Abstract
Background:
Exposure to parental chronic illness is associated with adverse developmental outcomes.
Objective:
We examined the association between parental multiple sclerosis (MS) and parental MS-related clinical factors on developmental health.
Methods:
We conducted a population-based cohort study in British Columbia, Canada, using linked health databases. The outcome was childhood development at 5 years of age, expressed as vulnerability on the Early Development Instrument (EDI). Adjusted odds ratios (aORs) and 95% confidence intervals (CIs) were estimated using conditional logistic regression.
Results:
MS-affected parents (n = 783) were older, more likely to be English speakers, and had higher rates of mental health morbidity (39.6% vs 22.2%, p < 0.001) than unaffected parents (n = 2988). In the adjusted models, children of mothers with MS (aOR = 0.62, 95% CI = 0.44–0.87), but not children of the fathers with MS, had a lower risk of vulnerability on the social development domain of the EDI. However, mental health comorbidity (aOR = 1.62, 95% CI = 1.05–2.50) and physical comorbidity (aOR = 1.67, 95% CI = 1.05–2.64) among mothers with MS were associated with increased vulnerability on the EDI.
Conclusion:
Maternal MS, but not paternal MS, was associated with lower rates of developmental vulnerability on the social development domain. However, mental and physical comorbidity among MS-affected mothers were associated with increased developmental vulnerability in children.
Introduction
Approximately 10% of children live in households where a parent has a chronic illness.1,2 Research often focuses on the ill child, but typically neglects children of chronically ill parents despite the evidence that these children have significantly more signs of anxiety and depression than children of healthy parents.1,3
Multiple sclerosis (MS) typically manifests between the ages of 20 and 40 years, when parenting is an important issue. 4 Although the relevant literature is sparse, some studies have suggested that children of parents with MS display higher rates of adjustment difficulties, particularly anxiety disorders and behavioral disturbances.5,6 Comorbidity is relatively common in MS with over one-third reporting at least one physical comorbidity, and approximately 50% of MS patients suffering from depression and anxiety disorders. 7 Overall, parental mental illness is an important predictor of child adjustment and has been associated with a detrimental effect on children’s early emotional development.8,9 As MS disease course changes over time and disability progresses, the family may experience more conflict and less cohesion, with parents paying less attention to the child’s needs. 4
Few methodologically rigorous studies have examined the association between parental MS and MS-related clinical characteristics and children’s developmental health. We therefore carried out a population-based study to investigate the association between parental MS and MS-related clinical factors such as the presence of mental and physical comorbidity, disability level, and disease duration on the child’s developmental outcomes.
Methods
Data sources
Information on study subjects was obtained from several population-based linked health and education databases in British Columbia. The publicly funded provincial health care program in British Columbia covers all residents and a lifelong unique personal health care number is assigned to each individual. Anonymized linked data included the: Medical Services Plan 10 database (fee-for-service physician billing claims); Discharge Abstract Database 11 (hospital admission and discharge records); PharmaNet 12 (prescription drug data); Census GeoData (socioeconomic status (SES) data expressed as average neighborhood income quintiles, using postal codes via Statistics Canada); Consolidation Files (demographic and residency data); Vital Statistics Birth 13 database (all births in the province); and the Early Development Instrument (EDI) 14 (early childhood developmental health data via the Human Early Learning Partnership). 15 MS-related clinical information (i.e. disease course and disability level) was obtained for a subset of individuals via linkage with the British Columbia MS database. 16
Study cohorts
Parents with MS were identified using a validated algorithm as those with ⩾3 records for a relevant hospital or physician claim between 1 April 1985 and 31 December 2011 or a relevant prescription claim after 1 April 1996 (Appendix Table 1—Supplementary Material). 17 Using the birth registry and the Consolidation File databases, individuals with MS were linked to their offspring. All persons with MS who had a child born between 1 January 1994 and 31 December 2006, with EDI data were included in the study cohort. These birth dates allowed each child to have reached his/her fifth birthday between 1999 and 2011 and to have been part of the EDI data collection. The onset date of MS was based on the first date for MS or a demyelinating condition identified in any hospital, physician, or prescription claims (Appendix Table 1—Supplementary Material). Up to four children with parents who were not known to have MS or any demyelinating condition, selected from the population of British Columbia, matched on year of birth and school district, formed the comparison group. The MS-affected parent was also matched by sex to a parent in the reference cohort. Matches for parental sex could not be obtained in 4% of instances and the next available parent in the database was selected in such cases. Children, both of whose parents had MS, were excluded from the study.
