Abstract
Friction and wear are unavoidable in mechanical systems with moving parts; however, their impact can be mitigated with the appropriate use of chemical additives in lubricants. Under severe conditions, these additives undergo tribochemical reactions, creating a low-shear strength tribofilm that reduces friction and wear. However, the tribofilm's growth and effectiveness can be adversely affected by the presence of water in the lubricant. This study proposes a new method for in-situ analysis of the effect of water on tribofilm chemical composition and growth, and its impact on friction and wear. A fully formulated oil containing Molybdenum dialkyldithiocarbamate (MoDTC) inorganic friction modifier has been tested using a bespoke pin-on-disc tribometer integrated with Raman spectroscopy. A cross-sectional analysis of in-situ grown tribofilm was carried out using Focused Ion Beam (FIB) and TEM-EDS to obtain the chemical information and thickness of the tribofilm. The results indicate that the presence of water not only negatively affects the tribological performance of MoDTC but also hampers the growth of MoDTC-derived low-friction tribofilm.
Introduction
Transport sector is one of the main contributors to worldwide carbon emissions, significantly contributing to global warming and air pollution. 1 This sector contributes approximately 1/5th (8 billion tonnes) of global carbon dioxide (CO2) emissions, of which 3/4th of total transport emissions originate from road transport. Figure 1 illustrates the breakdown of carbon emissions from the transport sector. To reduce or eliminate these emissions, a transition from internal combustion engines (ICEs) to electric vehicles (EVs) is becoming very prominent.
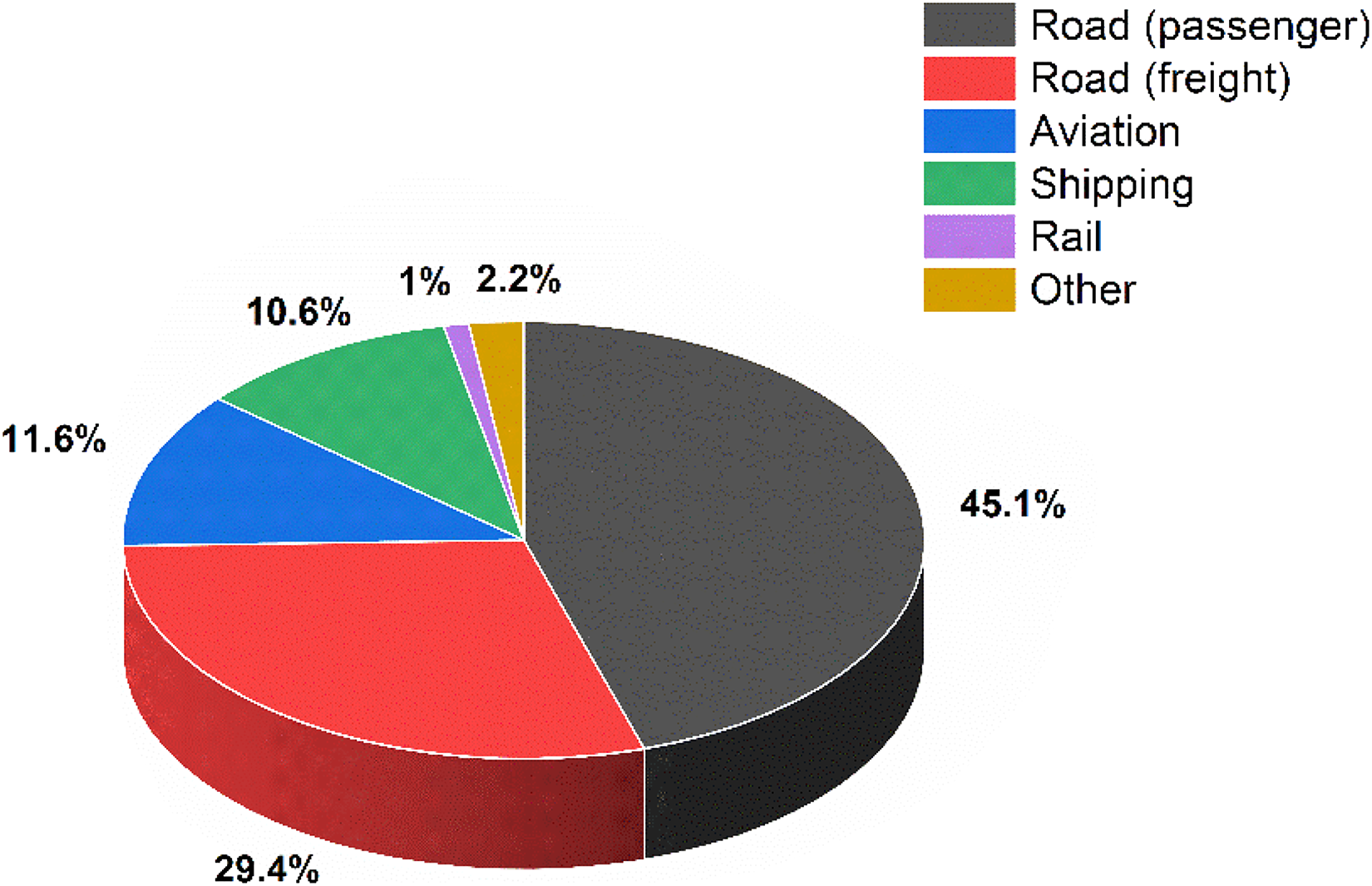
Emissions by the transport sector. 1
However, this transition is not as smooth as expected and possesses a number of tribological challenges 2 that needs to be addressed. Hybrid electric vehicles (HEVs) use both the conventional ICE and the battery-powered electric motor powertrain. 3 Due to the multiple start/stop capability, the duration of ICE engine operation is reduced, and the duration of ICE operation at low temperature increases significantly. This lower operating temperature can potentially lead to high levels of water accumulation in the oil sump, along with other contaminants (i.e., fuel dilution, etc.) and contaminate the engine oil. At high temperature, water, along with other contaminants, evaporates from the oil; however, at low temperature this tendency is less.3–5 Such water contamination of engine oil not only alters the functionality of additives but also reduces the service life, resulting in high friction and wear of the mechanical components.3,5,6
It is commonly known that friction modifiers (FMs) and anti-wear additives in engine oils (lubricants) help reduce the friction and wear of mechanical components in relative motion. They enhance the performance of the lubricants, ultimately increasing the engine component life while reducing fuel consumption, greenhouse emissions, and pollution.7,8 These additives work effectively in boundary and mixed lubrication regimes where friction and wear are dominant. Under the influence of contact pressure, temperature, and shear stress, decomposition of additives occurs, which promotes the formation of a low-shear strength tribofilm.8–10 The tribofilm works as a protective or sacrificial layer, resulting in wear reduction at the sliding interfaces.8,9
Molybdenum dialkyldithiocarbamate (MoDTC) and Zinc dialkyldithiophosphates (ZDDPs) are the most used FMs and anti-wear additives, respectively. However, contamination of oil with water can adversely impact the performance of these additives. The study by Dorgham et al. 6 reported structural changes in long phosphate chains, leading to a long-lasting detrimental effect on the ZDDP tribofilm. Other studies11,12 confirmed such detrimental effects and tribological performance degradation because of lubricant contamination with water. Although the effect of water contamination on ZDDP decomposition and its tribological performance is well established and documented, its effect on MoDTC tribofilm has not been investigated before.
This study aims to explore the effect of water contamination on MoDTC-derived low-friction tribofilm chemical composition, tribofilm growth, and the effect of tribofilm growth on friction and wear dynamics relevant to the lubrication of hybrid engines. Raman spectroscopy was successfully employed for in-situ monitoring of the effect of water contamination in lubricating oil on the MoDTC-derived MoS2 tribofilm formation and growth.
Materials and methods
Materials
The present testing was conducted using the as-supplied fully formulated baseline oil (dynamic viscosity = 14.5 mPa.s at 60 °C) that contained Group III oil and proprietary packages of additives with an added inorganic friction modifier, MoDTC. The additive package included typical engine oil additives, including antiwear additives, dispersants and detergents. The formulation, baseline oil with MoDTC is represented as Lubricant L1. To investigate the effect of water on friction modifier (MoDTC) tribological properties, we added 2 wt% of deionized (DI) water into the lubricant L1, and this formulation is represented as lubricant L2. Table 1 shows the lubricant's nomenclature. The lubricant mixture with water was prepared by stirring the oil-water mixture using a magnetic stirrer at 750 rpm for 30 min at room temperature to ensure proper mixing. AISI 1074 steel disc and AISI 52100 steel ball were used for the tribological tests. The hardness of the steel disc and ball was 47 and 64 Rockwell (HRC), respectively. The Young's modulus of the steel disc and ball was in the range of 190–210 GPa, and roughness (Ra) was 200 nm for the disc and 17 nm for the ball (5.5 mm diameter). The dimensions of the steel disc were 1 mm in thickness, 85 mm in inside diameter, and 110 mm in outside diameter.
