Abstract
Background:
Delayed-release dimethyl fumarate (DMF) demonstrates sustained efficacy and safety for relapsing forms of MS. Absolute lymphocyte count (ALC) is reduced initially, then stabilizes on treatment.
Objective:
PROCLAIM, a 96-week, prospective, open-label, phase 3b study, assessed lymphocyte subsets and immunoglobulin (Ig) levels during 48 and 96 weeks (W) of DMF treatment.
Methods:
Patients received 240 mg DMF BID. Endpoints: lymphocyte subset count changes (primary); Ig isotypes and ALC changes (secondary); adverse events and relationship between ALC changes and ARR/EDSS (exploratory); and neurofilament assessment (ad hoc).
Results:
Of 218 patients enrolled, 158 (72%) completed the study. Median ALC decreased 39% from baseline to W96 (BL–W96), stabilizing above the lower limit of normal (baseline: 1.82 × 109/L; W48: 1.06 × 109/L; W96: 1.05 × 109/L). CD4 + and CD8 + T cells correlated highly with ALC from BL–W96 (p < 0.001). Relative to total T cells, naive CD4 + and CD8 + T cells increased, whereas CD4 + and CD8 + central and effector memory T cells decreased. Total IgA, IgG, IgM, and IgG1–4 subclass levels remained stable. Adverse event rates were similar across ALC subgroups. ARR, EDSS, and neurofilament were not correlated with ALCs.
Conclusion:
Lymphocyte decreases with DMF were maintained over treatment, yet immunoglobulins remained stable. No increase in infection incidence was observed in patients with or without lymphopenia.
Support:
Biogen
Introduction
Delayed-release dimethyl fumarate (DMF), an approved oral therapy for relapsing forms of MS (RMS), has demonstrated sustained efficacy and a favorable benefit-risk profile.1–3 As of January 31, 2020, more than 445,000 patients have been treated with DMF worldwide, representing more than 875,000 patient-years of exposure. Of these, 6335 patients (14,241 patient-years) were from clinical trials. (Biogen, data on file). DMF treatment induces absolute lymphocyte count (ALC) reductions, typically in the first year of treatment, followed by stabilization. 4 Mean ALC reduction is ~30% from baseline and most patients remain above the lower limit of normal (LLN) (0.91 × 109/L). 5 Grade 3/severe lymphopenia (ALC < 0.5 × 109/L) persisting for ⩾ 6 months develops in ~2% of patients. 5 Absolute T-cell counts are more strongly impacted by DMF than B or natural killer (NK) cells, although most cell types are reduced to some degree. Within the T-cell compartment, CD8 + cells are reduced more profoundly than CD4 + cells.6–12 Despite ALC changes, DMF-treated patients mount an effective immune response to vaccination, including both T-cell–dependent and T-cell–independent antigens, comparable to interferon (IFN)-treated patients. 13 Although ALC does not appear to directly correlate with clinical response, functional shifts in circulating lymphocyte subsets favoring naive and anti-inflammatory phenotypes likely contribute to the effect of DMF.1,2,14,15
Previous immunophenotyping studies have been either cross-sectional6,9 or longitudinal, with few patients 10 or minimal data-collection time points. 16 PROCLAIM, the first large longitudinal study designed to assess the effects of DMF on lymphocyte subset counts and immunoglobulin (Ig) isotypes, may provide greater insight into DMF’s therapeutic benefit and inform the safety profile.
Methods
Study design
PROCLAIM (EUDRA CT 2015-001973-42) was a prospective, open-label, multicenter, phase 3b study assessing changes in immune cell compartments through 2 years of DMF treatment. The study period consisted of a screening visit, a 96-week treatment period, and a final follow-up visit 4 weeks after the last dose of DMF. All patients received 120 mg twice daily (BID) for the first 7 days, followed by a maintenance dose of 240 mg BID thereafter (approved dosing regimen for MS). Temporary dose reduction to 120 mg BID through 4 weeks was permitted for individuals who did not tolerate the maintenance dose due to flushing or gastrointestinal disturbance. Treatment compliance was monitored by study personnel via capsule counting at visits. Blood samples were collected at baseline and Weeks 4, 8, 12, 24, 36, 48, 72, and 96.
Patients
Eligible patients were aged 18–65 years with a confirmed diagnosis of RMS. 17 Key exclusion criteria were positive serology for HIV or hepatitis B/C; history of drug or alcohol abuse within 1 year before screening; clinically significant comorbid disorders or conditions, including infectious illness; leukocytes < 3.5 × 109/L; ALC values ⩽ LLN (0.91 × 109/L); or prior treatment with cladribine, mitoxantrone, total lymphoid irradiation, alemtuzumab, T-cell or T cell–receptor vaccination, or any therapeutic monoclonal antibody (except natalizumab or daclizumab). Concomitant treatment with alternative MS drugs or systemic steroid therapy was not allowed unless approved by the medical monitor for treatment of a protocol-defined relapse.
Study objectives and endpoints
The study objectives were to evaluate the effect of DMF on lymphocyte subset counts (primary) and the pharmacodynamic effect of DMF on ALCs and Ig isotypes (secondary) in patients with RMS during the first 48 weeks of treatment. Exploratory study objectives included evaluation of safety and tolerability over 96 weeks; lymphocyte subset counts through 96 weeks of DMF treatment; the relationship between changes in ALC and lymphocyte subsets and MS disease activity (measured by clinical relapse; annualized relapse rate (ARR)) or sustained clinical disease progression (CDP) (measured by the Expanded Disability Status Scale (EDSS)); and ad hoc evaluations of hypothesis-driven biomarker analyses, including neurofilament light chain(NfL).
