Abstract
Nickel-based superalloys (e.g., Inconel 718) are widely used in the aerospace industry due to their high temperature constancy in terms of mechanical properties and chemical stability. With the recent advances in sustainable aviation, there is a strong desire to better understand their compatibility with hydrogen combustion or water vapor in gas turbine engines. Therefore, this study investigated the tribological behavior of Inconel 718 under dry and water vapor conditions at various temperatures. The reciprocating ball on flat type tribometer was used to perform friction tests on Inconel 718 against alumina and Inconel 718 counterballs in the absence or presence of water vapor at room and elevated temperatures. The results showed that water vapor caused a reduction in the friction and wear at room and elevated temperatures against the Alumina mating surface, when compared to the tests performed under dry conditions. The low friction and wear in water vapor was attributed to the formation of an aluminum trihydroxide (bayerite—Al(OH)3) tribofilm. On the other hand, Inconel 718 vs. Inconel 718 showed higher wear in the presence of water vapor at RT compared to that in dry conditions. Conversely, water vapor decreased wear at HT compared to HT dry. The higher wear was attributed to the lack of sufficient lubricious oxides formation on the wear tracks for Inconel 718 vs. Inconel 718 at HT.
Introduction
Nickel-based superalloys, particularly Inconel 718, are widely used in the aerospace industry because of their remarkable mechanical properties, including high strength, excellent corrosion resistance, and stability at elevated temperatures.1,2 Their high strength, resistance to chemical degradation, and wear resistance make them well suited for use in extreme environments, especially at high temperatures. However, their ability to maintain these properties under extreme conditions is sometimes hampered by poor tribological properties. 3 Studies have shown that the tribological performance of Inconel 718 under dry sliding conditions was significantly influenced by the counterface material, temperature, and sliding speed. 4 Zhen et al. 5 showed that the coefficient of sliding friction was slightly lower against Si3N4 than Inconel 718, while the wear rate was nearly independent of the counterfaces of Si3N4 and Inconel 718. Thirugnanasambantham et al. 6 found that Inconel 718 tends to exhibit high coefficients of friction under dry sliding conditions due to adhesive wear mechanisms. The coefficient of friction was found to increase with sliding velocity and normal load. Samuel et al. 7 found that Inconel 718 tends to exhibit high coefficients of friction under dry sliding conditions due to the increased intensity of delamination wear and oxidation rate of non-lubricative NiO. However, the friction coefficient decreases with increasing temperature up to a certain limit, beyond which oxidation and softening of the material leads to increased friction, which was observed by Lei Li et al. 8 Dhananjay Dubery et al. showed that the mechanical and wear properties of Inconel-718 alloy were influenced by the γ’ and γ'’ precipitates, as well as the Laves and δ phases. By regulating the morphology and distribution of these precipitates within the alloy's microstructure, it was possible to effectively control the tribological performance of Inconel 718 alloy. 9 Stott et al. 10 demonstrated that the coefficient of friction of nickel-based superalloys (such as Nimonic 75, Nimonic C263, Nimonic 108, and Incoloy 601) decreases at elevated (i.e., 200°C to 800°C). This reduction in friction can be attributed to the formation of protective oxide layers of NiO, CoO, FeO, Cr2O3, and NiCr2O4 on the wear surface. Similarly, Laskowski et al. 11 found oxidation in nickel-based alloys at high temperatures (500°C and 800°C). Inconel X-750 exhibited the lowest coefficient of friction (0.27) when tested against Rene 41 at 800°C, while Inconel X-750 versus Inconel 909 exhibited the lowest pin wear (2.84 × 10−6 mm³/N-m) at 500°C, probably due to the development of a durable and lubricating oxide film (Ni-Cr oxide) on the wear tracks. Sun et al. 12 fabricated Inconel 718 coatings by high pressure cold spray deposition and investigated the microstructure and tribological behavior at room and elevated temperatures. Oxides began to form on the surface of Inconel 718 coatings when the ambient temperature exceeded 500°C, with a NiFe₂O₄ spinel oxide layer forming at 600°C. Despite the reduction in friction at higher temperatures, wear rates increased at 100°C and 200°C due to reduced hardness and abrasive wear. Xiao, Wei-han, et al. 13 synthesized Graphene/Inconel 718 composites by selective laser melting showed significant improvements in both mechanical and tribological performance. The addition of graphene nanoplatelets increased the yield strength and tensile strength by 42% and 53%, respectively, while the coefficient of friction and wear rate decreased by 22.4% and 66.8%, respectively, due to increased hardness and the formation of a protective graphene layer on worn surfaces. Another way of improving the wear resistance was to use of lubricants, such as oils or solid lubricants (e.g., graphite and molybdenum disulfide), which significantly reduced the coefficient of friction of Inconel 718 as reported by Zhen Jinming et al.. 14 The lubricants formed a protective layer that minimizes direct metal-to-metal contact. 14 In addition, Kaili Feng et al. 15 found that in Inconel 718 oxidation of spinel structure results in reduction of coefficient of friction and wear rate.
With the recent advancement in sustainable aviation, the demand for nickel-based superalloys (e.g., Inconel 718) to be compatible with sustainable aviation fuels such as hydrogen has been increasing recently. 16 However, the integration of hydrogen into propulsion systems, including gas turbine engines, poses unresolved challenges. One such challenge is the potential impact of hydrogen combustion and its product, water vapor or steam, on the materials used in these engines. Lai Ping et al. 17 conducted fretting test of Inconel 690 against 405 SS plate at RT, 90°C, 285°C, and 350°C. The wear depth of Inconel 690 first increased and then decreased as the water temperature increased from RT to 350°C, with the maximum value occurring at 90°C among these test temperatures. Similarly, Shengzan et al. 18 conducted fretting test of Inconel 690 against 304 SS plate at 100°C, 200°C, and 285°C. The authors’ found that the oxide film on Inconel 690 consists of an inner layer of Cr2O3 and an outer layer of spinel oxides such as NiFe2O4. In a vapor condition, the oxide layer continued to grow, reduced its protective effect, while in the presence of water, the growth of the oxide layer was limited, maintained a protective glaze layer. Additionally, in the presence of water in the contact, fretting wear damage was limited by the glaze layer, whereas in steam, the wear volume decreased up to 200°C and then increased, with more severe damage compared to water at all temperatures. 18 Additionally, Fretting wear of Inconel 690 steam generator tubes against AISI 420 pads in air was primarily caused by gross slip, with the maximum wear depth being a better indicator of wear progression than Archard's wear coefficients, and wear debris at 106 cycles consisting of (Ni,Fe)(Fe,Cr)₂O₄ nanoparticle agglomerates. 19 Jeong et al. 20 demonstrated that the wear of Inconel 690TT in air was primarily due to delamination, while in water the wear was affected by micro pitting. They also found that fretting wear rates in water increased with increasing water temperature. Furthermore, they found that in the sliding test, the friction coefficient in air was higher than that in water. In addition, the friction coefficient and wear rate increased as the temperature of the water increased in the water environment. Also, Guo et al. 21 investigated that with increasing sliding amplitude and normal load, the wear volume of alloy 690 tubes in high-temperature water increased, shifting the wear mode from abrasive to delamination wear, while corrosion accelerated as mechanical wear broke the protective oxide scale on the contact surface.
