Abstract
Background:
People with multiple sclerosis (MS) experience myriad symptoms that negatively affect their quality of life. Despite significant progress in rehabilitation strategies for people living with relapsing-remitting MS (RRMS), the development of similar strategies for people with progressive MS has received little attention.
Objective:
To highlight key symptoms of importance to people with progressive MS and stimulate the design and implementation of high-quality studies focused on symptom management and rehabilitation.
Methods:
A group of international research experts, representatives from industry, and people affected by progressive MS was convened by the International Progressive MS Alliance to devise research priorities for addressing symptoms in progressive MS.
Results:
Based on information from the MS community, we outline a rationale for highlighting four symptoms of particular interest: fatigue, mobility and upper extremity impairment, pain, and cognitive impairment. Factors such as depression, resilience, comorbidities, and psychosocial support are described, as they affect treatment efficacy.
Conclusions:
This coordinated call to action—to the research community to prioritize investigation of effective symptom management strategies, and to funders to support them—is an important step in addressing gaps in rehabilitation research for people affected by progressive MS.
Introduction
Multiple sclerosis (MS) is a chronic demyelinating disease that affects more than 2.8 million people worldwide.1,2 Several MS phenotypes exist—active disease including relapsing-remitting multiple sclerosis (RRMS) defined clinically as including acute or subacute episodes associated with new or increasing neurologic disability, followed by some recovery; and progressive disease including secondary progressive (SPMS) and primary progressive MS (PPMS) and defined as the accumulation of disability that is not associated with relapses. 3 In addition, SPMS follows an initial relapsing-remitting phase, which is not the case for PPMS. 3 To date, there are 22 disease-modifying therapies (DMTs) available to treat RRMS and active SPMS, with only one approved (ocrelizumab) for PPMS. 4 Unfortunately, this means that many patients with progressive disease have very few treatment options. Research is ongoing to identify biomarkers to detect the transition from RRMS to SPMS, yet this transition still relies primarily on expert opinion. However, there are important clinical implications associated with transition from RRMS to SPMS which need to be more rigorously studied. The clinical consequences of progressive MS are varied and cumulative, ranging from mild sensory or visual changes to profound cognitive and motor impairments. The dysfunction that results from progressive MS is worse than RRMS with far reaching implications including loss of jobs, stress to family, and financial strain.5,6 As in the research examining DMTs, the weight of evidence pertaining to symptom management comes overwhelmingly from studies predominantly or solely involving people with RRMS. 7 Collecting more data on progressive MS cohorts will allow for a more accurate and nuanced picture of progressive MS symptoms. A paucity of studies undertaken among people with progressive MS creates an immediate challenge for clinicians and researchers in the field to evaluate symptomatic treatments with specific importance to progressive MS.
Rehabilitation aims to optimize physical and cognitive function and quality of life, incorporating preventive, restorative, compensatory, and maintenance approaches. There is a strong need to study the effect of early preventive interventions and to evaluate management of existing symptoms. A challenge of rehabilitation is its complexity, requiring appropriate expertise for intervention delivery and persistent effort from the person working on it. Nonetheless, people living with MS find rehabilitation helpful and desire rehabilitative and wellness strategies that can help maintain a high quality of life. 8 Recent studies in mixed samples show, for example, disease-modifying effects of exercise that correlate with changes in the brain. 9 In addition, computer-assisted cognitive rehabilitation that improves attention and working memory is accompanied by changes in brain activation and connectivity between brain regions. 10 Capitalizing on redundancy and overlap among brain regions may offer the opportunity to derive rehabilitation-induced improvements in motor and cognitive performance. Whether these improvements are sustained over the longer term is uncertain especially in progressive MS. Much more research is required to decipher which interventions have the greatest potential to halt MS-related decline and even more importantly, determine the potential for pre-habilitation approaches that could prevent symptoms before they arise. 9
The International Progressive MS Alliance (The Alliance) has prioritized and is actively exploring prospects for rehabilitation, regeneration, recovery, prevention, and wellness for people with progressive MS. 11 The Alliance includes MS organizations from around the world, research experts, representatives from industry, and people affected by progressive MS, all dedicated to developing effective methods to treat and ultimately end progressive MS. In May 2018, the Alliance convened a Scientific Congress in Toronto, Canada, that focused on symptom management and rehabilitation in progressive MS. Scientists, industry members, and people affected by MS met to share evidence supporting rehabilitation interventions. This meeting emphasized that the existing evidence was broad but lacked sufficient depth and quality to meet the needs of people with progressive MS. As awareness of the need for faster and more efficient progress in the field grew through this meeting, the idea of highlighting a subgroup of common symptoms amenable to improvement with rehabilitation intervention, and that are important to people with progressive MS emerged as the first step toward improving rehabilitation research. This article describes the approach adopted by the Alliance to advance symptom management, with the urgent call to action to target research efforts aimed at optimizing rehabilitation and improving quality of life for individuals with progressive MS.