Outcomes and determinants
The primary outcome of interest was childhood development, as measured by the EDI. The EDI has routinely been administered province-wide every 1 to 3 years since the 1999/2000 school year, achieving at least 85% participation of kindergarten children from each school district in the province. Teachers completed the EDI for each child in their kindergarten class (age range = 5–7 years) in February. The EDI consists of 104 binary and three-category items designed to tap five core areas of early childhood development:14,18 physical health and well-being; social competence; emotional maturity; language and cognitive development; and communication skills and general knowledge (see Appendix 2—Supplementary Methods). 14 The EDI domains are highly skewed and are not amenable to simple transformation. Hence, children’s scores on the EDI outcomes were categorized as “developmentally vulnerable” if their scores fell below the 10th centile value 19 based on the national EDI cut-off scores for each of the five domains. These can be interpreted as the child demonstrating a lower-than-average ability (<10th centile) in the competencies measured in that domain. 20 Previous studies have shown that as the number of vulnerable domains increases, so does the future probability of failure to achieve basic competencies at ages 8, 10, and 12 years.21–23
The main determinant of interest was the presence (vs absence) of parental MS. Other variables of interest included were: the child’s sex, child’s first language at home (English vs other), age of the child at the time of the EDI assessment (in years), and SES. Parental characteristics of interest were parental age at the time of child’s birth, sex, and comorbidity. We studied all comorbidities affecting ⩾5% of the overall MS population. 24 Parental comorbid conditions examined included mental health disorders (either depression or anxiety disorder) 7 and physical comorbidity (i.e. presence of diabetes mellitus, hypertension, hyperlipidemia, or chronic lung disease) 25 before the EDI data collection. All comorbidities were identified using previously validated algorithms based on hospital, physician visit (using the International Classification of Diseases codes (ICD-9/10-CA)), and prescription information (Drug Identification Numbers and the Anatomical Therapeutic Chemical classification system).7,25
Statistical analyses
Conditional logistic regression was used to compare the characteristics of the parent-child dyads in the MS and matched reference cohorts. Multivariable conditional logistic regression models were used to determine the association between parental MS and vulnerability on one or more domains as well as on each domain of the EDI separately, adjusted for potential confounders. Generalized estimating equations, with an assumed unstructured correlation framework, were used to adjust the variance as some parents had more than one child in the cohort. Confounders were included in the final models based on the literature19,26 or statistical significance (p < 0.1). The full model included age of the child at the time of EDI completion (continuous), child’s sex (female vs male), SES (expressed as quintiles), parental age (continuous), parental mental morbidity (present vs absent), parental physical comorbidity (present vs absent) and parental sex (male vs female). Interactions between parental MS and confounders were examined and stratified results were presented when an interaction was present.
The association between MS-related clinical factors on the child’s developmental health was examined within the MS cohort. Analyses restricted to the MS cohort included an assessment of the association of parental mental comorbidity (present vs absent); parental physical comorbidity (present vs absent); the duration of parental MS (i.e. time from onset of MS to the child’s EDI assessment); and disability level (as measured by the Expanded Disability Status Scale (EDSS) score) 27 at the time of EDI data collection (±3 years) with each EDI domain. MS disease duration was categorized as: <5, 5 to <10, and ⩾10 years. MS disability was dichotomized <4.0 vs ⩾4.0 on the EDSS (an EDSS of ⩾4 signifies moderate or severe disability).
Results were expressed as odds ratios (ORs) with 95% confidence intervals (CIs). Regression model fit was assessed using the likelihood ratio test, and a two-sided p < 0.05 was used to determine statistical significance. Analyses were performed using SAS Version 9.2 (SAS Institute Inc., Cary, NC). The University of British Columbia’s Clinical Research Ethics Board approved the study.
Results
After applying the exclusion criteria, the final study population consisted of 783 children with an MS parent and 2988 parent-child dyads in the matched reference cohort (Figure 1). The children of parents with and without MS were similar in terms of sex, age, and SES at the time of EDI data collection (Table 1). Parents affected by MS were on average 1.5 years older, and were more likely to be English speakers. The frequency of physical comorbidity was similar among parents with and without MS, although mental health morbidity affected considerably more parents with MS compared with non-MS parents (39.6% vs 22.2%, p < 0.001).