This study investigates the impact of water vapor on the tribological behavior of Inconel 718, an area that has not been previously explored in detail. More specifically, the aim of this study was to evaluate the tribological behavior of a nickel-based superalloy, specifically Inconel 718, against various counterfaces at different temperatures under dry conditions and to compare these results with those obtained in the presence of water vapor. The specific focus was to elucidate the mechanisms underlying the lubricating behavior for water vapor conditions. Ex-situ analysis was performed using scanning electron microscopy (SEM), Micro-Raman spectroscopy, Fourier transform infrared spectroscopy (FTIR), etc.
Materials & method
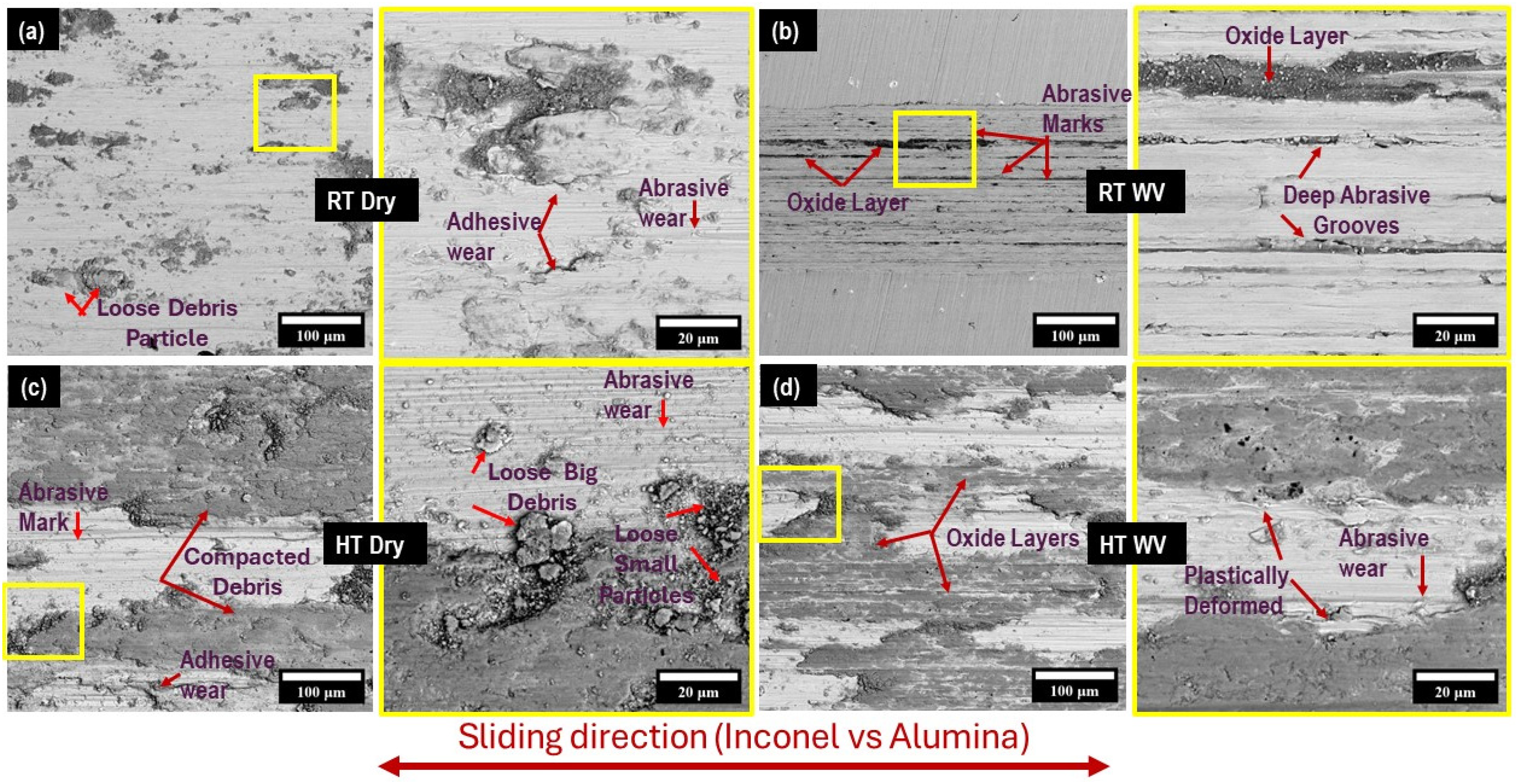
In this study, the nickel-based superalloy i.e., Inconel 718 softened annealed (McMaster, USA) with the dimensions of 6”×6”×0.25” sheet was procured and then precision cut into 1”×1”×0.25” samples using water jet cutting machine (Umake, Canada). The chemical composition of Inconel 718 is given in Table 1. 22
Chemical composition of Inconel 718 (reprinted with permission). 22
The Inconel 718 specimens were polished with 220 to 1200 grit SiC paper followed by 9 μm, 5 μm, and 1 μm water-based diamond suspensions in an automatic polishing machine (Struers, Canada). A 0.4 μm colloidal silica suspension was used as the final step in the polishing machine.