The purpose of this study was to identify and highlight common symptoms important to people with progressive MS and stimulate the design and implementation of high-quality studies focused on symptom management and rehabilitation. Specifically, we present four symptoms that are important to people with MS (fatigue, impairment of mobility, and upper limb function, pain, and cognitive impairment); review relevant literature, identify gaps in research, delineate key research questions, and provide a rationale to accelerate work in progressive MS. We quantify historic funding by member societies of the Alliance (the Italian MS Foundation, MS Research Australia, the MS Society of Canada, the UK MS Society, and the National MS Society) specific to these symptoms. Finally, we describe common factors that can affect symptom management and should be considered in MS research.
Symptom management receives limited research funds
To evaluate the extent of research being conducted regarding these symptoms, we examined the allocation of grant funding from the Alliance and its members. We conducted a landscape analysis of the research portfolios of the five managing members of the Alliance and the Progressive MS Alliance. In 2017, $228.7 million in multi-year grants was awarded by these organizations in total. Only 1.3% of total funds were spent on studies focused on fatigue or pain, 3% on mobility and/or upper extremity dysfunction, and 5% on cognitive dysfunction (Figure 1). The vast majority, 90% of funds, were spent on projects that were not focused on symptoms. This reflects a historical focus on projects that enhance the understanding of MS progression through genetics, immunology, and pathological mechanisms, biomarkers and drug discovery, among others. It is also important to acknowledge that a limited pool of applicants and laboratories focus on rehabilitation, symptom management, and overall well-being. The limited funding and focus on rehabilitation research are modifiable. One thing is clear, increased financial investment from the Alliance and other financers into studies of symptom management are needed if progress is to be made. We delineate in the following paragraphs where gaps exist and in Table 1 suggest important unanswered questions.
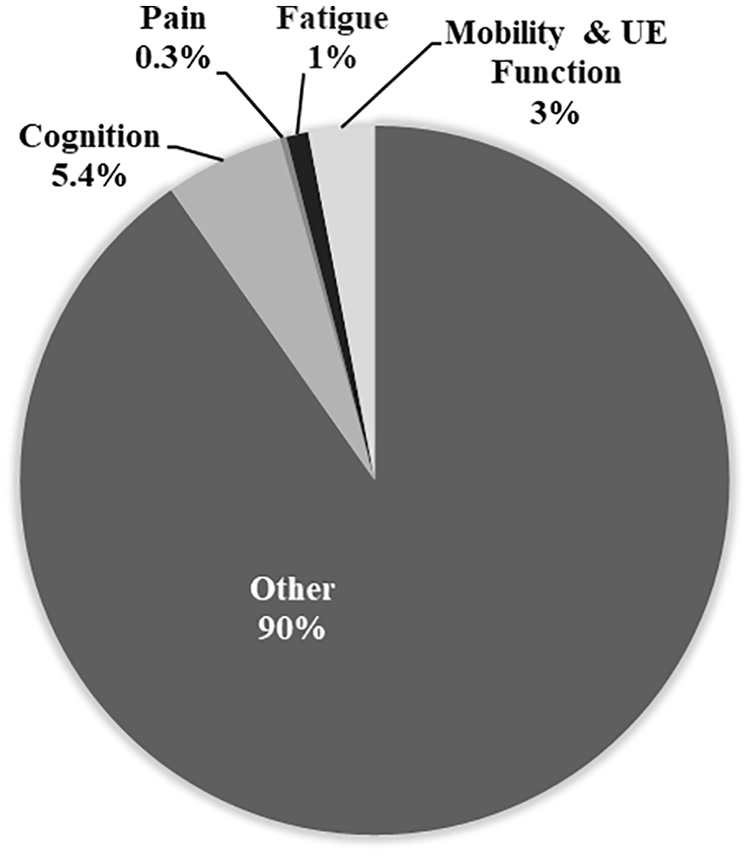
Percentage of total funds from the Progressive MS Alliance and member organizations spent on studies of the four highlighted symptoms. The section titled “Other” includes studies of the brain and spinal cord using imaging (MRI and PET), vision (including OCT and clinical measures), biomarker discovery and development.