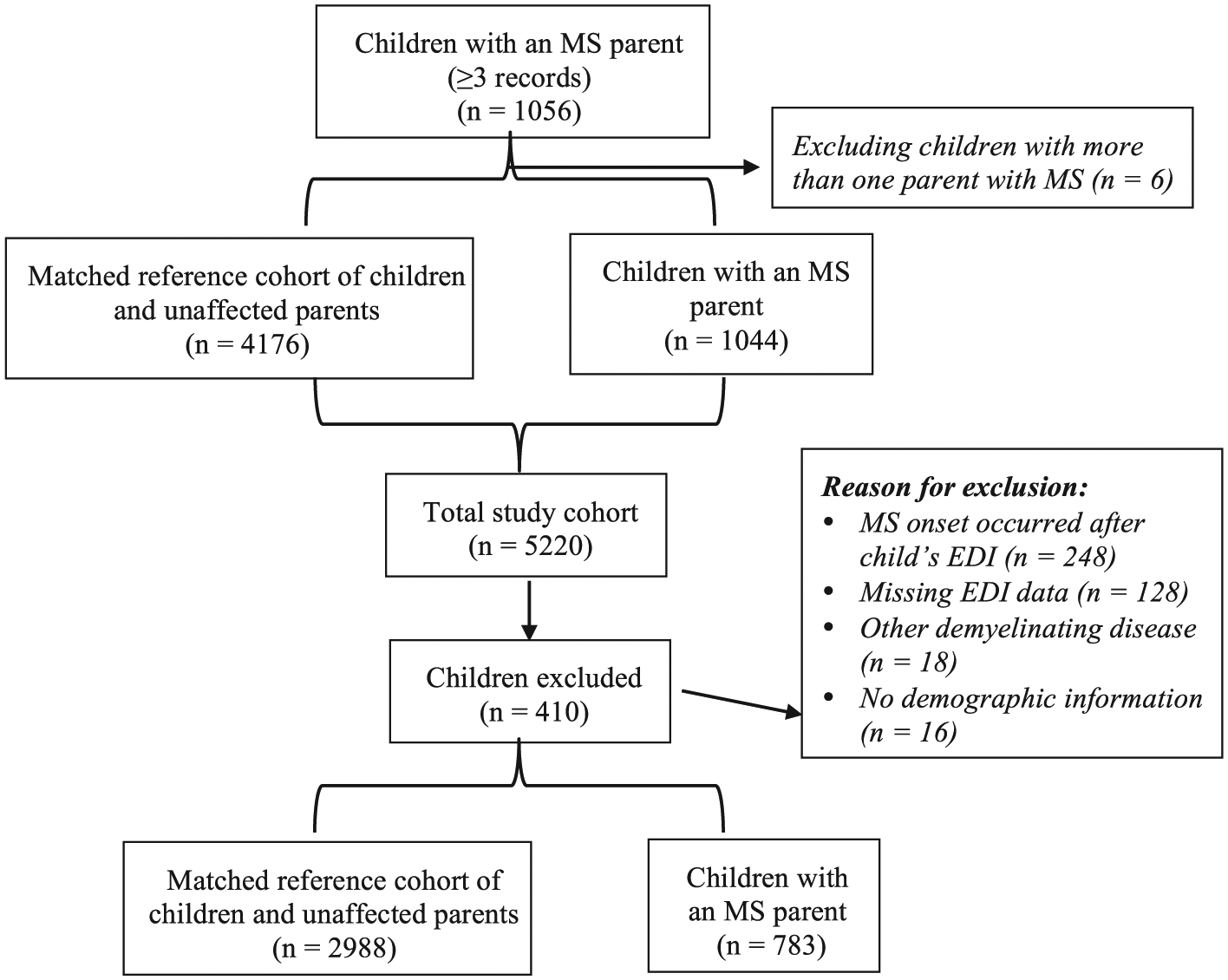
Schematic depiction of the cohort selection.
Characteristics of the children of parents with and without MS, British Columbia, Canada.
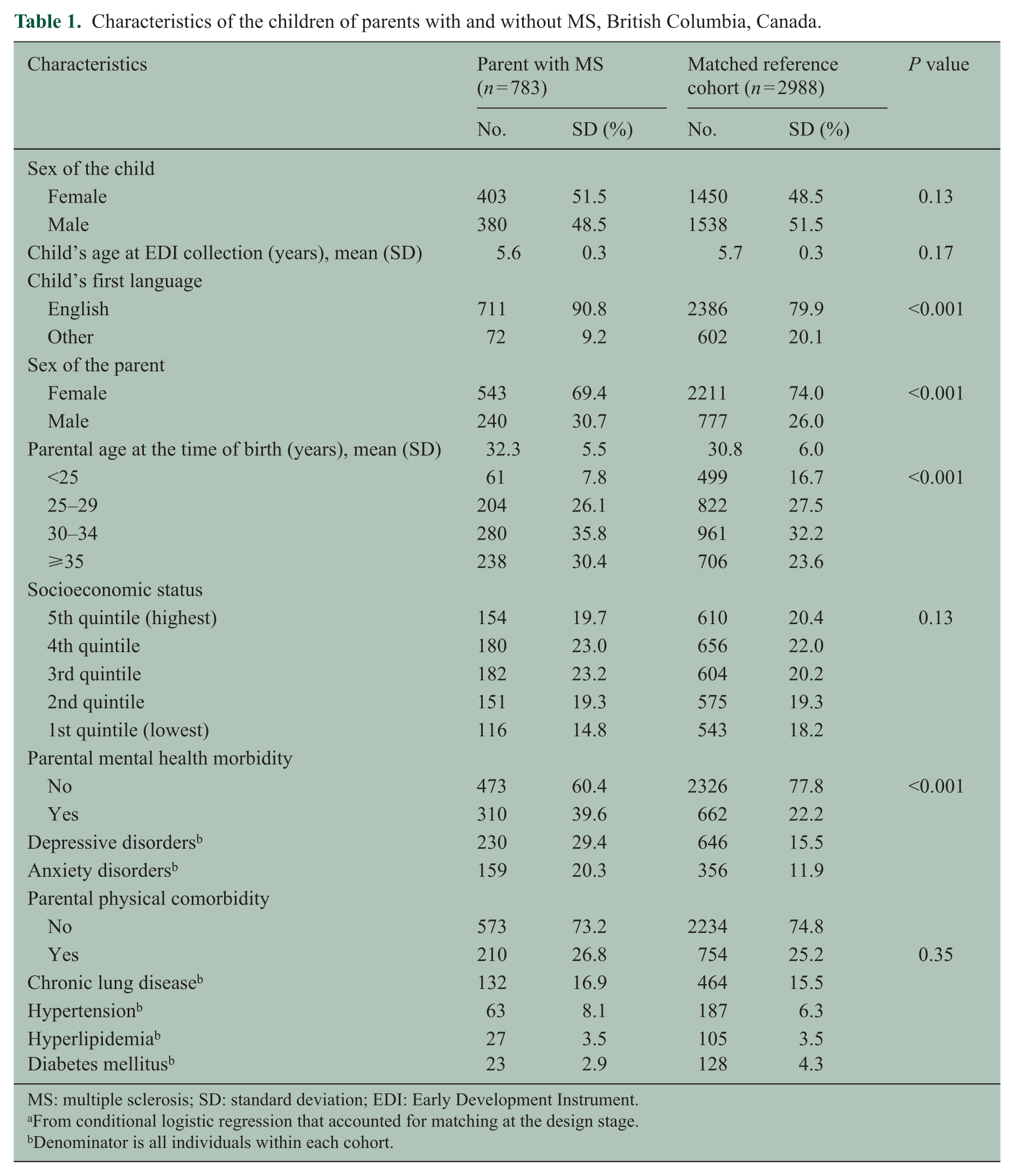
MS: multiple sclerosis; SD: standard deviation; EDI: Early Development Instrument.
From conditional logistic regression that accounted for matching at the design stage.
Denominator is all individuals within each cohort.
The disease characteristics of the MS parents are shown in Table 2. The median age at onset of MS was 31 years and 35% of the parents were ever on MS disease-modifying medication. The median disease duration at the time of EDI completion was 5.4 years (range = <1–26 years). Of the 280 (36%) MS parents for whom an EDSS score was available, most (81%) had minor neurological findings, while 19% had moderate or severe disability.
