Abstract
Neuromyelitis optica (NMO) is an inflammatory autoimmune disease of the central nervous system that preferentially targets the optic nerves and spinal cord. The clinical presentation may suggest multiple sclerosis (MS), but a highly specific serum autoantibody against the astrocytic water channel aquaporin-4 present in up to 80% of NMO patients enables distinction from MS. Optic neuritis may occur in either condition resulting in neuro-anatomical retinal changes. Optical coherence tomography (OCT) has become a useful tool for analyzing retinal damage both in MS and NMO. Numerous studies showed that optic neuritis in NMO typically results in more severe retinal nerve fiber layer (RNFL) and ganglion cell layer thinning and more frequent development of microcystic macular edema than in MS. Furthermore, while patients’ RNFL thinning also occurs in the absence of optic neuritis in MS, subclinical damage seems to be rare in NMO. Thus, OCT might be useful in differentiating NMO from MS and serve as an outcome parameter in clinical studies.
Keywords
Introduction
Neuromyelitis optica (NMO) is an immune-mediated disorder of the central nervous system (CNS) in which the optic nerves and the spinal cord are preferentially involved. 1 The disease-specific serum immunoglobulin (Ig)G targeting the astrocyte water channel aquaporin-4 (AQP4)2,3 has facilitated differentiation of NMO from multiple sclerosis (MS) and recognition of a broad phenotypic spectrum referred to as neuromyelitis optica spectrum disorders (NMOSD). 4 The use of the AQP4-IgG autoantibody in various in vivo and ex vivo models has led to an initial understanding of the pathogenic mechanisms that contribute to optic nerve injury. 5 While additional autoantibodies against myelin oligodendrocyte glycoprotein (anti-MOG) and aquaporin-1 have been reported in a small number of NMO patients, the specificity of these autoantibodies to NMO and their relationship to disease pathogenesis remain unclear.6–8
Techniques that provide information on the optic nerve structure and function are likely to prove useful to clinicians dealing with CNS diseases. Whereas a number of techniques evaluate visual function such as visual acuity, contrast sensitivity, color vision, visual fields, evoked potentials and pattern electroretinogram, others such as magnetic resonance imaging (MRI), optic coherence tomography (OCT), confocal scanning laser ophthalmoscopy (CSLO), and scanning laser polarimetry with variable corneal compensation (GDx-VCC), assess the anatomical integrity of the optic nerve and retina (Figure 1). OCT may have the advantage of requiring shorter acquisition times than CSLO,9,10 and is more sensitive than GDx-VCC 11 in detecting retinal nerve fiber layer (RNFL) thinning in nasal and temporal sectors. It has the additional capability of measuring retinal segmental thickness.12,13
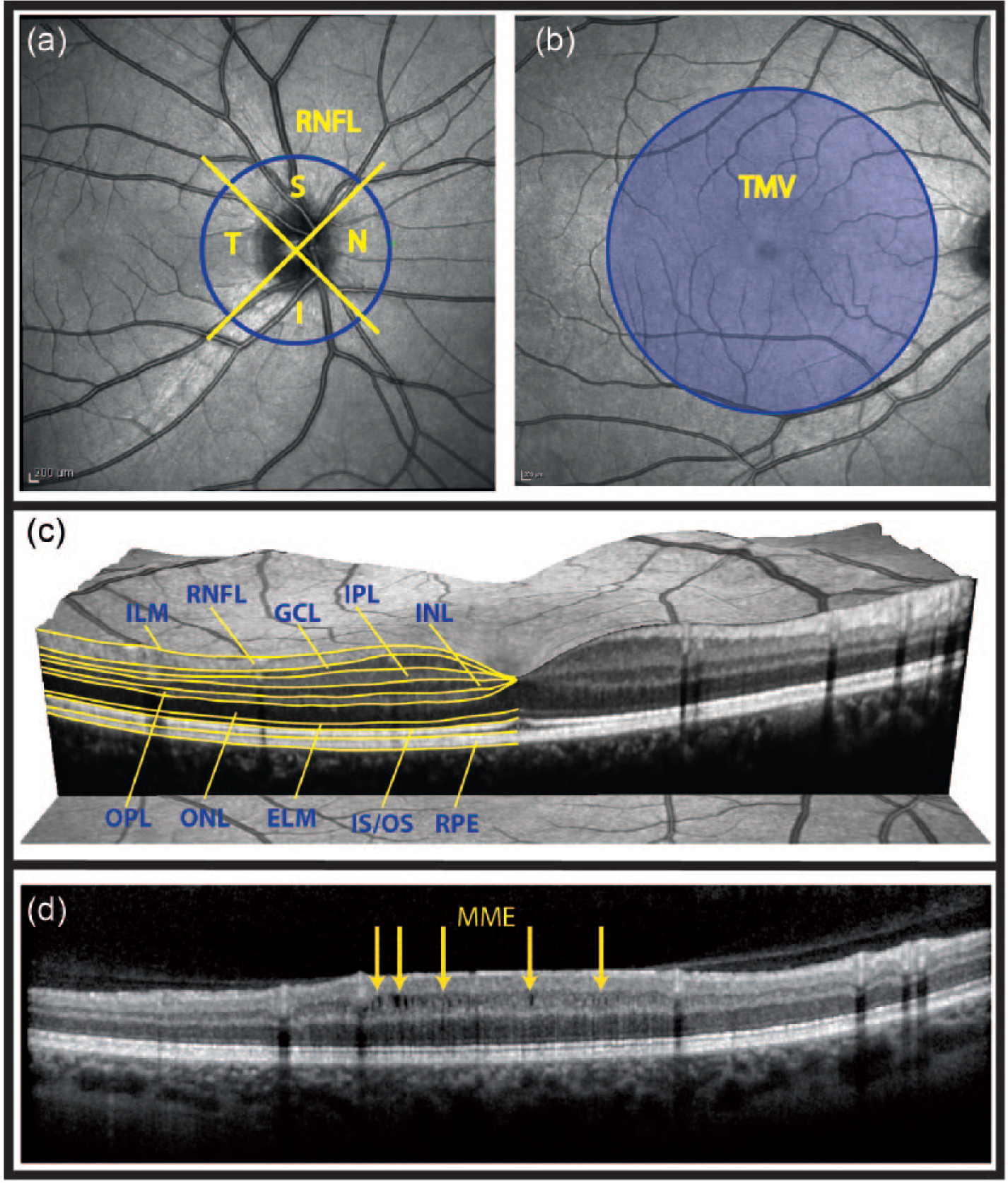