Prevalence and key research questions for the four highlighted symptom areas in progressive MS.
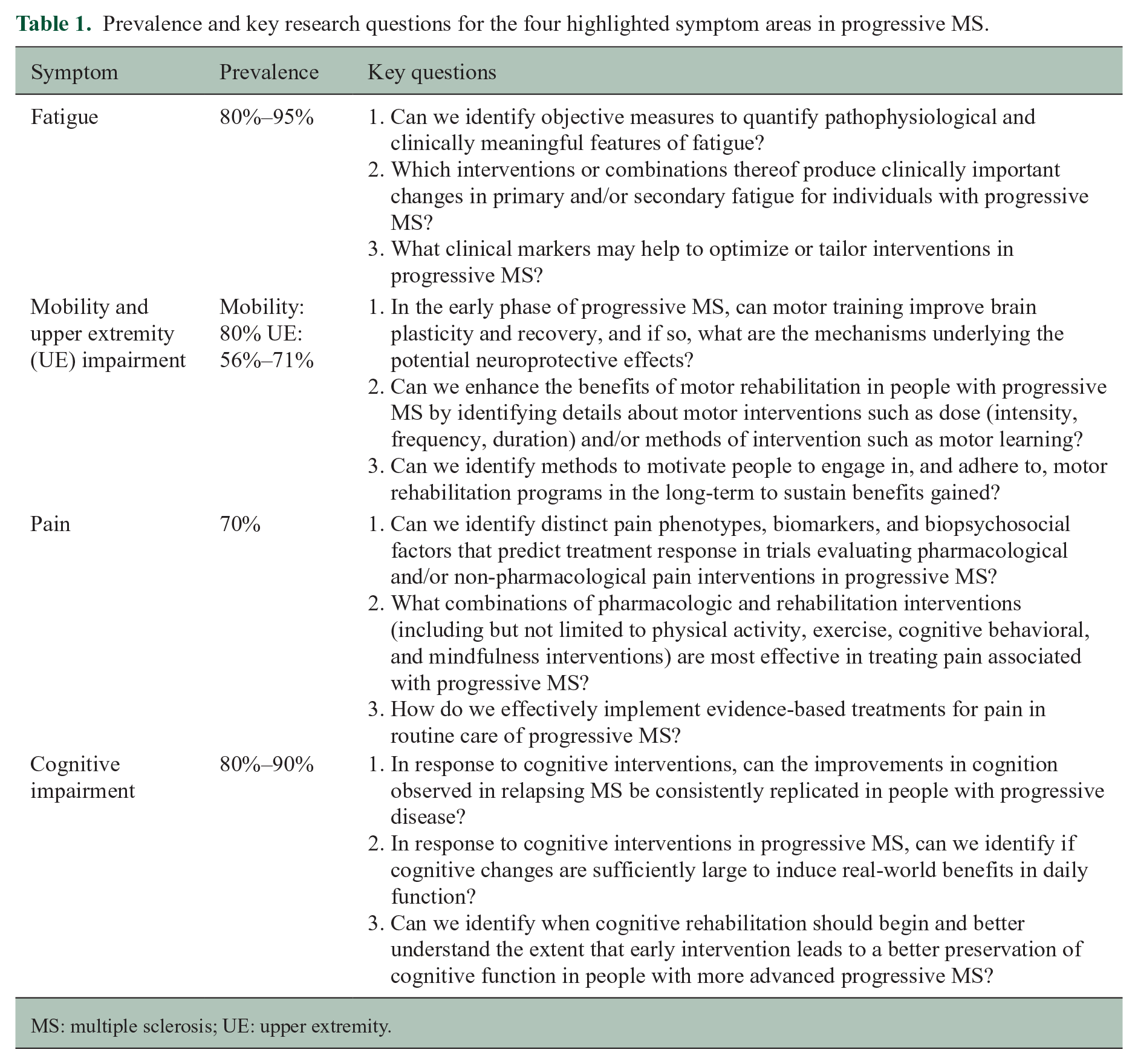
MS: multiple sclerosis; UE: upper extremity.