Lubricant nomenclature and composition.

Tribometry and Raman spectroscopy
The tribological testing was carried out on a bespoke unidirectional tribometer 13 attached to Raman Spectroscopy 14 , as schematically shown in Figure 2. All the tests were kept identical for both the lubricants (L1 and L2). The tests were conducted at room temperature and 60 °C. The oil and disc were heated using two cartridge heaters (300 W Firerod) procured from Watlow Electric Manufacturing Co., USA and K-type thermocouples were used to measure the temperature of the oil. 13 The applied Hertzian contact pressure was 0.99 GPa. The ball was fixed, and the disc was rotating against the ball. The motor (steel disc) speed was kept at 50 rpm (0.25 m/s) throughout all the tests. High pressure and low sliding speed were selected to ensure the boundary (lambda ratio = 0.077) lubrication condition. Before conducting the tribological test, lubricant was applied to the steel disc and the disc surface was ensured to be covered with lubricant. The tests were conducted at least two times and average values were reported. The total duration of the test was 60 min. Before conducting the friction tests, the steel disc and ball were cleaned using acetone to remove the contamination present on them. The friction coefficient was measured using the LabVIEW software installed on the computer connected to the tribometer. After completion of the tribological tests, worn steel surfaces were carefully rinsed using heptane for post-test surface analysis.
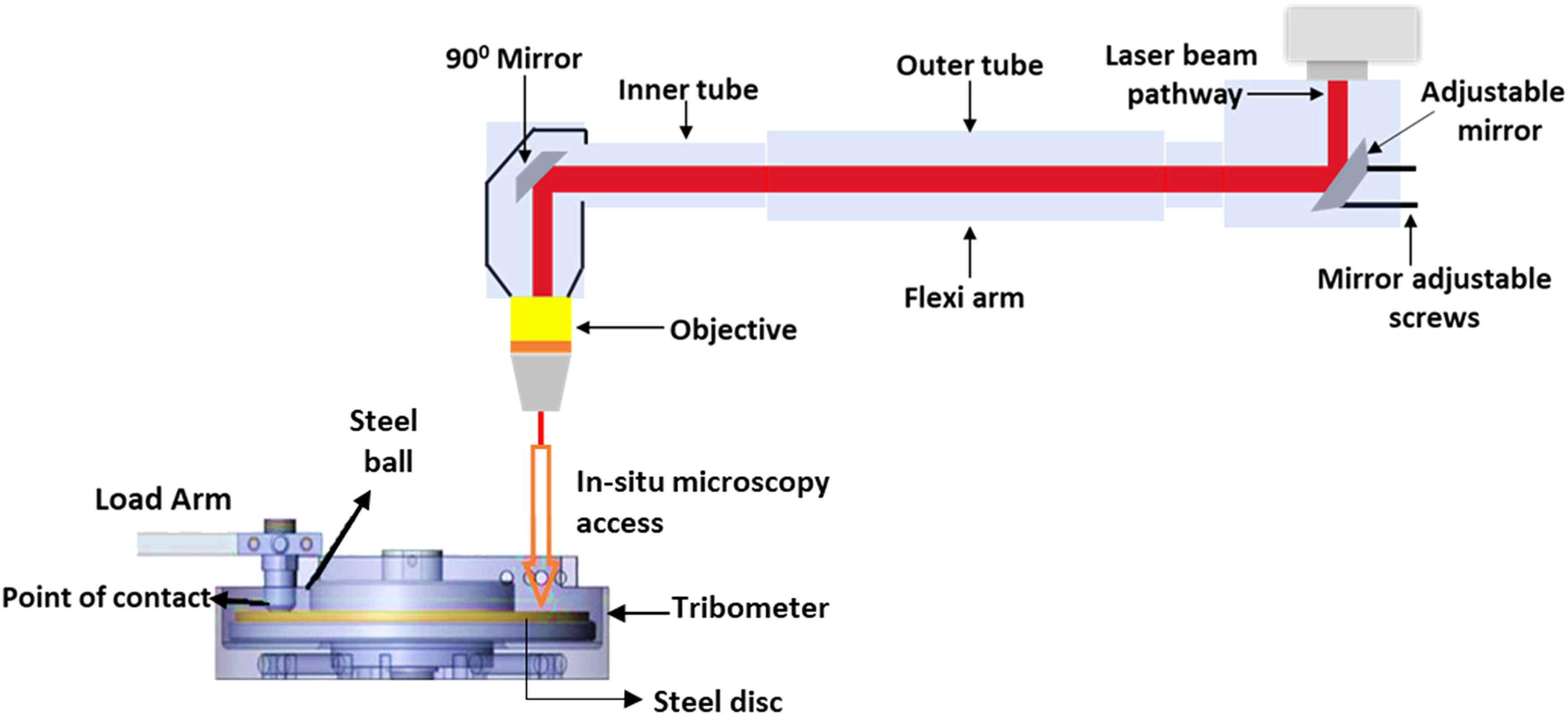
Schematic of the in-situ Raman tribometer (adapted with permission from [15]. Copyright {2018} American Chemical Society).
In-situ Raman analysis was conducted on a Renishaw Invia spectrometer (UK) integrated with a bespoke tribometer. A 488 nm laser wavelength, 5% laser power (0.5 mW), and 10 s exposure time were used for the chemical analysis of tribofilm. The laser power was kept low to avoid any damage to the tribofilm. The spectrometer with 1 cm−1 spectral resolution, 800 nm lateral resolution, 50 × L objective lens fastened with a flexi arm (Figure 2), and up to a 200 nm penetration depth 16 was employed for the chemical analysis. To conduct the Raman analysis of the generated tribofilm on steel discs, tribological tests were stopped after different rubbing intervals (2.5, 5, 10, 30, and 60 min). Raman analysis of the tribofilm was carried out without cleaning the steel discs.
Tribofilm TEM analysis
The cross-section and chemical analysis of the generated tribofilm were carried out using FEI Titan3 Themis 300 Transmission Electron Microscopy (TEM) and Energy-Dispersive X-ray spectroscopy (EDS). A focused ion beam (FIB), FEI Helios G4 CX DualBeam, was used to prepare the thin cross-section lamella of tribocontact for TEM analysis. To prepare the TEM sample, a 0.3 µm-thick platinum (Pt) electron beam (2 kV, 6 nA) was deposited on the tribocontact to protect it from the ion beam deposition. The sample was then tilted to 52 degrees for Pt (1 µm thick) ion beam deposition (0.25 nA) to protect the sample from the Gallium ion beam during bulk removal of material and polishing of the sample. Following this, bulk material removal (30 kV, 21 nA) was performed on the wear track to create a lamella. Cleaning of the lamella was carried out at 30 kV and 9 nA. Next, the sample was tilted to 0 degrees for an undercut at 30 kV and 9 nA. The sample was tilted again to 52 degrees for further cleaning of the lamella and then tilted back to 0 degrees. A needle was inserted and attached to the lamella, and milling was performed to remove the lamella from the wear track. Afterwards, the lamella was attached to the TEM grid, where it was polished at 30 kV using currents of 2 nA, 0.8 nA, and 0.24 nA. In the final stage, the lamella was cleaned at 8 kV and 0.21 nA until it became electron transparent. Afterwards, it was placed into the TEM chamber for cross-sectional and chemical analysis. Figure 3 shows the sample preparation steps (from step 1 to the final step) during FIB of the steel disc for TEM analysis.

Sample preparation by FIB for TEM analysis.
Optical microscope and profilometer
An optical microscope (Leica) and NPFLEX 3D (Bruker, USA) non-contact type profilometer (white light interferometry) were used to analyse the worn steel disc and ball, respectively. The wear scar diameter of the steel ball and its worn surface profile were assessed using the NPFLEX.
Results
Friction and in-situ Raman analysis
Figure 4 shows the variation in friction coefficient (µ) with different sliding (rubbing) times (i.e., 2.5, 5, 10, 30, and 60 min) and corresponding Raman spectra of MoDTC-derived tribofilm for lubricants L1 and L2 at 60 °C. In Figure 4(a) for lubricant L1, as sliding between the steel ball and disc initiates, the friction coefficient (µ) increases until 2.5 min, then it drops instantaneously with increasing rubbing time, reaching a steady state. The average value of µ after 2.5 min of rubbing is calculated to be 0.11. This period correlates with the running-in period, and high friction indicates that no low-friction tribofilm forms on the steel disc. However, after 2.5 min of sliding, a significant reduction in µ is observed until 10 min of sliding time. After 10 min of rubbing, a steady decrement in µ was observed throughout the test duration.
