Abstract
Introduction
Multiple sclerosis (MS) is twice as prevalent in females as in males, and has a typical onset between the ages of 20 and 40 years. 1 Accordingly, MS most commonly affects women of childbearing age. Interferon (IFN) beta is currently the most widely used therapy for MS. 2–4 There is no evidence that MS itself increases the risk of spontaneous abortion or congenital defects, 5 and studies that have examined the effects of IFN-beta therapy on pregnancy have so far yielded inconclusive or conflicting findings. 6–13 In primates, human IFN beta is associated with abortifacient, but not teratogenic, effects. 14
In the absence of conclusive data addressing this issue, women with MS are currently advised to take appropriate contraceptive measures while receiving IFN-beta treatment and to discontinue therapy when trying to conceive. 15 Similar warnings are in place for other immunomodulatory and immunosuppressive therapies for MS. 16,17 Nevertheless, unexpected or unplanned pregnancies do occur, and the risks related to exposure to IFN beta during pregnancy remain unclear.
We aimed to further investigate the effects of IFN beta on pregnancy by evaluating exposure and pregnancy outcomes among women with MS who were receiving subcutaneous (SC) IFN beta-1a treatment before or during their pregnancy, using data from a global drug safety database that included prospective and retrospective post-marketing surveillance data and cases from controlled clinical trials.
Methods
Global drug safety database and database search
All individual case safety reports for SC IFN beta-1a administered 44 µg or 22 µg three times weekly (Rebif®; Merck Serono S.A. – Geneva, Switzerland, an affiliate of Merck KGaA, Darmstadt, Germany), which were received in the post-marketing setting, as well as serious case reports from clinical trials with SC IFN beta-1a, are recorded in the Merck Serono global drug safety database.
Data on all pregnancy cases were retrieved by searching the database for verbatim terms, including ‘utero’ or ‘pregnan’, and by using the Medical Dictionary for Regulatory Activities (MedDRA) System Organ Class 10010331 (congenital, familial and genetic disorders) and 10036585 (pregnancy, puerperium and perinatal conditions).
All post-marketing surveillance data (n = 984) received between 1 February 1998 (the date of first market authorization for SC IFN beta-1a) and 3 November 2009, and case reports (n = 38) from manufacturer-controlled clinical trials between 1 September 2004 and 3 November 2009 were included in this analysis. Details of included clinical trials in which a pregnancy occurred are documented in the online Supplementary Table. Reports were received from healthcare professionals, health authorities and patients, and from published case reports. Pregnancy cases were categorized according to status as follows: ‘medically confirmed’ if the case was received from a healthcare professional, health authority, literature case report or clinical trial; and ‘not medically confirmed’ if it was received from a patient or relative, either spontaneously or when solicited (i.e. provided in response to a call-out to patients from the manufacturer support network). Reports included patients exposed to any dose of SC IFN beta-1a.
Time of foetal exposure to SC IFN beta-1a
The duration (days) of exposure to SC IFN beta-1a during pregnancy before therapy was discontinued was determined for all pregnancies for which the last menstrual period (LMP) and IFN beta-1a stop date (at least month and year) were available (n = 231, both prospective and retrospective cases). In utero exposure was calculated from the date of conception (day 0), which was estimated to be 15 days after the start of the LMP. Pregnancy cases where IFN beta-1a was discontinued before conception were not included in the calculation of exposure. Cases where the specific day of LMP and/or SC IFN beta-1a discontinuation was not recorded (n = 84) were arbitrarily assigned as the 15th day of the month reported.
Definitions of prospective/retrospective data for pregnancy outcomes
Classifications used to define pregnancy outcomes were based on European Medicines Agency (EMA) guidelines. 18 ‘Prospective’ data were defined as those acquired prior to either knowledge of the pregnancy outcome or the detection of a congenital malformation at prenatal examination (e.g. foetal ultrasound, serum markers). Conversely, data were classified as ‘retrospective’ if they were acquired only after the outcome of the pregnancy was known or after the detection of a congenital malformation on a prenatal test.