Characteristics of the cohort with MS (n = 783 parents), British Columbia, Canada.

MS: multiple sclerosis; EDI: Early Development Instrument; EDSS: Expanded Disability Status Scale.
n = 64 (23.5%) were exposed to glatiramer acetate and n = 208 (76.5%) to a beta-interferon.
Disease course and EDSS data were available on a subset of MS parents.
The presence of vulnerability within each of the EDI domains by parental MS is shown in Figure 2. Overall, 29% (1086/3771) of children were vulnerable on one or more domains of the EDI, with physical and social domains having the highest rates of vulnerability at 15% (547/3771) and 12% (452/3771), respectively.
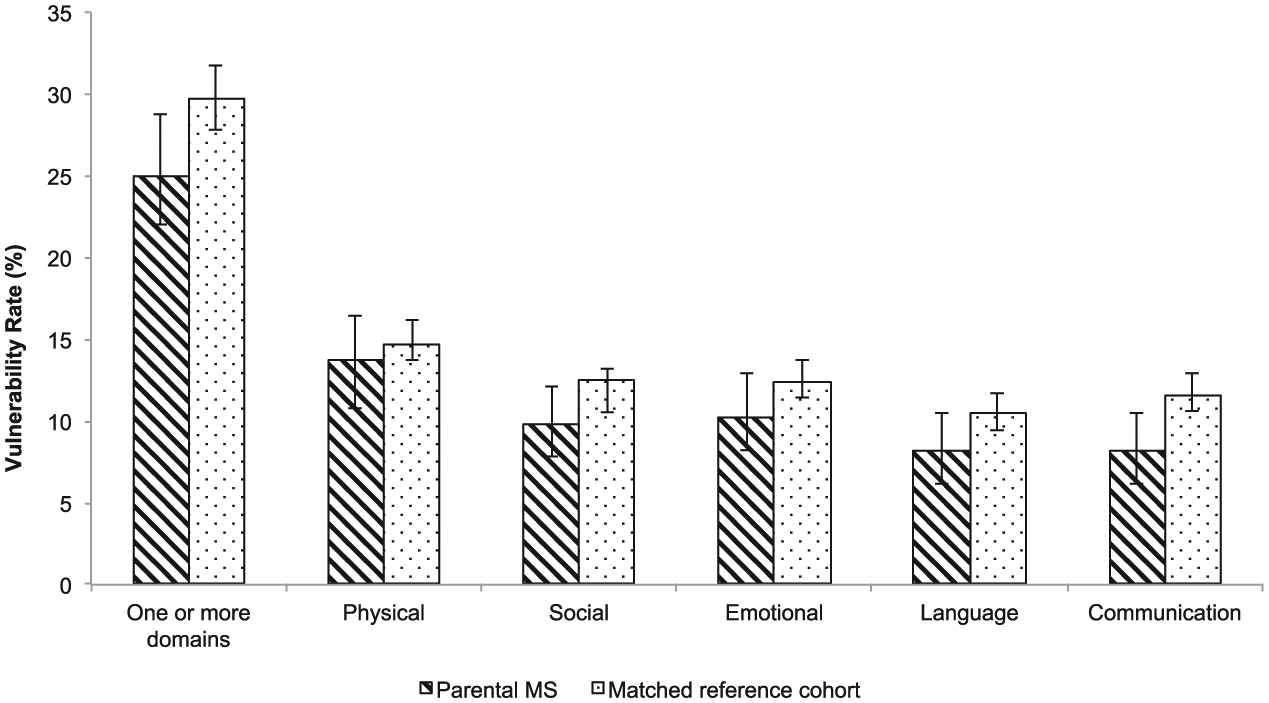
Rates of children’s vulnerability (%) within each Early Development Instrument (EDI) domain by parental multiple sclerosis (MS) status, British Columbia, Canada.