ALC, immune cell phenotyping, and Igs
ALC was measured using complete blood cell differential. Changes in lymphocyte subsets were assessed by flow cytometry utilizing cell surface markers (Supplementary Table 1). Serum Ig levels (total IgA, IgG, IgM, and subclasses IgG1, IgG2, IgG3, and IgG4) were measured using an immunoturbidimetric method on the Integra/Plus (Roche Diagnostics, Basel, Switzerland) or by immunologic methods (LOINC®, LabCorp, Burlington, North Carolina).
Clinical assessments
Safety outcomes were monitored throughout the study and safety assessments were performed at the post-treatment follow-up visit. Relapses were defined as new or recurrent neurologic symptoms not associated with fever or infection, lasting ⩾ 24 hours, accompanied by objective neurological findings and confirmed by a neurologist. Sustained CDP was defined as ⩾ 1.0-point increase on the EDSS from a baseline score of ⩾ 1.0, sustained for 24 weeks, or ⩾ 1.5-point increase on the EDSS from a baseline EDSS score of 0, sustained for 24 weeks.
ALCs were categorized as follows: always > LLN, 0.91 × 109/L; mild lymphopenia, < 0.91 × 109/L anytime, excluding patients with ALC < 0.8 × 109/L for ⩾ 6 months; moderate prolonged lymphopenia, ⩾0.5 × 109/L to <0.8 × 109/L for ⩾ 6 months; and severe prolonged lymphopenia, <0.5 × 109/L for ⩾ 6 months.
ALCs were stratified by age at baseline; younger patients (<50 years) versus older patients (⩾50 years). This cutoff was selected in order to explore the immune function in patients who may be prone to decreased immune function and immunosenescence based on age, while maintaining a sufficient sample size.18,19
Ad hoc analysis
Serum NfL (sNfL) levels were measured using single molecule array (Simoa™ NF-light® Advantage assay, Quanterix, Billerica, MA). Two patients with extremely high (> 10 standard deviations (SDs)) baseline sNfL values were excluded.
Statistical analyses
All patients who received ⩾ 1 dose of study treatment were included in safety and relapse assessments. All patients who received ⩾ 1 dose of study treatment and had ⩾ 1 post-baseline pharmacodynamic measurement were included in the immune cell phenotyping analysis; EDSS was assessed in those with ⩾ 1 post-baseline EDSS measurement. For the primary endpoint, the actual value, change, and percent change from baseline (CFB) were descriptively summarized. The Wilcoxon signed-rank test was used to assess if changes were different from zero. For secondary analyses, changes in levels of Ig isotypes and ALCs were descriptively summarized. Mixed-effect model repeated measurement assessed CFB in the Ig isotypes, and ALC CFB to Week 96 as the dependent variable. The model included visit, corresponding baseline value, age, and sex as fixed effects, and an unstructured variance-covariance matrix structure. Least squares mean, standard error, and 95% confidence intervals were reported for each visit.
ARR was estimated from a negative binomial regression model, adjusted for baseline covariates. No imputation was used for missing data; the missing values were skipped. The observed data were used for all analyses. EDSS metrics and the proportion of patients with CDP were descriptively summarized. All adverse events (AEs) were included.
Results
Study population
A total of 218 patients were enrolled from six countries (Table 1 and Supplementary Figure 1); 158 (72%) patients completed the study and 60 (28%) withdrew, primarily due to withdrawn consent (n = 22) and AEs (n = 17). Five patients withdrew due to lymphopenia: one with severe lymphopenia and four with moderate. The mean (SD) age in PROCLAIM was 42 (11) years and 69% of patients were female (Table 1). Patients were older in PROCLAIM than in the pivotal phase 3 clinical trials DEFINE/CONFIRM (NCT00420212/NCT00451451): 27% were aged ⩾ 50 years in PROCLAIM (Table 1) compared with 13% in the DMF 240 mg group in DEFINE/CONFIRM.1,2 Other demographic variables were consistent with the phase 3 studies.1,2
Patient baseline demographics and disease characteristics.

ALC: absolute lymphocyte count; DMT: disease-modifying therapy; EDSS: Expanded Disability Status Scale; NK: natural killer; SD: standard deviation.
Other DMTs used by ⩾ 3 patients: fingolimod (14), investigational drug (14), methylprednisolone (11), blinded therapy (7), dimethyl fumarate (5), interferon beta (5), fampridine (4), peginterferon beta-1a (3), teriflunomide (3).
DMF treatment reduced lymphocyte subset counts together with temporal changes in ALC
ALC decreases were evident as early as 8 weeks after DMF treatment initiation, continuing to decline for 6–12 months, then stabilizing; median ALC CFB was −41% by Week 48 and −39% by Week 96 (baseline, 1.82 × 109/L; Week 48, 1.06 × 109/L; and Week 96, 1.05 × 109/L) (Figure 1(a)). When stratified by younger patients (<50 years) versus older patients (⩾50 years) at baseline, median ALC CFB was −38% and −51% by Week 96, respectively. After 2 years of DMF treatment, 110/218 (50%) patients had never developed lymphopenia (all ALCs remained > LLN), 69 (32%) had mild lymphopenia, 32 (15%) had moderate prolonged lymphopenia, and 1 (< 1%) had severe prolonged lymphopenia.