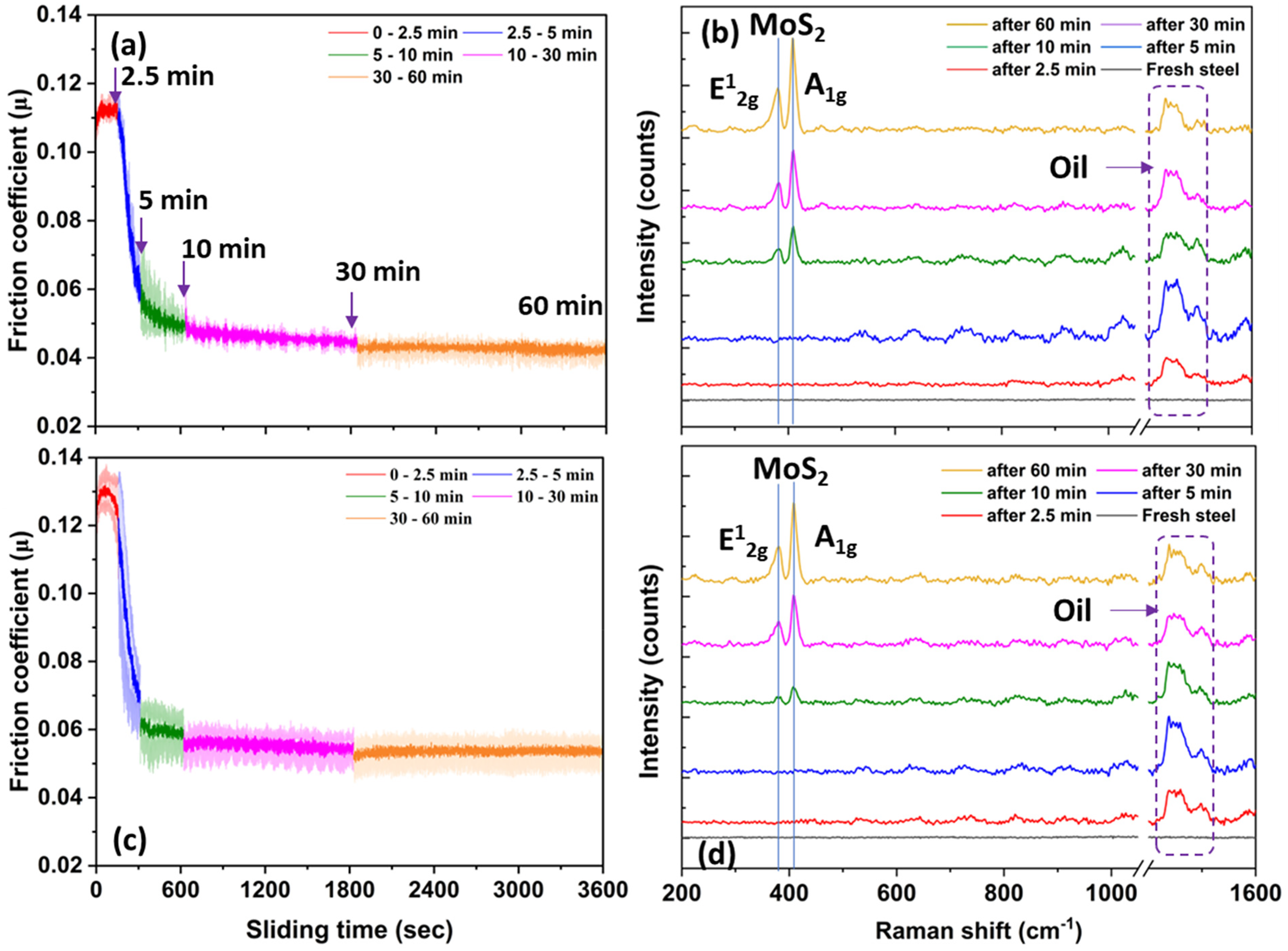
Variation in friction coefficient and corresponding Raman spectra of tribofilm with different rubbing times for lubricants L1 (a and b) and L2 (c and d) at 60°C.
Figure 4(b) shows the Raman spectra of MoDTC-derived tribofilm on the steel disc for lubricant L1 taken after different sliding times: 2.5, 5, 10, 30, and 60 min. Due to the patchy nature of the tribofilm, multiple Raman spectra were recorded on the wear track after different sliding times and averages of them are reported here. Figure 4(b) illustrates that after 2.5 and 5 min of rubbing, no MoS2 (A1g and E12g) peaks were observed on the steel disc. However, after 10, 30, and 60 min of rubbing, sharp and strong A1g peaks were observed at 409 cm−1, 409 cm−1 (± 0.60), and 408 (± 0.8) cm−1, respectively. Whereas broad E12g peaks were observed at 380 (± 2.4) cm−1, 381 (± 1.2) cm−1, and 381 (± 0.8) cm−1 after 10, 30, and 60 min of rubbing, respectively. The broadness in the peak could be due to the stress-induced disorder of the MoS2 formed on the wear scar. 17 The Raman spectra of the tribofilm for lubricant L1 show the two strong characteristic peaks of MoS217,18; (i) A1g (≈ 409) due to the out-of-plane vibration mode of sulphur (S) atoms away from molybdenum (Mo) atoms in both directions and (ii) E12g (≈ 381) due to the in-plane vibration mode of Mo and S atoms. Moreover, the broad peak from 1438 to 1500 cm−1 (marked in Figure 4(b) and (d)) is assigned to the oil (amorphous carbon 17 ).
In Figure 4(c) for lubricant L2 (with added water), the friction coefficient starts slightly higher than lubricant L1. After 2.5 min of rubbing, the average µ for lubricant L2 was found to be 0.13. Similar to lubricant L1, for lubricant L2, after 2.5 min of rubbing, µ begins to decrease notably until 10 min. After 10 min of rubbing, a gradual decrement in µ was also observed for lubricant L2 until the completion of the test.
Figure 4(d) shows the Raman spectra of MoDTC-derived tribofilm on steel disc for lubricant L2 (with added water) taken after different sliding times of 2.5, 5, 10, 30, and 60 min. Similar to lubricant L1, for lubricant L2, the presence of MoDTC-derived lubricious tribofilm or Raman peaks of MoS2 was not observed after 2.5 and 5 min of rubbing. However, for rubbing beyond 5 min, A1g peak of MoS2 can be distinctly observed at 409 (± 1.45) cm−1, 408 (± 0.8) cm−1, and 408 (± 0.8) cm−1 after 10, 30, and 60 min of sliding, respectively, and a broad E12g peak can be observed at 379 (± 3.3) cm−1, 381 (± 1.4) cm−1, and 380 (± 0.7) cm−1 after 10, 30, and 60 min of sliding, respectively. In Figure 4(b) and (d), the broad peak around 200 cm−1 for both lubricants could be assigned to crystalline disorder in the MoS2 structure, since it is subjected to high contact pressure and shear stress.8,17 Figure 4(b) and (d) also shows the slight shift of MoS2 peaks after different rubbing times. This could be due to the continuous change in MoS2 layers on the rubbing surfaces. 18 Furthermore, from Figure 4(b) and (d), it can be observed that MoDTC-derived MoS2 peak intensity increases with the rubbing time for both the lubricants (L1 and L2). No additional major shift or change was observed in Raman peaks of MoDTC-derived tribofilm for L2 (with added water) as compared to the lubricant L1 (as received). This indicates that the water is physically interacting with lubricant additives and altering their tribological performance.
Figure 5(a) shows variation in µ with sliding time for lubricants L1 and L2 at 60 °C. Lubricant L2 resulted in a higher value of µ than lubricant L1. At 60 °C, for both L1 and L2, µ starts with a higher value compared to µ at room temperature (refer Figure 6) then µ for L1 and L2 at 60 °C significantly reduces until 10 min and a steady decrement in µ was observed throughout the test duration. In Figure 5(b), the statistical difference in µ for the last 50 min of rubbing for lubricants L1 and L2 is significant. The average value of µ for lubricants L1 and L2 for the last 50 min of rubbing is calculated to be 0.044 and 0.055, respectively. The high value of µ for lubricant L2 compared to L1 demonstrates that the presence of water in lubricant L2 negatively affects its friction-reducing ability.