Retinal parameters acquired by OCT.
OCT is an analog of B-mode ultrasonography but uses infrared light instead of ultrasound to produce images based on the differential optical reflectivity. It can provide data on peripapillary and macular RNFL thicknesses and generate macular maps with segmental thicknesses and volumes (Figure 1). The recent development of spectral domain OCT (SD-OCT) allows enhanced resolution (2 µm), shorter acquisition times, three-dimensional scans, and video imaging.14,15 Also, eye tracking systems permit almost perfect repositioning in longitudinal studies, allowing investigators to capture subtle changes on the order of a few micrometers. Investigations in MS have demonstrated that OCT is easy to perform, reproducible, and provides downstream measures of secondary neurodegeneration (RNFL and macular thinning).12,16–19
Optic neuritis (ON) is a common condition involving primary inflammation, demyelination, and axonal injury in the optic nerve. 20 This process may lead to retinal ganglion cell (RGC) death, decreased macular volume, and to visual dysfunction or permanent visual loss. ON usually presents as an acute episode of unilateral or less frequently bilateral optic nerve inflammation, accompanied by ocular pain and decreased vision. ON is immune mediated 21 and, in NMOSD, is associated with detectable AQP4-IgG. 22 The pathological features of NMOSD include deposition of IgG and activated complement, loss of AQP4 expression, astrocytopathy, neutrophil accumulation and demyelination with axon loss.23,24 The frequent involvement of the optic nerve in MS and NMO may be caused by a more reduced blood-brain barrier (BBB) function.25,26 Furthermore, the optic nerve expresses high levels of supramolecular aggregates of AQP4. 27 The combination of enhanced AQP4 supramolecular aggregation and heightened BBB permeability may contribute to the specific pattern of tissue damage in NMOSD. Objective measures of the severity and etiology of optic nerve injury is important for the diagnosis, management, and treatment of ON. NMOSD-ON differs clinically from MS-ON; bilateral involvement is more common, and recurrent ON and severe residual visual dysfunction more likely.7,28–32 Common MRI imaging features include lesions extending over one-half the length of the optic nerve, posterior nerve involvement, and chiasmal inflammation.33,34 Whereas many characteristics of OCT in MS have already been established and are consistent with its pathophysiology,35–40 OCT features in NMOSD are currently ill-defined and the relationship of these abnormalities with disease pathophysiology remains unclear.41–51
This review analyzes the published data on OCT in NMOSD so that its ability to quantify optic nerve damage, facilitate diagnosis, monitor disease progression, evaluate therapeutic efficacy, and detect novel pathology might be assessed.