Important key knowledge gaps and research questions concerning four symptoms important to people with MS
Fatigue
Fatigue is one of the most common and debilitating symptoms of MS, affecting 80%–95% of people. 12 Whether persistent or sporadic, fatigue is associated with lower quality of life 13 and impaired work ability. 14 Although fatigue can be experienced throughout the disease course, levels are greatest in the progressive phase. 15 Results from four surveys that report symptoms of importance to people affected by MS show that fatigue ranks as a top symptom of importance.16–19 Yet, surprisingly few studies have investigated fatigue and its relationship with clinical features of progressive MS, highlighting an important knowledge gap.
The pathophysiological mechanisms underlying fatigue in MS are poorly understood, and have not been adequately investigated in progressive MS. MS fatigue has been proposed to be directly related to demyelination and axonal loss, 20 reduced cortical volumes and functional cortical reorganization, 21 asymmetric connectivity, 22 and immunological and neuroendocrine dysregulation. 23 Secondary mechanisms independent of MS pathophysiology such as depression/anxiety, disability, sleep disorder, or their treatments may also contribute to fatigue; 12 however, the literature generally lacks replication of any pathophysiological mechanism. Moreover, the effects of disease-modifying and symptomatic treatments on fatigue remain unclear and understudied.
In 2015, a panel of MS experts proposed including (self-reported) fatigue in the definition of disease activity, given its impact on quality of life. 24 However, the subjective and multifactorial nature of this symptom has posed challenges for the development of robust questionnaires which effectively measure fatigue; for example, some questionnaires measure fatigue impact while others measure fatigue severity (or combinations). Moreover, different manifestations of fatigue exist, including the perception of fatigue and performance fatigability; these distinctions and gaps in our understanding are important as they offer the opportunity for clarity and consistency in the study of fatigue and the search for effective treatments. 25
Clinical practice guidelines suggest using a combination of medication and rehabilitation interventions for managing MS-related fatigue. Pharmacological interventions are commonly used in practice, although their efficacy is poorly established in RRMS, and even less so in progressive MS.26,27 Rehabilitation interventions, such as exercise (aerobic, resistance, mixed and or other training) have shown the potential to improve fatigue, based on higher aerobic capacity 28 and reduced central muscle activation. 29 Furthermore, psychological/educational interventions (e.g. cognitive behavioral therapy) also reduce fatigue.26,30 Whether these benefits are sustained in the long-term (many months later) is not clear. Still, most intervention studies do not differentiate findings by MS type, thus the effectiveness of these interventions for progressive MS is uncertain, leaving a large gap in knowledge and practice. 31
Mobility and upper extremity impairment
Impaired mobility is a prominent concern for people with progressive MS, 32 with approximately 80% experiencing walking difficulties within 15 years of diagnosis, and 25% eventually becoming wheelchair dependent. 33 Surveys show that mobility impairment is a top symptom of importance to people with MS.16–19 Greater walking impairment, and more severe overall impairment, is experienced in the progressive compared with the relapsing remitting phase, with indications of greater impairments in primary compared with secondary progressive MS. 33 As mobility impairment increases, functional limitations can lead to safety concerns with increased likelihood of falls, and greater barriers to participation in rehabilitation. 34
Mobility studies investigating the biological mechanisms of impairment and recovery at molecular, cellular, and synaptic levels are encouraging; some focus exclusively on progressive MS. 35 These provide preliminary evidence of the potential impact of exercise on neuroprotection and regeneration in animal models 36 and humans.10,37 While these mechanistic studies vary in methodological quality, these inter-disciplinary collaborations provide a foundation for future work to inform the design of rehabilitation interventions. 38 We can learn valuable lessons from conditions such as stroke, where motor recovery trials have included the combination of pharmaceutical approaches with rehabilitation interventions and the incorporation of biological and behavioral recovery biomarkers (e.g. imaging measures of brain volume or white matter tracts) into rehabilitation trials. Although biomarkers have improved the understanding of how treatments may work and supported development of algorithms to inform clinical decision-making in MS,34,39 there is very little evidence validating biomarkers in progressive MS, highlighting an important gap in our knowledge.