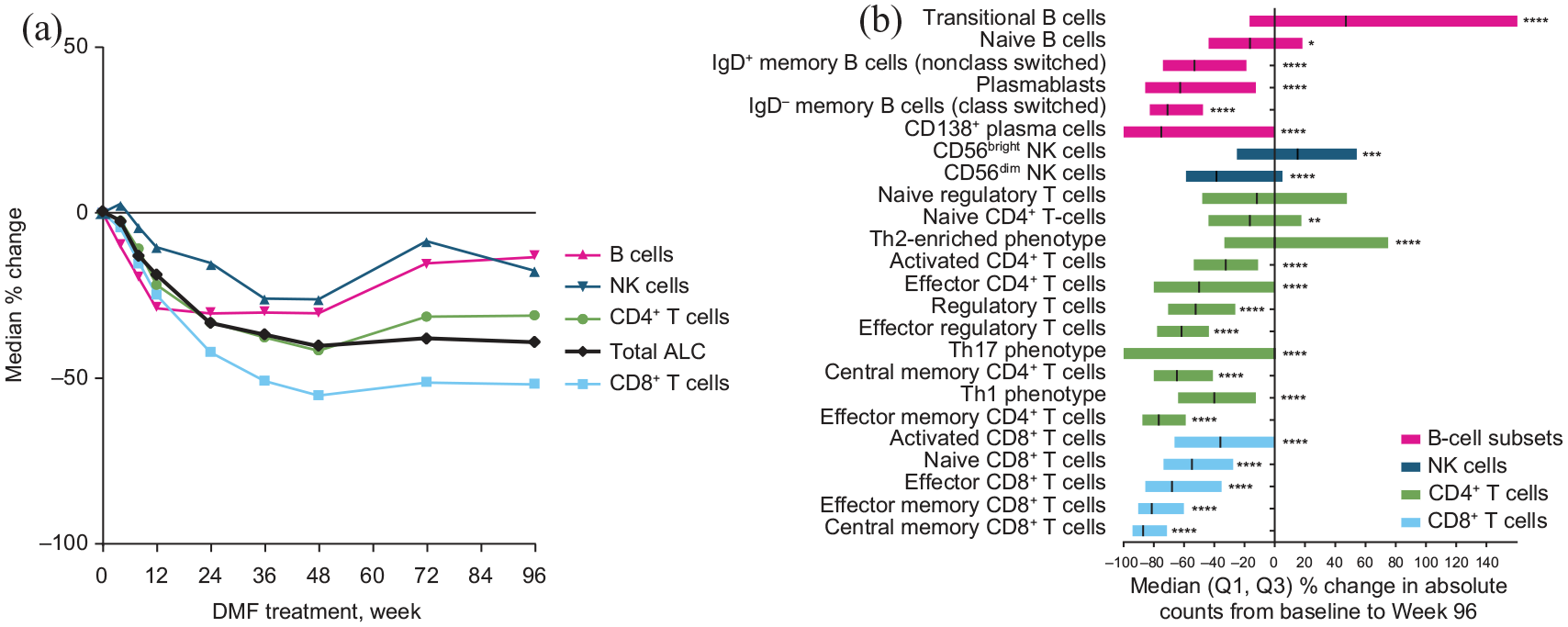
Median percentage change in ALC and major lymphocyte subsets with DMF treatment over time.
The degree of change in lymphocyte subset counts correlated with changes in ALC; CD4 + and CD8 + T, CD19 + B, and NK cell median CFB to Week 48 were −42%, −55%, −30%, and −26% (all p < 0.0001), respectively (Figure 1(b)). The changes were sustained through Week 96 (Figure 1(a)). Using a sequential, cross-sectional analysis of correlations over 2 years, changes in T-cell, B-cell, and NK cell subsets generally correlated with changes in ALC; the strongest correlations were observed in T cells (Pearson’s correlation coefficient range, 0.798–0.911; p < 0.001), though all correlations reached statistical significance. Among T cells, both CD4 + and CD8 + T cells correlated highly with ALC from baseline through Week 96 (Pearson’s correlation coefficient range, 0.559–0.880; p < 0.001) (Figure 2(a)). The correlations remained strong when stratified by age < 50 and ⩾ 50 years at baseline (Figures 2(b) and (c)).
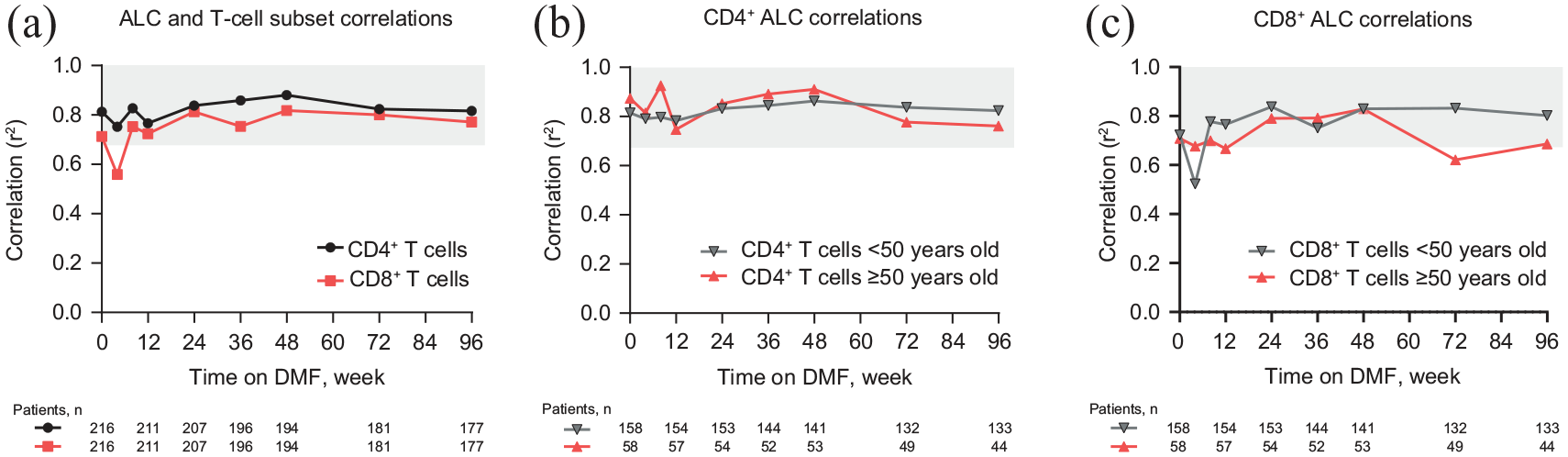
CD4 + and CD8 + T cells are highly correlated with ALC at all time points, regardless of age.
