Abstract
Introduction:
Lymphatic malformations (LM) are vascular anomalies that can be challenging to manage with new medical therapies emerging. This systematic review examines current medical therapies for pediatric patients with LMs that involve the soft tissues.
Materials and Methods:
MEDLINE, Embase, Cochrane Library, and SCOPUS were searched on April 12, 2024, using variations of the keywords “lymphatic malformation” AND “drug therapy” AND “pediatric.” Language was limited to English, and no date restriction was applied. Treatment success was defined as a reduction in lesion size of over 10%, with complete response (CR) defined as a reduction in size of over 90%.
Results:
Our review encompassed 4937 title/abstracts, 436 full-texts and ultimately included 77 studies. Reported success rates were variable, with notable results for alpelisib (oral) (n = 9/9), sirolimus (oral) (n = 257/287), sirolimus (topical) (n = 13/15), acetylsalicylic acid (oral) (n = 18/23), propranolol (oral) (n = 19/29), and sildenafil (oral) (n = 33/71). CR was reported with isotretinoin (n/a) (n = 1/1), cyclophosphamide (iv) (n = 1/2), acetylsalicylic acid (oral) (n = 4/23), sirolimus (topical) (n = 2/15), and sirolimus (oral) (n = 17/287).
Conclusion:
Overall, therapies such as alpelisib and sirolimus showed promising results in the reduction of pediatric LM size; however, additional long-term data are needed to validate their efficacy and safety profile. Limitations of our study include heterogeneity and a potential risk of bias.
Introduction
Lymphatic malformations (LM) are defined by the International Society for the Study of Vascular Anomalies as low-flow vascular anomalies of the lymphatic system. 1 Previously referred to as lymphangioma or when congenital, cystic hygroma, LMs can vary in their presentation. They can be simple or complex, microcystic, macrocystic, or mixed, they can occur in isolation or as part of various syndromes, and can be superficial, deep, or visceral. Additionally, LMs can be exclusively lymphatic in nature or can contain components of other vascular types (Table 1). 1
International Society for the Study of Vascular Anomalies Classification of LMs. 1
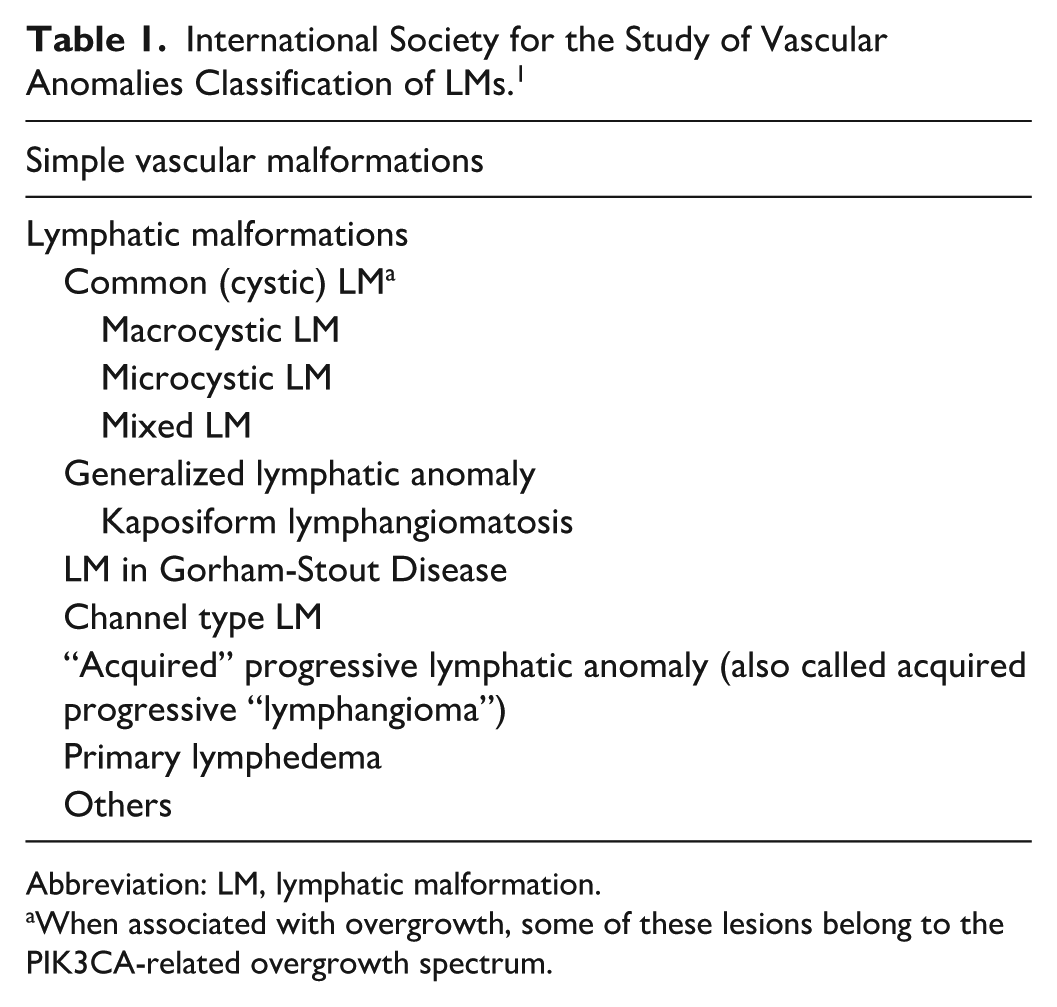
Abbreviation: LM, lymphatic malformation.
When associated with overgrowth, some of these lesions belong to the PIK3CA-related overgrowth spectrum.
The incidence of LMs is estimated to be between 1 in 6000 to 1 in 16,000 live births, affecting males and females equally. 2 Approximately, 50% to 60% of LMs are present at birth, while 90% manifest by 2 years of age. 3 Around 75% of LMs occur in the head and neck region, potentially resulting in significant comorbidities such as dysphagia, airway obstruction, and difficulty with speech. 