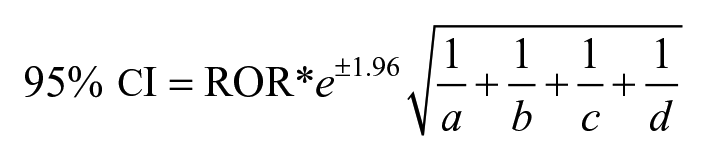Abstract
Background:
Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) are potentially life-threatening severe cutaneous adverse reactions (SCARs). Although the classical causal agents of SCARs (antibiotics, anticonvulsants, nonsteroidal anti-inflammatory drugs, and allopurinol) are well characterized, there has been little update to this list to account for newly marketed medications.
Objective:
To provide an updated and stratified list of medications with significant reporting odds ratios (RORs) of SCARs.
Methods:
A case/non-case analysis using the United States FDA Adverse Event Reporting System was performed.
Results:
As expected, the prototypical medication classes made up the majority of reported cases of SJS, TEN, AGEP, and DRESS (77%, 64%, 75%, and 72%, respectively). In addition, several infrequently or previously undescribed classes/medications implicated in SCARs were identified to have significant ROR signals, including acetylcysteine, anticoagulants, diuretics, immunotherapies, proton pump inhibitors, antivirals, and antifungals. Among these reported for SJS were acetylcysteine (ROR: 64.38) and fluconazole (ROR: 17.13). For TEN, we identified furosemide (ROR: 26.32), spironolactone (ROR: 14.45), fluconazole (ROR: 30.21), amphotericin B (39.06), and acetylcysteine (ROR: 93.12). For AGEP, we identified acyclovir (ROR: 61.72), valacyclovir (ROR: 30.76), and enoxaparin (ROR: 27.37). For DRESS, we identified vemurafenib (ROR: 17.35), acyclovir (ROR: 30.63), abacavir (ROR: 26.62), raltegravir (ROR: 23.27), and valacyclovir (ROR: 21.77) to have strong reporting odds.
Conclusion:
Our analysis provides an updated tool for physicians to reference when identifying suspected SCARs and a basis for future studies to investigate atypical medication causality.
Keywords
Introduction
Severe cutaneous adverse reactions (SCARs) are a series of rare, potentially life-threatening hypersensitivity-type adverse drug reactions (ADRs) affecting up to 2% to 3% of hospitalized patients and carrying high morbidity and mortality rates.1-4 SCARs include Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP), with mortality rates of 5% to 10%, 50%, 10%, and 5%, respectively.2,3,5-7 SJS and TEN present with widespread skin erythema, blistering, and sloughing.2,5 They are distinguished from one another by the percentage of body surface area involved (SJS < 10% and TEN > 30%). DRESS typically presents as a maculopapular eruption along with systemic symptoms including fever, lymphadenopathy, eosinophilia, and multiorgan involvement.6,8 Lastly, AGEP presents with pruritic, nonfollicular intraepidermal erythematous pustules, typically distributed on the trunk. 7
Several drugs/drug classes are classically implicated in SCARs, specifically antibiotics, antiepileptics, allopurinol, and nonsteroidal anti-inflammatory drug (NSAID) medications. 4 Although these classical drug associations are well established, the rising utilization of newer medications with rising association with SCARs necessitates the establishment of a comprehensive and updated reporting odds assessment of these drugs. Prior case-control studies in Europe (EuroSCAR) and smaller studies exploring the association of individual medications have been performed to establish the classically implicated medications in SCARs.8-17 However, a comprehensive analysis of medications with association with SJS, TEN, DRESS, and AGEP using data from The United States Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) has not been completed. The goal of this study is to examine all 4 categories of SCARs using FAERS and provide an updated and stratified list of medications with significant reporting odds ratios (RORs).
Materials and Methods
FAERS Database
The FAERS database was utilized in this study to collect post-market data on ADRs reported by healthcare workers, consumers, and pharmaceutical manufacturers to the US FDA. FDA regulations require that manufacturers report ADR events that they receive; however, reporting by healthcare workers and consumers is voluntary. 18 This database provides post-market surveillance of drug safety profiles for pharmacovigilance studies. FAERS categorizes ADRs by specified searchable Medical Dictionary for Regulatory Activities reaction terms and products that are coded with information on patient outcomes and demographic information (eg, age).