Pregnancies were categorized into one of the following outcomes: live birth (with or without congenital anomaly), spontaneous abortion (foetal defects/no foetal defects or unknown), elective termination (foetal defects/no foetal defects or unknown), stillbirth (foetal defects/no foetal defects or unknown), ectopic pregnancy. A congenital anomaly was classified as ‘major’ if it was either a life-threatening structural anomaly or one likely to cause significant impairment of health or functional capacity, and which would require medical or surgical treatment. Spontaneous abortion (comprising spontaneous abortion, miscarriage, missed abortion, incomplete abortion, or early foetal death) was defined as a pregnancy that ended spontaneously before 22 completed weeks of gestation (<24 weeks from the LMP). Stillbirth was defined as late foetal death (in utero, or during labour or delivery), ≥22 completed weeks of gestation (≥24 weeks from LMP). An elective termination with foetal defects was defined as a termination that had been medically induced because of a foetal defect. An elective termination without foetal defects was defined as a termination that had been medically induced for a reason other than a foetal defect (e.g. personal reason, medical condition in the mother). Miscarriage rates in the general population were derived from EMA guidelines. 18
Results
Number of individual case safety reports for pregnancy, by confirmation status, data collection type, and presence of outcome
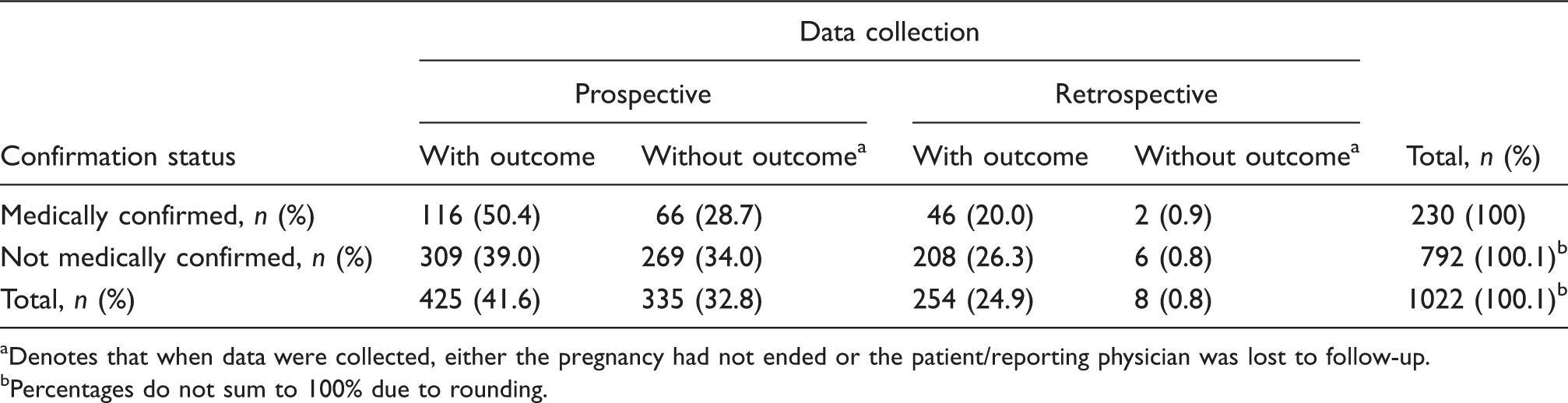
Denotes that when data were collected, either the pregnancy had not ended or the patient/reporting physician was lost to follow-up.
Percentages do not sum to 100% due to rounding.
Time of foetal exposure to SC IFN beta-1a
For the exposed pregnancies for which the date of LMP and SC IFN beta-1a stop date were available (n = 231, both prospective [n = 187] and retrospective [n = 44] cases), the mean (standard deviation) time of foetal exposure to SC IFN beta-1a was 28 (22) days (range 1 to 181 days) (Figure 1). In the vast majority of cases, patients discontinued treatment as soon as they discovered that they were pregnant and only seven patients remained on treatment beyond the first trimester (Figure 1). Most (86.1% [199/231]) foetuses were exposed for ≤45 days.