4 Superficial LMs, or those that involve the soft tissues, can result in pain, swelling, bleeding, oozing, recurrent infection, or aesthetic concerns for the affected individual. LMs must be evaluated by a multidisciplinary vascular anomalies team to determine the treatment that is most appropriate for each individual patient. Treatment options for LMs include observation, surgical resection, sclerotherapy, embolization, laser therapy, or radiofrequency ablation. The mainstay treatments have traditionally included sclerotherapy, surgical excision, or a combination of both. The therapeutic approach is determined by factors such as LM classification, size, position, growth rate, and response to previous therapies. For LMs that are purely macrocystic, only embolization ± surgery may be required. Mixed LMs necessitate additional investigations to assess for venous involvement and if present, treatment with coagulation and catered medical therapies may be needed, but are not addressed in this article. In cases where LMs are located in nonoperable anatomic regions, or the LM has been refractory to treatment, medical therapies are being explored as noninvasive alternatives. This systematic review was designed to evaluate the efficacy and safety profiles of medical treatment options for LM as alternatives to traditional invasive treatments.
Methods
The study focused on all original articles detailing pediatric patients (0-18 years) with LMs treated with medical treatments (intravenous, oral, topical). Primary outcome measures were reduction in size. Secondary outcome measures were reduction in symptoms (discharge, bleeding, and pain), and adverse events. A systematic review of the literature was conducted adhering to PRISMA reporting guidelines. 5 MEDLINE, Embase, SCOPUS, and Cochrane Library were searched on April 12, 2024, using variations of the keywords “lymphatic malformation” AND “drug therapy” AND “pediatric.” (Supplemental Table 1). No date restriction was applied, while language was limited to English. Title, abstract, and full-text screening was conducted by 2 independent reviewers using Covidence online systematic review software (www.covidence.org). Any conflicts between reviewers were resolved through discussion or with input from a senior reviewer until a consensus was reached. Inclusion criteria for our review included clinical studies that assessed the response of pediatric patients with LMs to medical therapy. This included microcystic (formerly called lymphangioma circumscriptum when on the skin surface), macrocystic (formerly called cystic hygroma when located on the neck), mixed or unspecified LMs/lymphangioma in addition to other conditions encompassing involvement of soft tissue LMs such as (kaposiform) lymphangiomatosis, lymphangioma circumscriptum, or generalized lymphatic anomaly. Syndromic causes of LM such as CLOVES syndrome (congenital lipomatous overgrowth, vascular malformations, epidermal nevi, and skeletal anomalies), PIK3CA-related overgrowth spectrum (PROS), and Gorham–Stout disease were screened; however, visceral or skeletal LMs were not included unless there was soft tissue involvement. Combined vascular malformations that were not exclusively LM in nature were excluded. Cellular, genetic, and animal studies were excluded.