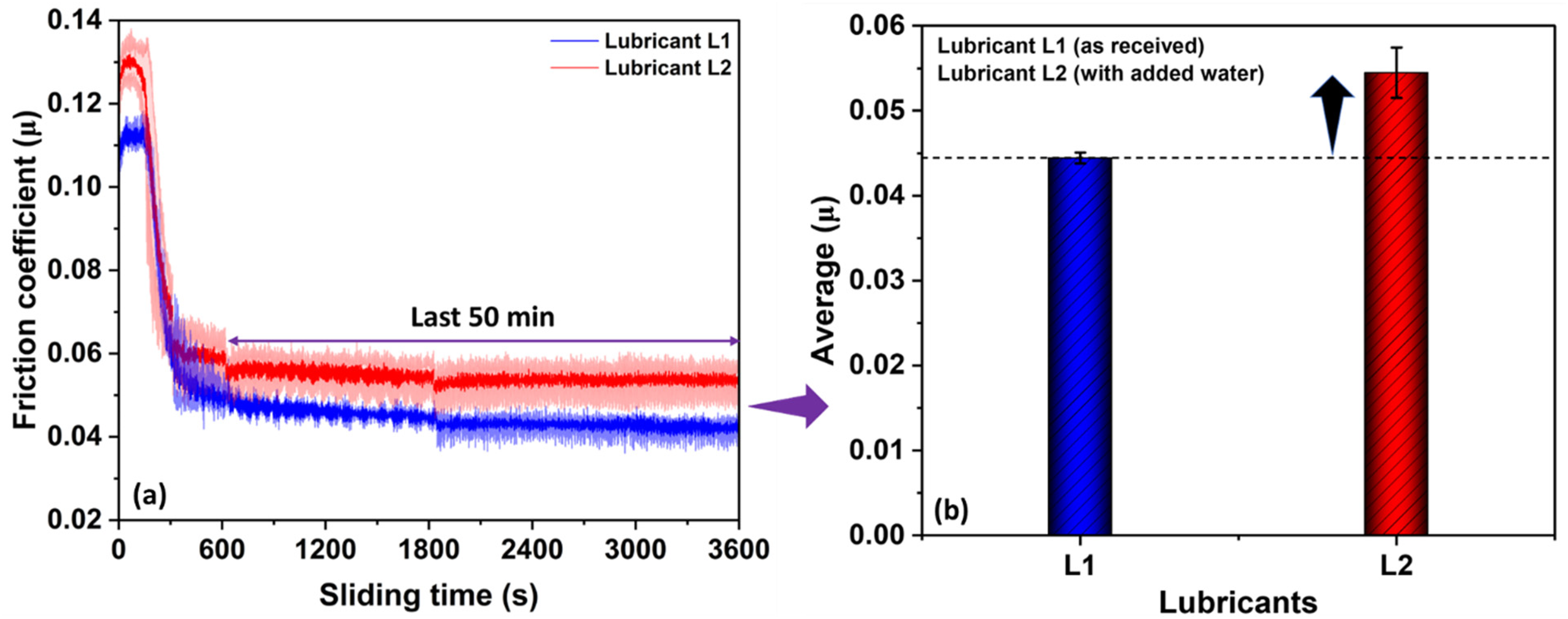
(a) variation in friction coefficient (µ) with rubbing time and (b) statistical difference in µ for the last 50 min of rubbing for lubricants L1 (as received) and L2 (with added water) at 60 °C.
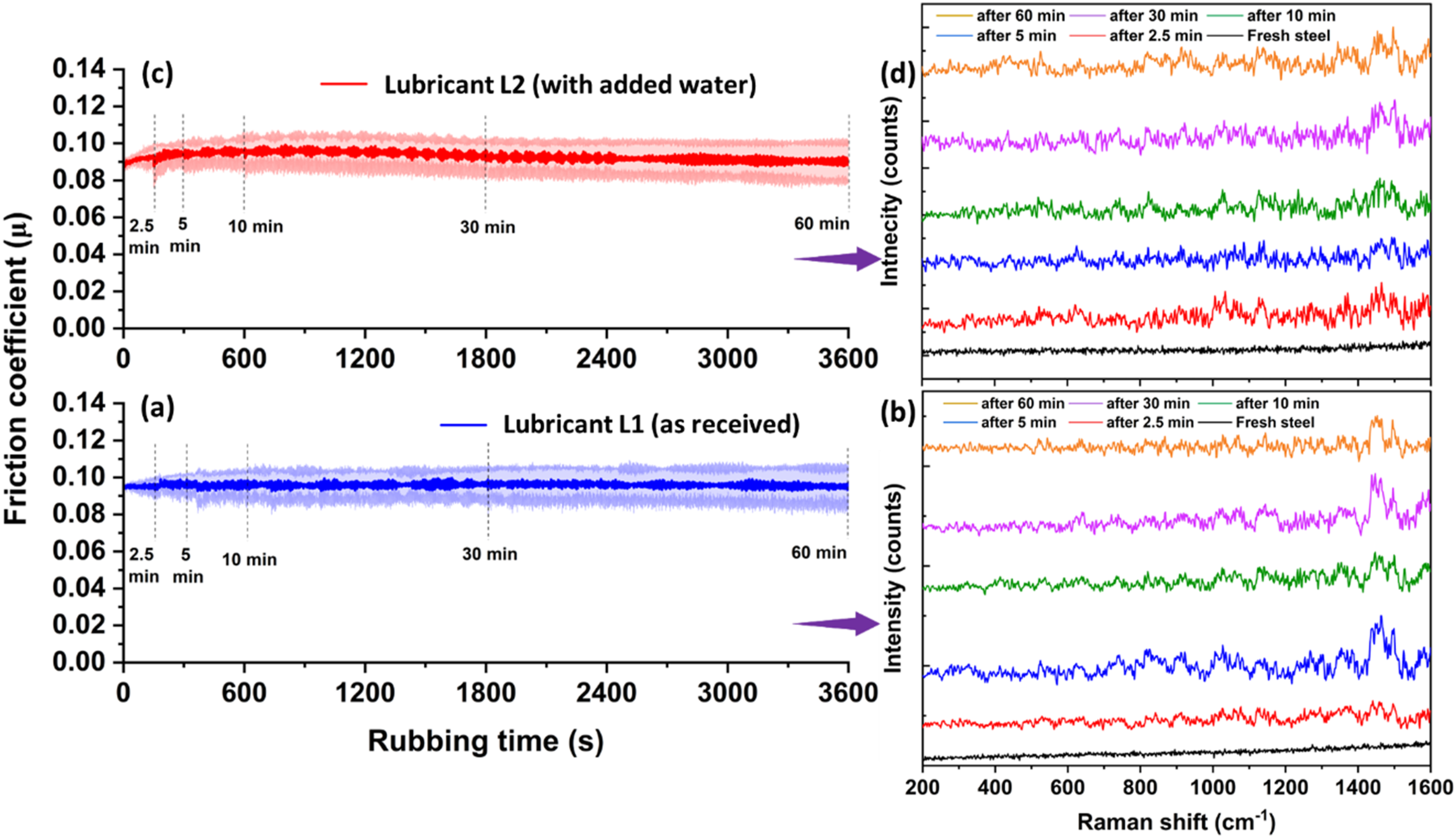
Variation in friction coefficient (a, c) and corresponding Raman spectra (b, d) with different rubbing times for lubricant L1 (a, b) and L2 (c, d) at room temperature.
It is also noticeable in Figure 4(b) and (d) that after 5 min of rubbing, no MoS2 peaks were observed. However, the sudden reduction in µ after 2.5 min of rubbing indicates the presence of a low-friction tribofilm on the steel disc for both lubricants. The absence of MoS2 A1g and E12g peaks could be due to; (i) after 5 min of rubbing, the formed tribofilm may be very thin and (ii) the presence of oil on the steel disc while taking the measurements could attenuate or weaken the Raman signal of this thin tribofilm. 8 To investigate this, the steel disc was carefully cleaned using heptane to remove the oil from the disc after 5 min of rubbing, followed by ex-situ Raman analysis. Figure 7 shows the results obtained. The analysis confirmed the presence of the MoS2 peaks from the MoS2 tribofilm, explaining the friction reduction. These MoS2 A1g and E12g peaks for lubricants L1 and L2 were observed at 409 cm−1 and 381 cm−1, respectively.