Length of foetal exposure to interferon (IFN) beta-1a, from time of conception (day 0) to treatment discontinuation in prospectively and retrospectively collected pregnancies. Pregnancy cases where IFN beta-1a was discontinued before conception were excluded from the calculation of exposure.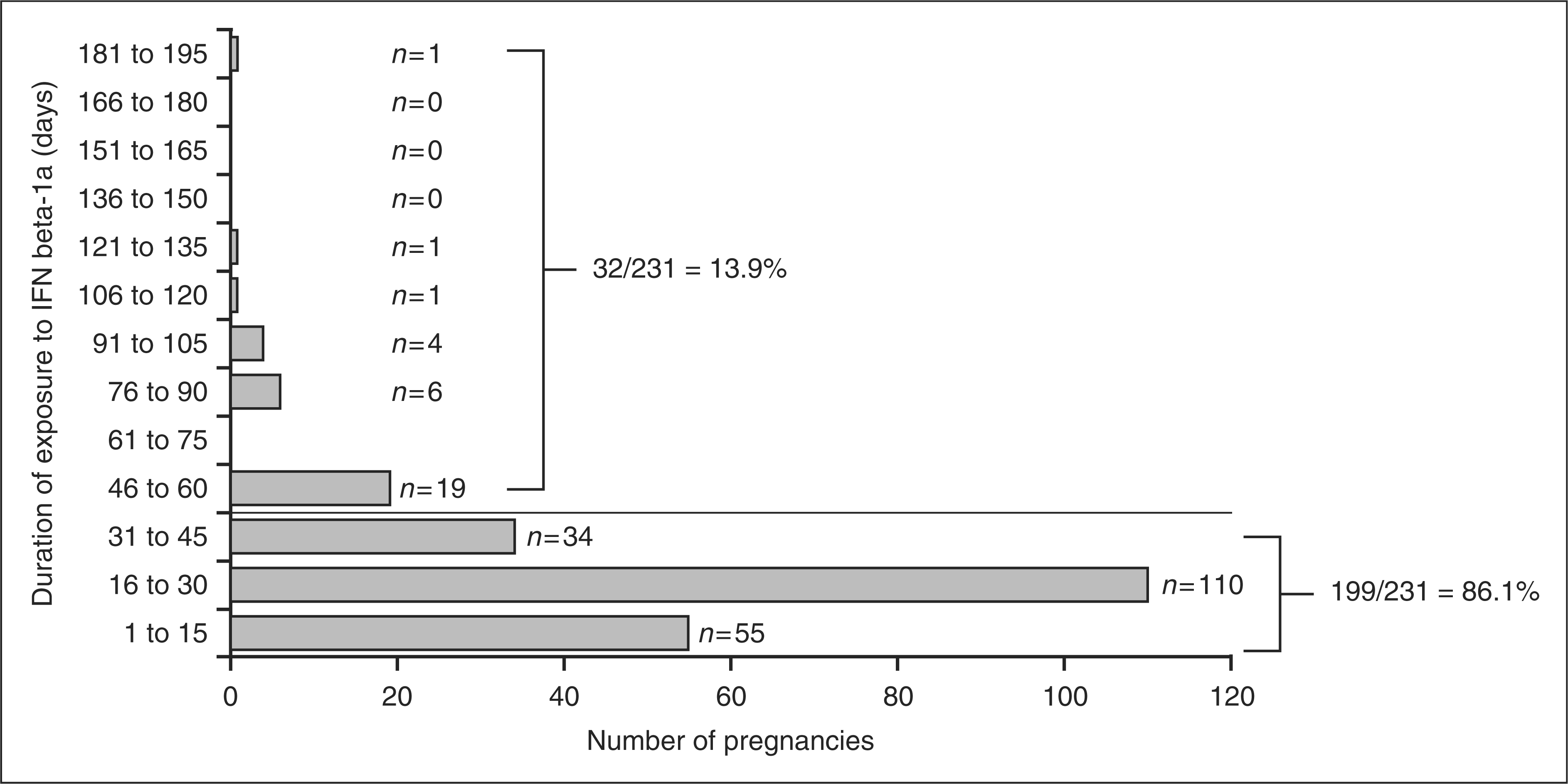
Pregnancy outcomes
Retrospectively reported pregnancies exposed to SC IFN beta-1a were approximately three times more frequently associated with an adverse outcome than prospectively reported pregnancies (Figure 2). Adverse outcomes included spontaneous abortion (with or without foetal defects), all other outcomes with foetal defects (for non-live births) and congenital anomalies (for live births), and ectopic pregnancy. Conversely, prospectively reported pregnancies exposed to SC IFN beta-1a (n = 425) were 1.5 times more likely to have an outcome of live birth without congenital anomaly (76.2% [324/425]) than the group with retrospectively reported data (49.6% [126/254]) (Figure 2).
