Abstract
Background:
Immunoglobulin A vasculitis (IgAV) is a systemic small-vessel vasculitis characterized by palpable purpura, often accompanied by joint, gastrointestinal, and/or renal involvement. Although cutaneous manifestations are usually mild and self-limited, they can occasionally present as persistent, severe, or recurrent skin lesions.
Objectives:
To assess the efficacy and safety of colchicine in the treatment of complicated cutaneous manifestations of IgAV.
Methods:
We conducted a retrospective chart review of patients treated with colchicine for skin manifestation of IgAV at our vasculitis clinic. This was complemented by a search of the PubMed and MEDLINE databases to identify additional reported cases.
Results:
A total of 30 cases were included, comprising 13 from our institution and 17 from the literature. Indications for colchicine use were persistent purpura lasting more than 4 weeks (n = 18), severe skin involvement including bullae, ulcerations, or widespread purpura (n = 9), and recurrent lesions after a symptom-free interval of at least 1 month (n = 8). Colchicine led to clinical improvement in 90% of patients (27/30), with complete resolution in 67% (20/30). All 12 patients treated with colchicine monotherapy responded favorably. Doses ranged from 0.6 to 1.8 mg/day, and the time to response varied from a few days to 3 months. No adverse effects were reported.
Conclusions:
Colchicine appears to be a safe and effective treatment for persistent, severe, or recurrent cutaneous manifestations of IgAV. These findings support its use in clinical practice and highlight the need for prospective controlled studies.
Introduction
Immunoglobulin A vasculitis (IgAV), formerly known as Henoch–Schönlein purpura, is a systemic vasculitis characterized by the deposition of IgA-containing immune complexes in the walls of small blood vessels. 1 The disease is 10 to 30 times more common in children than in adults with a pediatric incidence of 18.6/100 000.2 -4 Clinically, IgAV typically present with a triad of palpable purpura, arthralgia or arthritis, and gastrointestinal symptoms. 5 Renal involvement is the most serious long-term complication, leading to end-stage renal failure in 1.8% of cases. 6 Although the disease is usually self-limiting with spontaneous resolution in an average of 4 weeks, 7 symptoms persist for more than 1 month in 40% of patients, 8 and beyond 12 months in 7% of them. 9 Moreover, relapses occur in 14% to 51% of cases.8 -11
Whereas renal or severe gastrointestinal involvement typically requires systemic immunosuppression, cutaneous and joint-limited disease is generally managed with symptomatic treatment.12 -22 For patients with severe, recurrent, or persistent cutaneous symptoms, therapeutic options such as colchicine, dapsone, or methotrexate have been reported, although with variable success.23,24 Colchicine is a natural alkaloid that acts primarily on polymorphonuclear cells, and its off-label use has been reported for several dermatologic conditions including neutrophilic disorders and leukocytoclastic vasculitis. 25 The use of colchicine in IgAV, which is characterized by IgA deposition and neutrophilic destruction of small vessel walls in the skin, 26 is pathophysiologically relevant given its inhibition of neutrophil adhesion and migration. 25
We report a single-center case series along with a literature review of patients treated with colchicine for cutaneous manifestations of IgAV. We aim to evaluate the indications, efficacy, and tolerability of colchicine in this context.
Materials and Methods
Monocentric Retrospective Case Review
We conducted a retrospective chart review of pediatric patients aged <18 years with IgAV who were seen at the specialized vasculitis clinic at CHU Sainte-Justine between June 2019 and March 2023. The study was approved by the CHU Sainte-Justine ethics committee (2024-5771). Included patients had a diagnosis of IgAV based on the EULAR/PRINTO/PreS criteria 5 and received colchicine for cutaneous involvement. We collected data on demographics (age, sex), comorbidities, number of flares, symptom duration, clinical features, description and location of skin lesions, colchicine indications, dosage and treatment duration, concomitant therapies, treatment efficacy, and adverse events. For patients requiring dose escalation, the dose achieving therapeutic response—defined as improvement in skin lesions as judged by the patient and/or physician—was recorded. Outcomes were categorized as complete resolution, improvement, or no improvement. Severe skin involvement was defined as the presence of bullae, necrosis, ulceration, or widespread purpura beyond the lower legs. Persistent skin involvement referred to lesions lasting more than 4 weeks, while recurrence was defined as reappearance of skin lesions after at least 4 weeks of remission.