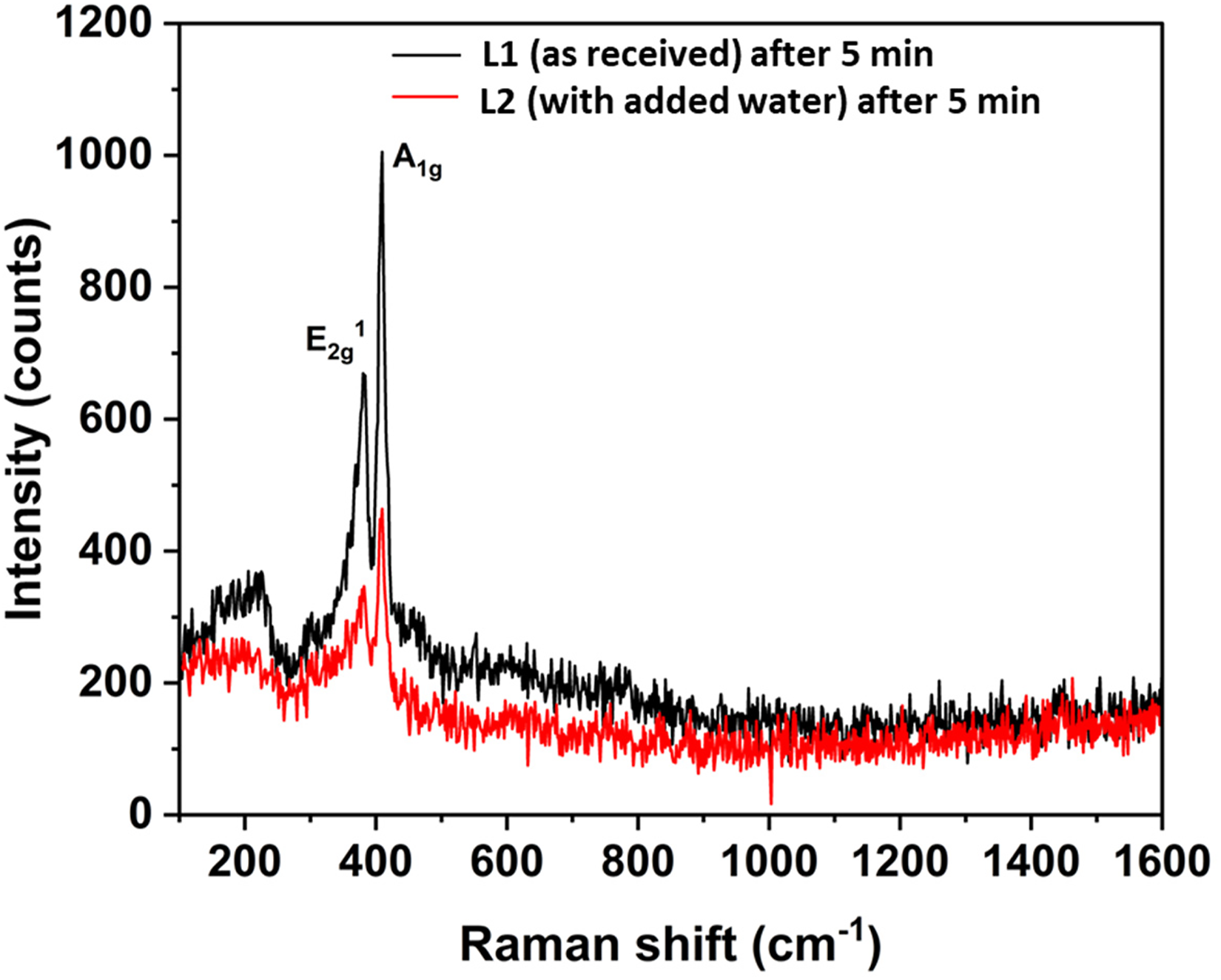
Raman spectra of MoDTC-derived lubricious tribofilm on steel disc after 5 min of rubbing at 60 °C for lubricants L1 and L2.
Growth of the MoS2 tribofilm
Figure 8 shows the growth in the MoDTC-derived MoS2 obtained through the A1g peak area intensity as a function of rubbing time. The increment in the A1g peak area intensity after different rubbing times (10, 30, and 60 min) at 60 °C can be attributed to acceleration in MoDTC decomposition rate, rise in MoS2 concentration or growth in tribofilm formation on the steel disc with time. 17 In Figure 8, the increasing trend in peak area intensity was observed for both lubricants L1 and L2 after different rubbing times at 60 °C. Furthermore, the optical micrographs in Figure 9 are evidence of the in-situ grown tribofilm on the steel disc after 60 min of rubbing at 60 °C for lubricants L1 and L2.
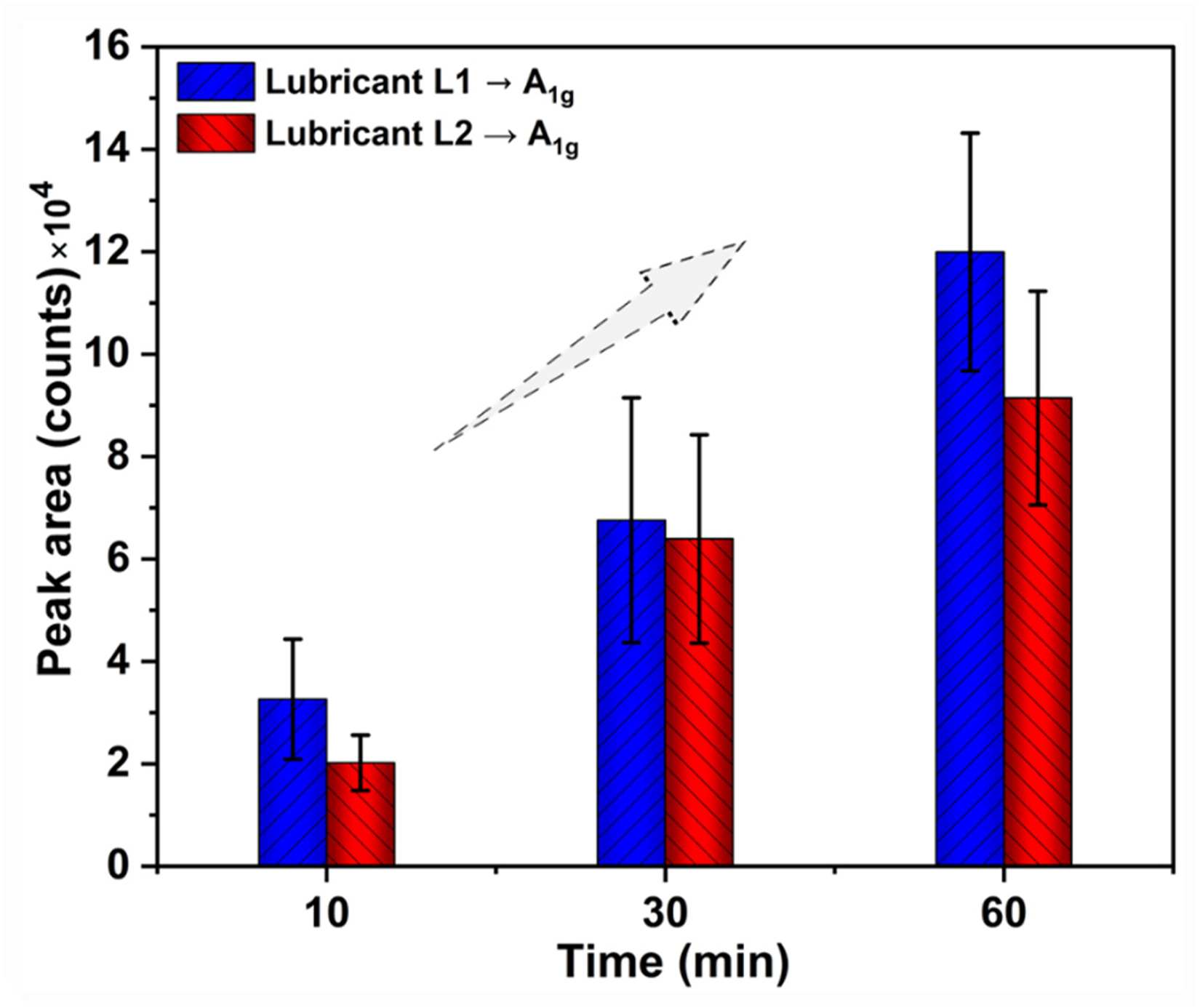
Comparison of the growth of MoDTC-derived tribofilm on the steel disc after different rubbing times at 60 °C for lubricants L1 (as received) and L2 (with added water).

Optical micrographs; (a) fresh steel disc surface, (b) tribofilm on steel disc for lubricant L1 (as received) and (c) lubricant L2 (with added water), after 60 min of rubbing at 60 °C.
Friction and in-situ Raman analysis at room temperature
Figure 6 shows the friction responses and Raman spectra for lubricants L1 and L2 after different rubbing times: 2.5, 5, 10, 30, and 60 min at room temperature (RT). No significant difference in friction coefficient (µ) was observed for both lubricants. From the start of sliding to 2.5 min of rubbing, µ increases; however, it becomes steady for the rest of the test time. During the running-in period at RT for lubricants L1 and L2, µ starts from a lower value (0.09) compared to lubricants L1 (0.11) and L2 (0.13) at 60 °C. This may be because the viscosity of oil is higher at room temperature than at high temperature. Higher viscosity will reduce severe metal-metal (asperities-asperities) contact at the tribo-pair interface, thus reducing the friction. It is evident from Figure 6(a) and (c) that, unlike the test at 60 °C, there is no drop in µ after 2.5 min of the test time, which is expected as MoDTC decomposition and the formation of lubricious MoS2 sheets do not take place at room temperature. 8 In Figure 8(b) and (d), in-situ Raman spectra also confirm the absence of MoS2. The presence of broad Raman peaks between 1438 and 1500 cm−1 for lubricants L1 and L2 at RT is assigned to amorphous carbon present in oil17,18 and the other unidentified peaks are assigned to fully formulated oil (containing packages of additives). The sudden drop in µ during the intermediate state (from 2.5 to 10 min) was observed only at 60 °C, and that has been seen to correlate with the formation of lubricous MoS2 tribofilm on the rubbing surfaces. The Raman peaks observed for MoS₂ at 60 °C during the intermediate state confirm its presence; refer to Figure 4 and 7.