Number of individual case reports for all pregnancies documented by outcome in (a) prospectively collected pregnancies, and (b) retrospectively collected pregnancies. Multiple pregnancies, e.g. triplet pregnancy, counted only once as one pregnancy outcome. The following outcomes from individual case safety reports were discarded: ‘Not reported’ (pregnancy was still ongoing) and ‘Unknown’ (patient was lost to follow-up). a‘Unknown’ indicates no information was available on whether a foetal defect was noticed. bThree of four were major anomalies (classified as life-threatening structural anomaly or one likely to cause significant impairment of health or functional capacity, and that required medical/surgical treatment): VACTERL syndrome, solitary kidney, tetralogy of Fallot. cOne baby died shortly after birth (lived for 20 minutes). dAll were single occurrences. Four of five were major anomalies: Sturge-Weber syndrome, Down's syndrome, hydrocephalus, possible genetic mitochondrial defect ('major, to be conservative'). Minor anomaly (classified as a relatively frequent structural anomaly not likely to cause any medical or cosmetic problems): congenital corneal anomaly. eOne baby died shortly after birth (lived for 'minutes to hours').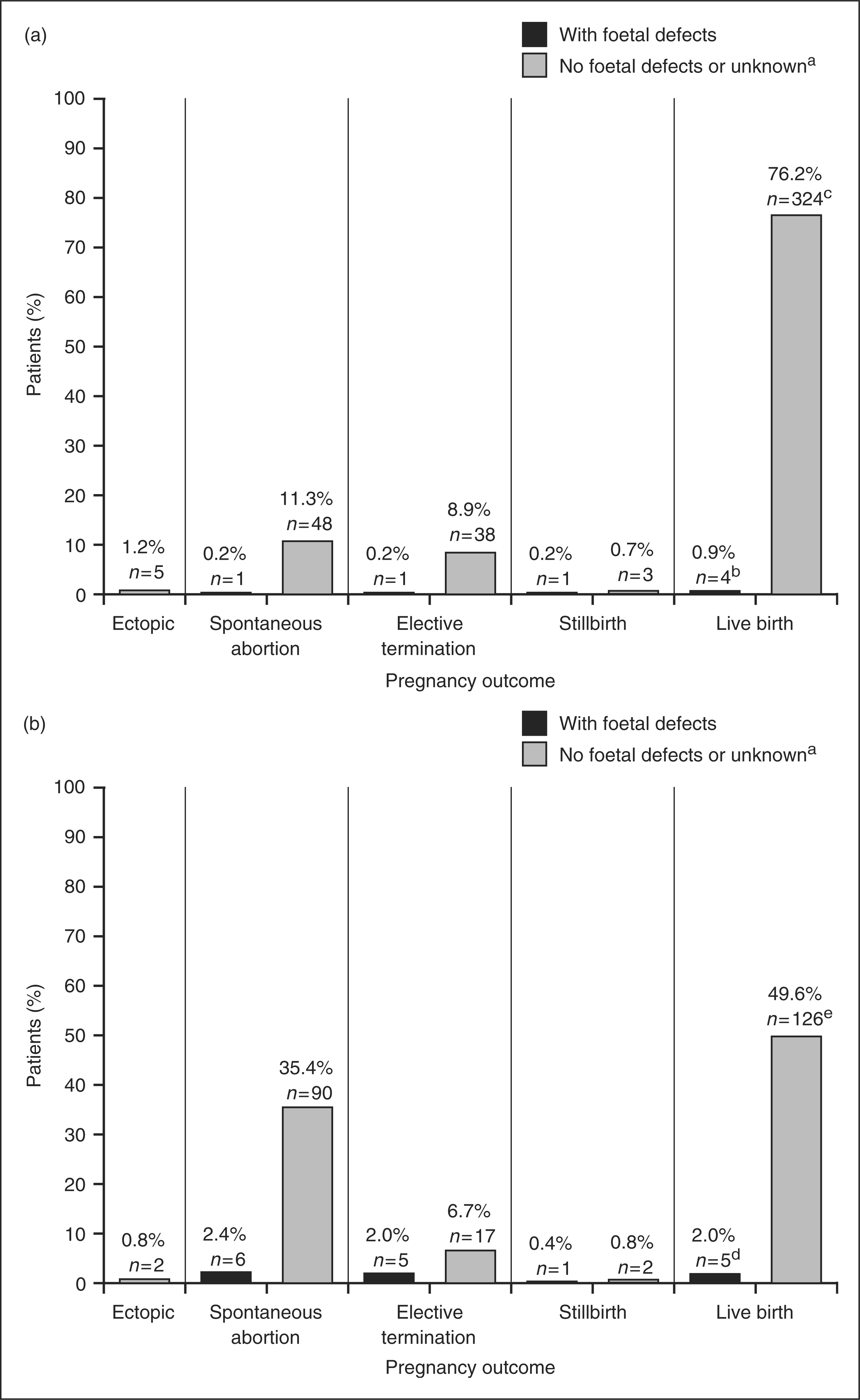
Congenital anomalies detected in live birth pregnancies exposed to IFN beta-1a
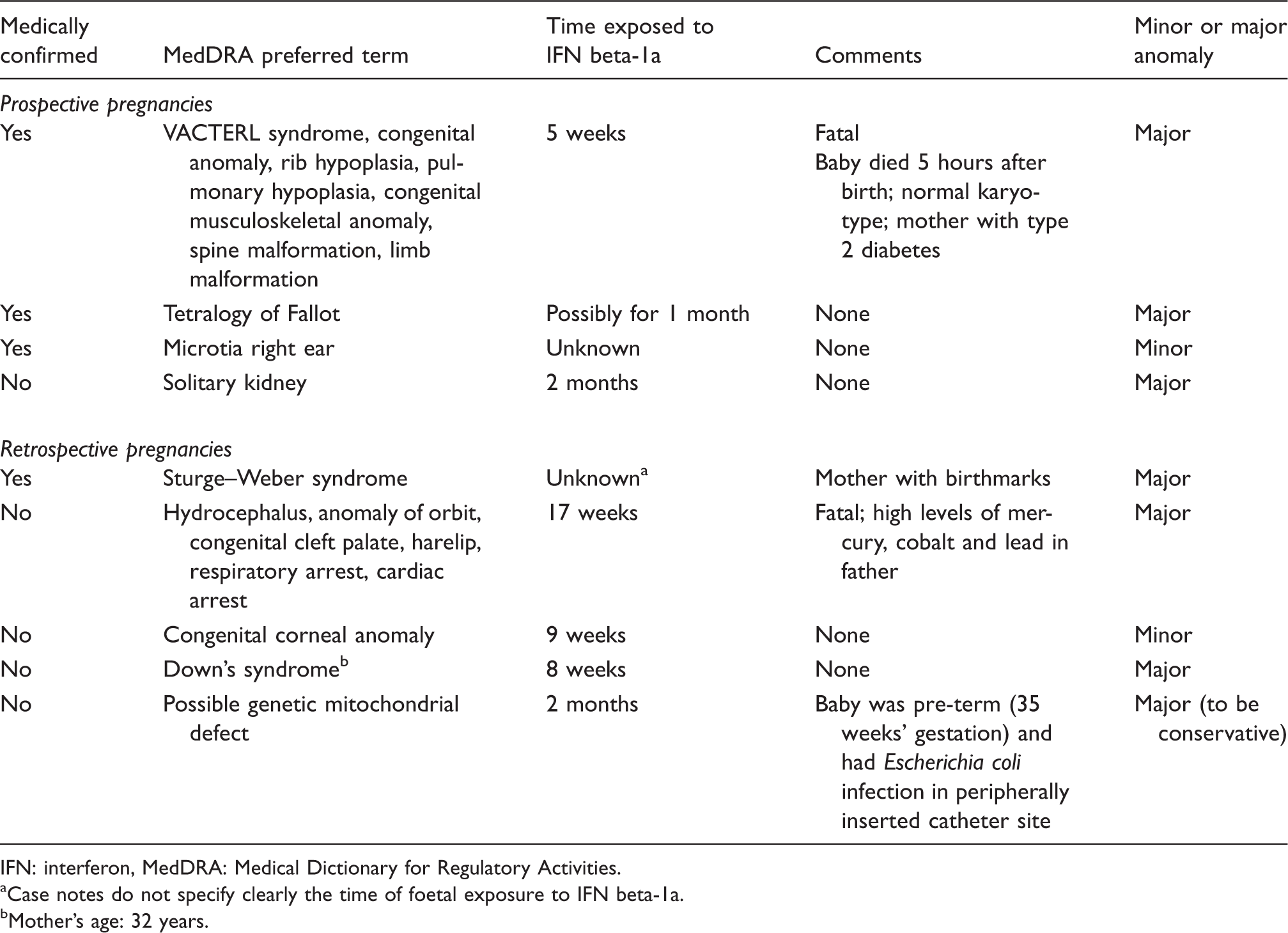
IFN: interferon, MedDRA: Medical Dictionary for Regulatory Activities.