Data extraction was completed by 2 independent reviewers and subsequently analyzed. If any data items were incomplete, additional information was obtained by contacting study authors where possible.
The strength of clinical data of included studies were graded with the Oxford Centre for Evidence-Based Medicine levels of evidence by 2 authors independently, with any conflicts resolved after collective discussion. 6 Levels of evidence included level 1 properly powered and conducted randomized clinical trial; level 2 well-designed, controlled trial without randomization or prospective comparative cohort trial; level 3 case–control studies or retrospective cohort studies; level 4 case studies with/without intervention or cross-sectional studies; level 5 case reports. Grades of recommendation include A, multiple level 1 studies; B, multiple level 2 or 3 studies or extrapolations from level 1 studies; C, multiple level 4 studies or extrapolations from level 2 or 3 studies; and D, level 5 or inconclusive studies.
Results
Our literature search yielded 4969 articles, with 1 article added from citation verification. Of the 4970 articles, 4537 were excluded based on title and abstract review (Supplemental Table 2). After retrieving 429 studies for full-text screening, 351 were excluded. Ultimately, 77 studies were included in the review, of which 8 were conference abstracts (Supplemental Table 3).
The included studies were published between 1995 and 2024 and comprised 2 randomized controlled trials, 30 prospective or retrospective cohort studies, 6 case series, and 39 case reports. The majority were conducted in the United States (n = 18), followed by China (n = 10), Turkey (n = 8), Spain (n = 7), Japan (n = 5), France (n = 5), Austria (n = 3), and Italy (n = 3). The evidence quality and grade recommendations are summarized in Supplemental Table 4, with grades of B given to 4 treatments [alpelisib, propranolol, sildenafil, and sirolimus (oral)], a grade of C given to 1 treatment [sirolimus (topical)], with the remainder receiving a grade of D level evidence.
In total, our pooled analysis includes 449 individuals, 156 males, 174 females, 119 gender unspecified, with a mean age of 5.5 years, ranging from newborns to 18 years of age.
The primary outcome measured in this study was volume reduction in response to treatment (Supplemental Table 4). Volume reduction was determined by radiography, most commonly via magnetic resonance imaging, or subjective clinical assessment (Supplemental Table 5). A partial treatment response was defined as a reduction in volume over 10% and less than 90%, while a complete response was defined as a volume reduction greater than 90%. Treatment success was defined as either a partial or complete response (ie, greater than 10% volume reduction). In the event a study provided results that mentioned a reduction in size but failed to quantify the change or mention a complete/near complete resolution, the results were categorized as a partial response.