Worn surface analysis
Generally, wear occurs at the commencement of the sliding over a certain period, until a tribofilm is formed. To get insights of the wear during the test, the scars on the steel balls were analysed using the 3D profilometer. Figure 10 shows the wear scar diameter (WSD) of the steel ball for lubricants (L1 and L2) both at room temperature (RT) and 60 °C. The results show that at 60 °C, lubricant L2 (with added water) showed a large WSD compared to lubricant L1 (as received). While at room temperature, the WSD for lubricants L1 and L2 can be given as L’1 < L’2. The WSD for lubricants L1 and L2 at RT was observed between the WSD of L1 and L2 at 60 °C. The WSD for different lubricants after 60 min of rubbing can be given in ascending order, L1 < L’1 < L’2 < L2. The inset images in Figure 10 show the wear scar profile of the steel ball for lubricants L1 and L2 after 60 min of rubbing at 60 °C and RT.
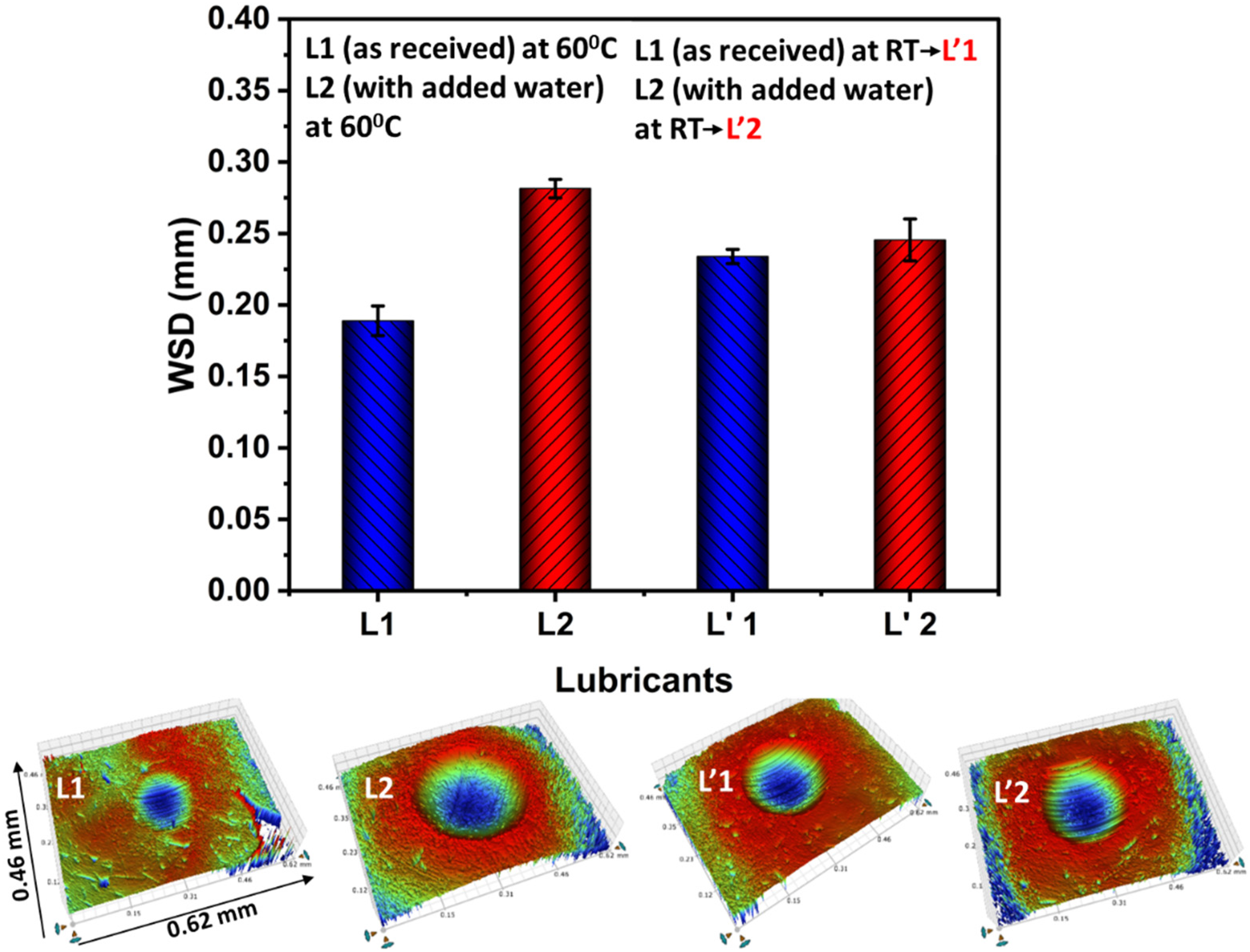
Wear scar diameter (WSD) of steel ball for lubricants L1 and L2 at room temperature (RT) and 60 °C after completion of tribo tests.
Tribofilm formation
Figure 9 shows the presence of tribofilm (dark colour areas) on the steel disc for lubricants L1 (as received) and L2 (with added water) after 60 min of rubbing. The optical micrographs of the steel disc in Figure 9 show the patchy nature of the tribofilm for both the lubricants (L1 and L2). However, for lubricant L2, tribofilm looks patchier (fewer dark areas/less surface coverage) and for lubricant L1, tribofilm looks more continuous and evenly distributed (higher surface coverage) over the wear track. This indicates that the presence of water hinders the tribofilm growth/coverage on steel discs for lubricant L2.
To know more about the characteristics of the tribofilm, such as structure, composition and thickness, the FIB-TEM cross-section analysis was also conducted. The high-magnification TEM micrographs of the tribofilm cross-section in Figure 11 confirm the formation of MoS2 (interplanar spacing 0.61 nm) sheets with a layer-like structure. These TEM micrographs show a thinner tribofilm for lubricant L2 (t ≈ 25 nm) compared to lubricant L1 (t ≈ 50 nm). This confirms that the presence of water hinders tribofilm growth.

FIB-TEM cross-section of MoDTC-derived tribofilm on steel disc after 60 min of rubbing for lubricants L1 and L2 at 60 °C.
The thinner tribofilm allows more asperities-asperities contact, causing a higher friction and wear than the thicker tribofilm. Besides this, in FIB-TEM cross-section micrographs, for lubricant L1 (as received) tribofilm appeared to be more continuous than lubricant L2 (with added water); see Figure 11(a) and (d). For both the lubricants (L1 and L2), lubricious MoS2 layers/sheets are embedded in the tribofilm, while for lubricant L1, MoS2 sheets are distributed in the tribofilm compared to lubricant L2, where MoS2 sheets appear to be bundled together.
Moreover, TEM-EDS elemental mapping of the tribofilm cross-section in Figure 12 indicates that the tribofilm is composed of molybdenum (Mo), sulphur (S), zinc (Zn), phosphorus (P), carbon (C), and oxygen (O). The presence of other elements such as Zn and P, could be from the additive packages present in the oil (fully formulated). The presence of these elements suggests the composite nature of the tribofilm or two-phase (MoDTC/ZDDP) tribofilm. Where the presence of MoS2 sheets reduces friction and the mixed Zn/Mo phase imparts antiwear ability. 10 For lubricant L1 (as received), the elements such as Mo, S, P, and Zn are aligned and arranged over each other layer by layer. The P and O are either deposited at the bottom of the tribofilm or near the iron surface, followed by Zn, S, and Mo. While carbon is distributed evenly from the bottom to the top of the tribofilm. However, for Lubricant L2 (with added water), not such an arrangement for elements Mo, S, P, Zn, and O was observed.
