Abstract
Introduction
Multiple sclerosis (MS) requires long-term treatment with immunomodulating or immunosuppressive agents. Therefore, it is important to assess potential risks that may be related to the continuous use of these therapies over the long term. 1 Agents currently licensed for the treatment of MS include the immunomodulators interferon (IFN) beta and glatiramer acetate, which are generally considered first-line therapies for relapsing forms of MS. By contrast, the currently available immunosuppressive agents, natalizumab and mitoxantrone, are usually used to treat patients with aggressive forms of MS. 2,3 The use of IFN beta in MS is supported by safety and efficacy data from large, randomized clinical trials. 4–7 However, none of these trials were statistically powered to assess rare adverse events such as malignancy.
The background risk of malignancy in patients with MS has been evaluated in population-based cohort studies. 8–16 Most of these studies indicate that the risk of any type of malignancy does not differ substantially between patients with MS and the general population. However, one Swedish study 12 found a slightly reduced overall risk of malignancy in patients with MS, particularly for lymphomas and digestive, respiratory, ovarian and prostate cancers relative to the general population. By contrast, a slightly increased risk was reported for other types of malignancy in patients with MS, such as for urinary tract and brain tumours. None of these findings were statistically significant. In addition, it was not possible to determine whether the differing risks for malignancy between patients with MS and the general population reported in the Swedish study were due to disease-related factors or behavioural differences between the two groups. Also, the elevated risk of brain tumours may possibly have been due to surveillance bias resulting from frequent monitoring of patients with MS using magnetic resonance imaging (MRI).
Two published studies have assessed the risk of malignancy in patients with MS receiving immunomodulatory drugs. 17,18 The first study 17 compared the risk in patients with MS in Israel with that in the general Israeli population and additionally analysed the relative risk of malignancy associated with treatment versus non-treatment using a time-dependent Cox model. It reported no significantly elevated risks of malignancy in patients treated with immunomodulatory drugs. However, this study did not precisely define the patient population or how the MS clinic population may have differed from the general Israeli population, making the elimination of possible confounding factors difficult. Further, it did not report whether data from deceased patients were included in the analysis, or comprehensively define the duration of follow-up. The second study, 18 which compared the incidence of cancer in patients with MS in France with the general French population, also concluded that there was no heightened malignancy risk associated with immunomodulatory therapy. However, it is possible that this study may have been compromised by incomplete registration of cancer cases in the clinical database used, which could have led to inaccuracy in the estimated incidence of malignancy. Further, the analysis did not adjust for confounding risk factors.