Methods
A literature search of Ovid MEDLINE (1946–2014) was conducted using the search terms “neuromyelitis optica” combined with “optical coherence tomography.” Given that OCT is a newer technology, all articles were current (2008–present). Review articles and case reports were excluded; however, the references contained in such articles were reviewed for completeness. PubMed was also searched using similar methodology. Only studies available in English were included. Authors were not contacted for unpublished data. A comprehensive overview of the individual studies is given in the supplementary table.
OCT findings in NMOSD
RNFL and macula
OCT has been applied to NMOSD cohorts, initially in cross-sectional studies46–48 and, more recently, in prospective longitudinal investigations. 52 Cross-sectional studies have consistently shown that the RNFL is significantly altered in NMOSD patients with ON compared to healthy controls and that RNFL thinning may be an early and frequent phenomenon. NMOSD-ON affects the entire peripapillary RNFL with particular involvement of the superior and inferior quadrants 43,45 (Table 1 and Figure 2). This may reflect a lower preference for small-diameter axons, which are more abundant in the temporal quadrant and are preferentially affected in MS-ON.36,53 Macular thinning is more severe in NMOSD with ON than in MS-ON, in line with poorer visual recovery observed following ON in NMOSD. NMOSD patients with a history of ON tend to have significantly lower RNFL thicknesses than patients with MS-ON42–45 (Table 1 and Figure 2). A meta-analysis showed that ON significantly affects RNFL integrity and, on average, leads to a loss of approximately 20 μm in the affected eye in relapsing–remitting MS (RRMS) compared to healthy controls. 