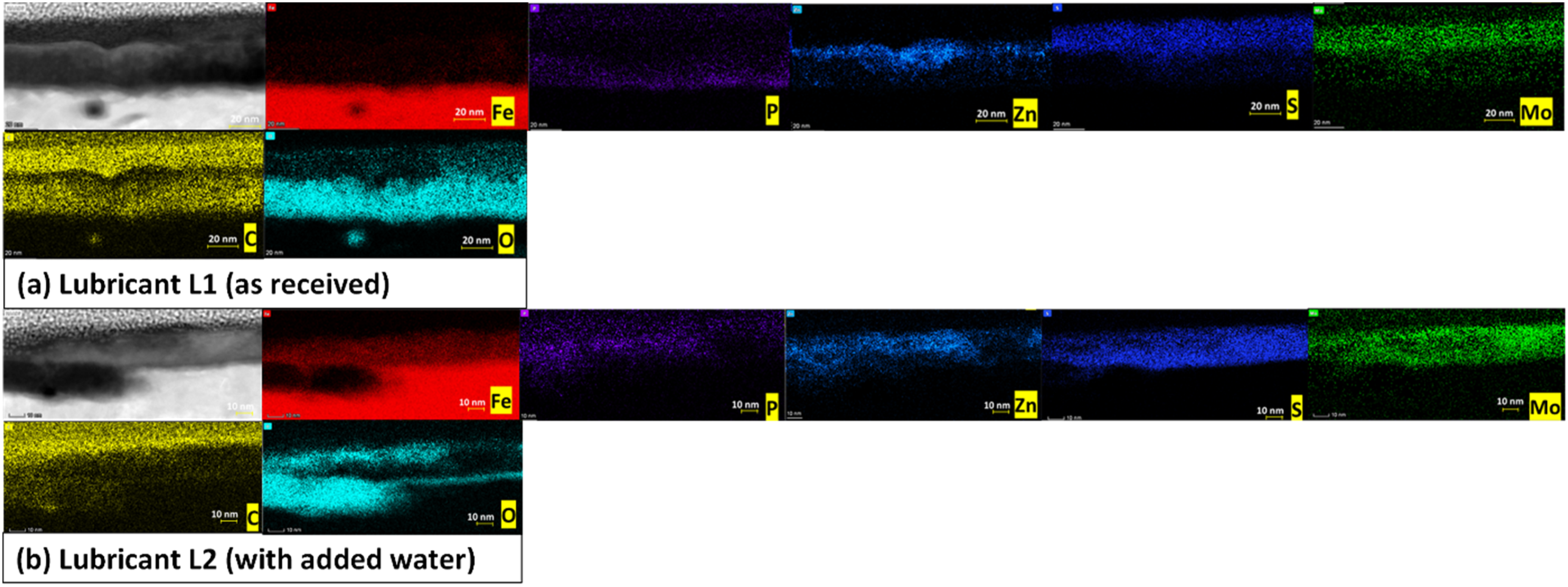
TEM-EDS mapping of in-situ grown tribofilm on steel disc after 60 min of rubbing time for lubricants (a) L1 and (b) L2 at 60 °C.
Discussion
Friction response and MoDTC decomposition
The friction response for lubricants L1 and L2 in Figure 13 can be divided into three different sections: (I) high friction section (running-in period), (II) Intermediate (drop in friction), and (III) low friction section. The reduction in friction coefficient for lubricants containing MoDTC is due to the decomposition or autocatalytic reaction of MoDTC at the steel-steel interface and formation of MoS2-rich lubricious tribofilm at the tribo-pair interface under high temperature, contact pressure, and shear stresses (rubbing).8,19 It has been proposed using XPS analysis that the decomposition of MoDTC takes place in two stages, resulting in the formation of MoS2 (lubricious entity) and MoO2 (which then oxidises to MoO3). 19
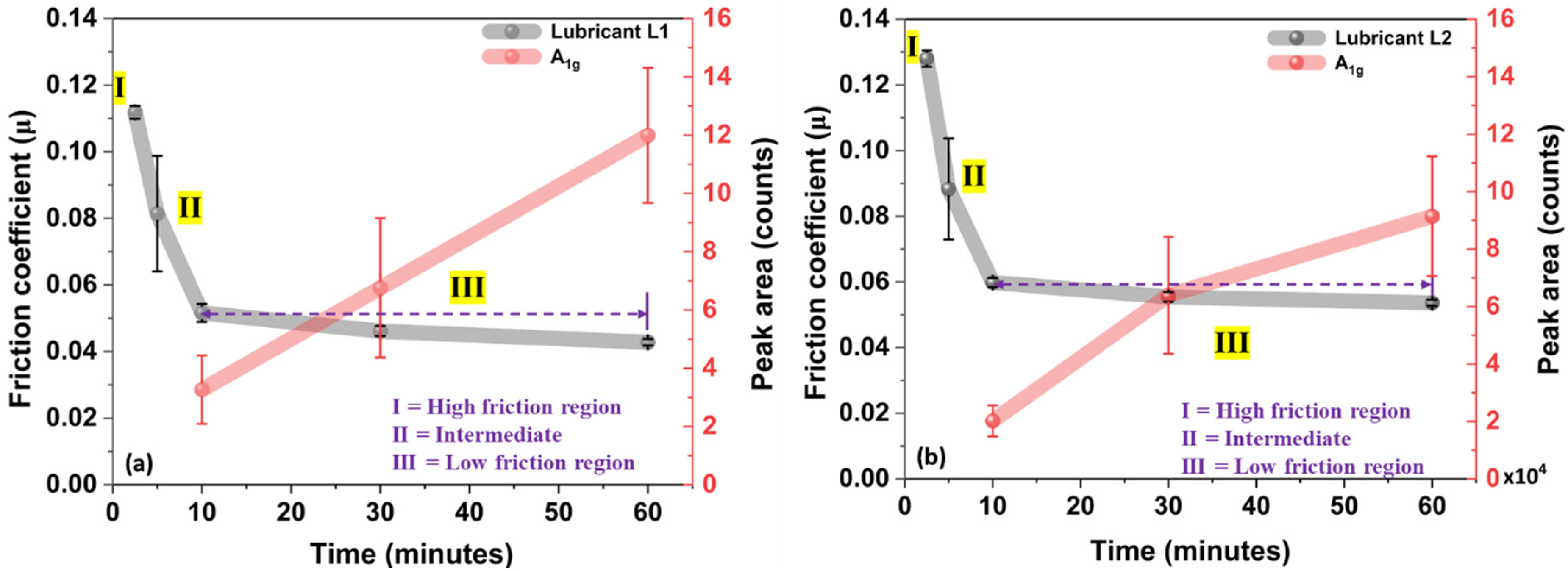
Correlation between the average friction coefficient and tribofilm growth after different rubbing times at 60 °C for (a) lubricant L1 (as received) and (b) lubricant L2 (with added water).
In the current in-situ Raman study, the presence of distinctive Raman peaks related to MoO3 at 820 cm−1 and 990 cm−1 17 was not observed on the worn steel disc throughout the tribo tests, in agreement with previous Raman studies of MoDTC tribofilms.7,8,18,20 It has also been proposed that the decomposition of MoDTC is sensitive to the test conditions, therefore, it may decompose during the steel-steel contacts to form MoS2, FeMoO4, and MoSx (converts to MoS2 at high temperature under tribological sliding). 8 Similarly, another in-situ Raman study of MoDTC-containing lubricant by Okubo et al. 18 did not observe the presence of MoO3 on steel surfaces throughout the test. They estimated that the formation rate of MoS2 from MoDTC-containing lubricant is higher than MoO3 and suggested that MoO3 might be present on the worn steel surface, but in amorphous form and may not be Raman active. 18
The absence of lubricious MoS2 (no MoS2 Raman peaks were observed after 2.5 min of rubbing) tribofilm explains the high friction during the running-in period in Figure 13(a) and (b) for lubricant, L1 and L2 at 60 °C. The intermediate friction drop after 2.5 min of rubbing (Figure 13(a) and (b)) can be explained by the initiation in MoDTC decomposition and the formation of lubricious MoS2-rich tribofilm on the sliding interface (refer to Figure 7). Moreover, after 10 min of rubbing for lubricants L1 and L2, the low friction coefficient can be explained by the growth in MoS2-rich tribofilm (formation of more MoS2 sheets on the steel surface) over the rubbing time.
In Figure 8, the increasing trend for MoS2 A1g peak area intensity with increasing rubbing time was observed for both the lubricants L1 and L2. This indicates the conversion of more MoDTC into the MoS2 sheets or MoS2 tribofilm growth at the steel disc. 15 MoS2 assists in friction reduction due to its layer-lattice structure. 10 The weak Van der Waals attraction force between the lamellae of MoS2 imparts low shear strength and easy sliding between them. Under high contact pressure and shear stress, these lamellae easily slide over each other, thus enabling the reduction of boundary friction. 10
Effect of water on MoDTC performance
The effect of water on friction modifier (MoDTC) performance can be understood in terms of (i) friction response, (ii) interaction between water and MoDTC-derived MoS2 sheets, and (iii) tribofilm thickness. In Figure 13, the drop in friction coefficient (µ) with rubbing time is seen to correlate with the formation of lubricious MoS2 sheets rich tribofilm on the steel surface. In Figure 13, the further reduction in µ for both tested lubricants corresponds to the growth of MoS2 tribofilm, as indicated by A1g peak area intensity. For the low friction section (from 10 to 60 min), the gradual decrease in µ and increase in MoS2 A1g peak area intensity were observed for both lubricants (L1 and L2). However, for lubricant L2 (with added water), the higher friction coefficient (µ = 0.054) and lower MoS2 A1g peak area intensity were observed compared to lubricant L1 (µ = 0.044). This higher friction coefficient and lower peak area intensity for lubricant L2 indicate that the presence of water hinders the growth of MoS2 lubricious tribofilm in contact. TEM micrographs of tribofilm for lubricants L1 and L2 confirmed that the presence of water hampers the tribofilm growth, as a thinner tribofilm was observed for L2 compared to L1.