Differing rehabilitation strategies are used to optimize mobility function, including restorative, compensatory, preventive, maintenance, and combined approaches. The evidence base supporting their benefit is growing but varies in methodological quality and is largely confined to mixed participant samples. 7 Steady progress is being made regarding exercise, with a substantial rise in randomized controlled trials (RCTs) over the past two decades. These have investigated mobility (albeit mostly as a secondary outcome and in mixed samples), with convincing evidence of benefit in RRMS. 40 Yet, even in this focused area, the quality and volume of research specific to progressive MS is low, 41 and limited to pilot, feasibility, or case series studies.42–45 Well-powered, robustly designed RCTs exclusively recruiting people with progressive MS targeting improvements in motor function as their primary outcome are scarce,46–48 and more studies are therefore needed to address this gap. Furthermore, although evidence-based physical activity guidelines exist for RRMS, the same cannot be said to guide people with progressive MS. 49
Having a similar prevalence to mobility impairment, upper extremity impairment is more common in progressive MS than RRMS, 50 and worsens over time.51,52 As mobility deteriorates, there is increased reliance on the upper extremities to manipulate ambulatory aides such canes and walkers, and to propel wheelchairs. People affected by progressive MS, including the authors, emphasize and highlight the importance of better understanding upper extremity impairment so treatments can improve. Upper extremity impairment remains under-recognized and under-studied relative to mobility impairment. 46 Few studies have addressed mechanisms of upper limb impairment or recovery. 53 Larger RCT studies rarely include upper extremity function with only a few underpowered trials in mixed samples. 46 Despite these gaps, we know that both mobility loss and upper extremity impairment are strongly correlated with reduced quality of life and are emotionally, financially, and socially costly. 54
Pain
Pain is a multidimensional experience involving intensity, interference (impact of pain on function), quality, temporality, affect (e.g. unpleasantness and emotional responses such as fear), and behavior. Chronic pain is one of the most prevalent, disabling, and persistent symptoms associated with MS. 55 People affected by MS indicate that pain, including numbness, tingling, and muscle spasms—is an important and particularly disabling symptom.16–19 A meta-analysis indicated that 7 in 10 adults with SPMS and PPMS experience pain. 56 In a prospective study, progressive MS was associated with a greater risk for disruptive pain relative to relapsing MS. 57 Chronic pain has been associated with poorer health, sleep disruption, fatigue, depression, physical inactivity, more falls, poorer cognitive functioning, increased healthcare utilization, social disruption, and vocational dysfunction in those with MS.58–61 Notably, these studies have primarily included participants with RRMS, leaving a large gap in our understanding of pain in progressive MS.