The wear test was performed on the polished Inconel 718 samples (i.e., < 1 μm) against Alumina and Inconel 718 counterballs (i.e., Ø ∼ 6.35 mm or radius 3.175 mm) in dry and in the presence of water vapor at two different temperature conditions (25°C-RT, 450°C-HT) using a ball-on-flat reciprocating tribometer (Anton Paar TRB, 3 Switzerland). A cool mist humidifier was used to turn the distilled water into water vapor which was then redirected to the contact point of the counterball and substrate using a flexible tube as shown in Figure 1. The supply was controlled by the humidifier, which was set as a default rate of water vapor production, i.e., 100 ml of water-to-water vapor in one hour. The wear test parameters (i.e., load 5 N, track length 10 mm, and total sliding distance 100 m) and temperatures (i.e., 25°C and 450°C) were determined based on the conditions of the maximum capability of the equipment and based on experience with prior testing of materials for tribological interfaces in gas turbine engines. The hardness of the Inconel 718 was Rockwell B95 23 whereas the Alumina was Rockwell 45N 83. 24

Ball on flat reciprocating type tribometer in the presence of water vapor.
The room temperature tribological tests in dry conditions and in the presence of water vapor were designated RT Dry and RT WV, respectively. In addition, the friction tests at elevated temperatures in dry and in the presence of water vapor were designated HT Dry and HT WV, respectively. The test parameters were a load of 5 N, a frequency of 1 Hz, a stroke length of 10 mm, and a total sliding distance of 100 m (5000 cycles). The parameters were selected based on the contact conditions in aerospace engines.25,26 The detailed procedure of the friction tests at room and elevated temperatures can be found elsewhere. 27 After the sliding tests, the wear depths were measured using confocal laser microscopy (Olympus LEXT, Japan). A total of four repeats were performed, and the average value was presented in this study.
Field emission scanning electron microscopy (FESEM) (Hitachi, Japan) coupled with energy dispersive X-ray spectroscopy (EDS) was used to observe the wear tracks and counterballs after the friction test. The wear track samples were mounted and cut perpendicular to the length of the wear track. Then, the polished cross-sections were analyzed under SEM to reveal the changes in the subsurface.
Micro-Raman spectroscopy (CNI MGL-U-532, China) was performed on the unworn and worn surfaces using a laser with a wavelength of λ = 532 nm to detect the oxide phases, and the spectra were identified based on published literature. Additionally, FTIR spectroscopy using Attenuated Total Reflectance (ATR) (Nicolet IS20, Thermo Fisher Scientific, USA) was performed on the samples to detect the presence of phases. A fresh unworn Alumina and Inconel counterball was used as a reference baseline to identify the different peaks.
Results
Friction behavior
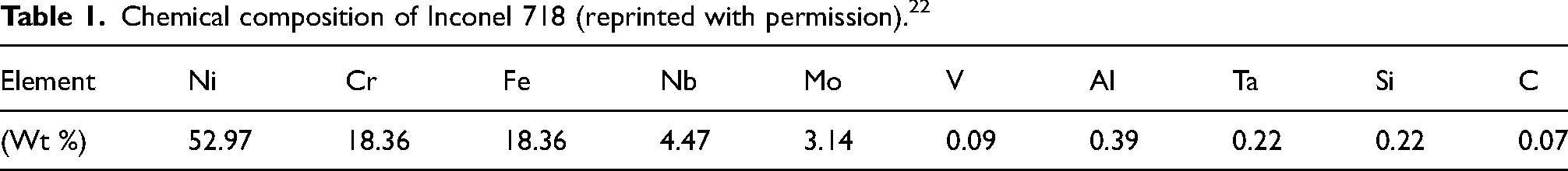
Figure 2 shows the friction characteristics of Inconel 718 vs. Alumina and Inconel 718 vs. Inconel 718 samples in dry and in the presence of water vapor at room and elevated temperatures. First, focusing on Inconel 718 vs. Alumina in Figure 2 (a), at RT dry conditions a steady and consistent friction value of ∼ 0.8 was observed. However, when water vapor was introduced at RT, the friction coefficient dropped to an average of ∼ 0.35. Also, the friction remains quite smooth and steady throughout the test at this condition, similar to RT dry. On the other hand, the coefficient of friction for the HT samples was not stable over the test duration. The coefficient of friction for HT dry and water vapor exposed samples were ∼ 0.75 and ∼ 0.70 respectively at the beginning of the test and increased to ∼ 0.90 and ∼ 0.80 at 500 cycles. Subsequently, the coefficient of friction decreased to ∼ 0.65 and 0.55 at 1500 cycles, and finally, both reached the plateau of ∼ 0.65 at 3500 cycles, which was stable until the end of the friction test. In summary, the sample exposed to water vapor at room temperature exhibited the lowest average coefficient of friction compared to dry and elevated temperature conditions. In addition, the friction curve was smooth in the presence of water vapor compared to dry conditions.
Coefficient of friction of (a) Inconel 718 vs. Alumina counterball, (b) Inconel 718 vs. Inconel 718 in dry and in the presence of water vapor (Dry and WV), at room and elevated temperatures (RT and HT).
The friction behavior of Inconel 718 when tested against Inconel 718 counterballs in dry and in the presence of water vapor at room and elevated temperatures is shown in Figure 2 (b). It was observed that the coefficient of friction of Inconel 718 vs. Inconel 718 at RT was ∼ 0.85, which was slightly higher than that of Inconel 718 vs. Alumina at RT (∼ 0.88). In the presence of water vapor, the friction decreased to ∼ 0.75 and remained constant throughout the friction test. While the frictional behavior of Inconel 718 vs. Inconel 718 at HT decreased with increasing sliding and reached a steady state after 2000 cycles. In the HT conditions, in dry and in the presence of water vapor, the friction was initially high, then decreased and remained steady from 1800 cycles to the end of the test at 5000 cycles. It can be seen that at HT, in the presence of water vapor, the frictional behavior of Inconel 718 vs. Inconel 718 was higher than the Inconel 718 vs. Alumina conditions.