36 A recent study by Green and Cree showed an average RNFL loss in MS-ON of 17.6 μm compared to an average 31.1 μm reduction in NMOSD-ON. 44 Several studies showed that RNFL thickness in NMOSD patients after ON is reduced to 55–83 µm, compared to 93–108 µm in the respective control groups.42,43,45–48,50,54–57
Summary of neuro-ophthalmological parameters in neuromyelitis optica compared to multiple sclerosis.
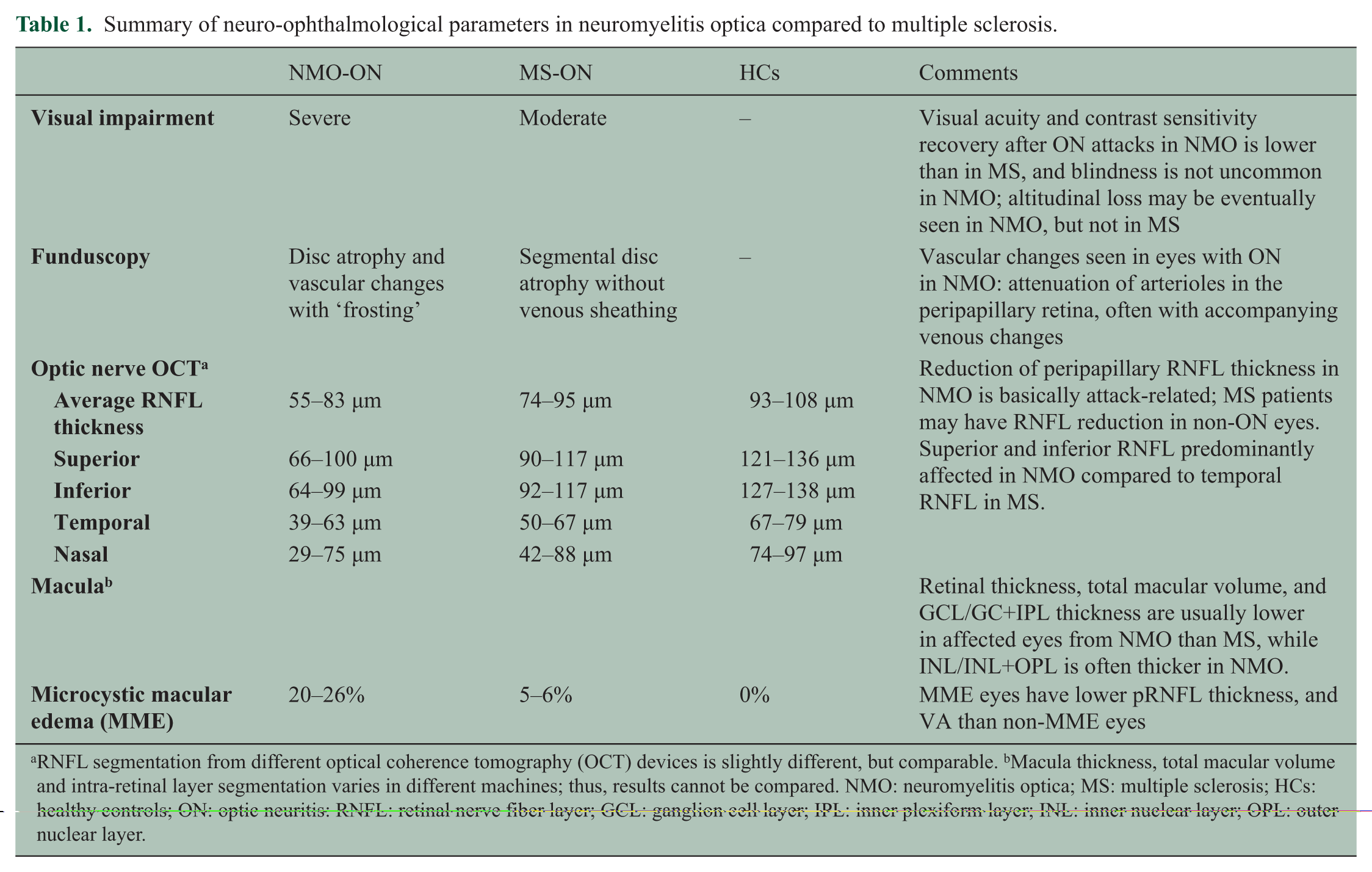
RNFL segmentation from different optical coherence tomography (OCT) devices is slightly different, but comparable. bMacula thickness, total macular volume and intra-retinal layer segmentation varies in different machines; thus, results cannot be compared. NMO: neuromyelitis optica; MS: multiple sclerosis; HCs: healthy controls; ON: optic neuritis: RNFL: retinal nerve fiber layer; GCL: ganglion cell layer; IPL: inner plexiform layer; INL: inner nuclear layer; OPL: outer nuclear layer.
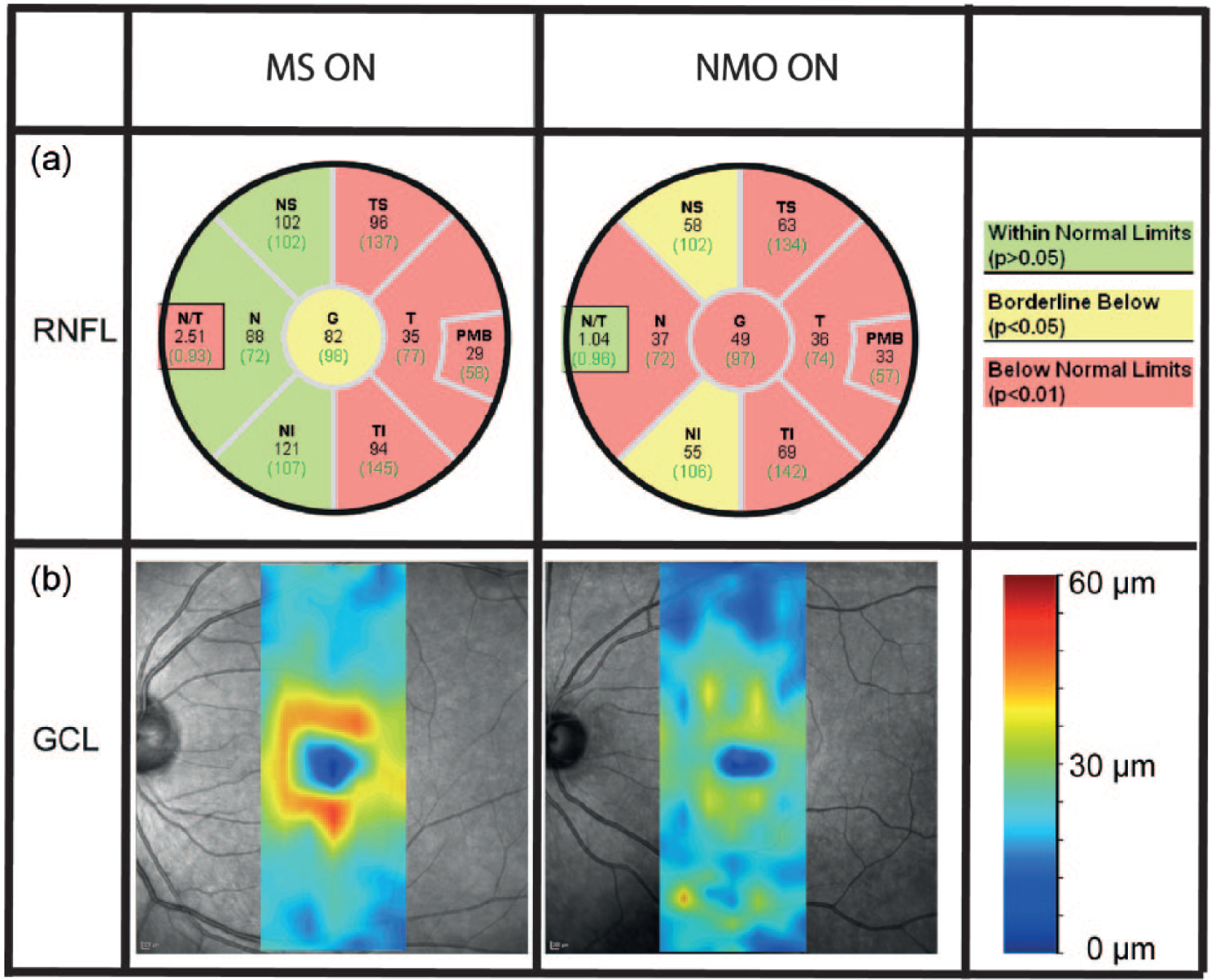
Typical differences in retinal damage between NMO-ON and MS-ON.
OCT measures and visual function
As in patients with MS, OCT in NMOSD correlated well with results from visual acuity testing.42,45 Using 1.25% low-contrast visual acuity charts in monocular testing, the average number of correct letters read by NMOSD patients with ON was four; MS-ON patients and healthy controls averaged 6.5 and 16, respectively. 