The assessment of the risk of malignancy using clinical trial data is limited by the relatively small sample sizes, short duration of follow-up and restricted study population. Spontaneously reported adverse events in the post-marketing setting present a greater dataset obtained from real-life clinical practice; however, their assessment is also limited – the true incidence of malignancy cannot be calculated due to potential under-reporting and uncertainty of the denominator. In an attempt to overcome the limitations of each of these approaches, the present analysis aimed to evaluate the risk of malignancy in patients with MS receiving subcutaneous (sc) IFN beta-1a treatment by assessing both pooled data from key clinical trials and data from the Merck Serono Global Drug Safety database.
Methods
The data sources for the two analyses are shown in Figure 1. Data on serious adverse events recorded in the 12 key clinical trials are also captured in the Global Drug Safety database, and were therefore included in both analyses. However, the number of such cases was relatively small. Other subsets of data were analysed separately as shown.
Datasets used for the analyses of risk of malignancy in patients with multiple sclerosis receiving sc IFN beta-1a treatment – pooled data from the 12 key clinical trials and safety data from the GDS database. AE, adverse event; GDS, global drug safety; ICSR, individual case study report; SAE, serious adverse event.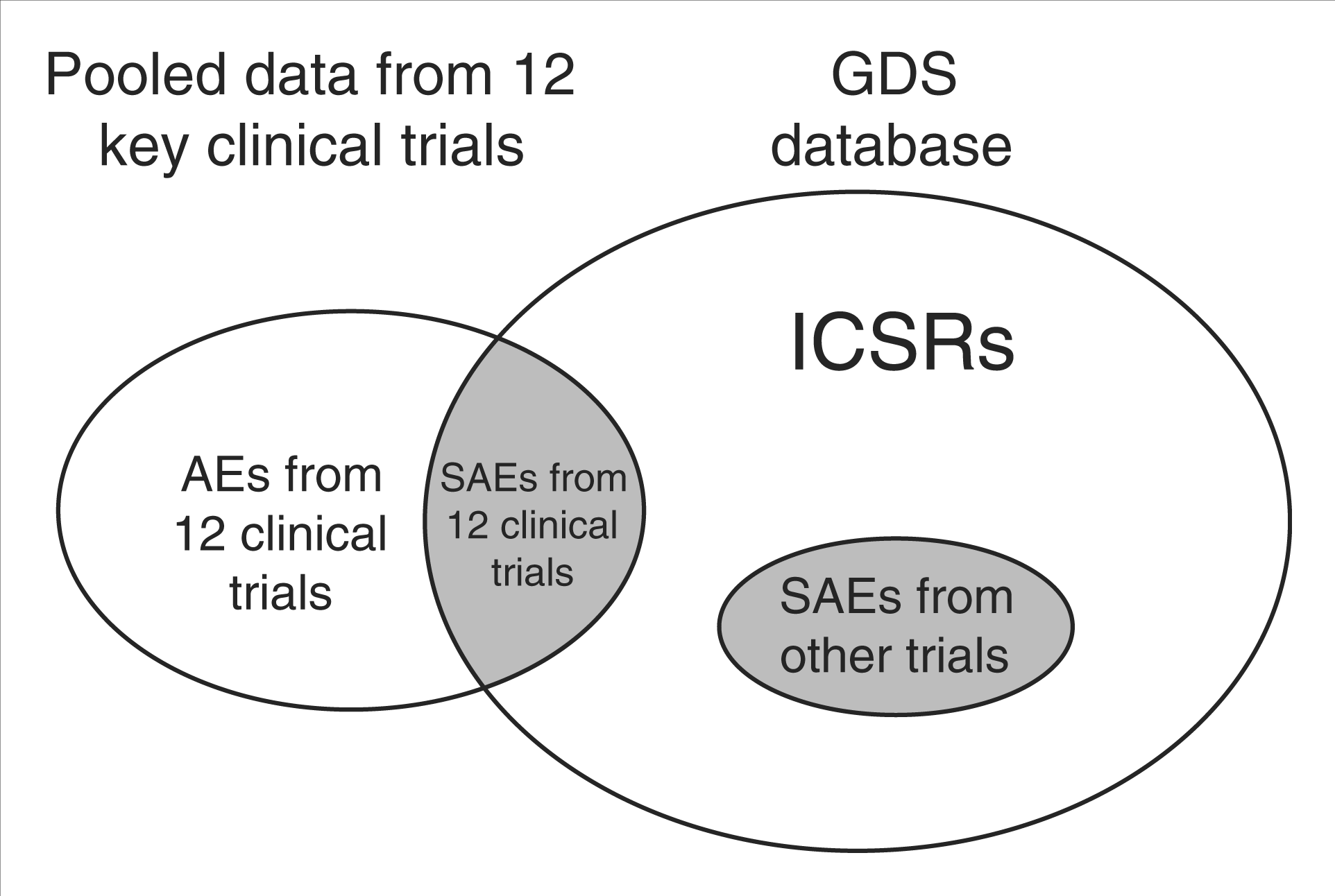
For both data sources, data were retrieved using the Medical Dictionary for Regulatory Activities (MedDRA) standard query 20000090 for “malignancies”. This query comprises four sub-queries, covering more than 1500 preferred terms relating to different aspects of the evaluation of malignancy. The data are coded in MedDRA version 12.1.
Data sets analysed
Pooled data from key clinical trials of sc IFN beta-1a
Key clinical trials included in the pooled analysis

ETOMS, Early Treatment Of Multiple Sclerosis; EVIDENCE, EVidence of Interferon Dose-response: European North American Comparative Efficacy; IFN, interferon; im, intramuscular; LTFU, long-term follow-up; OWIMS, Once Weekly Interferon for Multiple Sclerosis; PRISMS, Prevention of Relapses and disability by Interferon beta-1a Subcutaneously in Multiple Sclerosis; qd, daily; qw, weekly; REGARD, Rebif® versus Glatiramer Acetate in Relapsing Multiple Sclerosis Disease; sc, subcutaneous; SPECTRIMS, Secondary Progressive Efficacy Clinical Trial of Recombinant Interferon beta-1a in Multiple Sclerosis; SPMS, secondary progressive multiple sclerosis; tiw, three times weekly.
Analysis cohorts for pooled clinical trial data
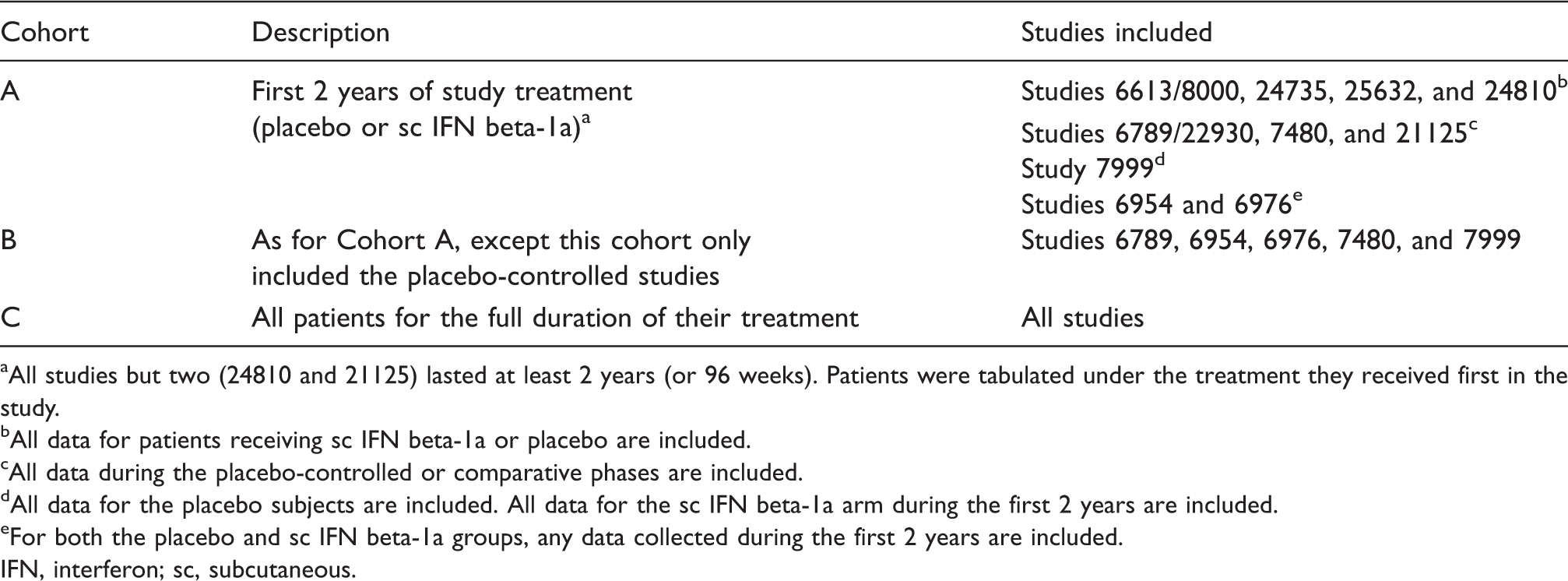
All studies but two (24810 and 21125) lasted at least 2 years (or 96 weeks). Patients were tabulated under the treatment they received first in the study.