DMF drives a general shift toward a naive/anti-inflammatory repertoire
Decreased total CD19 + B-cell counts were observed at Week 4 after treatment initiation (−10% CFB; p < 0.001), whereas reductions were observed for ALCs and CD4 + and CD8 + T-cell subsets by Week 8. Total B cells reached nadir at Week 24 (−31% CFB; p < 0.0001). After Week 48, a gradual increase in total B cells was observed, though levels did not return to baseline, remaining decreased through Week 96 (−13% CFB; p < 0.05). Total CD14 + monocytes appeared stable during DMF treatment, with modest reductions during Year 2 (median CFB at Week 96: −11.6%, p = 0.005). Circulating classical monocytes (CD14 + CD16-) remained generally stable during the treatment period (−0.88%, p = 0.948 median CFB to Week 96) while the nonclassical (CD14DIMCD16 +) monocyte numbers declined beginning at Week 12 (median CFB: −25.5%, p < 0.0001) and remained at a lower level for the duration of the treatment period. As measured by flow cytometry, granulocytes slightly increased at Week 96 (median CFB: 13% (p < 0.001). When assessed by hematology, neutrophil shifts of potential clinical significance were infrequently reported (< 1.5 cells × 109/L: 11/218 patients; ⩽ 1.0 cell × 109/L: 0 patients; ⩾ 12.0 cells × 109/L: 10/218 patients).
At Week 96, the memory compartment was selectively reduced compared with naive cells for CD4 + and CD8 + T cells and B cells. CFB: CD4 + CD45RA−, −70% (p < 0.0001), versus naive CD4 + CD45RA + CCR7 + T cells, −16% (p < 0.005); CD8 + CD45RA−, −85% (p < 0.0001), versus naive CD8 + CD45RA + CCR7 + T cells, −55% (p < 0.0001); and CD27 + IgD + B cells (non-class switched) −53% (p < 0.0001), and CD27 + IgD–/B cells (class switched), −71% (p < 0.0001), versus naive CD27−IgD+ B cells, −16% (p < 0.05) (Figure 1(b)). Only immunoregulatory CD56bright NK and transitional CD24hiCD38hi B-cell numbers increased over 96 weeks:
Compartmental relative shifts within lymphocyte subsets
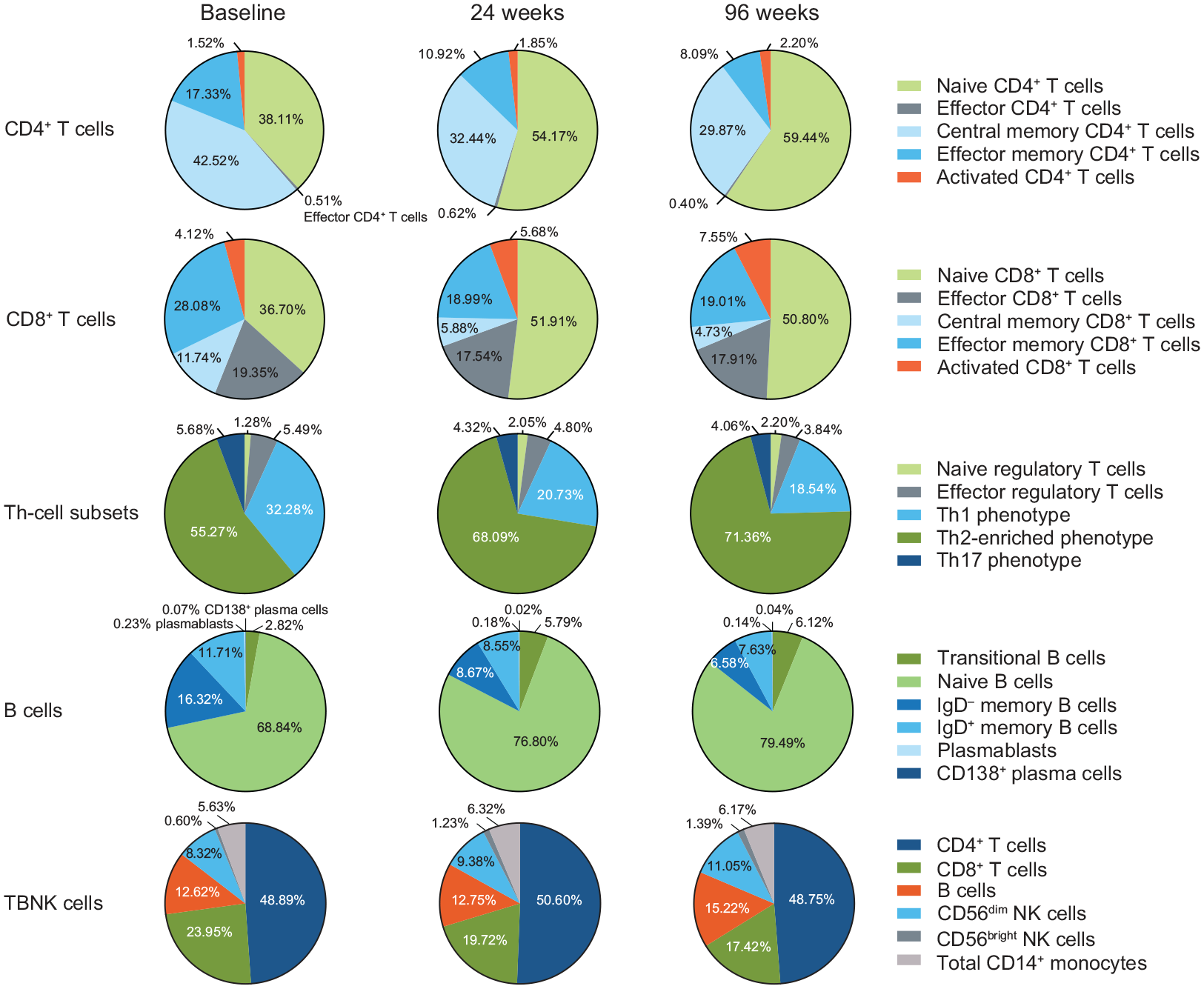
When relative changes were assessed within the total T-cell compartment, naive CD4 + and CD8 + T-cell populations demonstrated a relative increase, whereas CD4 + and CD8 + central memory and effector memory populations decreased (Figure 3). Within T helper (Th) cell subsets, there was a relative decline in pro-inflammatory Th1 and Th17 cells, a relative increase in anti-inflammatory Th2 cells, and stable total regulatory T cells. The relative proportion of regulatory T cells remained stable compared with effector T cells. The relative proportion of the naive and transitional B-cell populations increased, compared with a decrease in the proportion of memory B cells (Figure 3). There was a relative increase in the immunoregulatory CD56bright NK cell population.