The unadjusted analysis (Table 3) showed that children of parents with MS had lower odds of vulnerability on one or more domains of the EDI compared with children in the matched reference cohort (OR = 0.79, 95% CI = 0.66–0.95). Greater odds of vulnerability on one or more domains of the EDI were also observed among boys, children who were younger at completion of the EDI, children who were in the lower income quintiles (vs highest), and those whose parents had a mental or physical comorbidity (Table 3). In the adjusted model (Table 3), parental sex modified the relationship between parental MS and vulnerability on the EDI (p for interaction 0.05). In analyses stratified by parental sex, the odds of vulnerability on one or more domains of the EDI were significantly lower among children of mothers with MS compared with the children of mothers without MS (adjusted odds ratio (aOR) = 0.75, 95% CI = 0.60–0.95). Paternal MS did not show this association.
Unadjusted and adjusted odds ratios and 95% confidence intervals showing the association between parental MS and the child’s vulnerability on the EDI, British Columbia, Canada.
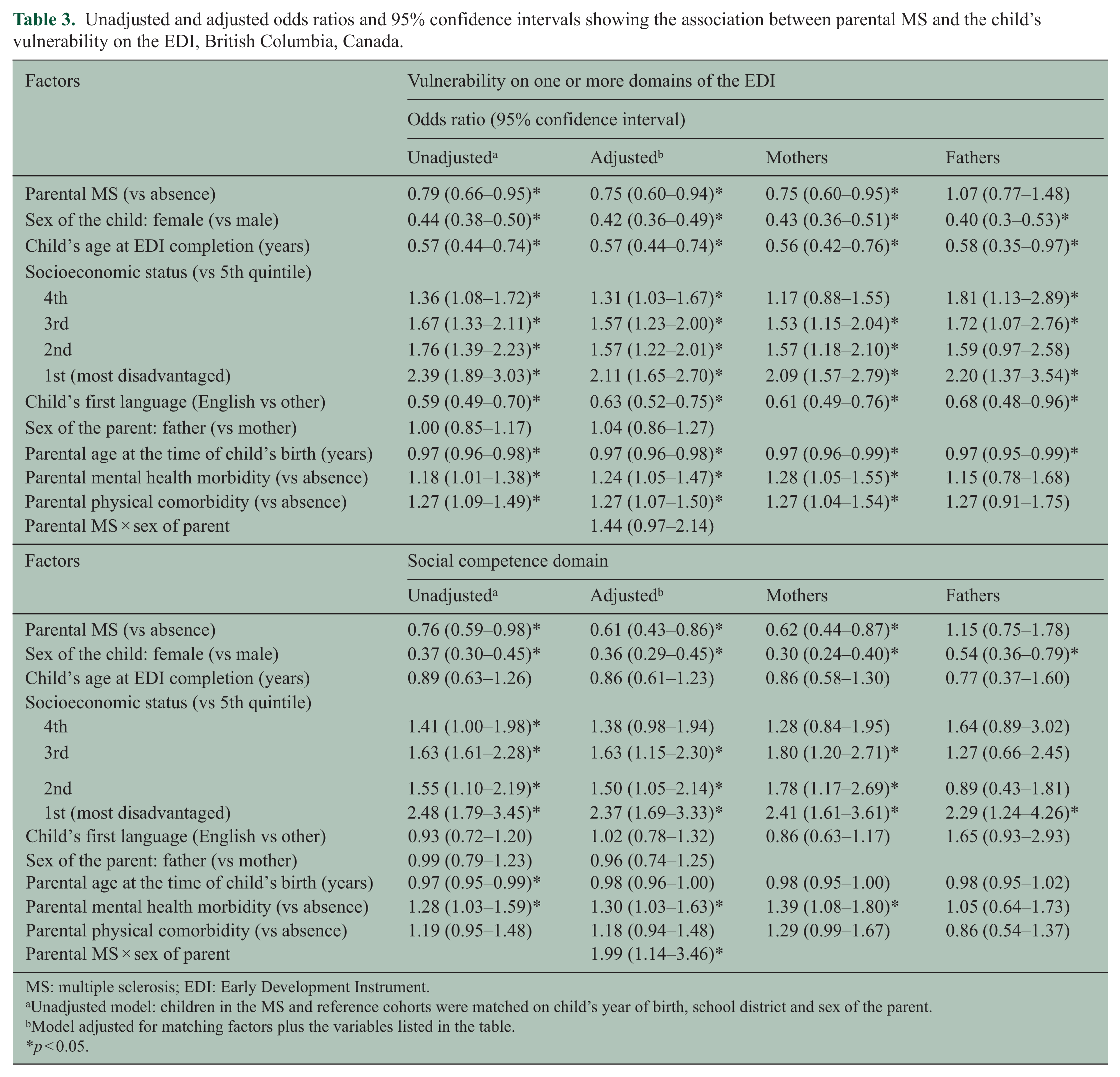
MS: multiple sclerosis; EDI: Early Development Instrument.