All data for patients receiving sc IFN beta-1a or placebo are included.
All data during the placebo-controlled or comparative phases are included.
All data for the placebo subjects are included. All data for the sc IFN beta-1a arm during the first 2 years are included.
For both the placebo and sc IFN beta-1a groups, any data collected during the first 2 years are included.
IFN, interferon; sc, subcutaneous.
Incidence rates of malignancies per 1000 patient-years were calculated for each treatment group in each cohort. The design of some of the trials included in Cohort C allowed patients to switch from one treatment arm to another. In such cases, the patient was counted twice – once for treatment, once for placebo. For calculations of treatment exposure in these specific cases, the time the patient spent in each treatment arm was considered separately. Any events were linked to the treatment the patient was receiving at the time of the event onset, and calculations of overall incidence rates took into account individual patient exposure.
Data from the Global Drug Safety database
Subcutaneous IFN beta-1a (Rebif®) is manufactured and marketed by Merck Serono S.A. – Geneva, Switzerland (a division of Merck KGaA, Darmstadt, Germany). In compliance with international regulations, all individual case safety reports (ICSRs) received in the post-marketing setting are recorded in the Global Drug Safety database. In addition, the database records all ICSRs from clinical trials that have been classified as serious events. It should be noted that only 1.7% of all events reported in the database originate from clinical trials; the remaining 98.3% relate to the spontaneous reporting in the post-marketing setting.
The ICSRs included in this analysis were received between 1 February 1998 and 3 November 2009. These ICSRs comprised information gathered from healthcare professionals, health authorities, patients, published case reports and clinical trial reports. The ICSRs included in this analysis were limited to those from patients who had been exposed to sc IFN beta-1a (at any dose). Although the database gathers data on patients receiving placebo in clinical trials, reports from these patients are excluded from the Global Drug Safety analyses. Selected ICSRs were categorized according to their origin as follows: reports were classified as ‘medically confirmed’ (MC) if the ICSR was received from a healthcare professional, health authority, literature case report or clinical trial; or ‘not medically confirmed’ (NMC) if the ICSR was received from a patient or relative, either spontaneously or when solicited, i.e. provided in response to a call out to patients from the company support network. When a patient or relative reported a serious event, medical confirmation was then sought from the treating physician. However, if upon follow-up the physician considered the event as unrelated to sc IFN beta-1a, the ICSR remained categorized as NMC.
Retrieved ICSRs were grouped by the body system or location of the malignancy. They were then counted to obtain the number of reported (observed) cases. Since the reporting rate was the endpoint of the analysis, if a patient presented with two or more malignancies that had no obvious pathophysiological link between them, each malignancy was counted as a separate event in the appropriate category. For example, for one patient diagnosed with Hodgkin’s lymphoma and lung cancer, two malignancy events were counted.
The exposure of patients to sc IFN beta-1a in the post-marketing setting was estimated according to sales volume; however, this estimation was based on several assumptions and thus cannot provide a precise denominator. In addition, spontaneous reporting may under-report the incidence of adverse events. Therefore the analyses presented refer to the reporting rate rather than incidence. For each malignancy type examined as part of the Global Drug Safety database analysis, the number of reported cases was compared with the number of expected cases in the general population. The expected figures for all malignancy types except leukaemia subtypes were based on the incidence rates in ‘more developed regions’ as defined and reported in the 2002 GLOBOCAN report. 19 The GLOBOCAN subdivision ‘more developed regions’ was selected as it was considered to best represent a suitable comparator population, representative of regions where patients with MS may be treated with sc IFN beta-1a. Since GLOBOCAN does not provide detailed statistics for leukaemia subtypes, for cases of acute and chronic myeloid leukaemia and acute and chronic lymphocytic leukaemia, expected numbers were based on the 2002–2006 USA Surveillance Epidemiology and End Results (SEER) Cancer Statistics review. 20
The majority (88%) of the NMC malignancies identified in the Global Drug Safety database originated from the USA, where a support programme is available to all patients starting or continuing sc IFN beta-1a. All patients who participate in the programme are contacted regularly by certified MS nurses, either by telephone or in person (depending on individual patient need), and if an adverse event is reported, it is captured and incorporated into the Global Drug Safety Database. Most reports of NMC cases were thus considered to be ‘solicited’. For this reason, the expected figures for comparison with NMC cases were based on general population data for the USA only.