42 The same study indicated that high-contrast acuity became very poor in eyes of NMOSD patients with ON when the average RNFL thickness fell below 60 µm. 42 More recent studies applying macular multilayer segmentation analyses have provided evidence that not only peripapillary RNFL but also neuronal layers like the ganglion cell layer (GCL) or the combined ganglion cell/inner plexiform layer (GCIP) are significantly thinned in patients with MS-ON and NMOSD-ON; the NMOSD patients showed greater thinning57,58 (Figure 2).
OCT measures in NMOSD eyes without ON
While in eyes of MS patients without a history of ON RNFL thickness is on average reduced by 7 µm, 36 several studies showed that NMOSD patients without a history of ON had normal RNFLs.42,46,48,50 This suggests that subclinical ON in NMOSD is uncommon and contrasts with both the subclinical visual-evoked potential (VEP) abnormalities and OCT abnormalities recognized in clinically isolated syndrome (CIS) patients with or without clinical ON. 19 Interestingly, another study reported that NMOSD eyes without a history of ON had prolonged P100 latencies on VEP compared to normal controls; however, the absolute latencies were still within the normal range. 59 Other groups have reported thinning of the combined GCIP in NMOSD eyes without a history of ON.55,56 This discrepancy may result from difficulties in accurately ascertaining clinical ON events in a retrospective approach, differences in OCT devices, segmentation techniques, and the smaller variance of the GCIP metric. Some differences in segmental retinal thinning has been observed in NMOSD-ON and MS-ON eyes, suggesting that SD-OCT may be useful in distinguishing NMO from MS following ON. 51 Additional investigations are needed, however, to confirm these preliminary observations.