Literature Search and Study Selection
We conducted a search in PubMed and Ovid MEDLINE databases between June 2021 and September 2022. The search terms used, without any filters or date restrictions, were: ([[[[[[henoch schonlein [MeSH Terms]] OR [henoch schonlein purpura [MeSH Terms]]] OR [IgA vasculitis [MeSH Terms]]]] AND [colchicine [MeSH Terms]]] OR [henoch schonlein purpura AND colchicine]] OR [henoch schonlein AND colchicine]) OR (IgA vasculitis AND colchicine). Two independent reviewers selected publications in English and French describing cases of IgAV diagnosed according to EULAR/PRINTO/PReS 5 criteria for children and the American College of Rheumatology (ACR) 27 criteria for adults, who were treated with colchicine, regardless of age, sex, comorbidities, concomitant treatments, and duration of follow-up. Both full-length articles and letters were included. We excluded reports where colchicine was used for indications other than IgAV (e.g., familial Mediterranean fever) and review articles. Clinical data collected were the same as those recorded in our case series.
Statistical Analysis
Data from both the retrospective case review and the literature search were summarized descriptively. Results were compiled and reported as frequencies, percentages, means, or medians with ranges, as appropriate.
Results
Single-Center Retrospective Case Review
Of the 124 consecutive children with IgAV seen at the vasculitis clinic between 2019 and 2023, 13 were treated with colchicine for skin involvement. Patient characteristics are presented in Table S1. Female to male ratio was 1.6 and mean age was 11 years. Colchicine was prescribed for persistent, recurrent, or severe purpura; 3 patients had more than 1 indication for colchicine use. Skin improvement was observed in 12 of the 13 patients. The only patient who did not respond—a 15 year-old girl—had notably poor adherence to treatment and was later found to have a positive urea breath test, suggesting a potential contribution of Helicobacter pylori infection to her persistent symptoms.
Seven patients received concomitant systemic corticosteroids, which were tapered prior to colchicine discontinuation in all cases. One patient also received hydroxychloroquine, with resolution of lesions within 3 weeks. Among the 5 patients treated with colchicine monotherapy, cutaneous lesions resolved within 1 to 6 weeks.
Three patients experienced a relapse of purpura while on colchicine, which resolved with continued treatment. In contrast, 4 patients had a relapse after colchicine discontinuation, including 2 with associated abdominal pain. Among the 3 patients for whom timing was reported, the mean time to relapse following colchicine withdrawal was 1 month (range: 0.5-1.5 months). Colchicine was reintroduced in all 4, leading to resolution of purpura in each case.
Literature Review
Of the 59 articles retrieved from our literature review, we analyzed 16 articles including 2 retrospective studies and 14 case reports. The article selection process is shown in the flowchart (Figure S1). Given 2 larger retrospective reviews included subsets of patients treated with colchicine, and lacked granularity of data sought for description, we opted to limit inclusion of these studies to the discussion rather than formulating an analysis with the included case reports.28,29 We found 17 patients treated with colchicine for cutaneous involvement of IgAV, detailed in Table S2. Colchicine was prescribed for persistent, severe, or recurrent purpura. Three patients initially described as recurrent were reclassified as persistent according to our definition. One patient met all 3 indications. One author reported a dose of 10 mg/day, which we considered likely erroneous; as we were unable to contact the author for clarification, this value was excluded from the mean. 30 The median treatment duration was 6 months. No side effect was experienced with colchicine in the reported patients.
Seven patients were treated with colchicine monotherapy, while 10 received concomitant treatments, most commonly corticosteroids (n = 8), followed by azathioprine (n = 2), aspirin (n = 2), and hydroxychloroquine (n = 1). All patients receiving concomitant treatments had gastrointestinal or renal involvement, except for one with widespread purpura. All 7 patients treated with colchicine alone had a favorable response. Time to response was generally <2 weeks (n = 11/15); response time was unknown in 2 cases.
Five patients relapsed between 3 days and 10 months after colchicine discontinuation. One patient relapsed during treatment (at 6 weeks), but after colchicine was continued for 5 months, no further relapse occurred up to 6 months after stopping treatment.
Two patients failed to respond to colchicine: A 12 year-old boy with skin, gastrointestinal, and renal involvement did not respond to colchicine 1 mg/day and to methylprednisolone for 4 months plus hydroxychloroquine for 2 months, but eventually responded to methotrexate. The second patient was a 47 year-old woman with a 24 year history of recurrent purpura and IgA nephritis refractory to multiple systemic agents, though dose and duration of colchicine were not specified. She ultimately improved with omega-3 fatty acids.
Table 1 compares our pediatric cohort with pediatric and adult cases from the literature.
Characteristics of Pediatric Patients From CHU Sainte-Justine, Pediatric and Adult Cases From the Literature Treated With Colchicine for Cutaneous Manifestations of IgA Vasculitis.