The reported to expected (R:E) ratio, with 95% confidence intervals (CIs), for each type of malignancy was calculated using the methods for standardized morbidity ratio calculations. 21 Benign tumours were excluded from the analysis. Non-melanoma skin cancers were also excluded from the analysis, as these cancers are incompletely reported and thus incidence figures are not available from cancer registries. Cases in which sc IFN beta-1a had been used in an oncology indication in clinical trials were also excluded to avoid confounding the analysis by indication bias.
Results
Pooled data from key clinical trials of sc IFN beta-1a
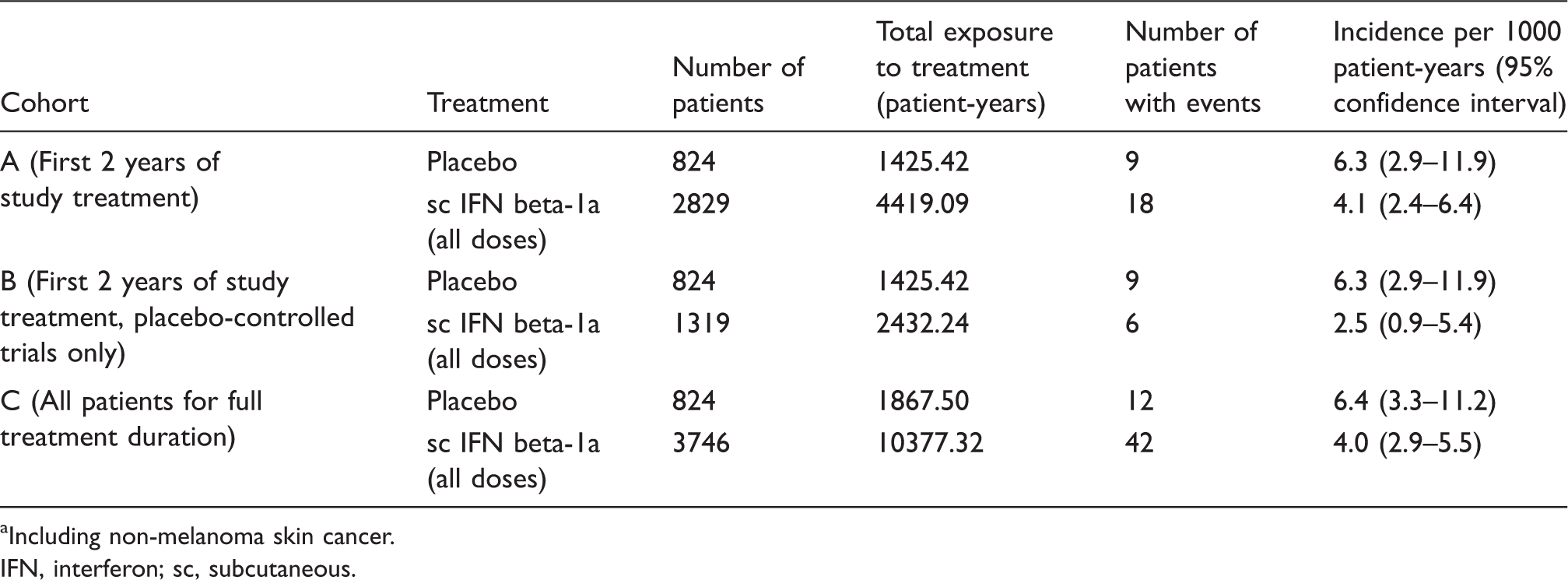
Data for a total of 3746 patients treated with sc IFN beta-1a were included in this analysis. The mean (range) patient exposure to sc IFN beta-1a was 29.6 (0.0–96.0) months; 1096 patients had more than 3 years of exposure and 568 had more than 5 years. Furthermore, the mean (range) duration per patient of safety follow-up was 33.5 (0.0–98.0) months, with 1257 and 737 patients having more than 3 and 5 years’ safety follow-up, respectively. Analysis of data from the first 2 years of placebo-controlled studies (Cohort B) revealed that there was a lower incidence of malignancies per 1000 patient-years in patients who had received sc IFN beta-1a (2.5; 95% CI: 0.9–5.4) compared with those who had received placebo (6.3; 95% CI: 2.9–11.9) (Table 3), although this was not statistically significant. This trend for lower malignancy incidence in patients treated with sc IFN beta-1a was also observed for Cohorts A and C, although the difference was smaller in these two cohorts than in Cohort B (Table 3). Figure 2 shows the incidence per 1000 patient-years over time, which indicates that the incidence of malignancies did not increase with the length of follow-up.
