initial reports suggest that CSF GFAP may emerge as a potential marker of PMS disease severity. 38 We observed inconsistent changes in CSF neurodegenerative biomarkers, which may be related to the small sample size, variability between patients, duration of measurement, or inherent responsiveness of these biomarkers in the evaluation of neuroprotective therapies in PMS.
Intrathecal delivered cell therapies may offer specific advantages by directly addressing unresolved compartmentalized inflammation 14 and/or failure of neuroprotective mechanisms in PMS and have shown superior outcomes compared to IV administration in clinical studies8,39 and in the experimental autoimmune encephalomyelitis preclinical model. 40
MSC-NTF cells have unique primed cargo, through culture-based differentiation, including enhanced secretion of neuroprotective factors while maintaining immunomodulatory functions, including increased T/B regulatory function. 41 The combined activity of immunomodulation and neuroprotection may be relevant to PMS.
Other small non-randomized clinical trials have demonstrated preliminary evidence of safety and efficacy in participants with progressive or advanced MS.42 –46 This study adds to the growing body of evidence supporting additional investigation of intrathecal MSC therapy to potentially address the unmet medical need in PMS.
This small open-label study did not directly compare treatment outcomes with a randomized placebo-treated group; therefore, the interpretation of efficacy data may be limited by expectation bias. A limitation of the biomarker analysis was that the third CSF specimen was obtained just prior to the third treatment and, therefore, only reflects the effect of the first two treatments.
In summary, we report the safety and preliminary clinical and biomarker outcomes from a Phase II clinical trial of MSC-NTF cells in participants with stable, non-relapsing PMS. In view of the open-label uncontrolled design, the clinical observations will require confirmation in a placebo-controlled trial.
Supplemental Material
sj-pdf-1-msj-10.1177_13524585221122156 – Supplemental material for Evaluation of neurotrophic factor secreting mesenchymal stem cells in progressive multiple sclerosis
Supplemental material, sj-pdf-1-msj-10.1177_13524585221122156 for Evaluation of neurotrophic factor secreting mesenchymal stem cells in progressive multiple sclerosis by Jeffrey A Cohen, Fred D Lublin, Christoper Lock, Daniel Pelletier, Tanuja Chitnis, Munish Mehra, Yael Gothelf, Revital Aricha, Stacy Lindborg, Chaim Lebovits, Yossef Levy, Afsaneh Motamed Khorasani and Ralph Kern in Multiple Sclerosis Journal
Supplemental Material
sj-pdf-2-msj-10.1177_13524585221122156 – Supplemental material for Evaluation of neurotrophic factor secreting mesenchymal stem cells in progressive multiple sclerosis
Supplemental material, sj-pdf-2-msj-10.1177_13524585221122156 for Evaluation of neurotrophic factor secreting mesenchymal stem cells in progressive multiple sclerosis by Jeffrey A Cohen, Fred D Lublin, Christoper Lock, Daniel Pelletier, Tanuja Chitnis, Munish Mehra, Yael Gothelf, Revital Aricha, Stacy Lindborg, Chaim Lebovits, Yossef Levy, Afsaneh Motamed Khorasani and Ralph Kern in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors thank the study participants and their families, without whom this study would not have been accomplished. Brainstorm Cell Therapeutics wishes to thank Brainstorm’s R&D, Manufacturing, Quality Control, and Quality Assurance teams. They also thank the Dana–Farber Cancer Institute (DFCI) in Boston, MA for work carried out in the Connell and O’Reilly Families Cell Manipulation Core Facility (CMCF) led by Dr Jerome Ritz, who manufactured autologous MSC-NTF cells for participants enrolled in this trial.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.A.C. reports personal compensation for consulting for Bio-gen, Bristol—Myers Squibb, Convelo, Genentech, Janssen, NervGen, Novartis, and PSI; speaking for H3 Communications; and serving as an Editor of Multiple Sclerosis Journal. F.L. reports research funding from: Novartis, Actelion, Biogen, Sanofi, NMSS, NIH, Brainstorm Cell Therapeutics; consulting agreements/advisory boards/DSMB activities with: Biogen, EMD Serono, Novartis, Teva, Actelion/Janssen, Sanofi/Genzyme, Acorda, Roche/Genentech, MedImmune/Viela Bio/Horizon Thera-peutics, Receptos/Celgene/BMS, TG Therapeutics, Medday, Atara Biotherapeutics, Mapi Pharma, Apitope, Orion Biotechnology, Brainstorm Cell Therapeutics Jazz Pharmaceuticals, GW Pharma, Mylan, Immunic, Population Council, Avotres, Neurogene, Banner Life Sciences, Labcorp, Entelexo Biotherapeutics, Neuralight; stock Options from Avotres; and speaking for Sanofi (nonpromotional). C.L. reports personal compensation for speaking, advisory boards, or consulting for Biogen, Sanofi, EMD Serono, Alexion Pharmaceuticals, Bristol Myers Squib, Greenwich Biosciences, InterX Inc., and Diagnose Early. D.P. reports consulting compensation for advisory board activities from Biogen, Sanofi/Genzyme, EMD Serono, and Roche/Genentech. T.C. reports personal compensation for consulting for Biogen, Novartis, Genentech, and Sanofi-Genzyme, and research funding from Brainstorm Cell Therapeutics, Novartis and Verily. M.M. is an employee of Tigermed. Y.G. is an employee of Brainstorm Cell Therapeutics. R.A. is an employee of Brainstorm Cell Therapeutics. S.L. is an employee of Brainstorm Cell Therapeutics. C.L. is an employee of Brainstorm Cell Thera-peutics. Y.S.L. is an employee of Brainstorm Cell Therapeutics. A.M.K. reports personal compensation for consulting for Argenix, Atara Biotherapeutics, and Brainstorm Cell Therapeutics. R.K. is an employee of Brainstorm Cell Therapeutics.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Brainstorm Cell Therapeutics. A grant (grant no: FF181232990) was received from the National Multiple Sclerosis Society, through its commercial funding Fast Forward program, to support the biomarker analyses in this clinical trial.