Pain syndromes are complex and may include trigeminal neuralgia, central neuropathic pain, painful tonic spasms, and optic neuritis-associated pain; 62 these chronic pain syndromes are often associated with brain and spinal cord lesions. 63 Mechanical musculoskeletal pain syndrome including low back pain is generally considered a secondary pain syndrome 63 due to muscle weakness, sensory changes, immobility, structural malalignment, or fall-related injuries. While the pathophysiology of chronic pain in MS involves a complex interplay of neural and non-neural mechanisms, multiple studies have confirmed the role of psychosocial factors (including distress, negative thoughts/beliefs about pain, insufficient coping skills, and activity avoidance) in pain intensity and pain-related disability in people with MS.64,65
The narrow therapeutic window of non-opioid pharmacologic treatments 58 requires that non-pharmacologic management including rehabilitation (e.g. cognitive behavioral, 58 mindfulness-based interventions,66–68 self-hypnosis training,69–71 imagery, 72 physical activity interventions, and stretching or exercise) 73 be incorporated into the overall analgesic strategy, 74 yet they often are underutilized for pain management in MS. 75 The need for strategies that are focused on identifying biomarkers (including neuromodulatory markers), somatosensory and psychosocial factors, and clinical outcomes assessments are recognized for a variety of painful syndromes globally (e.g. in low back pain), 76 and should be applied to pain mechanisms research in MS. In the United States, the Federal Pain Research Strategy, an effort of the Interagency Pain Research Coordinating Committee (IPRCC) and the Office of Pain Policy (National Institutes of Health), also includes the need for precision medicine research to prevent and treat pain syndromes. In addition, high-quality population-based studies of the prevalence and characteristics of acute and chronic pain in progressive MS are needed to fill the gap in knowledge. Research that examines the development of effective models of care delivery that integrate evidence-based pain interventions into routine care of the patient with progressive MS, and how to tailor these treatments for people with progressive MS is lacking.
Cognitive impairment
Cognitive impairment affects approximately 43% of people with MS overall, but up to 80% with SPMS and 90% with PPMS. 77 Processing speed, learning and memory, and executive function are most frequently affected. 78 Cognitive impairment adversely affects employment, relationships, leisure pursuits, and quality of life. 79 Given how common cognitive dysfunction is in MS and its negative consequences it is no surprise that it was identified as an important symptom to study by people affected by MS.16–19 What may be more surprising, however, is the limited amount of funding dedicated to the study of this common symptom in progressive MS (Figure 1), illustrating the dire need for funding to alleviate this gap in knowledge.
Cognitive rehabilitation may reverse some cognitive decline. 78 A double-blind, placebo-controlled RCT of 10 sessions of a behavioral intervention, the Story Memory Technique, revealed improved learning only in the treated group, which persisted for six months post-intervention. 80 A subsequent study using similar methods in people with progressive MS reported comparable results, with sustained improvement 3 months post-intervention. 81 Impairments in processing speed, considered the quintessential cognitive problem in people with MS (RRMS and progressive MS), may also respond to cognitive intervention. A pilot RCT of a computerized intervention improved processing speed elicited in the tester’s office and in a real-world setting. 82 Such studies pave the way for more work in progressive MS.
Group-based cognitive rehabilitation may benefit processing speed, working memory, and executive function in MS.83,84 Given the large number of people with progressive MS who are cognitively impaired, and the resources needed for cognitive rehabilitation, the benefits of successful group intervention take on added significance. Computer-administered cognitive rehabilitation may support more widespread treatment; increasing evidence indicates its effectiveness when administered generally 85 or in a personalized digital application. 86
Of particular interest is a nascent literature reporting the benefits of combined interventions such as cognitive rehabilitation and motor rehabilitation.87,88 This interdisciplinary approach builds on findings from a large literature showing the benefits of exercise for multiple symptoms, including cognitive impairment.89,90 This also opens the possibility that there are rehabilitation approaches that can improve multiple impairments.
Although the mechanisms driving treatment-induced cognitive improvement require further elucidation, functional MRI studies have shown a pattern of enhanced cerebral activation and/or connectivity that correlates with beneficial changes in memory 91 and processing speed. 10
The cognitive rehabilitation findings and complementary imaging data raise optimism that people with MS who are cognitively impaired can be helped. The positive findings come overwhelmingly from people with RRMS. Only two underpowered studies, both with positive outcomes but without imaging data, provided evidence among participants with progressive disease, leaving a gap in knowledge and practice that must be filled.81,89
Factors that may impact the four highlighted symptoms
There are several important factors which may influence symptomatic management and rehabilitation in progressive MS, and which, in themselves, warrant further research investment; these include depression, resilience, other comorbidities, and psychosocial support. All rehabilitation studies should take account of these factors.