Case notes do not specify clearly the time of foetal exposure to IFN beta-1a.
Mother’s age: 32 years.
Foetal defects detected in non-live birth pregnancies a exposed to IFN beta-1a
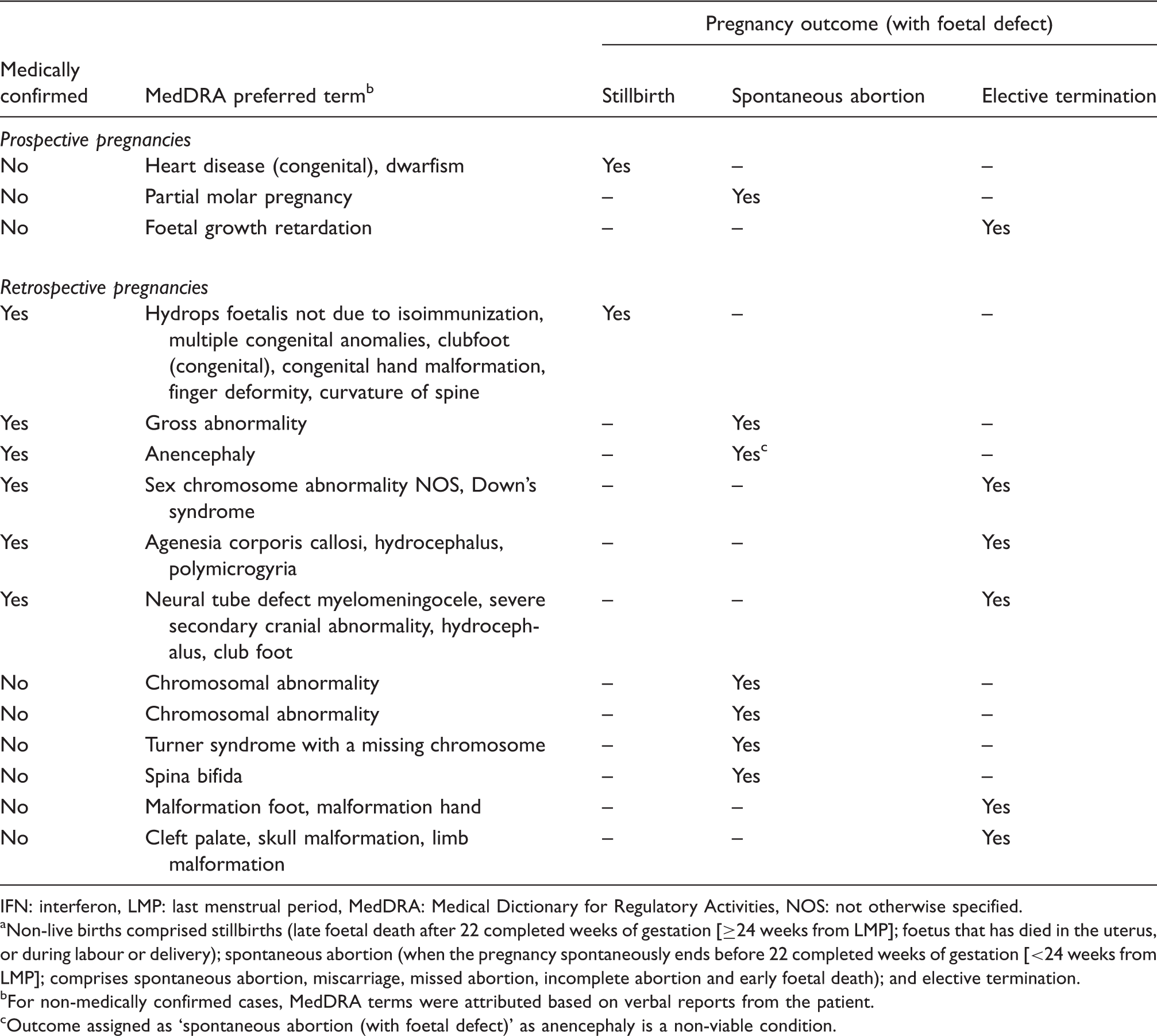
IFN: interferon, LMP: last menstrual period, MedDRA: Medical Dictionary for Regulatory Activities, NOS: not otherwise specified.