Macular changes including microcystic macular edema (MME)
Some but not all OCT studies have also highlighted macular changes following MS-ON and RNFL changes following ON in NMOSD. These differences could reflect different pathogenic processes. 54 MME has recently been described in patients with MS and other optic neuropathies (Figure 1(d)). In MS patients, MME seems to be associated with higher levels of clinical disability 60 and disease activity as measured by the frequency of clinical relapses and MRI activity (T2- and gadolinium-enhancing T1 lesions). 61 Although the underlying mechanisms of MME are still a matter of debate, 62 clinical reports have shown that MME is not MS-ON specific but can also be detected in NMOSD-ON eyes and other optic neuropathies (chronic relapsing inflammatory optic neuropathy, ischemic, Leber disease, etc.). Microcystic inner nuclear layer abnormalities can be detected in 20%–26% of NMOSD patients,49,56,58 and in up to 40% of ON-affected eyes in AQP4-IgG-positive patients but not in unaffected eyes. 63 This is much higher than the 5% previously reported in MS patients.60,49
OCT measures and clinical disability
Similar to studies of MS-ON, visual function correlates well with RNFL thickness36,46–48 in NMOSD patients. However, in contrast to some studies of MS-ON,13,64,65 RNFL thickness in NMOSD has only sporadically correlated with the overall Expanded Disability Status Scale (EDSS). 46 Although this may not be surprising in light of the limited amount of visual function captured in the EDSS, the weaker correlation between RNFL thickness and EDSS in NMOSD as compared to MS may be the product of both distinct pathophysiology and involvement of a more limited spectrum of the CNS neuroaxis. The development of a validated disability scale specific to NMOSD will be important in determining whether OCT parameters may extend beyond the visual system to prove a clinical biomarker of disease activity.
ON and OCT in pediatric NMO
While most clinical characteristics of pediatric NMO are similar to adult-onset disease, a comprehensive clinical evaluation of NMOSD-ON in children has not been conducted. Approximately 50% of pediatric NMO cases have severe residual visual impairment in one or both eyes. 66 However, the vast majority of cases involve recurrent episodes of ON. Since MRI examinations and VEPs can be challenging in children because of a lack of patient cooperation, OCT may be a promising tool to differentiate pediatric NMO from other causes of ON. A longitudinal analysis of NMOSD-ON in the pediatric population with accompanying OCT analysis will be critical for understanding whether visual outcomes and corresponding neuro-anatomic measures of injury vary in the pediatric age group and powering clinical trials for acute and prophylactic therapies.
Association of OCT measures with brain volume
Animal models have demonstrated that RNFL thinning as measured by OCT reflects retinal axonal loss.67–69 Recent studies on OCT/MRI correlations in MS have shown good correlation between RNFL measures and both white and gray matter atrophy17,70 and brain parenchymal fraction. 71 Another recent study in a cohort of CIS and early RRMS patients with short disease duration (3.2 years) and mild levels of disability (median EDSS 1.5) indicated that in early stages of relapsing forms of the disease, OCT-derived retinal measures reflect white matter damage, with variability in gray matter being an age-related effect. 72 von Glehn et al. demonstrated cortical thinning in NMO patients (1.55 mm) compared to healthy controls (1.62 mm, p = 0.027) and a positive correlation between RNFL and cortical thickness. 73 When stratified by disease duration, RNFL and cortical thinning both progressed with time. Additional MRI analyses demonstrated global white and gray matter volume loss.