Analysis of pooled clinical trial data: incidence of malignancy over time in patients treated with subcutaneous interferon beta-1a. Incidence of neoplasms
a
in clinical trials of sc IFN beta-1a Including non-melanoma skin cancer. IFN, interferon; sc, subcutaneous.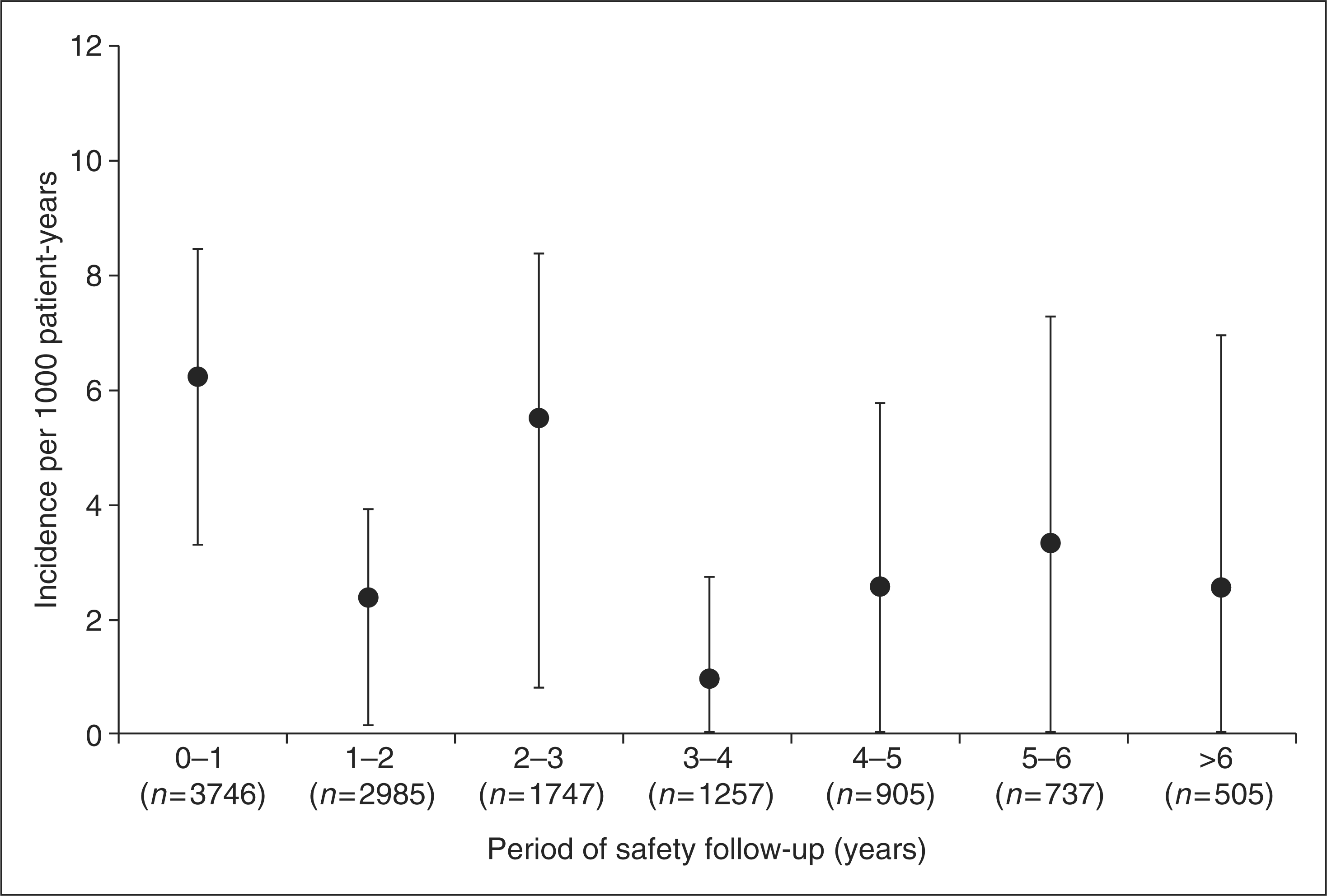
Data from the global drug safety database in the post-marketing setting
As of 3 November 2009, the cumulative patient exposure to sc IFN beta-1a was estimated at 720,123 patient-years, based on sales volume. The exposure to sc IFN beta-1a in clinical trials is negligible in comparison and therefore was not taken into account.
A total of 741 ICSRs reporting malignant events were retrieved from the Global Drug Safety database. The following reports were excluded from the analysis: two non-cancer cases (erroneously identified by the database query); 13 benign nodules; 53 non-serious neoplasms; 35 cases of non-melanoma skin cancer and 11 cases in which sc IFN beta-1a had been used in clinical trials for an oncology indication. Some 27 patients experienced two types of malignancies and their reports were therefore counted twice as explained above.