Study Design
This study utilizes case/non-case design to examine the association between medications with reported cases of SCARs and identify their RORs in FAERS. For a particular drug of interest, “cases,” defined as those where exposure to the drug results in an ADR of interest (eg, SJS), are compared to “non-cases,” defined as those where drug exposure resulted in all other ADRs. The case-to-non-case comparison is used to calculate a ROR, where cases of exposure to a drug of interest (a), non-cases of exposure to a drug of interest (b), cases of all other drugs (c), and non-cases of all other drugs (d) are used to create a 2 × 2 contingency table. ROR can thus be calculated using the contingency table and the equation (a*d)/(c*b) to determine the disproportionality of a drug exposure resulting in the reaction of interest compared to all other reactions. A 95% confidence interval (CI) is also calculated for each ROR using the equation:
Case Selection Criteria
For each of the 4 categories of SCARs, data were obtained from FAERS containing all reported drug exposures from 1968 to 2021 by searching reaction terms “Stevens-Johnson syndrome,” “toxic epidermal necrolysis,” “drug reaction with eosinophilia and systemic symptoms,” and “acute generalized exanthematous pustulosis.” Medications were ordered by the number of cases reported for each category and exposures with less than 125 cases were excluded. Medications with a calculated ROR < 5.00 were excluded from Supplementary Figure S1, to eliminate medications with less significant associations in each SCARs category (the expanded table is reported as Supplementary Table 1).
Results
SCARs Cases
From 1969 to 2021, there were 23,667,594 reported ADRs, 63,083 of these being SCARs reported to FAERS. Of these, 23,761 were cases of SJS, 14,349 were TEN, 5498 were AGEP, and 19,244 were DRESS. The cases per year are mapped in Figure 1a, with reported cases of SCARs demonstrating an upward trend since 1969 in congruence with the upward trend in overall reports to FAERS (Figure 1b).
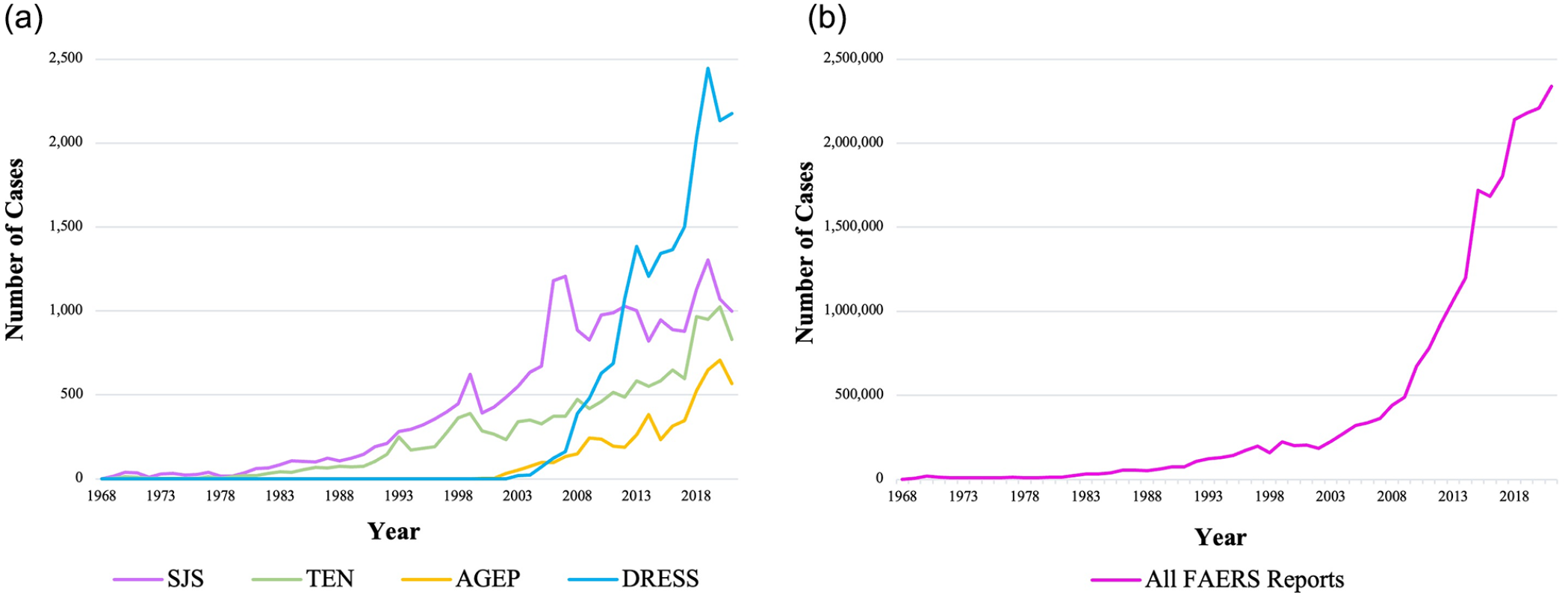
(a) Cases of SJS, TEN, AGEP, and DRESS by year reported in the FAERS database with the first case reported in 1969 to 2021. (b) All FAERS reports are plotted by year from 1968 to 2021. AGEP, acute generalized exanthematous pustulosis; DRESS, drug reaction with eosinophilia and systemic symptoms; FAERS, FDA Adverse Event Reporting System; SJS, Stevens-Johnson syndrome; TEN, toxic epidermal necrolysis.