A small, consistent, literature suggests that clinically significant depression adversely affects cognition, reducing memory and attentional capacity or slowing processing speed. 92 Untreated depression can also have a strong debilitating effect on a person’s ability and motivation to maintain their own health and participate in rehabilitation programs. Furthermore, depression also commonly co-occurs and has a bi-directional relationship with chronic pain. 93 Further investigation of depression in the context of progressive MS is needed.
Resilience mediates the relationships between common MS symptoms, including fatigue and emotional well-being, and thus quality of life. 94 Increases in resilience are associated with reduced symptoms of pain and fatigue. 95 Resilience is considered modifiable; 96 however, the extent to which resilience affects well-being and moderates symptom intervention outcomes in progressive MS is unknown.
A wide variety of comorbidities, complications, and secondary conditions including cerebrovascular disease, diabetes, urinary tract infections, anxiety, and depression are common in MS, and influence the prevalence, severity, progression and characteristics of cognitive and motor impairments, pain, and fatigue.97,98 The prevalence of comborbidity increases with age in RRMS and progressive MS, as does disability. Therefore, symptom management studies must consider the complexities and potential impact of comorbidities. 97
Psychosocial factors influence pain intensity and pain-related disability.61,64,65 In people with MS, access to social support improves quality of life and mitigates depression, anxiety, and stress. 99 The rapid changes in our environment due to the COVID-19 pandemic increase the relevance of these factors and may intensify barriers to receiving rehabilitation interventions. 100 The potential contribution of psychosocial factors on symptoms, feelings of self-efficacy, and adherence to interventions should not be underestimated.
Conclusions and future directions
Despite the progress in managing RRMS, effective symptom management and rehabilitation remain far behind in progressive MS. This reflects multiple factors. Little empirical rehabilitation data pertaining to progressive MS exists, and our understanding of mechanisms underlying symptoms and treatment responses is incomplete. Clinical rehabilitation trials continue to be designed with strategies used in pharmacotherapy trials, despite important differences related to control groups, blinding, outcome measures, and other factors. For example, in some intervention studies, we observed that eligible participants did not always have the symptom of interest measurable at a clinical level at baseline so in fact any changes due to the intervention could not be measured. Clinical trials often fail to include measures to establish the ecologic validity of their findings, or the long-term effects of rehabilitation interventions. Pragmatic trials designed to evaluate complex health interventions in real-world settings and using cost-effectiveness analyses when appropriate, as an alternative to classical clinical trials of efficacy are needed. Such trials need to be powered to detect clinically meaningful effects and incorporate patient-reported outcomes to support coverage by payers. Equally important is the use of transparent reporting guidelines, such as CONSORT and TiDieR, to ensure that studies can be validated and appropriately applied in clinical practice. Greater collaborative efforts that make use of data aggregation and data harmonization to increase sample sizes and allow examination of heterogeneity according to sociodemographic and clinical characteristics are also needed. Rehabilitation and quality-of-life research could be strengthened by partnering with other scientists such as geneticists, immunologists, and neuroimaging experts. Welcoming collaborative opportunities that include industry and the involvement of individuals affected by progressive MS in the design and implementation of studies is essential to the growth of understanding in this area.
Despite these challenges, progress has been made. For example, recommendations for exercise have shifted over two decades from being contraindicated to enthusiastic endorsement. We also know that, in some cases, successful symptom treatment can change the brain and the biology of MS. Central to all interventions is our understanding that benefits are more likely to accrue when the person is actively engaged with the intervention and when interventions are started early. Emerging technologies, including opportunities for brain-computer interfaces, telerehabilitation and remote monitoring in rehabilitation have great potential. Wearables and new data processing techniques to analyze data flows, such as artificial intelligence, can be leveraged to collect and monitor quantitative and continuous data in real-life circumstances, and potentially facilitate function. 101 Progress in symptom management for other chronic illnesses provide hope and guidance. Successful efforts such as those achieved in cardiac rehabilitation, which is now an accepted method to reduce heart failure symptoms and prevent worsening, provide a powerful example of how rehabilitation can produce improved quality of life and be supported by diverse payers and stakeholders.