Analysis of the Global Drug Safety database – medically confirmed malignancies: reported versus expected cases
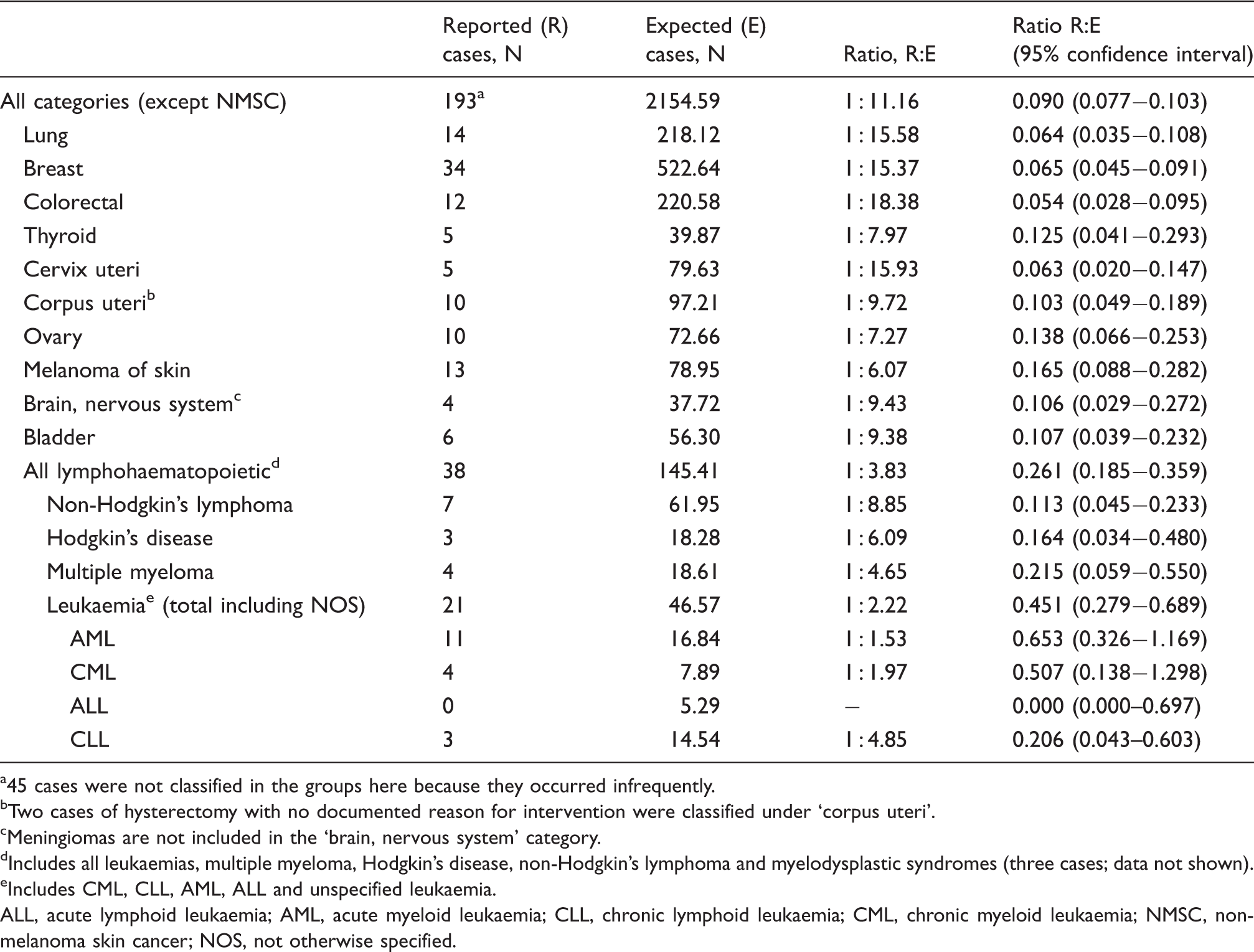
45 cases were not classified in the groups here because they occurred infrequently.
Two cases of hysterectomy with no documented reason for intervention were classified under ‘corpus uteri’.
Meningiomas are not included in the ‘brain, nervous system’ category.
Includes all leukaemias, multiple myeloma, Hodgkin’s disease, non-Hodgkin’s lymphoma and myelodysplastic syndromes (three cases; data not shown).
Includes CML, CLL, AML, ALL and unspecified leukaemia.
ALL, acute lymphoid leukaemia; AML, acute myeloid leukaemia; CLL, chronic lymphoid leukaemia; CML, chronic myeloid leukaemia; NMSC, non-melanoma skin cancer; NOS, not otherwise specified.
Analysis of the Global Drug Safety database – serious, non-medically confirmed malignancies in the USA population: reported versus expected cases
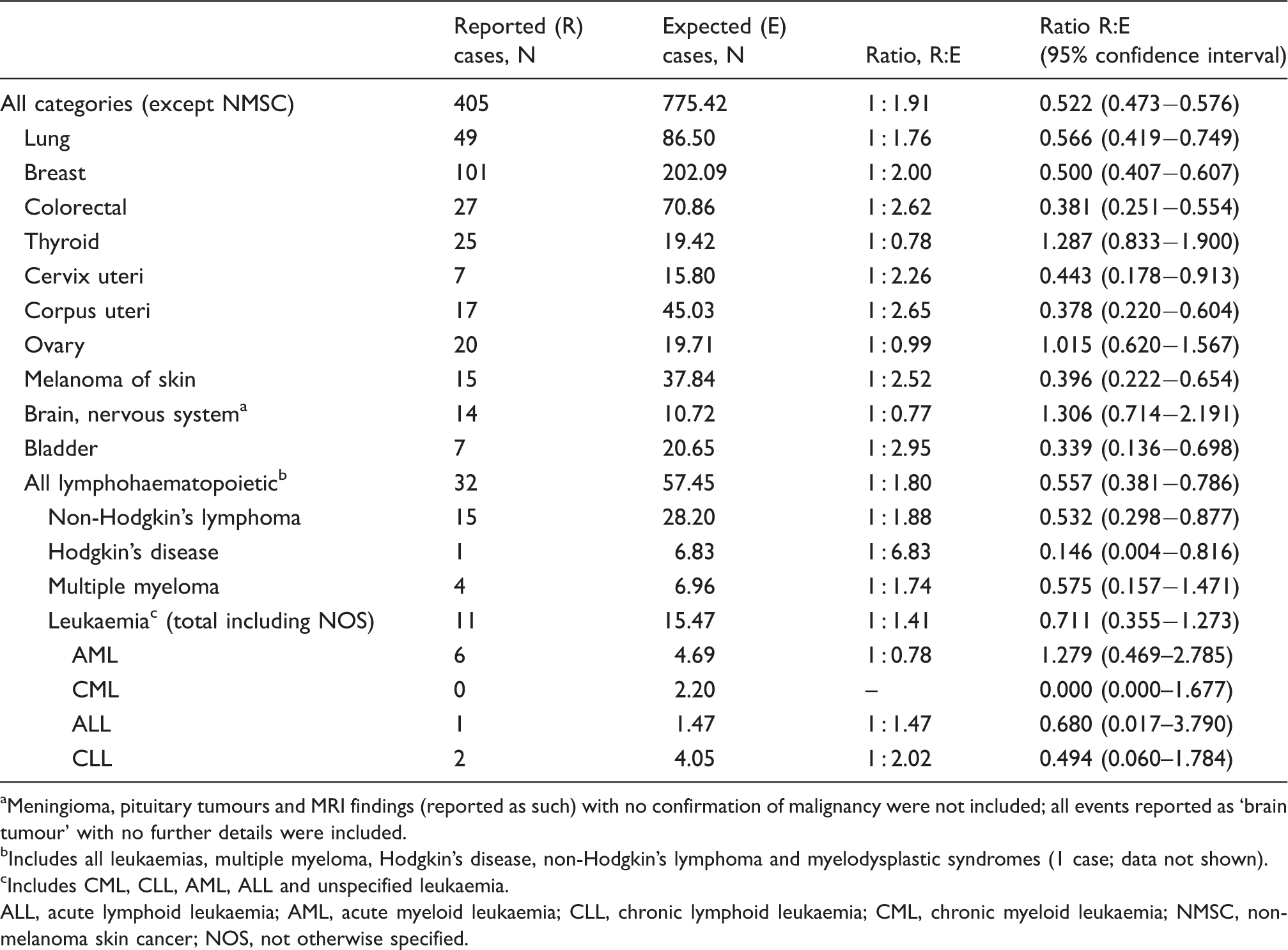
Meningioma, pituitary tumours and MRI findings (reported as such) with no confirmation of malignancy were not included; all events reported as ‘brain tumour’ with no further details were included.
Includes all leukaemias, multiple myeloma, Hodgkin’s disease, non-Hodgkin’s lymphoma and myelodysplastic syndromes (1 case; data not shown).
Includes CML, CLL, AML, ALL and unspecified leukaemia.
ALL, acute lymphoid leukaemia; AML, acute myeloid leukaemia; CLL, chronic lymphoid leukaemia; CML, chronic myeloid leukaemia; NMSC, non-melanoma skin cancer; NOS, not otherwise specified.
In all MC malignancy categories and most of the NMC malignancy categories the reported numbers were lower than the expected numbers. As shown in Table 5, the R:E ratios were around 1 for NMC reports of brain, thyroid and lymphohaematopoietic malignancies. These three types of malignancies are discussed in detail below.
Central nervous system malignancies