Wear depth
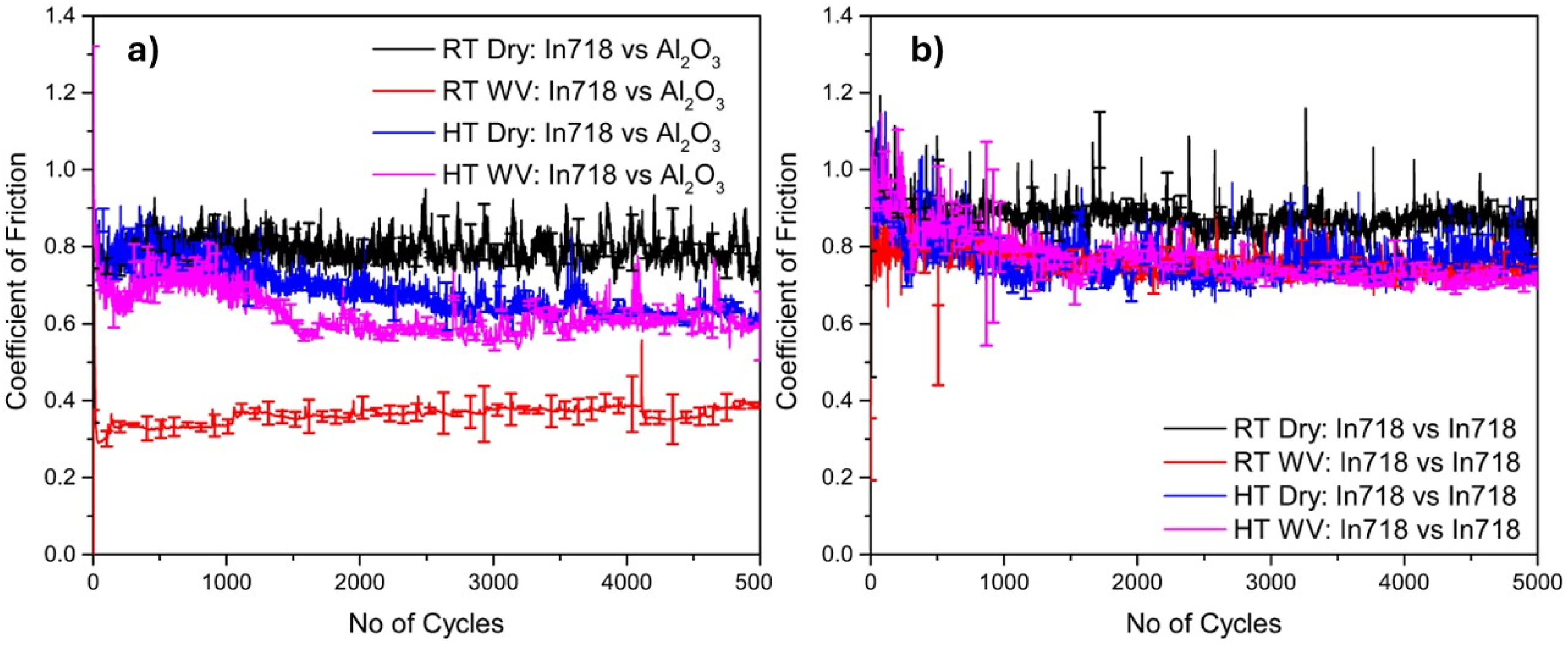
Figure 3 (a) shows the wear profiles for the Inconel 718 vs. Alumina and Inconel 718 vs. Inconel 718 at the end of the 5000-cycle friction test. At RT dry, the wear depth was ∼ 61 µm, which significantly decreased to a depth of ∼3 µm when exposed to water vapor at the same RT. In addition, at HT dry condition, the wear depth was ∼ 69 µm, which was the highest among the four conditions. However, at HT WV condition, the wear depth decreased to z ∼ 39 µm, which followed a similar trend as in RT water vapor condition.
Wear depth of (a) Inconel 718 vs. Alumina counterball, (b) Inconel 718 vs. Inconel 718 in dry and in the presence of water vapor (Dry and WV), at room and elevated temperatures (RT and HT).
The wear depth graph for Inconel 718 vs. Inconel 718 in dry and in the presence of water vapor at two different temperature conditions is shown in Figure 3 (b). It can be seen that at RT under dry condition, the wear depth was the lowest of all samples, ∼ 20 µm. Also, at RT, the wear depth increased to ∼ 39 µm when exposed to water vapor. On the other hand, at HT, the wear depth was generally higher than at RT, and decreased from ∼103 to ∼76 µm in the presence of water vapor compared to HT dry conditions. In all conditions, significant variations in wear depth along the respective wear tracks were observed in the Inconel 718 vs. Inconel 718 conditions that were not observed in the Inconel 718 vs. Alumina conditions.
Worn surface morphologies
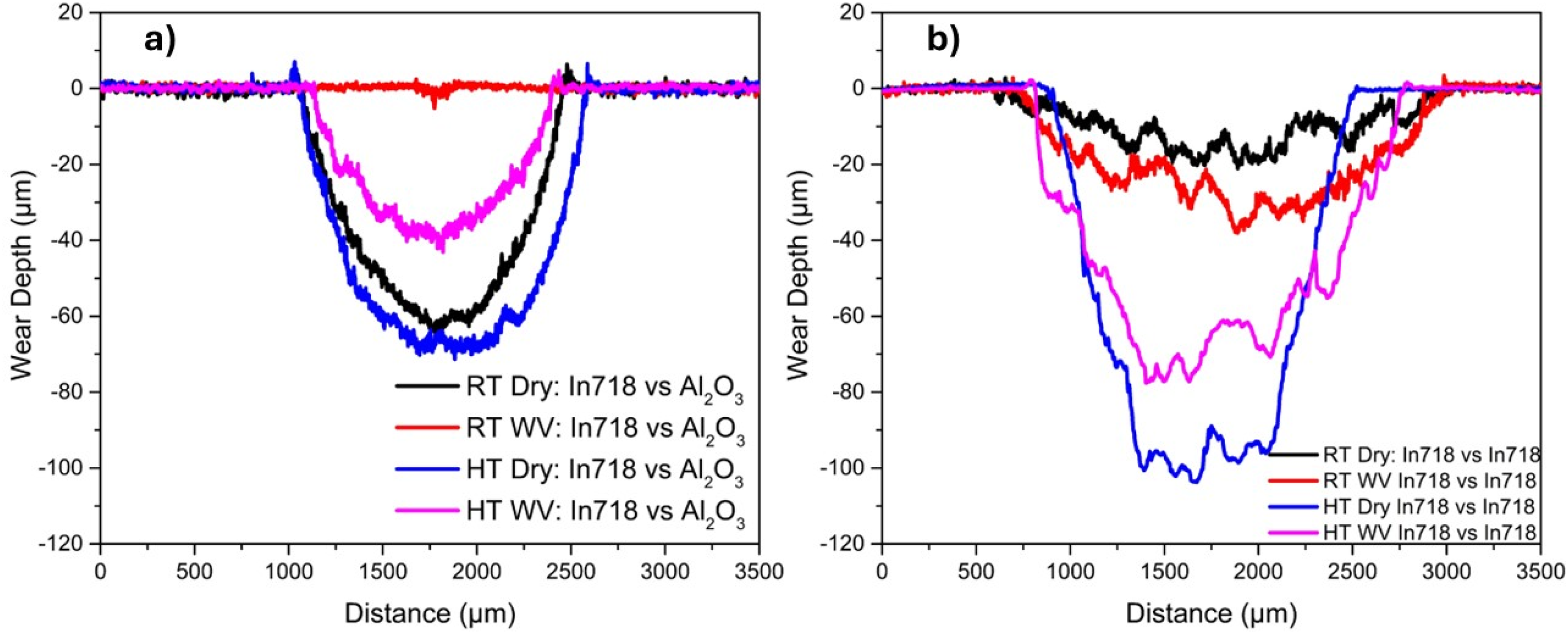
The SEM images of the wear tracks after testing under different conditions are shown in Figure 4. Figures 4(a-b) shows results from RT Dry and WV Inconel 718 vs. Alumina conditions. Note that the width of the wear track (i.e., 220 µm) of the RT WV conditions was much smaller than the width (i.e., 1400 µm) of the RT Dry conditions, as observed in Figure 3 (a). Under RT Dry, [Figure 4(a)], the SEM image shows the presence of wear debris particles throughout the wear track. In addition, the smeared layers as well as the presence of the black debris, which may be an oxide, can be seen in the wear track. On the other hand, at RT WV conditions [Figure 4(b)], the presence of deep abrasive grooves in the sliding directions and oxide layers along the deep grooves can be seen. Overall, it was appeared that abrasive type of wear was dominant in these RT WV conditions [Figure 4(b)], while adhesive wear was likely the dominant wear mechanism in RT dry conditions [Figure 4(a)]. The darker debris were the oxides of the nickel-based superalloy, as confirmed by SEM/EDS (not shown here).