Non-live births comprised stillbirths (late foetal death after 22 completed weeks of gestation [≥24 weeks from LMP]; foetus that has died in the uterus, or during labour or delivery); spontaneous abortion (when the pregnancy spontaneously ends before 22 completed weeks of gestation [<24 weeks from LMP]; comprises spontaneous abortion, miscarriage, missed abortion, incomplete abortion and early foetal death); and elective termination.
For non-medically confirmed cases, MedDRA terms were attributed based on verbal reports from the patient.
Outcome assigned as ‘spontaneous abortion (with foetal defect)’ as anencephaly is a non-viable condition.
Discussion
This study assessed outcomes in women with MS who became pregnant while receiving SC IFN beta-1a therapy, using data from a global drug safety database.
It has been shown that rates of adverse outcomes are higher when ascertained retrospectively rather than prospectively. 19 To avoid any potential reporting bias generated by over-reporting of adverse outcomes, this discussion focuses only on prospectively reported pregnancies, unless otherwise specified.
The majority of pregnancies exposed to SC IFN beta-1a (76.2%) were associated with normal, live births. The percentage of pregnancies resulting in spontaneous abortions (11.5%), with or without foetal defects, was in line with the reported rate in the general population (known to be relatively common, 10–20% before 20 weeks of pregnancy), 20 suggesting that an association between SC IFN beta-1a and spontaneous abortion is unlikely.
The proportion of live births resulting in a major congenital anomaly after exposure to IFN beta-1a (0.9%) was lower than the incidence of major anomalies among live births in the general population in many published studies (2–4%). 18 Among non-live births with reported outcomes there were three with foetal defects. The reported defects did not cluster around any specific body system or organ, suggesting a coincidental occurrence, rather than the involvement of an IFN beta-1a-induced mechanism. Taken together, these results suggest that an association between IFN beta-1a and the incidence of congenital anomalies or foetal defects is unlikely. Recent findings from a prospective registry of pregnant women exposed to intramuscular IFN beta-1a during the first trimester of pregnancy also showed that the rate of spontaneous abortions and major/serious birth defects was not increased when compared with rates in the general population. 21
Results from previous studies 6–13 of pregnancy outcomes in women with MS receiving other immunomodulatory or immunosuppressive therapies have been inconclusive and, consequently, exposure to these agents during pregnancy is not advised. Use of mitoxantrone during pregnancy is contraindicated due to its mechanism of action. Post-marketing surveillance studies of glatiramer acetate have reported an incidence of spontaneous abortion of 21%. 22 Animal studies involving the immunosuppressive agent natalizumab have also demonstrated harmful effects on foetuses. 17 In a recent prospective evaluation of pregnancy outcomes in women, preliminary data showed that the rate of spontaneous abortions with natalizumab treatment was comparable to the rate in the general population. 23
Currently, advice against use of IFN beta during pregnancy is based on findings from animal studies, which report higher-than-expected abortion rates in primates exposed to human IFN beta during pregnancy. 16,17 However, it should be noted that across the IFN-beta drugs assessed, these studies used up to 40 times the recommended human dose, based on surface area. 24
The analyses presented here were subject to several limitations. Outcomes were unavailable for 111 pregnancies collected prospectively that were subsequently lost to follow-up, which may have introduced bias. Further, estimation of time of exposure to IFN beta-1a may have been inaccurate; patients may have been uncertain of the date of LMP or the date when IFN beta-1a treatment was discontinued.