In total, 18 events were reported in the category ‘brain and nervous system’, excluding one non-USA NMC case (Tables 4 and 5). All events were located in the brain. The R:E ratio was 1 : 9.38 for MC and 1 : 0.77 for NMC cases. Most of these cases (15/18; 83%) occurred within 2 years of sc IFN beta-1a treatment initiation. Only two cases were reported after a treatment period longer than 2 years; one additional case had an unknown latency period. In four of 18 cases, brain tumours had already been present before initiation of sc IFN beta-1a therapy. Four further cases were associated with potential confounding factors. These were identified as: ‘benign brain lesion prior to sc IFN beta-1a treatment’; ‘unspecified brain lesion prior to sc IFN beta-1a treatment’; ‘human immunodeficiency virus (HIV)’ and ‘history of melanoma’.
Thyroid malignancies
A total of 30 cases of thyroid cancer were reported, excluding one non-USA NMC case (Tables 4 and 5). The R:E ratio was 1 : 7.97 for MC cases and 1 : 0.78 for NMC cases. Of these malignancies, only 4/30 (13.3%) occurred after more than 2 years’ exposure to sc IFN beta-1a.
Lymphohaematopoietic malignancies
A total of 74 lymphohaematopoietic malignancies were reported. The R:E ratio was 1 : 3.83 for MC cases and 1 : 1.80 for NMC cases. More than half (43/74; 58.1%) of these malignancies were reported less than 2 years after the start of sc IFN beta-1a therapy. A total of 20 patients (27%) had potential confounding factors, including previous treatment with agents carrying an increased risk of haematopoietic malignancy, such as azathioprine or mitoxantrone. 22
Discussion
Results from these analyses do not suggest an association between sc IFN beta-1a treatment and occurrence of malignancy in patients with MS. However, while reported rates were lower than expected rates, R:E ratios in the brain, thyroid and lymphohaematopoietic categories were higher than those in other categories. Therefore risks relating to these types of malignancies warrant further examination.