SEM images of the worn surfaces of Inconel 718 vs. Alumina counterball (a) RT Dry, (b) RT WV (c) HT dry, (b) HT WV. Conditions in dry and in the presence of water vapor (Dry and WV), at room and elevated temperatures (RT and HT).

SEM images of the worn surfaces of Inconel 718 vs. Inconel 718 (a) RT Dry, (b) RT WV (c) HT dry, (b) HT WV. Conditions in dry and in the presence of water vapor (Dry and WV), at room and elevated temperatures (RT and HT).
Figure 4 (c-d) show the SEM images of Inconel 718 vs Alumina HT condition. The width of the wear track in HT dry (i.e., 1530 µm) was slightly higher than HT WV (i.e., 1260 µm) (as observed in Figure 3 (a)). In Figure 4(c), at HT Dry conditions, the abrasive marks along the sliding direction accompanied by a large area of compacted debris as well as wear debris particles were observed. In the case of HT WV, abrasive marks were present but less dominant than that of HT Dry condition. Moreover, the oxide layers were scattered all across the wear track [Figure 4(d)]. It can be said that both of conditions (i.e., Inconel 718 vs Alumina in RT & HT with WV) the oxide layers were present.
Figure 5 shows the SEM images of the wear tracks for Inconel 718 vs. Inconel 718 conditions after the sliding test in dry and in the presence of water vapor at room and elevated temperatures. The RT dry condition showed evidence of deep abrasive grooves along the sliding direction and with potentially embedded debris particles throughout the wear track [Figure 5 (a)]. On the other hand, in the presence of water vapor, potential oxide layer formation was visible on the wear tracks along with abrasive marks and embedded zone [Figure 5 (b)]. The RT WV sample wear track was also plastically deformed and has significant oxide layer black zone. More or less similar features to the RT conditions were observed in all wear tracks for Inconel 718 vs. Inconel 718 HT Dry and HT WV conditions. However, the wear tracks for the HT conditions were much smoother than the RT conditions [Figure 5(a-b)]. In all conditions, the wear tracks were plastically deformed.
Raman analysis
Micro-Raman spectroscopy was performed on the Inconel 718 wear tracks after the room and elevated temperature sliding tests, in dry and in the presence of water vapor, for both the Inconel 718 vs. Alumina and Inconel 718 vs. Alumina conditions, as shown in Figure 6. For Inconel 718 vs Alumina shown in Figure 6 (a), characteristic peaks were observed at wavenumbers of 500 cm−1 for RT & HT dry and water vapor condition, and 570 cm−1 in HT dry condition, corresponding to NiO and Cr2O3, respectively. 18 In addition, the observed characteristic peaks at the Raman shift of 670–680 cm−1 correspond to NiFe2O4.14,18
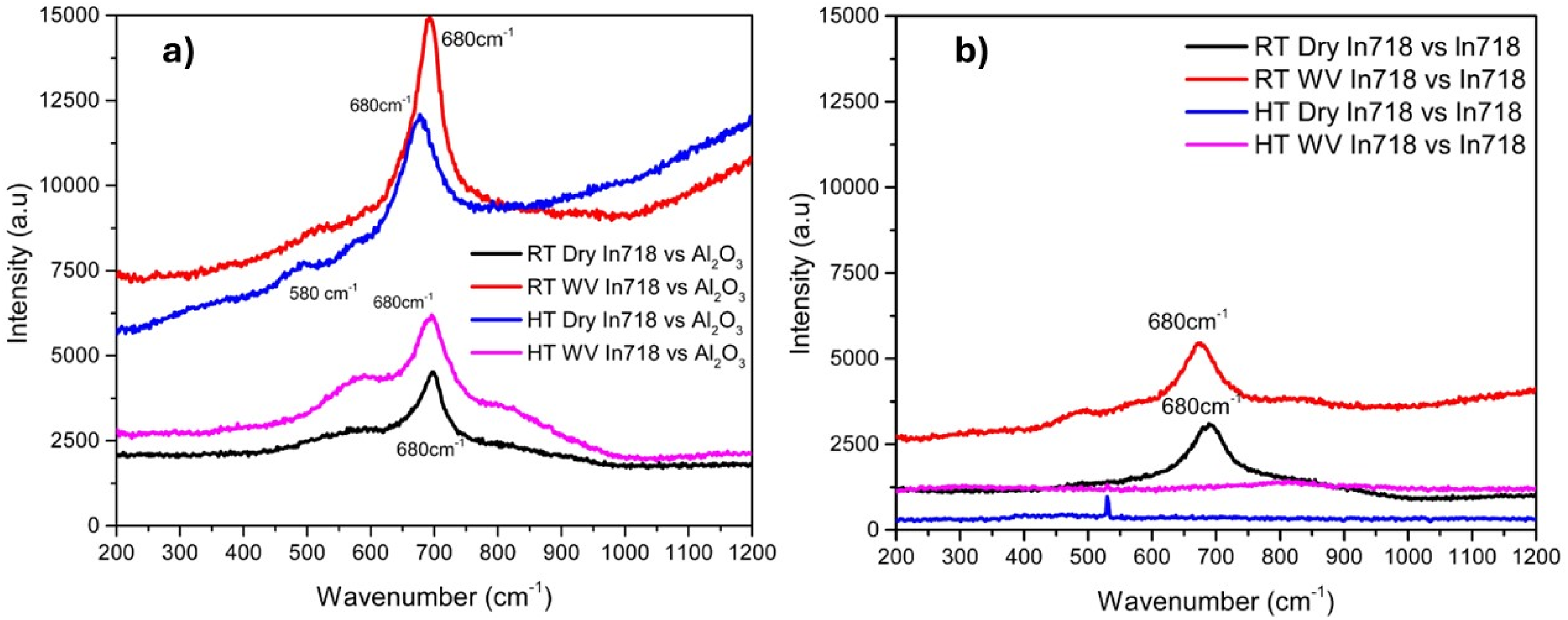
Raman analysis of (a) Inconel 718 vs. Alumina counterball, (b) Inconel 718 vs. Inconel 718 in dry and in the presence of water vapor (Dry and WV), at room and elevated temperatures (RT and HT).