Unadjusted model: children in the MS and reference cohorts were matched on child’s year of birth, school district and sex of the parent.
Model adjusted for matching factors plus the variables listed in the table.
p < 0.05.
Analyses examining the association between parental MS and each domain of the EDI demonstrated that parental MS was associated with statistically significantly lower odds of vulnerability on the social competence domain of the EDI (Table 3). Parental MS was not significantly associated with vulnerability on any of the other four EDI domains (Appendix Table 2—Supplementary Material). Furthermore, parental sex modified the relationship between parental MS and vulnerability on the social domain (p for interaction = 0.01). The odds of vulnerability on the social competence domain was 38% lower among children of mothers with MS compared with the children of mothers without MS (aOR = 0.62, 95% CI = 0.44–0.87; Table 3). Paternal MS did not show the same association.
Within the MS cohort (n = 783), children with an MS mother who also had mental health comorbidity had 62% greater odds of vulnerability on one or more domains of the EDI (aOR = 1.62, 95% CI = 1.05–2.50; Table 4) compared with MS mothers without mental health comorbidity. This effect was significant for two domains: language and cognitive development and physical health. Paternal MS with mental health comorbidity was associated with higher odds of vulnerability on the emotional domain of the EDI (aOR = 2.52, 95% CI = 1.01–6.26) compared with paternal MS without mental health comorbidity. Presence of physical comorbidity in mothers with MS put children at significantly higher odds of vulnerability on one or more domains of the EDI (aOR = 1.67, 95% CI = 1.05–2.64). There was a two-fold greater odds of vulnerability on the physical health domain (aOR = 2.58, 95% CI = 1.10–6.05) and five-fold greater odds of vulnerability on language and cognitive development (aOR = 5.02, 95% CI = 1.67–15.1) domain of the EDI, among children of parents with moderate to severe disability compared with those with less severe disability (Appendix Table 3—Supplementary Material). Although disease duration was not significantly associated with vulnerability on any of the EDI domains, children of mothers with a disease duration of ⩾10 years had non-significantly higher odds of vulnerability on all domains of the EDI compared with mothers with a disease duration of <5 years (Table 4).
Associations between parental characteristics and the child’s vulnerability on the EDI domains stratified by parental sex among parents with MS, British Columbia, Canada (n = 783 children).
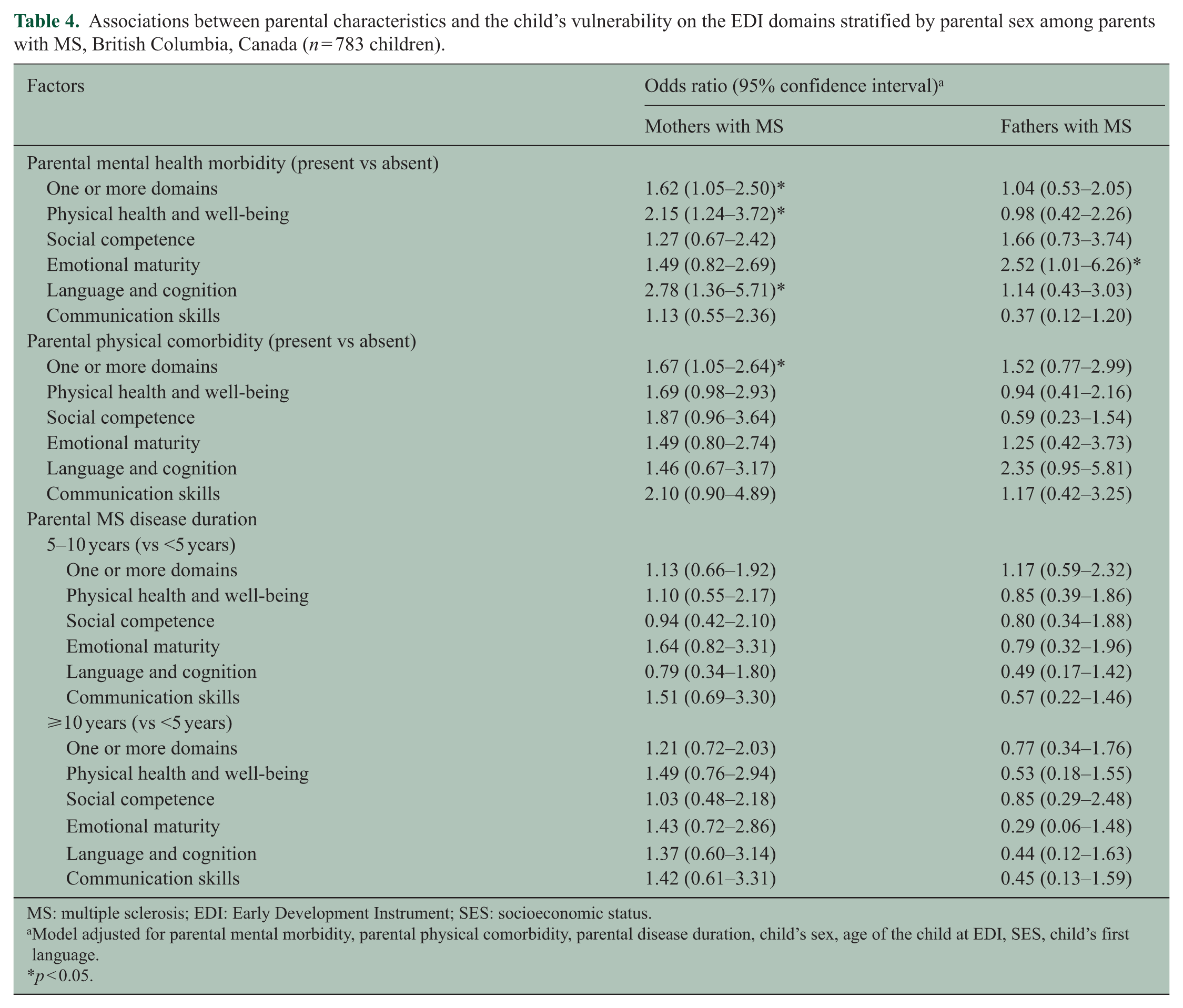
MS: multiple sclerosis; EDI: Early Development Instrument; SES: socioeconomic status.
Model adjusted for parental mental morbidity, parental physical comorbidity, parental disease duration, child’s sex, age of the child at EDI, SES, child’s first language.
p < 0.05.
Discussion