Despite a small sample size, alpelisib demonstrated a 100% treatment success rate across 5 studies involving 9 subjects. Notably, in 10 of the included studies, patients had previously experienced treatment failure with other pharmacologic agents including propranolol (n = 5), sirolimus (n = 4), oral steroids (including prednisone) (n = 2), imiquimod (n = 1), tadalafil (n = 1), and cantharidin (n = 1).7-16 Other therapeutic agents also reported 100% treatment success rates including celecoxib 17 (published 2019), cyclophosphamide 18 (published 1995), isotretinoin 19 (published 2016), tacrolimus 20 (published 2021), as well as sirolimus, and propranolol in combination 8 (published 2020). However, these findings were based on case reports with fewer than 4 patients per study and include no current indications to treat LMs. Sirolimus, administered both orally and topically, had the most robust evidence supporting its use with a combined 49 studies and 302 patients. Efficacy rates were comparable between the oral and topical formulations with a 90% and 86% success rate in size reduction greater than 10%, respectively. Acetylsalicylic acid (ASA) showed a success rate of 78% in a single study on 23 patients. 21 Propranolol, an approved therapy for infantile hemangioma, was used in 4 studies on 29 patients and also reported a relatively low rate of treatment success at 66% of patients. Sildenafil was the second most well-studied therapy, with 10 studies and 71 patients. However, the reported treatment success rate was considerably lower at only 46% of patients. These findings highlight the need for higher-quality studies assessing the efficacy and safety profile of the use of these therapies to treat pediatric LMs.
Many studies assessed improvement in patient function and reductions in symptom severity, including bleeding, infection, swelling, and pain. The majority of included studies documented adverse events experienced by patients associated with pharmacologic agents, although the reported rates of these events varied widely (Supplemental Table 5).
Discussion
Our systematic review focused on medical treatment of pediatric LMs and highlighted promising results for several therapeutic options. Therapies assessed in studies with >5 patients included sirolimus (oral) (n = 287), sildenafil (oral) (n = 71), propranolol (oral) (n = 29), ASA (oral) (n = 23), sirolimus (topical) (n = 15), and alpelisib (oral) (n = 9). Excellent response rates were seen in both alpelisib and sirolimus, both of which target the PI3K/AKT/mTOR intracellular pathway.
The PI3K/AKT/mTOR pathway represents a major intracellular signalling pathway established to play a significant role in tumor cell growth and proliferation. 22 Subunit p110α of the phosphatidylinositol 3-kinase (PI3K) gene, referred to as phosphatidylinositol 3-kinase catalytic subunit alpha (PIK3CA) is involved in the regulation of cell growth and proliferation.23,24 Pathogenic PIK3CA gene mutations are believed to be implicit in most LMs. 25 Somatic gene mutations in PIK3CA are believed to play a role in the developmental impairment of lymphatic channels as a result of abnormally functioning endothelial cells. 26 Alpelisib, an α-selective PI3K inhibitor, selectively targets PIK3CA mutations and represents an attractive treatment option for PROS LMs. 27
Similarly, mammalian target of rapamycin (mTOR), a protein kinase involved in the mediation of cell growth and proliferation, is also believed to be inherent in the formation of LMs. 28 Sirolimus (rapamycin), classified as a macrolide antibiotic, exhibits an inhibitory downstream effect on the mTOR portion of the PIK/mTOR/AKT pathway.11,29
Interconnected to the PIK3CA pathway, the RAS/MAPK (rat sarcoma virus oncogene homolog/mitogen-activated protein kinase) pathway plays a major role in cell proliferation and differentiation, but with different downstream targets than the PIK3CA pathway. The MAPK pathway includes RAS, RAF (rapidly accelerated fibrosarcoma), MEK (mitogen-activated protein kinase), and ERK (extracellular signal-regulated kinase). Inhibition of the MEK step of this cascade is currently being explored for treatment of syndromic lymphatic malformations and tumors. 30
Cystic dilation in LMs may result from increased intramural pressure following the contraction of thickened muscular linings. 31 Phosphodiesterase-5 (PDE5) facilitates the breakdown of guanosine monophosphate, resulting in contraction of vascular smooth muscle and vasodilation. 32 Sildenafil selectively inhibits PDE5 to prevent activation of this pathway which may lead to the relaxation of vascular smooth muscle and cystic decompression in addition to normalization of lymphatic endothelial function due to opening of secondary lymphatic spaces. 33
A retrospective cohort study by Wang et al directly compared the use of sildenafil (n = 24) and sirolimus (n = 31) in the treatment of pediatric LMs. 34 The study found that 54% (n = 13/24) of patients in the sildenafil group achieved a partial response, while 94% (n = 29/31) of patients in the sirolimus group experienced either a partial or a complete response. Sildenafil had no patients experience a complete response while sirolimus had one. Symptom improvement was reported in 79% of patients in the sildenafil group and 97% of patients in the sirolimus group. Symptom improvement often included reductions in discharge, bleeding, or pain, all of which may provide a significant impact on quality of life. Lastly, only 17% (n = 4/24) of sildenafil patients experienced adverse events (transient diarrhea), while 74% (n = 23/31) of patients in the sirolimus group experienced adverse events including oral mucositis (n = 15), upper respiratory tract infection (URTI) (n = 9), hepatic dysfunction (n = 9), elevated cardiac enzymes (n = 9), hypertriglyceridemia (n = 6), hypercholesterolemia (n = 1), and fever (n = 1). Despite sirolimus showing greater efficacy in volume reduction, it was also associated with higher rates of adverse events. However, all reported adverse events were deemed to be mild and controllable.