It should be noted that there is potential for surveillance bias regarding brain malignancies in patients with MS, as these patients undergo regular MRI scans for the assessment of MS disease progression. As a result, tumours are more likely to be detected in patients with MS than in the general population. In addition, for almost half of brain malignancy events assessed in this analysis (8/18; 44%), relevant confounding factors, such as pre-existing tumours and HIV were known to be present.
The comparatively high number of thyroid cancer cases reported in this analysis could also be explained by potential surveillance bias. Patients treated with sc IFN beta-1a are closely monitored for thyroid disorders, in accordance with treatment guidelines. Some 20% of treated patients presented with thyroid nodules before treatment onset and thus may have been monitored even more closely by their healthcare professionals. Some 84% of the thyroid malignancies were reported in female patients, which is in line with the sex distribution of thyroid cancer (more common in females than males with a ratio of 3 : 1), 23 and of MS (70% of patients are females). Lastly, at the time of this analysis the most recent records to estimate the expected number of cases that were available dated from 2002, while most reported cases occurred between 2005 and 2009. As the incidence of thyroid cancer is increasing over time (according to the USA cancer registries), this may have also have led to an over-estimation of the R:E ratio for thyroid neoplasms. 24
The R:E ratio for solid tumours was much lower for MC events compared with NMC events. However, the ratios are comparable between MC and NMC events for lymphohaematopoietic malignancies. One possible explanation for this difference is that healthcare professionals may monitor patients more closely for haematopoietic neoplasms due to a perceived link between these types of malignancies and the pharmacological mechanisms of immunomodulatory therapies. Haematological tests are performed regularly on treated patients with MS in order to monitor blood cell counts, which may also increase the likelihood of early detection of haematopoietic neoplasms, relative to the general population. In addition, in most of the MC cases of acute myeloid leukaemia, the patients had previously been treated with immunosuppressive therapy. Chronic use of such therapies may be associated with a heightened long-term risk of malignancy. 25 It is known that other agents used in the treatment of cancer carry a risk of secondary, therapy-related leukaemias, which increases by up to 1% per year from 2 years after treatment initiation. 26 Most brain, thyroid and lymphohaematopoietic malignancies in our analysis were reported within 2 years of initiation of sc IFN beta-1a treatment. Therefore a causal relationship between these malignancies and IFN beta-1a treatment was deemed unlikely.
The analyses reported here are subject to limitations. When comparing the number of malignancies reported in patients exposed to sc IFN beta-1a with those expected in the general population, it should be noted that adverse events may be under-reported in the post-marketing setting. In general, the under-reporting of drug-associated adverse events to market authorization holders has been estimated to be 94% for all events and between 80–85% for serious events only. 27 In the case of the drug safety database analysis, the true incidence of malignancies cannot be calculated from spontaneously reported events due to uncertainty of the size of both the numerator and denominator. However, due to the comprehensive patient support programme for patients receiving sc IFN beta-1a in the USA, the reported figures in these analyses are likely to be close to the actual occurrences of malignancies. Hence, these data are considered to be highly relevant, although medical details for individual cases may be incomplete.
Analysis of pooled clinical trial data is also limited by the inclusion of short-term clinical trials with relatively small sample size, and by the short duration of the placebo-controlled phases of some of the studies. It is recognized that malignancies can occur after long latency periods. It should be noted that in the analysis of pooled clinical trial data, the rate of incidence of malignancies did not increase during 6 or more years of follow-up. Indeed, the upper limit of CI for incidence rate is lower at 6 years and beyond than that at 0–1 years. However, the trial data analysis did not include assessment of the risk of malignancy following discontinuation of treatment.
In conclusion, these analyses evaluated the risk of malignancy in patients with MS treated with sc IFN beta-1a during the clinical development programme, and in the post-marketing setting. Overall, the data do not indicate any increased risk of malignancy in these patients. These findings confirm and extend those of previous studies examining sc IFN beta-1a in patients with MS. However, while there was no clear association between sc IFN beta-1a therapy and brain, thyroid or lymphohaematopoietic malignancies, these topics remain an area for close pharmacovigilance surveillance.
Footnotes
Acknowledgements
The authors thank Steve Smith of Caudex Medical, Oxford, UK (supported by Merck Serono S.A. – Geneva, Switzerland, an affiliate of Merck KGaA, Darmstadt, Germany) for assistance in preparing the initial draft of the manuscript, collating the comments of authors and other named contributors, and assembling tables and figures; and Joanne Tang (Caudex Medical, supported by Merck Serono S.A. – Geneva, Switzerland), for assistance in editing for English, formatting manuscript to meet journal guidelines, and coordinating submission requirements.
Funding
This work was supported by Merck Serono S.A. – Geneva, Switzerland (an affiliate of Merck KGaA, Darmstadt, Germany).
Conflicts of interests
MS-W has received honoraria from Serono Symposia International Foundation and sanofi-aventis (lectures); from Merck Serono (lectures, work in data safety monitoring boards DSMB); from Genentech (DSMB); from Elan (advisory board); from the Swedish bank SEB Enskilda (lectures); from Bayer Health Care (contribution of articles to Swedish health website). She has also received honoraria for serving on the board of directors of Active Biotech in Lund, Sweden, and as external reviewer of a PhD thesis at the University of Copenhagen.
GK, DB, MSM, BH and EA are employees of Merck Serono S.A. – Geneva, Switzerland, an affiliate of Merck KGaA, Darmstadt, Germany.