Micro-Raman analysis of the Inconel 718 vs Inconel 718 wear tracks is shown in Figure 6 (b). Interestingly, only RT dry and water vapor conditions showed the Raman peak, but no peaks were observed for HT conditions. In RT water vapor conditions, the characteristic Raman shifts at 500 cm−1 and 680 cm−1 correspond to NiO and NiFe2O4, respectively.18,28,29
SEM analysis of counterball
SEM images of the Alumina counterball running against Inconel 718 are shown in Figure 7 after sliding tests under RT Dry and RT WV conditions. The RT Dry conditions show greater elliptical contact compared to the RT WV conditions, seen in Figure 7 (a-b). The RT WV showed the smeared transfer film along with the cracks on it, while the RT Dry counter ball has larger and loose material transfer from the Inconel 718 substrate [Figure 7(a-b)]. In Figure 7 (c-d) it can be seen that the HT Dry conditions resulted in a smeared transfer film on the Alumina counter ball, whereas for the HT WV conditions the transfer film was within the contact and outside of the contact zone. This transfer film outside of the contact may have spread away from the contact zone during sliding.

SEM images of the worn surfaces of Counterball of Inconel 718 vs. Alumina counterball (a) RT dry condition, (b) RT Water Vapor condition (c) HT dry condition, (b) HT Water Vapor condition.
SEM images of the Inconel 718 counterball running against Inconel 718 are shown in Figure 8 after sliding tests under RT Dry and RT WV conditions. It can be seen from Figure 8(a-b) that the RT Dry sample has a slightly larger contact area than the RT WV sample and both were circular in shape. Both samples have deep abrasive grooves along the sliding direction. Remarkably, the RT WV sample has much more loose debris on the worn surface. From Figure 8(c-d), the HT conditions resulted in a counter ball contact point with an elliptical shape. In this case, the HT Dry specimen has smaller contact area than HT WV specimen. In HT Dry conditions, the contact was in elliptical shape with smeared transfer film, whereas HT WV counterball has the embedded debris particles on the counterface.

SEM images of the worn surfaces of Counterball of Inconel 718 vs. Inconel 718 counterball (a) RT dry condition, (b) RT Water Vapor condition (c) HT dry condition, (b) HT Water Vapor condition.
FTIR analysis
The FTIR analysis was performed on the Alumina and Inconel 718 counterball after sliding against Inconel 718 substrate in dry and in the presence of the water vapor at room and elevated temperatures. Figure 9 (a) shows the FTIR graph on the Alumina counterball at RT Dry and RT WV at different temperatures. The wavenumber 3270 cm−1 represents the characteristic of –OH stretching vibration which was bonded to Al. The -OH stretching existed from 3200–3700 cm−1.30,31 The Alumina counterball after sliding in RT WV & HT WV conditions exhibited the -OH stretching [Figure 9(a)]. The unworn Alumina counter ball having wavenumber 3400 cm−1 also represents the characteristic of –OH stretching vibration is bonded to Al (the usual stretching is from 3200–3700 cm−1). All the conditions except Inconel 718 in 450°C dry condition have a peak at wavenumber 1080 cm−1 representing Al-O. 32
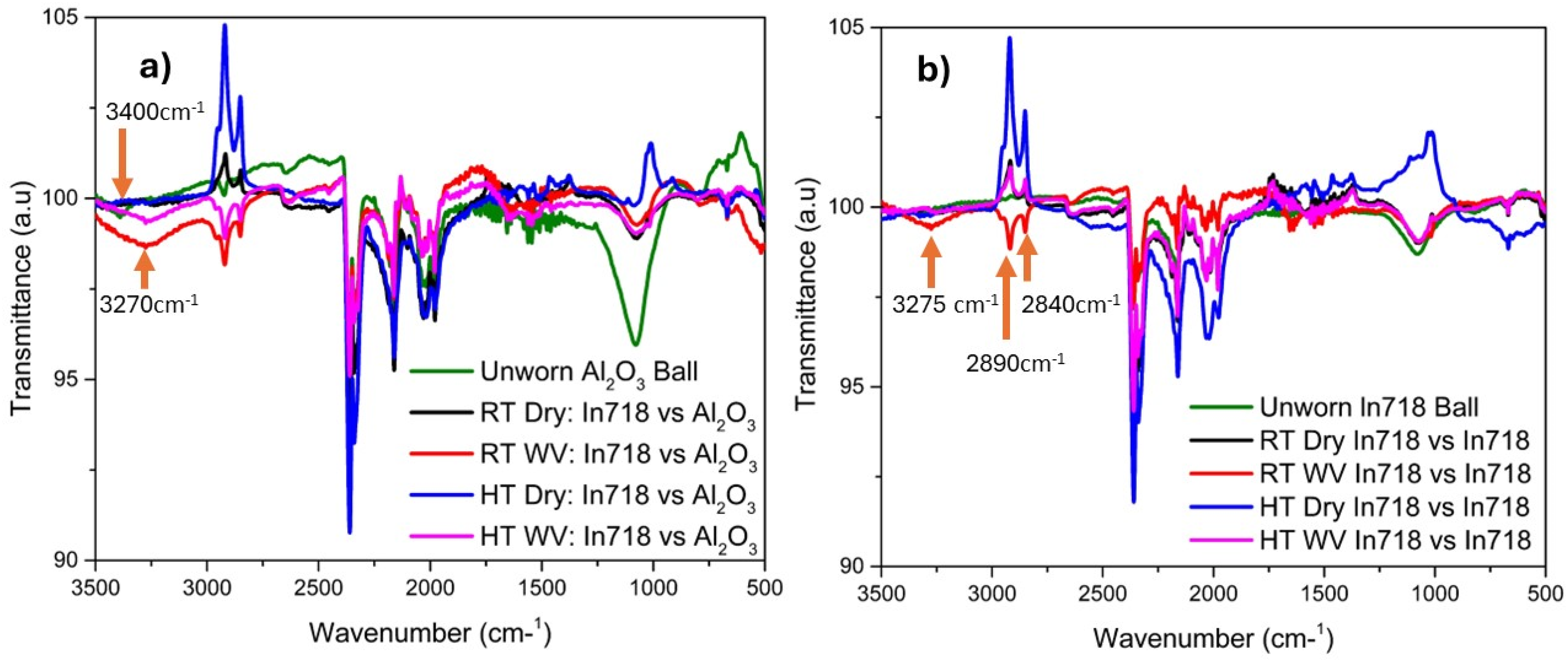
FTIR analysis of counterball (a) Inconel 718 vs. Alumina counterball in dry and in the presence of water vapor at room and elevated temperature, (b) Inconel 718 vs. Inconel 718 in dry and in the presence of water vapor, at room and elevated temperatures.