Registration and protocol
The study was registered on Clinicaltrials.gov (registration # NCT03799718). First Posted: January 10, 2019. First patient (09-101-001) was screened at Cleveland Clinic on March 13, 2019.
The details of the review committees and approvals are listed in the table below for BCT-101-US Phase II Multiple Sclerosis review committee approvals.
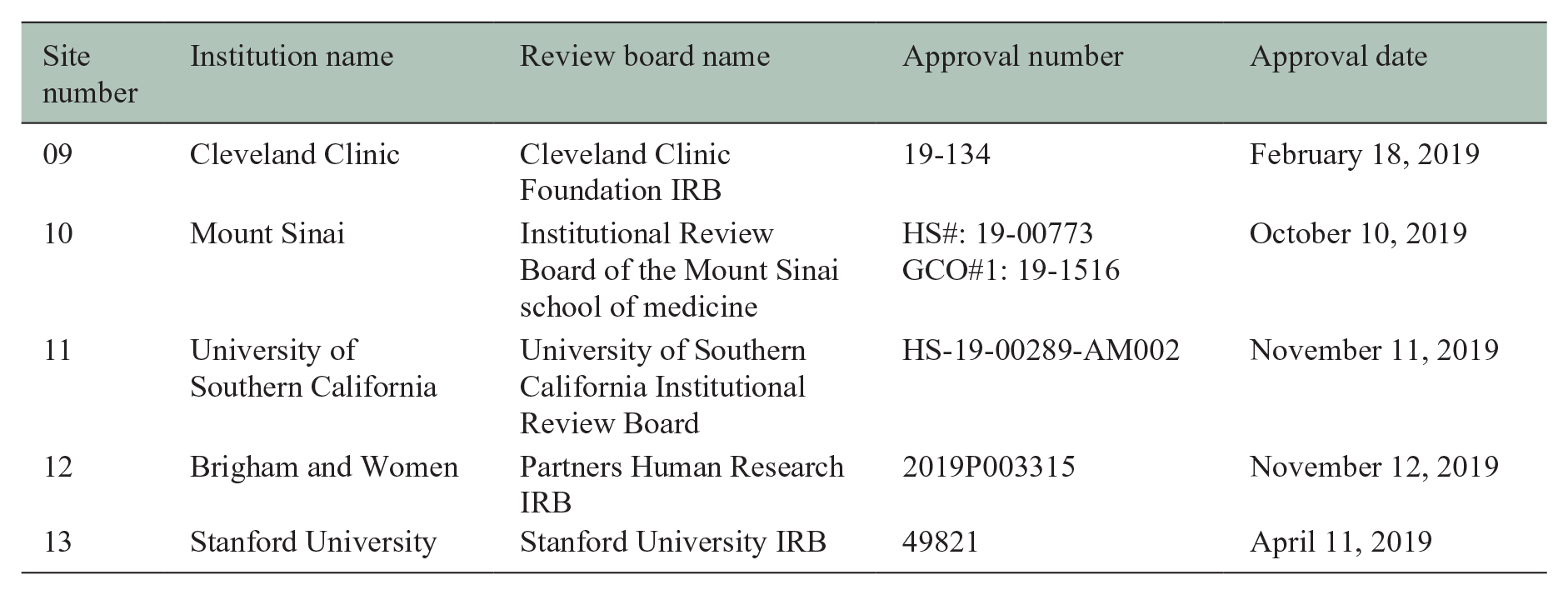
| Site number | Institution name | Review board name | Approval number | Approval date |
|---|---|---|---|---|
| 09 | Cleveland Clinic | Cleveland Clinic Foundation IRB | 19-134 | February 18, 2019 |
| 10 | Mount Sinai | Institutional Review Board of the Mount Sinai school of medicine | HS#: 19-00773 GCO#1: 19-1516 |
October 10, 2019 |
| 11 | University of Southern California | University of Southern California Institutional Review Board | HS-19-00289-AM002 | November 11, 2019 |
| 12 | Brigham and Women | Partners Human Research IRB | 2019P003315 | November 12, 2019 |
| 13 | Stanford University | Stanford University IRB | 49821 | April 11, 2019 |
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