The International Progressive MS Alliance, encompassing a convergence of people affected by progressive MS, researchers, clinicians, and industry leaders, is uniquely positioned to focus and lead the conversation and galvanize the research community, to elucidate mechanisms, define appropriate outcomes, and guide the implementation of rehabilitation treatment into clinical practice. We recommend here key research questions to advance symptom management in four important areas (Table 1) and propose a rationale for progressing this work scientifically as well as highlighting the importance of financial investment from stakeholders to fund research in these areas. This analysis is intended to facilitate the development of treatment interventions for these and other challenging and important symptoms affecting people with progressive MS.
Footnotes
Acknowledgements
The authors thank Dr. Lenka Ortner for her consistent help with editing this document and formatting references.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kathleen M. Zackowski, none. Jennifer Freeman, none. Giampaolo Brichetto, none. Diego Centonze is an Advisory Board member of Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck KGaA Serono, Novartis, Roche, Sanofi-Genzyme, and Teva, and received honoraria for speaking or consultation fees from Almirall, Bayer Schering, Biogen, GW Pharmaceuticals, Merck KGaA Serono, Novartis, Roche, Sanofi-Genzyme, and Teva. He is also the principal investigator in clinical trials for Bayer Schering, Biogen, Merck KGaA Serono, Mitsubishi, Novartis, Roche, Sanofi-Genzyme, and Teva. His preclinical and clinical research was supported by grants from Bayer Schering, Biogen Idec, Celgene, Merck KGaA Serono, Novartis, Roche, Sanofi-Genzyme, and Teva. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results. Ulrik Dalgas reports grants from Genzyme, grants from Canadian MS Society, grants from Trygfoundation, outside the submitted work. John DeLuca reports grants and personal fees from Biogen, personal fees from Celgene, personal fees from Novartis, personal fees from Sanofi-Genzyme, personal fees from Medrhythms, outside the submitted work. Dawn Ehde, none. Sara Elgott, none. Vanessa Fanning, none. Peter Feys is a Member of the editorial boards of the following journals: Multiple Sclerosis Journal and Neurorehabilitation and Neural Repair. Marcia Finlayson reports grants from MS Society of Canada, during the conduct of the study; other from Biogen, other from Sanofi-Genzyme, outside the submitted work. Stefan M. Gold reports personal fees from Almirall, personal fees from Celgene, personal fees from Mylan, grants from Biogen, grants from GAIA, outside the submitted work. Matilde Inglese reports grants from MS Society Canada, grants from NIH, grants from NMSS, grants from TEVA Neuroscience, grants from FISM, grants from Horizon_2020 EU, outside the submitted work. Ruth Ann Marrie, none. Michelle Ploughman, none. Christine N. Sang reports consulting activities with Alkermes, Arena, Evidation, Genentech, Heron, Lilly, Nevakar; grant from and consulting activities with Merck; grants from Abbvie, Vivozon, and Helixmith; and an issued patent, “Treatments of central neuropathic pain,” which has not yet been licensed. Jaume Sastre-Garriga reports grants from Fondo de Investigaciones Científicas (FIS), other from Multiple Sclerosis Journal, other from Revista de Neurologia, personal fees from Sanofi-Genzyme, personal fees from Merck, personal fees from Celgene, personal fees from Novartis, personal fees from BIAL, personal fees from BOPASS, personal fees from ORCHID, personal fees from BIOGEN, outside the submitted work. Jonathan Strum reports grants, personal fees, and non-financial support from EMD Serono, grants from Novartis, grants from Sanofi-Genzyme, personal fees from Celgene, personal fees from Janssen Pharmaceuticals, outside the submitted work. Johan van Beek is an employee and shareholder of F Hoffmann–La Roche Ltd, Biogen, Sanofi-Genzyme, Novartis, Roche, outside the submitted work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