Effect of relapse treatment on OCT measures and visual function
Recent therapeutic studies found that relapse activity in NMO patients responded better to immunosuppressive drugs than to immunomodulators.74–77 Immunosuppressive drugs are known to have anti-inflammatory effects, but to date there are no data regarding their effect on neuronal and axonal loss. One study compared intravenous methylprednisolone (IVMP, 2 g per day for three to five days) to IVMP in conjunction with plasma exchange (PE, five consecutive exchanges) in individuals with a first attack of ON due to NMO or NMOSD. 78 In the PE group, 75% recovered to 20/40 or better. While the Snellen equivalent did not improve in the IVMP-only group (20/400 at initial and end of study exams), the visual acuity in the PE group improved from 20/400 at baseline to 20/50 at the final visit. While high-contrast visual acuity scores were better in the PE group, there were no statistical differences in RNFL thickness (mean thickness 63 µm in the IVMP group vs. 70 µm in the IVMP plus PE group, p = 0.16). A second study performed in patients who had failed to improve with high-dose corticosteroids also examined the use of PE. 79 In this small study, RNFL thickness was preserved at one year, with one patient followed longitudinally and demonstrating stable RNFL thickness over four years. Another small retrospective study from Japan suggested that early IVMP treatment after an acute ON event may help preserve RNFL thickness in NMO. 45 Eyes with RNFL thicknesses exceeding 71.41 µm had a significantly earlier treatment with IVMP than those eyes below this cutoff. Average RNFL thickness after an ON attack was inversely correlated with the period from onset of clinical symptoms to IVMP therapy. Thus, because of its sensitivity, OCT is a noninvasive tool that is ideally suited to provide information on potential neuroprotection in ON clinical trials.45,80
Value of OCT for differential diagnosis
In light of different treatment strategies for NMOSD and MS, an early and accurate diagnosis is key for optimal patient management but may remain challenging in cases of seronegative NMO. In this regard, OCT may be of potential value to help the clinician discriminate between MS-ON and NMOSD-ON, particularly when ON is the initial clinical presentation. Some studies have analyzed the ability of OCT measures to distinguish between MS and NMOSD. Naismith et al. reported that the odds of falling into the NMOSD group increased by 8% for every 1 µm decrease in RNFL thickness. 43 In a multilayer segmentation study, Park et al. found that ONL thickness greater than 83 µm at the inner temporal location (from the foveal center) and GCIP thickness of less than 62 µm at the outer superior location (from the foveal center) were suggestive of NMO. 57 Schneider et al. reported that both peripapillary RNFL thickness and the ratio of nasal to temporal peripapillary RNFL thickness may be helpful in distinguishing NMOSD-ON and MS-ON. 50 However, each of these findings has to be interpreted with caution given the low sample sizes and the exploratory nature of the analyses. Confirmatory studies using larger patient populations are needed before they can be used to guide clinical decision making.
Future prospects
Major advancements in the management of brain disease are likely to depend on the identification of imaging or molecular biomarkers. Such biomarkers are needed for an improved understanding of the pathogenesis, the stratification of patients based on the prognosis or response to therapy, and as surrogate endpoints in clinical trials. As such, one of the main initiatives from the National Institutes of Health (NIH) in Alzheimer’s disease is a multicenter study for validating biomarkers (MRI, positron-emission tomography (PET), beta-amyloid or Tau in cerebrospinal fluid (CSF)). For this reason, OCT for quantifying and monitoring axon damage of the optic nerve and the related retrograde degeneration of the GCL is likely to improve assessment of optic nerve tissue damage in NMO-ON. Although OCT will not capture non-optic nerve damage, its exquisite ability to quantify changes in the retina after optic nerve damage may allow prediction of visual recovery, general assessment of therapeutic efficacy as a surrogate of disability after inflammatory attacks or as a measure of neuroprotection or regeneration, and stratification of patients based on different patterns of damage.
Multimodal and comprehensive evaluation of the anterior visual pathway will provide even better understanding of NMOSD patients with ON. It may deliver clinically useful instruments for patient analysis. The good agreement between morphology captured by OCT, functional assessments such as multifocal visual-evoked potentials, and clinical outcomes such as visual fields, visual acuity and visual quality of life, may enable a comprehensive assessment of ON in patients with NMOSD. Moreover, multimodal assessments of patients provide the opportunity to integrate all factors participating in the disease, from molecular and cellular processes to visual system performance. New laser technologies are being developed that will allow molecular analysis of the retina changes, single-cell visualization of the RNFL and RGC, assessment of neural activity by imaging (which can be integrated with electroretinography), changes in blood flow and other retina fluids. NMO, an AQP4-astrocytopathy that commonly affects the optic nerves, is likely to benefit from these developments. For this reason, NMO can effectively indicate the potential of this new technology in assessing CNS damage and of neuroprotective or regenerative treatments.
Footnotes
Acknowledgements
The authors thank The Guthy-Jackson Charitable Foundation for its support in organizing the NMO International Clinical Consortium & Biorepository.
Conflict of interest
None declared.
a
Members of The Guthy-Jackson Charitable Foundation (GCJF) NMO International Clinical Consortium and Biorepository (ICC&BR) and the GJCF NMO Biorepository Oversight Committee (BOC) are recognized as affiliated authors of this study and listed below in alphabetic order by institution:
Funding
This work was supported by the German Ministry for Education and Research (Competence Network Multiple Sclerosis) (to FP).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