In this large population-based study, we showed that children of parents who had MS had a lower risk of developmental vulnerability at 5 years of age, as measured by the EDI. However, this relationship was modified by the sex of the affected parent. Children of mothers with MS had lower risk of vulnerability on the social development domain of the EDI compared with the matched children of mothers without MS. This association was not observed for fathers. However, even though parental MS did not independently increase the risk of vulnerability in children, mental health morbidity and physical health, comorbidity in the mothers with MS were both associated with a higher risk of developmental vulnerability on the language and cognition and the physical health and well-being domains (compared with children of mothers who had MS but no mental health comorbidity). On the other hand, children of fathers with MS who had a co-existing mental health condition were at an elevated risk of vulnerability on the emotional maturity domain. Finally, children whose MS parent had moderate to severe disability had a higher risk of vulnerability on the physical health, and language and cognition development domains of the EDI compared with children whose MS-affected parent had less severe disability.
The lower risk of developmental vulnerability in children with parental MS in this study extends previous work by our group based on a smaller cohort from Manitoba where we were unable to find a significant positive association. 9 Although similar trends were observed, the relative small size of the MS cohort in the Manitoba study (n = 153 vs 783 in this study) likely limited our ability to detect differences. 9 Other qualitative and cross-sectional studies have also shown that parental MS imposes a greater burden of domestic/household duties on children and this results in an increase in pro-social behaviour.28,29 Children from such families describe having higher personal competence, feeling more empathetic to others and more “grown-up,” as the benefits to having a parent with MS. 30 Even though these studies were focused on older children, such findings are consistent with the hypothesis that mild, intermittent stressors (positive stress) in a child’s life may have beneficial effects and are likely helpful for child development. 31 However, we cannot rule out adverse effects in later childhood and adolescence especially if the stress increases measurably. 32
In our study, children of mothers with MS (but typically not those of fathers with MS) had lower rates of overall vulnerability on the EDI and specifically on the social domain of the EDI. To the best of our knowledge, there are no similar studies on the issue of the parental sex (or gender), MS, and child development. Nevertheless, the broader literature does suggest that children who have a mother with a chronic condition, such as MS, do not experience serious academic difficulties unless the burden of illness and associated stress reach an extreme level. 33 Although, we were able to demonstrate a difference between the effects of paternal MS on developmental vulnerability, our study was likely underpowered to examine the possible association(s) between paternal MS and early childhood development.