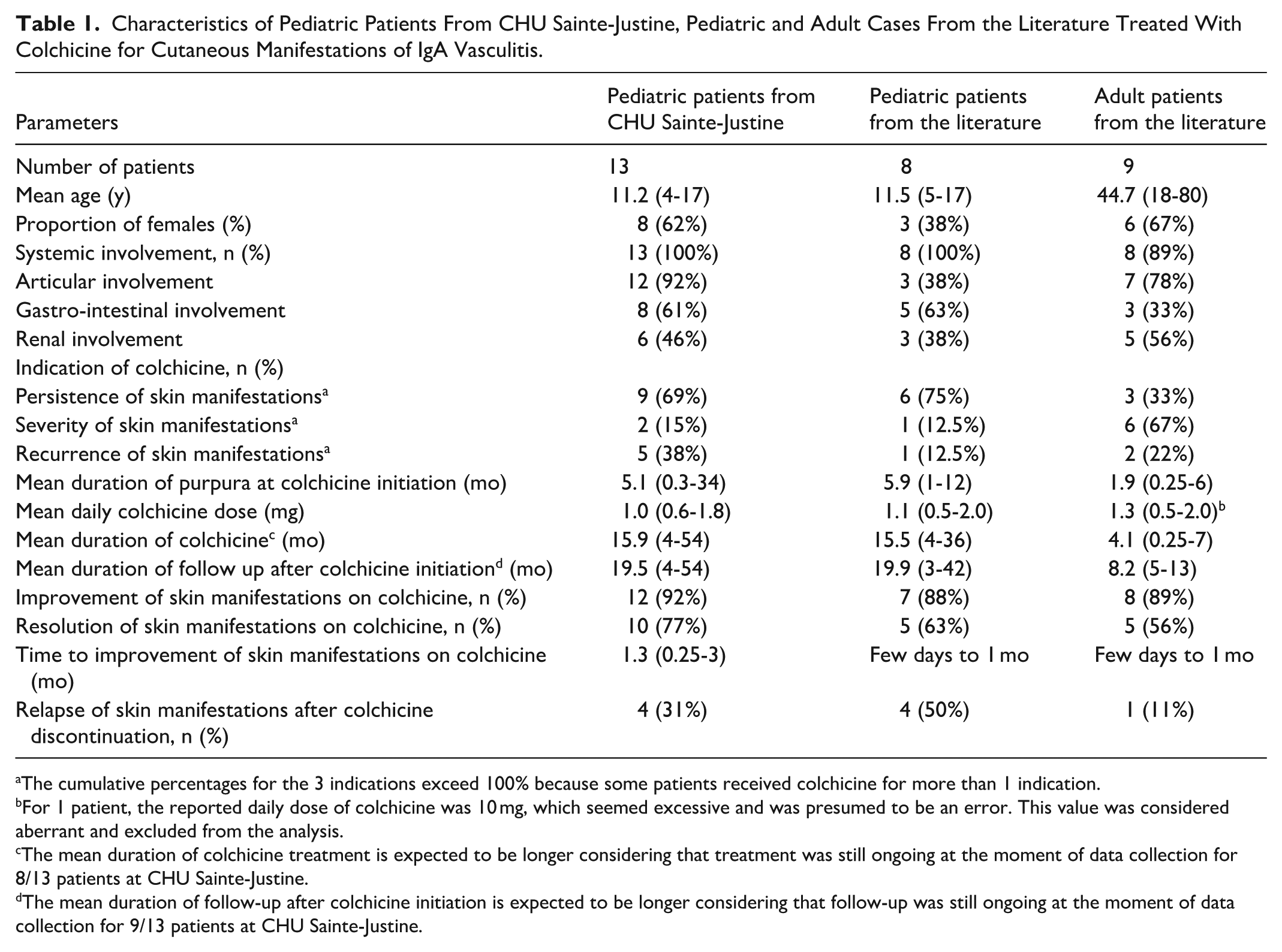
The cumulative percentages for the 3 indications exceed 100% because some patients received colchicine for more than 1 indication.
For 1 patient, the reported daily dose of colchicine was 10 mg, which seemed excessive and was presumed to be an error. This value was considered aberrant and excluded from the analysis.
The mean duration of colchicine treatment is expected to be longer considering that treatment was still ongoing at the moment of data collection for 8/13 patients at CHU Sainte-Justine.
The mean duration of follow-up after colchicine initiation is expected to be longer considering that follow-up was still ongoing at the moment of data collection for 9/13 patients at CHU Sainte-Justine.
Discussion
To the best of our knowledge, this is one of the largest case series reporting the use of colchicine for cutaneous manifestations of IgAV. Our findings suggest that colchicine is a safe and effective treatment option for persistent, severe, or recurrent purpura in both pediatric and adult patients. Overall, cutaneous improvement was observed in 90% of patients (27/30), with complete resolution in 67% (20/30). Among those treated with colchicine as monotherapy, all responded favorably. Furthermore, patients who received concomitant treatments appeared to benefit from the addition of colchicine, suggesting its specific cutaneous efficacy.