Reported rates of adverse events varied widely amongst these therapies. Notably, there were no adverse events reported with alpelisib use and only 29% of patients using sildenafil while 89% of patients taking ASA had reported an adverse event. The remaining 4 therapies had a range of reported adverse events between 11% to 40%. Local irritation was commonly experienced by patients using topical sirolimus. Metabolic side effects were often reported in patients taking oral sirolimus such as dyslipidemia, hypercholesterolemia, and hypertriglyceridemia, in addition to several reports of hepatic dysfunction. Oral mucositis or stomatitis was the most reported adverse event of oral sirolimus, followed by URTI. No major adverse events were noted in the includable studies, however, Arya et al reported a case of a 32-week preterm infant with massive cervicofacial LM who was treated with sclerotherapy, surgical debulking, and systemic sirolimus who died at 4 months of age due to acute pulmonary hemorrhage. 35 It was believed that the patient’s death may have been related to sirolimus given the absence of other associated organ involvement or derangement.
Several clinical trials are currently ongoing with no reported results yet. A clinical trial by Teng et al (NCT02335242) was included in our study and reported successful treatment of only 25% of patients (n = 2/8) using sildenafil to treat pediatric LM. 36 Adams et al reported a clinical trial of oral sirolimus in complicated vascular anomalies (including 22 patients with LM) in pediatric and adult patients. 37 Of the 57 total patients studied, none reported complete response, while 47 out of 57 had a partial response. Multiple Phase 2 trials are ongoing, assessing the use of topical sirolimus to treat cutaneous LM (NCT03972592), 38 microcystic LM (NCT05050149), 39 and lingual microcystic LM (NCT04128722). 40 Percutaneous administration of sirolimus is also being studied to treat superficial complicated vascular anomalies, including LM (NCT04921722). 41 Additionally, an ongoing Phase 2 trial is looking at sildenafil in the treatment of LMs (NCT02335242). 36 Promising results from a retrospective chart review (EPIK-P1) led to the U.S. Food and Drug Administration (FDA) granting accelerated approval of alpelisib for use in severe pediatric PROS in 2022 (NCT04589650). 42 Similarly, the results from another Phase 2 trial (SELVA) led to the FDA granting breakthrough therapy designation in 2023 for 3.9% sirolimus anhydrous gel to treat microcystic LM, with Phase 3 trials currently ongoing (NCT 06239480). 43
Sirolimus has been used in fetal therapy in 2 studies that diagnosed cervicofacial LM in utero.44,45 In one of the studies, the mother was started on sirolimus followed by a transition to sirolimus in the neonate, with both studies reporting positive outcomes.
Conclusion
LMs represent complex congenital vascular anomalies that require the expertise of a multidisciplinary vascular anomalies team to evaluate and manage appropriately. Our findings highlighted the potential efficacy of various medical treatments for pediatric LMs, particularly in terms of volume reduction and symptom management. Limitations within our study include notable heterogeneity and risk of bias inherent with several included studies in our analysis.