Median percentage change in T-cell subsets, B-cell subsets, NK cells, and monocytes during delayed-release dimethyl fumarate treatment.
Igs remain stable during DMF treatment
At baseline, mean (SD) total IgM, IgA, and IgG levels were 133 (71) mg/dL, 212 (91) mg/dL, and 1051 (233) mg/dL, respectively. Total IgM, IgA, IgG, and IgG1-4 subclass levels remained stable over 2 years of DMF treatment (Figure 4(a)). Ig levels remained stable during the study whether patients experienced relapses (n = 41) or not (n = 170) while on DMF treatment (Figure 4(b)). Baseline serum IgM, IgA, or IgG levels were below the LLN in 7% (16/218) of patients. Most of these patients had been previously treated with ⩾ 1 DMTs, although three had no previous DMT exposure. Seven of the 16 patients with low baseline Ig levels reached a normal value during DMF treatment. In general, when patients were stratified by ALC category, baseline Ig concentrations and median CFB to Weeks 48 and 96 were similar across categories. Data were available for only one patient with severe prolonged lymphopenia and therefore this category was not included in the analysis.
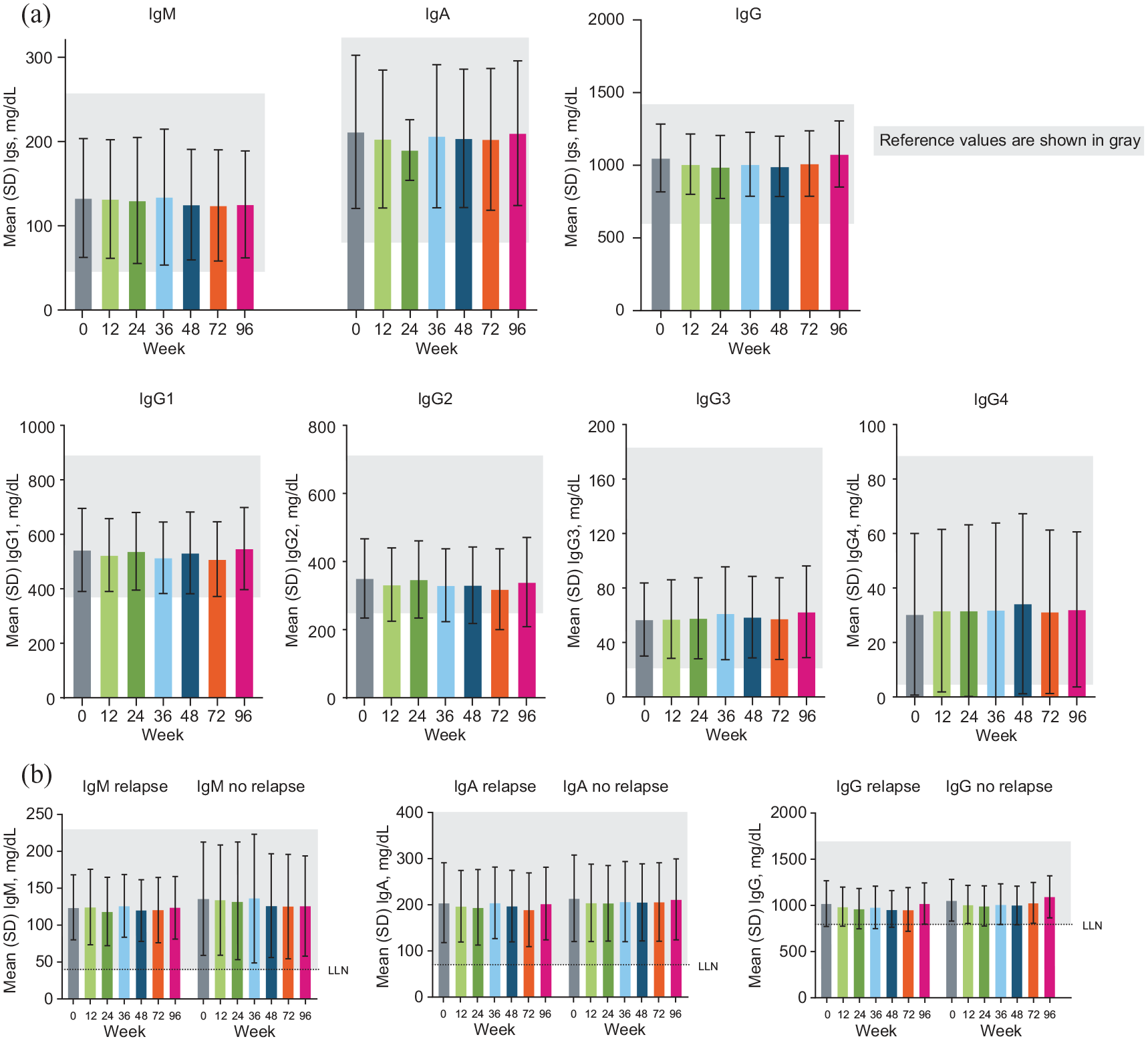
Mean Ig levels (a) over time and (b) in patients with or without relapse during delayed-release dimethyl fumarate treatment.