The most common indication for colchicine use was persistent purpura lasting more than 4 weeks (60%), followed by severe skin manifestations (30%) and recurrent lesions (27%). Time to response varied from a few days to <3 months. The efficacy of colchicine for persistent skin lesions was high, with 89% (16/18) of patients showing improvement, including complete resolution in 9 patients. This high response rate is unlikely to be explained by natural disease course or concomitant therapies, given that these patients had a prolonged duration of skin lesions prior to colchicine initiation (mean 6.9 months). Notably, over half of these patients improved within 2 weeks (9/16, 56%) and received colchicine as monotherapy (9/16, 56%). Four patients with persistent purpura despite corticosteroid treatment experienced dramatic improvement following colchicine introduction, suggesting that corticosteroids alone were insufficient for skin lesion resolution. Furthermore, purpura recurrence after colchicine discontinuation, with subsequent improvement upon rechallenge, reinforces the beneficial effect of colchicine in managing persistent cutaneous symptoms of IgAV.
In addition to persistent lesions, colchicine also showed promise in patients with recurrent skin manifestations demonstrating effectiveness in 88% of patients (7/8). This conclusion is supported by the absence of relapse during colchicine therapy in 6 patients, and the observation that 4 patients experienced relapse after colchicine discontinuation but achieved resolution of purpura upon reintroduction of the treatment. These findings align with a retrospective study of 12 children with recurrent IgAV, in which 11 patients showed favorable outcomes following colchicine treatment. 28 In that study, colchicine doses ranged from 0.5 to 1 mg daily, with treatment durations between 6 and 24 months. Only 1 patient experienced recurrence during follow-up. 28
In severe skin lesions, interpretation of colchicine’s effectiveness is more challenging. Although all patients showed a favorable response, with complete resolution observed in 67% (6/9), spontaneous improvement or the impact of concomitant corticosteroid treatment cannot be ruled out. 14 Most patients (8/9) presented with severe skin lesions during the acute phase (<4 weeks), a period when IgAV typically has a favorable natural course after 4 to 6 weeks. Additionally, 6 patients received corticosteroids alongside colchicine. Nonetheless, 5 patients experienced rapid clearance of skin lesions within days to 2 weeks following colchicine initiation, including 2 who were not on corticosteroids, suggesting a potential benefit of colchicine in this context.
Interestingly, 30% of patients overall (9/30) had a recurrence of purpura after colchicine discontinuation. When reported, the mean time to relapse after colchicine discontinuation was 2.8 months (range 0.1-10). Relapses improved or resolved on reintroduction of colchicine, confirming the efficacy of this treatment.
A retrospective study of adult patients with IgAV 29 reported an overall response rate of skin symptoms to colchicine of 59% (n = 10/17), defined as 50% to 100% improvement. This value is lower than our overall response rate (90%), but colchicine dose and duration were unspecified in this retrospective study.
Colchicine was well tolerated at doses ranging from 0.5 to 2 mg daily, with no reported adverse effects both in our cohort and in the literature. The most common side effects of colchicine are nausea, vomiting, and diarrhea, which are dose-dependent and occur in 5% to 10% of patients. 25 Potential toxicity including myelosuppression and multiorgan failure can occur in the setting of renal or hepatic impairment. 25
Due to its retrospective design and literature review, limitations of this study include lack of a control group as well as publication bias. In addition, comparability of patients is hampered by the lack of consensus in the definition of recurrent or persistent purpura used in the literature.
Conclusion
Our review suggests that colchicine may be a safe and effective treatment option for severe, persistent, or recurrent cutaneous lesions of IgAV in both adult and pediatric patients. Further prospective controlled studies are needed to better evaluate colchicine’s role in cutaneous manifestations of IgAV. In the near future, 2 ongoing studies may provide some guidance: a clinical trial comparing the efficacy of colchicine, dapsone, and azathioprine in patients with isolated skin vasculitis, including IgAV. 31 Another study aims to compare the efficacy of colchicine to placebo in preventing cutaneous relapses of IgAV in adult patients. 32
Supplemental Material
sj-docx-1-cms-10.1177_12034754261433521 – Supplemental material for Colchicine for Skin Involvement in Immunoglobulin A Vasculitis: A Single-Center Case Series and Review of the Literature
Supplemental material, sj-docx-1-cms-10.1177_12034754261433521 for Colchicine for Skin Involvement in Immunoglobulin A Vasculitis: A Single-Center Case Series and Review of the Literature by Maha El Barch, Meryem Safoine, Selma Ouali and Maryam Piram in Journal of Cutaneous Medicine and Surgery
Footnotes
Acknowledgements
We would like to thank Claude Belleville for assistance with the submission of the ethics protocol.
Ethical Considerations
This study was approved by the CHU Sainte-Justine ethics committee (2024-5771).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon request.
Supplemental Material
Supplemental material for this article is available online.