The negative association between mental and physical comorbidity and vulnerability on the EDI is consistent with findings from previous work demonstrating that children whose parent had both MS and a mental health condition (vs MS, but no mental health comorbidity) were at an increased risk of vulnerability on emotional maturity and social competence domains of development.9,34 Further, in our subgroup of MS parents with information on disability (as measured by the EDSS), a greater level of MS-related disability in the parent was associated with an increased risk of developmental vulnerability among children. The literature suggests that disability in parents may disrupt parenting and constrain them from accessing timely care for their children. 35 Indeed, studies with older children show that parental MS-related disability has adverse effects on youth adjustment and family functioning through the higher caregiving demand on the youth and the potential stigma related to the parent’s disability. 36
The strengths of our study include the large study size, the comprehensive population-based nature of our cohort, and the use of previously validated case definitions for both MS and other morbidity. Approximately one-third of MS parents were ever on a MS disease-modifying medication which is compatible with rates of such treatment seen in the MS population in British Columbia 37 (despite being lower than that observed in some MS specialty clinic-based cohorts). The EDI assessment by teachers avoided reliance on parental or self-report of the child’s developmental health. Although the EDI has undergone significant psychometric testing to confirm validity and reliability as a research tool,14,38 there may be some individual differences in teachers’ ability to evaluate developmental health. 39 The relatively small number of MS-affected fathers limited our ability to draw robust conclusions about the association between paternal MS and developmental health in children. Also, we were only able to identify morbidity among subjects whose condition was recognized by the health care system, and hence morbidity rates may have been underestimated. Finally, we did not have access to ethnicity and family-specific information on SES, and used neighborhood-level average income as a proxy for families’ SES.
In summary, our population-based study demonstrates that maternal MS is associated with lower rates of developmental vulnerability, specifically vulnerability on the social development domain, in children at 5 years of age. However, although parental MS was not independently associated with developmental vulnerability in children, presence of mental and physical comorbidity, and greater disability in MS-affected parents are associated with higher risk of developmental vulnerability in children. Parents affected by MS who also have co-existing mental and physical comorbidity may benefit from appropriate support strategies and services that will help them mitigate adverse developmental effects on their children.
Footnotes
Acknowledgements
The authors gratefully acknowledge the late Professor Clyde Hertzman for providing guidance and helpful feedback in conceptualizing and planning this program of research and for his role in obtaining funding. They thank the BC Ministry of Health, BC Vital Statistics Agency, and BC PharmaNet for approval and support for accessing provincial data and Population Data BC for facilitating approval and use of the data.
They gratefully acknowledge the BC MS Clinic neurologists who contributed to the study through patient examination and data collection (current members listed here by primary clinic):
UBC MS Clinic: A Traboulsee (UBC Hospital MS Clinic Director and Head of the UBC MS Programs); A-L Sayao, V Devonshire, and S Hashimoto (UBC and Victoria MS Clinics); J Hooge (UBC and Prince George MS Clinic); L Kastrukoff (UBC and Prince George MS Clinic); J Oger.
Kelowna MS Clinic: D Adams, D Craig, and S Meckling.
Prince George MS Clinic: L Daly.
Victoria MS Clinic: O Hrebicek, D Parton, and K Atwell-Pope. The views expressed in this paper do not necessarily reflect the views of each individual acknowledged.
Conflict of interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Canadian Institutes of Health Research (MOP-119393, PI Helen Tremlett).
Neda Razaz is funded by the Multiple Sclerosis Society of Canada (PhD Research Studentships). She has received travel grants from the Canadian Institutes of Health Research and the endMS Research and Training Network/Multiple Sclerosis Society of Canada.
KS Joseph is the recipient of a Chair in maternal, fetal, and infant health services research from the Canadian Institutes of Health Research. His work is also supported by the Child and Family Research Institute.
W Thomas Boyce is funded by a grant from the Canadian Institute for Advanced Research.
Martin Guhn is funded by a grant from the Lawson Foundation, and he is PI/Co-PI on research grants from the Canadian Institutes for Health Research (CIHR) and the Social Sciences and Humanities Research Council (SSHRC) of Canada.
Ruth Ann Marrie is supported by a Don Paty Career Development Award from the MS Society of Canada and by a Manitoba Research Chair from Research Manitoba. Dr Marrie has received research support from CIHR, the MS Society of Canada, the National MS Society, Research Manitoba, and the MS Scientific Research Foundation. She has conducted clinical trials for sanofi-aventis.
Helen Tremlett is the Canada Research Chair in Neuroepidemiology and Multiple Sclerosis. She has received research support from the National Multiple Sclerosis Society, the Canadian Institutes of Health Research, and the UK MS Trust; speaker honoraria and/or travel expenses to attend conferences from the Consortium of MS Centres (2013), the National MS Society (2012, 2014), Bayer Pharmaceuticals (2010), Teva Pharmaceuticals (2011), ECTRIMS (2011, 2012, 2013, 2014), UK MS Trust (2011), the Chesapeake Health Education Program, US Veterans Affairs (2012), Novartis Canada (2012), Biogen Idec (2014), and American Academy of Neurology (2013, 2014, 2015). Unless otherwise stated, all speaker honoraria were either donated to an MS charity or to an unrestricted grant for use by her research group.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