Safety outcomes
AEs and serious AEs were reported in 185/218 (85%) and 26/218 (12%) patients, respectively (Table 2), consistent with the known safety profile of DMF;1,2 flushing was the most common (46%). Treatment-emergent infections were reported in 89/218 (41%) patients. When stratified by ALC subgroup, a similar percentage of infections were reported across subgroups: 0.5 ⩽ ALC < 0.8 × 109/L (14/32 (44%) patients); 0.8 × 109/L ⩽ ALC < LLN (29/69 (42%) and ALC ⩾ LLN (43/110 (39%)). No infections were reported in the patient with severe, prolonged lymphopenia.
Adverse events.

Treatment-emergent events by preferred term.
Serious treatment-emergent infections (nasopharyngitis and cellulitis) were reported in two patients, neither considered related to study treatment. Nasopharyngitis occurred in a patient with mild lymphopenia (ALC 0.8 × 109/L to < LLN) and cellulitis occurred in a patient without lymphopenia (ALC ⩾ LLN); IgG concentrations were > LLN (700 mg/dL) for both patients. A malignancy, stage 1 breast cancer, was reported in one patient, considered unrelated to study treatment. No deaths or treatment-emergent opportunistic infections were reported.
ALC is not a marker of treatment response
The overall unadjusted ARR was 0.153. When patients were stratified into quartiles by percentage CFB in ALC over 96 weeks, ARR for each quartile was generally consistent, relapse rates were low across all groups, and there were no patterns associating ALC change with relapse rate (Figure 5(a)). Similarly, changes in EDSS score and the proportion of relapse-free patients were not associated with ALC quartiles (Figure 5(b) and (c)). The low rate of disability progression observed in this study (13 patients with CDP events) prevented assessment of CDP by ALC quartile.
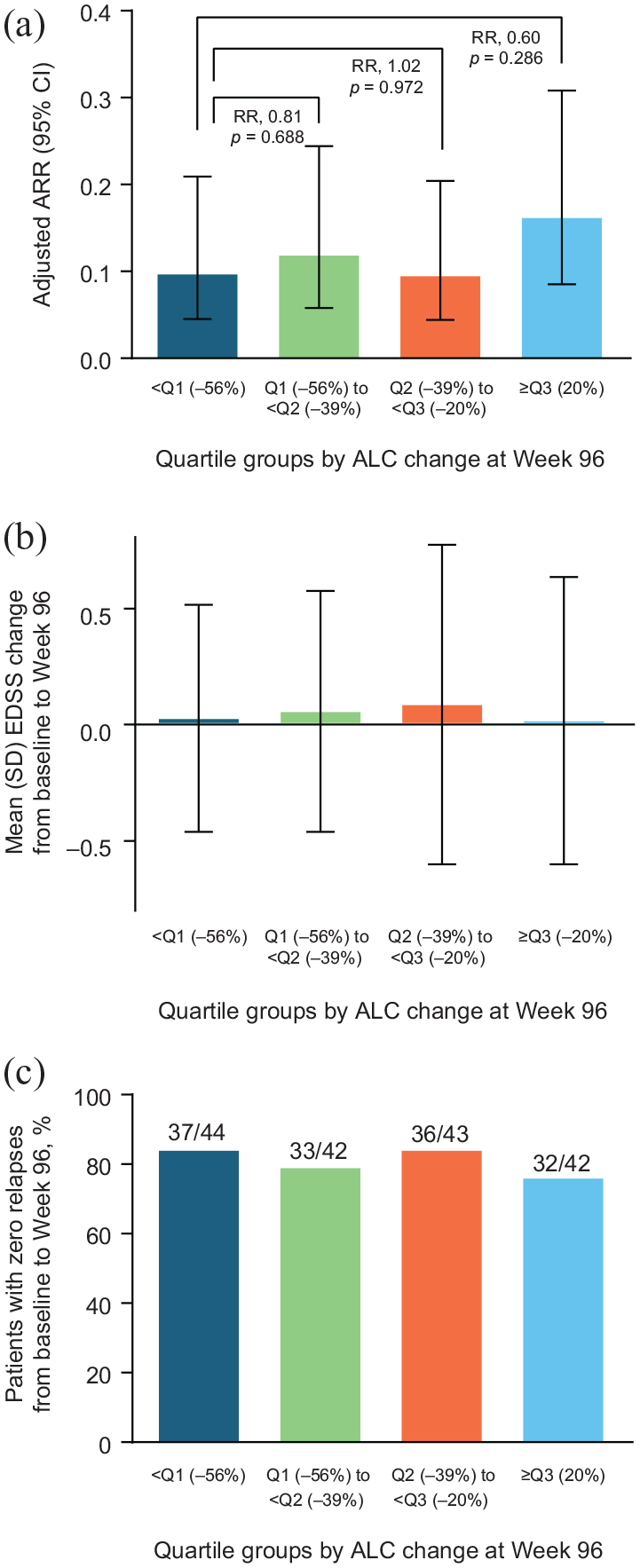
(a) Adjusted ARR, (b) change in EDSS score, and (c) patients with zero relapses by ALC change, from baseline to Week 96. (a) Both protocol-defined and non-protocol–defined relapses are included in the analysis. Based on negative binomial regression, adjusted for baseline EDSS score (⩽ 2.0 vs > 2.0) and baseline age (< 40 vs ⩾ 40 years). If the negative binomial regression model did not converge, a Poisson regression model with the same covariates was used. (b) Includes patients in the clinical assessment population with a baseline EDSS score (n = 183). (c) Percentage of patients with 0 relapses from baseline to Week 96.