Figure 9 (b) shows Inconel 718 in room temperature WV condition has wavenumber 3270 cm−1 represents the characteristic of –OH stretching vibration that is bonded to Al30–32 (the usual stretching is form 3200–3700 cm−1). All the condition except Inconel 718 against Inconel 718 in 450°C dry condition have wavenumber 1080 cm−1 representing Al-O. 32
SEM cross-section of wear tracks
SEM images of the wear track cross section perpendicular to the sliding direction for the Inconel vs. Alumina condition are shown in Figure 10. For the RT Dry condition, the cross section of the wear tracks appeared with pits, debris embedment near the surface after the sliding tests [Figure 10 (a)]. These phenomena could be due to the appearance of debris, striations mark and adhesive behavior of the wear track surfaces [Figure 4 (a)]. On the other hand, cracks and peeling were observed on the cross section of the wear tracks after wear tests at RT water vapor conditions [Figure 10 (b)], which could be due to the presence of oxide layers and deep abrasive grooves on the wear track [Figure 4 (b)]. Similar wear mechanisms were observed for Inconel 718 samples when sliding against silicon nitride ball by Xu et al.. 33 The detachment of the compacted large debris and the ploughing phenomena were observed for the HT dry condition [Figure 10 (c)], which correlated well with the observations from the worn surfaces [Figure 4 (c)]. The presence of irregularities (peaks and valleys of the surface of the wear tracks) [Figure 10 (d)] can be correlated with the irregular distribution of oxide layers on the surface of the wear tracks [Figure 4 (d)].

SEM image of cross section of wear track of Inconel 718 substrate vs Alumina counter ball in (a) RT dry condition (b) RT exposed to water vapor condition, (c) HT in dry condition (d) HT exposed to water vapor condition.
Figure 11 shows the SEM wear track cross section after sliding in the Inconel 718 vs. Inconel 718 conditions. The SEM wear track cross section of RT dry conditions shows the smooth and flat features, which could be due to the continuous removal of materials during sliding as the smooth, plastic deformation behavior appeared on the wear track surface [Figure 5 (a)]. On the other hand, the irregularities of the cross section of the wear track [Figure 5(b)] could be related to the compacted and smeared layer on the wear surface when water vapor was introduced during sliding at RT [Figure 5 (b)]. Both conditions (HT dry and HT WV) appeared to have grooves, pits [Figure 11 (c-d)], which could be related to the deep abrasive grooves and plastically deformed wear surface [Figure 5 (c-d)].

SEM image of cross section of wear track of Inconel 718 substrate vs Inconel 718 counter ball in (a) Room temperature (RT) dry condition (b) Room temperature (RT) exposed to water vapor condition, (c) Elevated 450°C (HT) in dry condition (d) Elevated 450°C (HT) exposed to water vapor condition.
Discussion
Friction and wear behavior of Inconel against alumina counterface
The coefficient of friction of the Inconel 718 in the dry sliding condition was ∼0.8 with high fluctuation at RT [Figure 2(a)]. The high friction and fluctuation could possibly be explained by the formation of NiO, NiFe2O4, and Cr2O3 as well as the increased amount of adhesion due to the lack of continuous formation of a protective tribofilm (e.g., in the form of lubricious oxides) on the wear tracks during sliding. 33 The presence of non-lubricious oxides i.e., NiO, NiFe2O4, and Cr2O3 as well as the high amount of adhesion contributed to the transfer of material to the mating surfaces, which may have further contributed to the high wear [Figure 4 (a); Figure 7(a); Figure 10(a)]. Indeed, the high friction and wear at room temperature was likely due to the formation of non-lubricating oxides (i.e., NiO, NiFe2O4, and Cr2O3) on the wear tracks as observed by Raman analysis [Figure 6 (a)] and claimed by the previously published articles.28,29
On the other hand, the friction coefficient of the Inconel vs Alumina in the presence of water vapor was ∼0.35 at RT, which was approximately 60% less friction than in the dry condition at RT. The reduced friction can be attributed to the formation of a lubricious tribofilm (i.e., aluminum trihydroxide (bayerite—Al(OH)3)) as shown in Figure 12, which minimized the direct contact between two bodies, resulting in reduced friction and wear. Additionally, the lubricious film can contribute to a decrease in the real contact area and high contact stress [Figure 4; Figure 7] which corelated well with the smooth steady state coefficient of friction throughout the test [Figure 2(a)]. Similar results were found by Gates RS et al. 34 where the authors observed a significant reduction in friction and wear when water was used as a lubricant in a four-ball apparatus. Moreover, their study showed that transition (gamma) Alumina reacted with water to form aluminum hydroxides. 34 These can be correlated with the black oxide zones and abrasive grooves on the wear tracks [Figure 4 (b); Figure 10 (b)] and the Al-O phases on the counterball which was observed by the FTIR analysis [Figure 9 (a)].

Lubricating mechanisms of Inconel against Alumina counterface in the presence of water vapor at room temperature.