General population data were used for comparisons, although patients with MS who were not receiving disease-modifying drugs may have been a more accurate control population. However, treated patients with MS are closely monitored and comparative data from monitored but untreated patients would be difficult to obtain.
The mean foetal exposure time for both prospectively and retrospectively reported pregnancies (for cases for which duration of exposure was available; n = 231) was 28 days (range 1 to 181 days). For the vast majority of these pregnancies (86.1%), foetal exposure time was short, ranging from 1 to 45 days, and nearly all cases of foetal exposure occurred during the first trimester. Other studies have reported a mean foetal exposure time of 4–9 weeks (28–63 days). 7,11,12 It is possible that the difference in length of exposure to IFN beta-1a may account for differences in pregnancy outcomes between different studies. In the current analysis, the vast majority of women were not exposed to IFN beta-1a during the whole period of foetal organogenesis or in later trimesters. However, although the length of exposure was short, exposure occurred during a critical period of foetal development.
It should be noted that in a previous study of pregnancy outcomes occurring during controlled clinical trials in women with MS, a case of hydrocephalus was described in a live birth after the foetus had been exposed to a low dose of IFN beta-1a throughout the pregnancy. 12 However, the overall results of the study showed that there was no overall risk associated with use of SC IFN beta-1a. 12
The apparent lack of detrimental effect of IFN beta-1a on pregnancy outcomes could be attributed to the large size of the IFN molecule (approximately 23 kDa for IFN beta-1a), which may inhibit the degree to which IFN crosses the placental barrier. The degree of transferral across the placenta is not known, although the related molecule IFN alpha (19.5 kDa) was not detectable in the foetal blood or amniotic fluid in one study involving two women. 25
In conclusion, no increased risk was found for a detrimental effect of SC IFN beta-1a treatment on pregnancy outcomes in women with MS when compared with pregnancy outcomes in the general population. The women who did not discontinue IFN beta before conception, but who stopped therapy as soon as pregnancy was recognized, were not at a higher risk of adverse pregnancy outcomes. The analyses described here add to previous research 7–13,26 and should be taken into account when considering options during pregnancy and exposure to SC IFN beta-1a. Future analyses of long-term safety should come from large, prospective registries, ideally with adjustment for potential confounders such as maternal age and prior miscarriages.
Footnotes
Acknowledgements
The authors thank Clare McNulty (Caudex Medical, Oxford, UK, supported by Merck Serono S.A. – Geneva, Switzerland, an affiliate of Merck KGaA, Darmstadt, Germany) for her assistance in preparing the initial draft of the manuscript, collating the comments of authors and assembling tables and figures; and Carol Cooper, PhD (Caudex Medical, Oxford, UK, supported by Merck Serono S.A. – Geneva, Switzerland) for her assistance in editing for English, formatting the manuscript to meet journal guidelines and coordinating submission requirements. Members of the Merck Serono Drug Safety team gathered the data in the global drug safety database, performed the database searches, and agreed with the corresponding author regarding interpretation of the findings. All authors had access to the study data. MSW, as corresponding author, takes final responsibility for the conclusions drawn from these data and the decision to submit for publication.
Funding
This work was supported by Merck Serono S.A. – Geneva, Switzerland (an affiliate of Merck KGaA, Darmstadt, Germany).
Conflict of interest statement
MS-W has received honoraria from Serono Symposia International Foundation (lectures); from Merck Serono (lectures, work in data safety monitoring boards [DSMB]); from Genentech and Roche (DSMB); from Elan (advisory board); from the Swedish bank SEB Enskilda (lectures); from sanofi-aventis (lectures); and from Bayer Health Care (contribution of articles to Swedish health website). She has also received honoraria for serving on the board of directors of Active Biotech, Lund, Sweden, and as an external reviewer of a PhD thesis at the University of Copenhagen. EA, MSM and GK are salaried employees of Merck Serono S.A. – Geneva, Switzerland.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