Overall mean (SD) percentage change in sNfL from baseline to Week 96 was −19% (34). When stratified by age < 50 and ⩾ 50 years at baseline, sNfL mean percentage CFB to Week 96 were −22% (34) and −8% (33), respectively. Mean percentage change in sNfL did not vary significantly based on on-treatment ALCs (always ⩾ LLN, −17%; moderate prolonged lymphopenia, −21%; all other lymphopenia, −17%). Similarly, sNfL mean percent CFB to Week 96 were similar regardless of CD4 + and CD8 + levels: CD4 + < 200 cells/mm3 (n = 24), −15%, versus CD4 + ⩾ 200 cells/mm3 (n = 141), −19%, and CD8 + < 100 cells/mm3 (n = 59), −20%, versus CD8 + ⩾ 200 cells/mm3 (n = 106), −18%.
Discussion
The PROCLAIM study results demonstrate that DMF treatment of up to 2 years produced temporal changes in ALC and lymphocyte subsets, consistent with the known effect of DMF treatment. A median 39% ALC reduction from baseline to Week 96 and stabilization ⩾ LLN for most patients were observed; this decline is slightly higher than that observed in previous phase 3 and extension studies1,2,5 but is similar to an observational study of patients who were also older at baseline. 20 The number of circulating lymphocytes within most major subsets, including CD4 + and CD8 + T, CD19 + B, and CD56 + NK cells, declined following DMF initiation, consistent with overall ALC. Overall, monocytes were generally stable, which is expected given that classical monocytes, which comprise the majority of the compartment, were not changed with treatment. Interestingly, the nonclassical monocytes, which have been implicated in various inflammatory autoimmune disorders,21,22 declined and remained low. While likely not a primary driver of DMF efficacy, this change may be another reflection of the mechanism by which DMF shifts circulating cells from an inflammatory to an anti-inflammatory repertoire. Granulocytes, specifically neutrophils, were generally not decreased. These findings are consistent with clinical observations of DMF-treated patients in that the rate of serious and opportunistic infection is generally low. The relative proportion of regulatory T cells remained stable compared with effector T cells, suggesting immune tolerance mechanisms were maintained. Increased immunoregulatory CD56bright NK cells with DMF treatment confirm previous observations.23,24 Overall, DMF treatment induced a general shift toward a circulating naive/anti-inflammatory cell profile and away from memory/pro-inflammatory phenotypes, in both the T-cell and B-cell compartments. Similar reductions in memory B cells have been previously observed.9,14 including concomitant reductions in the pro-inflammatory cytokines granulocyte macrophage colony-stimulating factor (GM-CSF), tumor necrosis factor (TNF)-α, and IL-6.15,25 This shift was established within the first 3–6 months of treatment and maintained over 2 years.
Total IgA, IgG, IgM, and subclass IgG1–4 levels remained stable over 96 weeks of DMF treatment, regardless of ALCs, and were similar to Ig levels in healthy adult and MS populations.26,27 This is consistent with prior studies demonstrating that DMF-treated patients mount an effective T cell–independent and T cell–dependent immune response to recall and neoantigens comparable with that of IFN-treated patients. 13 Previous IFN studies have shown no decrease in serological response to vaccination when vaccines were co-administered during treatment with IFN;28,29 therefore, the similar rise in immunoglobulin levels in patients who were vaccinated while treated with IFN or DMF suggests similar seroprotection potential for the two agents. In PROCLAIM, which included patients slightly older at baseline than pivotal trials1,2 and those previously treated with DMTs, there was no change in serum IgG levels, in contrast to the decrease noted during treatment with other DMTs.27,30
These data do not support changes in ALC as a biomarker of treatment efficacy, consistent with previous reports. 5 Although T-cell numbers, specifically CD8 + T cells, are impacted by DMF treatment,6–12 the magnitude of the reduction does not correlate with relapse rate or changes in EDSS score. This prospective study and prior observational studies agree that relapse status, unadjusted ARRs, and change in EDSS scores were similar across ALC subgroups. Ig isotypes were also not differentially affected by relapse status or disability progression. The exploratory marker sNfL was decreased by DMF treatment, similar to other studies, 34 but the magnitude of reduction was not associated with ALC. Only younger age was associated with higher baseline ALC.
Despite ALC changes not being indicative of the magnitude of treatment response, it is purported that changes in the peripheral lymphocyte compartment toward an anti-inflammatory phenotype may contribute to reducing disease activity in DMF-treated patients with RMS.4,11,14,15,35 In addition, a decrease in interleukin-17–producing CD8 + T cells in DMF-treated patients, 36 together with the reduced IFN-gamma production by CD4 + T cells,37,38 dendritic cells, and monocytes,37,39,40 and greater reduction of CD8 + T cells,6–11 suggests DMF impacts MS through multiple mechanisms.