Under HT conditions (i.e., Dry and WV), the coefficient of friction was initially high within the first 1000 cycles and then decreased as sliding continued, reaching ∼0.65 at the end of the test. The high friction upon initial sliding could be due to the direct contact between the two bodies at high temperatures, which could increase the amount of adhesion. Such a behavior has been previously observed for two body contacts resulting in a sharp increase in friction coefficient. 35 Subsequently, the low friction after 2000 cycles was likely due to the formation and depletion of hard oxides on the wear tracks. 36 The presence of hard oxides could be related to the high hardness resulting in the reduction of the coefficient of friction, which has been observed previously.37,38 However, the formation of hard oxides, as seen in the images of the wear tracks, could cause two or three body abrasion during sliding and consequently high wear, which corelated well with previous studies [Figure 4 (c-d), Figure 6 (a)]. 39
Interestingly, the wear depth of Inconel 718 against Alumina was less under the HT water vapor conditions than under the HT dry conditions. The lower wear with smooth profile of the HT WV may be due to the formation of lubricious aluminum oxide hydroxide. The presence of the Al-O phase could be correlated with the aluminum oxide hydroxide (boehmite-AlO(OH)) [Figure 9 (a)] 40 . A similar phase i.e., AlO(OH), was observed by Gates et al. above 200°C in the interaction of water vapor with Alumina 31 . However, it can be seen that aluminum trihydroxide (bayerite—Al(OH)3) formed at RT WV conditions was much more effective than AlO(OH) in improving the tribological performance for Inconel 718.
Friction and wear behavior of Inconel against Inconel counterface
Under RT dry conditions, the coefficient of friction was approximately ∼ 0.9 on average over 5000 cycles [Figure 2 (b)]. Higher contact area [Figure 8 (a)] and the formation of hard ternary oxides, i.e., NiFe2O4 [Figure 6 (b)], on the wear track could lead to high coefficient of friction. Furthermore, the presence of non-lubricious oxides can lead to the formation of grooves, release of wear debris and plastic deformation [Figure 5 (a); Figure 11 (a)]. When water vapor was introduced into this condition, the friction was reduced to ∼ 0.8 [Figure 2 (b)], which may be due to reduced frictional heating at the contact interfaces. Additionally, the formation of non-lubricious oxides i.e., NiFe2O4 [Figure 6 (b)] on the wear tracks caused a relatively high wear depth of about ∼ 39 µm [Figure 3 (b)]. Similar results were obtained by R.S. Gates et al., 34 where they performed four ball tests with water vapor. In addition, the excessive plastic deformation, dominant adhesive behavior compared to abrasion, and release of large amount of loose debris contributed to the high wear [Figure 5 (b); Figure 11(b)]. Overall, the friction of the Inconel 718 vs. Inconel 718 combinations was higher than the Inconel 718 vs. Alumina combinations at both room and elevated temperatures. This could be due to the variation in the mechanical properties of the mating surfaces, i.e., the higher hardness of the Alumina resulted in a lower contact area than Inconel 718 which resulted in a higher contact area,41,42 apart from the reasons explained above and in section 4.1. In addition, the chemical behavior of the tribo-pairs could alter the friction and wear characteristics of Inconel 718. Rabinowicz et al. 43 showed that dissimilar materials (i.e., Inconel 718 vs. Alumina) have low adhesion compared to similar materials (i.e., Inconel 718 vs. Inconel 718) resulting in low wear.
For the HT dry and water vapor conditions, the coefficient of friction behaved almost similarly from the beginning to the end of the test. For both conditions, the coefficient of friction dropped steadily from ∼0.95 to ∼0.8 at about 1500 cycles [Figure 2]. It then remained constant at about 0.8 up to 5000 cycles. However, in the water vapor condition, the fluctuation of the friction curve was smaller than in the dry condition. This could be due to the presence of water vapor in the contact conditions, whereas in the dry conditions, the disruption or removal of the oxides from the contact caused a higher fluctuation. In the case of the wear behavior, the wear depth of HT dry was ∼26% higher than that of HT water vapor conditions. The high wear and irregular profiles of HT dry could be due to the absence of lubricating oxides in the contact conditions as the oxides were continuously removed during sliding. Also, the high wear could be due to the direct metal to metal contact (i.e., Inconel 718 to Inconel 718), which increased the adhesion during sliding due to their high solubility. 43 This can be well correlated with the presence of plastic deformation, smeared layer and the deep abrasive grooves on the wear track and counterballs under HT dry conditions [Figure 5(c); Figure 8 (c)]. In addition, the removal or delamination of the oxide layer as shown in [Figure 11 (c)] could increase the wear during sliding. This corelates well with the Raman analysis where no peaks of oxides were observed [Figure 6 (b)]. On the other hand, for water vapor condition, almost similar features as HT Dry were observed on the wear tracks, cross section of the wear tracks, Raman and FTIR analysis [Figure 6(b), Figure 9(b)].
Conclusions
Tribological evaluation was performed on the nickel-based superalloys (e.g., Inconel 718) in dry and in presence of water vapor at room and elevated temperatures. It was shown that the Inconel 718 vs. Alumina condition in the presence of water vapor at RT was superior to the other seven conditions in terms of tribological performance.
Friction and wear were significantly lower in the RT WV condition than in the RT dry condition. This was due to the formation of an aluminum trihydroxide (bayerite—Al(OH)3) tribofilm on the wear tracks and the small contact area, whereas the absence of lubricious oxides and greater adhesion caused the high friction and wear for RT dry. Similar trends were observed for HT conditions in dry and water vapor environments, although wear for HT WV was less than HT dry due to the presence of aluminum oxide hydroxide (AlO(OH)). However, friction and wear were higher for HT than for RT. Inconel 718 vs. Inconel 718 showed higher wear in the presence of water vapor at RT than at RT dry conditions. This may be due to less frictional heating at the contact interfaces. Conversely, water vapor decreased wear at HT compared to HT dry. The higher wear was attributed to the lack of sufficient lubricious oxides formation on the wear tracks for Inconel 718 vs. Inconel 718 at HT. This may be due to the dominant adhesive nature of the Inconel vs. Inconel conditions rather than the abrasive wear mechanisms of Inconel vs. Alumina.
Footnotes
Acknowledgements
The authors thank the Taiho Kogyo Tribology Research Foundation (TTRF) for supporting this project. The authors also thank Dr. Fadhel Ben Ettouil for his assistance with characterization techniques.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Taiho Kogyo Tribology Research Foundation, (grant number N/A).