The negative effect of water on MoDTC friction response could be understood in two ways: (i) competition between water molecules and MoDTC molecules to interact with the steel surface, and (ii) interaction between water molecules and MoDTC-derived MoS2 sheets, as schematically shown in Figure 14. The friction-reducing properties of MoDTC depend on the formation of MoS2 sheets; specifically, lower concentrations of MoDTC result in a lower quantity of MoS2.15,21 Water (polar nature) molecules show the tendency to adsorb on steel surfaces preferentially, prevent additives from interacting with the steel surface and slow down the tribofilm growth.5,22–24 Therefore, the presence of water could hinder MoDTC molecules from effectively interacting with the steel surface under tribological conditions. It may cause a delay in the tribochemical reaction between MoDTC molecules and the steel surface and the formation of lubricious MoS2 sheets, which in turn leads to higher friction. Moreover, literature17–21 reported that the presence of water affects the MoS2 tribological performance. It has also been reported25–28 that water molecules physically interact with MoS2 sheets, adsorb on them, and promote bridging between the MoS2 sheets/layers. This increases inter-layer (lamellae) shear strength and prevents the easy sliding between MoS2 sheets and the formation of lubricious long MoS2 sheets, causing high friction and wear.25,29,30
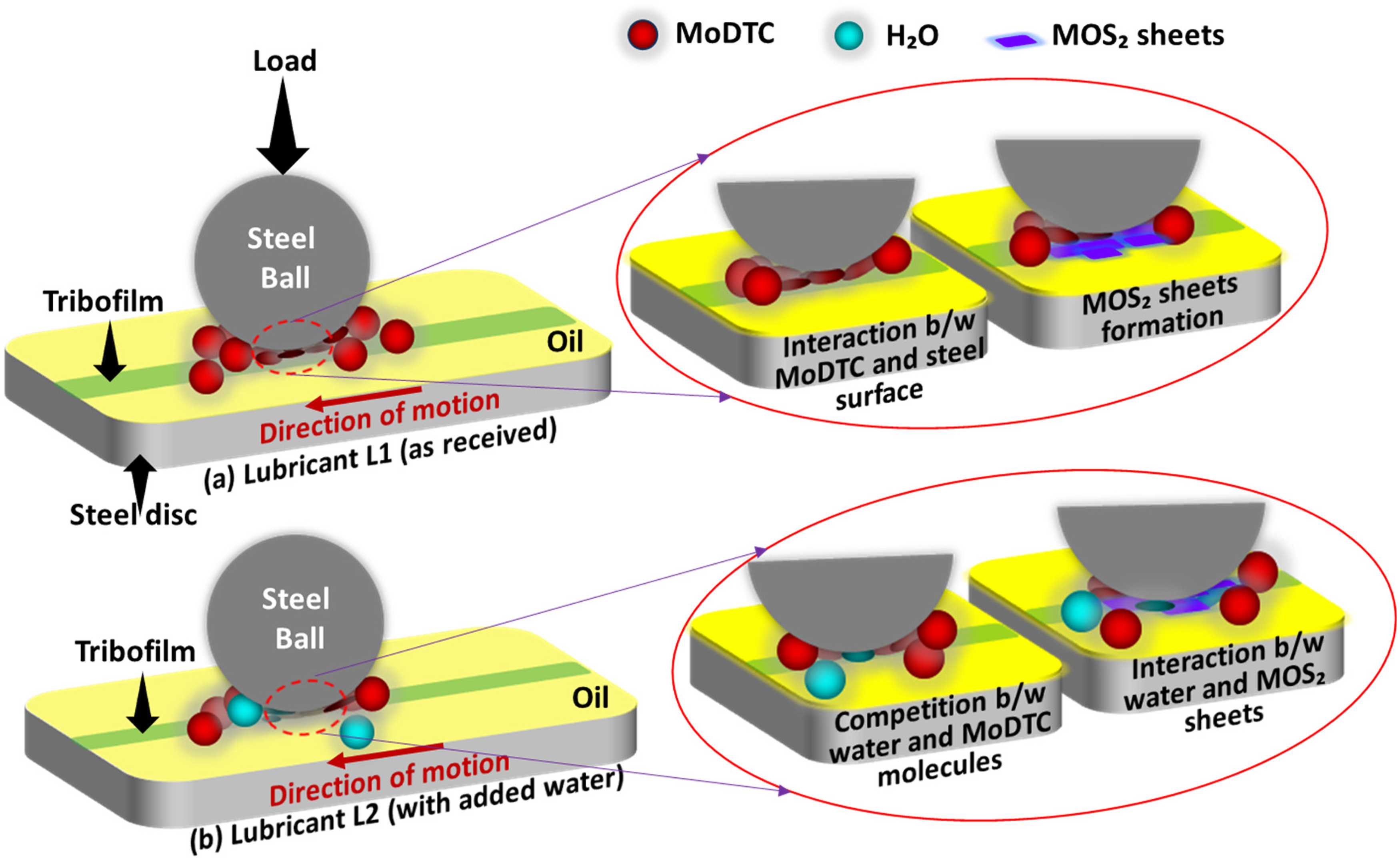
Schematic of possible interaction mechanism between (a) MoDTC (lubricant L1) and (b) MoDTC + water (H2O) (lubricant L2) with the steel surface.
Therefore, in the current study, it is anticipated that the delayed MoDTC decomposition and increased inter-layer shear strength between the MoS2 layers are responsible for higher friction for lubricant L2 (with added water) compared to lubricant L1 (as received).
The highest wear scar diameter (Figure 10) and thinner tribofilm (Figure 11) were observed for lubricant L2 (with added water) after 60 min of rubbing at 60 °C. The highest WSD for L2 could be due to the ineffectiveness of the ZDDP (anti-wear agent) present in lubricant additive packages; TEM-EDS analysis indicates the presence of ZDDP in lubricants. These findings are in line with previous studies.6,12,31 Dorgham et al.6,31 reported that water negatively interacts with the ZDDP and hinders the polymerization of the lubricious long phosphate chain (accelerates the depolymerization of the phosphate chain). This impacted the ZDDP performance in terms of tribofilm formation and anti-wear property. Similarly, Cen et al. 12 reported that the presence of water in oil is detrimental to the ZDDP and causes higher wear.
Conclusions
In this work, the effect of water contamination into lubricating oil on tribochemistry and friction properties of MoDTC is investigated using a bespoke ball-on-disk tribometer coupled with Raman spectroscopic analysis of the worn steel. Results indicate that water has a detrimental effect throughout the process of lubrication by affecting the growth of the MoS2 tribofilm and its physical structure. Other key conclusions are:
MoS2 A1g peak area intensity measured over sliding time for lubricant containing water as a contaminant indicates that the presence of water hinders the MoS2 tribofilm growth. TEM micrograph of the cross-section of tribofilm formed at high temperature confirmed that the presence of water hampers the tribofilm growth. A relatively thinner tribofilm was observed for lubricant L2 (with added water) as compared to that of the lubricant L1 (as received). Raman analysis indicated that the water physically interacts with MoDTC-derived MoS2 sheets, as no additional peak or shift in MoS2 peaks was observed. The competitive participation between water and MoDTC molecules and increased inter-layer shear strength between the MoS2 layers is responsible for high friction and wear. The increment of 22.5% in friction coefficient for the last 50 min of rubbing and 48.9% in wear scar was observed for the lubricant L2 (with added water) compared to the lubricant L1 (as received) at 60 °C.
Footnotes
Acknowledgements
The authors acknowledge the funding received from the EPSRC Prosperity Partnership Preventing Surface Degradation in Demanding Environments (EP/R00496X/1) and Tribology as an Enabling Technology (TRENT, EP/S030476/1) grants to support this work. The authors are also grateful to Dr Zabeada Aslam, Mr Stuart Micklethwaite, and Leeds Electron Microscopy and Spectroscopy Centre (LEMAS), Bragg Centre for Materials Research, University of Leeds, for assisting in conducting FIB and TEM-EDS analysis.
Author contributions
Ajay Pratap Singh Lodhi: Conceptualization, Visualization, Methodology, Investigation, Formal analysis, Data curation, Writing-original draft. Alaaeddin Al Sheikh Omar: Methodology, Investigation, Formal analysis, Data curation, Writing-original draft. Chun Wang: Methodology, Investigation, Formal analysis, Data curation, Writing-original draft, Khurshid Ahmad: Methodology, Investigation, Formal analysis, Data curation, Writing-original draft and Ardian Morina: Supervision, Conceptualization, Visualization, Methodology, Writing-original draft.
Data availability
Data included in this work will be made available upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the EPSRC Prosperity Partnership Preventing Surface Degradation in Demanding Environments, EPSRC Tribology as an Enabling Technology, (grant number EP/R00496X/1, EP/S030476/1).