The PROCLAIM dataset provides additional context for the safety implications of T-cell subset changes in DMF-treated patients. No deaths or treatment-emergent opportunistic infections were reported over the 2-year study period. Rates of AEs were similar across all ALCs, consistent with the known safety profile of DMF; rates of serious AEs were low. Taken together with the strong correlation between ALC and T-cell subsets, these data continue to support that additional monitoring of T-cell subsets is not required for safety surveillance in routine clinical practice for DMF-treated patients. 11 However, the correlation coefficient is not 1.0; therefore, a small proportion of patients will have lower than expected T-cell subset counts despite normal ALCs.
To our knowledge, PROCLAIM is the first large prospective longitudinal study evaluating immunophenotypic changes among patients treated with DMF. Exclusion criteria for this study were limited, allowing enrollment of patients of a wider age spectrum with prior exposure to DMT. The study population therefore more closely reflects clinical practice, and the data reported here correspond well with the observational data previously reported. The discontinuation rate (28%) in PROCLAIM was higher than the assumption of 10% but in line with the pivotal clinical trials (31% and 30%).
This study was not powered to assess changes that occur in only a small percentage of patients, for example., 2%–3% of patients with severe prolonged lymphopenia; this population has been assessed in other studies.11,41 Moreover, this open-label prospective study did not include a comparator group, limiting the interpretation of some results. Disease activity at baseline was not controlled for in this study and only 13 patients experienced disability progression, limiting conclusions for changes in ALC in relation to disease activity. The clinical significance of this study is also limited by the lack of MRI data.
Conclusion
DMF modulates a shift in circulating lymphocytes away from memory cells and toward a naive repertoire that does not impair protective humoral immunity. This may contribute to the therapeutic benefits of DMF in MS. Overall, the relative proportion of naive and anti-inflammatory–type (Th2-enriched) cells increase, whereas central and effector memory and pro-inflammatory–type (Th17) cells decrease over 2 years of DMF treatment. These shifts do not affect Ig isotype concentrations. The magnitude of DMF-mediated changes in ALC were not associated with greater efficacy or increases in serious infections, as similarly noted in patients treated with DMF over many years.4,35 Additional T-cell subset monitoring is not required for safety surveillance in routine clinical practice based on the longitudinal correlation between T cells and ALC, regardless of T-cell subset type or age. Overall, the lymphocyte subset changes and Ig stability observed, together with the safety profile, indicate that DMF is generally well tolerated as evidenced by 72% of patients remaining on the study at 2 years. Protective humoral immune function is maintained over 96 weeks of treatment.
Supplemental Material
MSJ937282_supplemental_material – Supplemental material for Dimethyl fumarate treatment shifts the immune environment toward an anti-inflammatory cell profile while maintaining protective humoral immunity
Supplemental material, MSJ937282_supplemental_material for Dimethyl fumarate treatment shifts the immune environment toward an anti-inflammatory cell profile while maintaining protective humoral immunity by Erin E Longbrake, Yang Mao-Draayer, Mark Cascione, Tomasz Zielinski, Eris Bame, David Brassat, Chongshu Chen, Shivani Kapadia, Jason P Mendoza, Catherine Miller, Becky Parks, Diana Xing and Derrick Robertson in Multiple Sclerosis Journal
Footnotes
Acknowledgements
Biogen (Cambridge, MA) provided funding for medical writing support in the development of this paper; Miranda Dixon from Excel Medical Affairs (Horsham, UK) copyedited and styled the manuscript per journal requirements. Biogen reviewed and provided feedback on the paper. The authors had full editorial control of the paper and provided their final approval of all content.
Data Sharing Statement
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.E.L. has received consulting fees from Alexion, Biogen, Celgene, EMD Serono, Genentech, Genzyme, and Teva; research support from the Nancy Davis Center Without Walls and National Institutes of Health K23NS107624. Y.M-D. has received consulting/speaker fees from Bayer, Biogen, Celgene, Chugai, EMD Serono, Genentech, Novartis, Sanofi-Genzyme, and Teva; research support from Biogen, Chugai, National Institute of Neurological Disorders and Stroke R01-NS080821, National Institute of Allergy and Infectious Diseases Autoimmune Center of Excellence UM1-AI110557, Novartis, and Sanofi-Genzyme. M.C. has received consulting fees from Biogen, EMD Serono, Genentech, Novartis, and Sanofi-Genzyme; speaker fees from Acorda, Biogen, EMD Serono, Novartis, and Sanofi-Genzyme; grant support from Adamas, Biogen, Genentech, Novartis, and Sanofi-Genzyme. E.B., D.B., C.C., S.K., J.P.M., C.M., B.P., and D.X. are employees of and hold stock/stock options in Biogen. D.R. has received consulting fees from Alexion, Biogen, Celgene, EMD Serono, Genentech, Novartis, Sanofi-Genzyme, and Teva; speaker fees from Acorda, Alexion, Biogen, Celgene, EMD Serono, Genentech, Mallinckrodt, Novartis, Sanofi-Genzyme, and Teva; grant support from Actelion, Biogen, EMD Serono, Genentech, Mallinckrodt, MedDay, Novartis, Patient-Centered Outcomes Research Institute, Sanofi-Genzyme, and TG. T.Z. has nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Biogen provided funding for the study.
Research Ethics and Patient Consent
The PROCLAIM study was conducted in accordance with The International Conference on Harmonization Guidelines on Good Clinical Practice, the ethical principles outlined in the Declaration of Helsinki, and all applicable local laws and regulations. Written informed consent was obtained from each patient prior to eligibility evaluations.
Trial Registration
2015
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