Our analysis suggests that both topical and oral sirolimus may provide a noninvasive option to treat pediatric LM, with high reported rates of treatment success, symptom improvement, and only mild adverse events. Despite a small sample size, alpelisib showed promise with the highest reported rate of treatment success, with low rates of adverse events. Alpelisib has already received FDA approval for treatment of severe pediatric PROS and remains the treatment of choice in patients positive for the PIK3CA gene mutation. Other treatments such as sildenafil and propranolol, showed less promise with lower reporter rates of successful treatment. Several ongoing clinical trials represent a concerted effort toward developing on-label medical treatment for LMs, while further high-quality studies are needed to explore the efficacy and safety profile of the treatment options identified in our study.
Supplemental Material
sj-pdf-1-cms-10.1177_12034754251386785 – Supplemental material for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review
Supplemental material, sj-pdf-1-cms-10.1177_12034754251386785 for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review by Dylan Hollman, Harleen Cheema, Adam C. Yu, Newsha Mahinpey and Fatmah AlZahrani in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-pdf-2-cms-10.1177_12034754251386785 – Supplemental material for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review
Supplemental material, sj-pdf-2-cms-10.1177_12034754251386785 for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review by Dylan Hollman, Harleen Cheema, Adam C. Yu, Newsha Mahinpey and Fatmah AlZahrani in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-pdf-3-cms-10.1177_12034754251386785 – Supplemental material for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review
Supplemental material, sj-pdf-3-cms-10.1177_12034754251386785 for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review by Dylan Hollman, Harleen Cheema, Adam C. Yu, Newsha Mahinpey and Fatmah AlZahrani in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-pdf-4-cms-10.1177_12034754251386785 – Supplemental material for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review
Supplemental material, sj-pdf-4-cms-10.1177_12034754251386785 for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review by Dylan Hollman, Harleen Cheema, Adam C. Yu, Newsha Mahinpey and Fatmah AlZahrani in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-pdf-5-cms-10.1177_12034754251386785 – Supplemental material for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review
Supplemental material, sj-pdf-5-cms-10.1177_12034754251386785 for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review by Dylan Hollman, Harleen Cheema, Adam C. Yu, Newsha Mahinpey and Fatmah AlZahrani in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-pdf-6-cms-10.1177_12034754251386785 – Supplemental material for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review
Supplemental material, sj-pdf-6-cms-10.1177_12034754251386785 for Medical Therapies for Pediatric Lymphatic Malformations: A Systematic Review by Dylan Hollman, Harleen Cheema, Adam C. Yu, Newsha Mahinpey and Fatmah AlZahrani in Journal of Cutaneous Medicine and Surgery
Footnotes
Acronyms & Abbreviations
ASA—Acetylsalicylic Acid
CLOVES—Congenital Lipomatous Overgrowth, Vascular Malformations, Epidermal Nevi, and Skeletal Anomalies
CT—Computed Tomography
ERK—Extracellular Signal-Regulated Kinase
FDA— Food and Drug Administration
GLA—Generalized Lymphatic Anomaly
GSD—Gorham-Stout Disease
ISSVA—International Society for the Study of Vascular Anomalies
LM—Lymphatic Malformation
MAPK—Mitogen-Activated Protein Kinase
MEDLINE—Medical Literature Analysis and Retrieval System Online
MEK—Mitogen-Activated Protein Kinase
MRI—Magnetic Resonance Imaging
mTOR—Mammalian Target of Rapamycin
PDE5—Phosphodiesterase-5
PI3K—Phosphatidylinositol 3-Kinase
PIK3CA—Phosphatidylinositol 3-Kinase Catalytic Subunit Alpha
PRISMA—Preferred Reporting Items for Systematic Reviews and Meta-Analyses
PROS—PIK3CA-Related Overgrowth Spectrum
RAF—Rapidly Accelerated Fibrosarcoma
RAS—Rat Sarcoma Virus Oncogene Homolog
RCT—Randomized Controlled Trial
URTI—Upper Respiratory Tract Infection
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Previous Publication
Interim data from the study was published as an abstract and presented at the 2024 Canadian Dermatology Association annual conference in Ottawa, Ontario, Canada.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