The case characteristics for each reaction are listed in Table 1. The mean ages were 46.48 for SJS, 50.68 for TEN, 56.75 for AGEP, and 49.16 for DRESS. There was a higher percentage of cases reported for females than males across each reaction. The majority of reports were submitted by healthcare professionals, making up 64.67%, 74.12%, 88.01%, and 92.89% for SJS, TEN, AGEP, and DRESS, respectively.
Demographic and Reporting Characteristics of SJS, TEN, AGEP, and DRESS Cases in FAERS From 1968 to 2021.
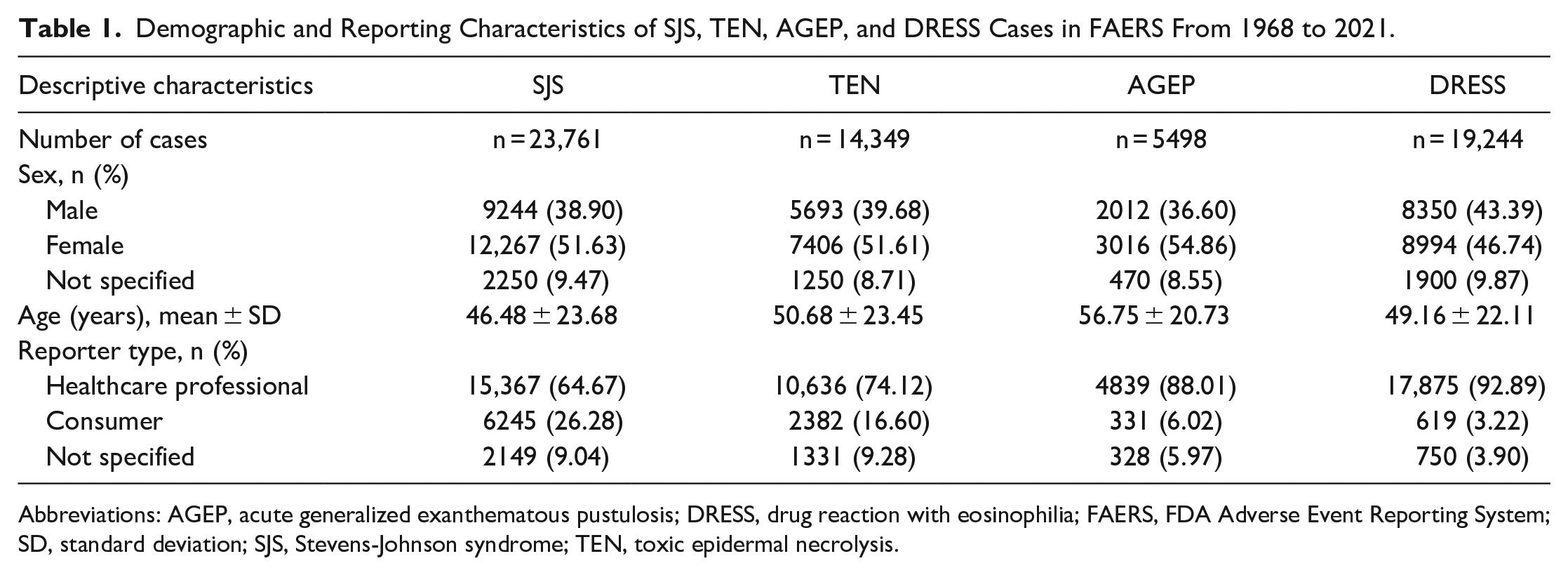
Abbreviations: AGEP, acute generalized exanthematous pustulosis; DRESS, drug reaction with eosinophilia; FAERS, FDA Adverse Event Reporting System; SD, standard deviation; SJS, Stevens-Johnson syndrome; TEN, toxic epidermal necrolysis.
When the number of SCAR cases with a particular drug exposure is compiled by class (Figure 2), antibiotics have the highest proportion of cases across all 4 categories with 28%, 32%, 51%, and 43% for SJS, TEN, AGEP, and DRESS, respectively. The drugs with the next highest proportions vary across categories, but each category of SCARs includes antiepileptics, NSAIDs/analgesics, antigout medications, proton pump inhibitors (PPIs), antivirals, antifungals, diuretics, and steroids.
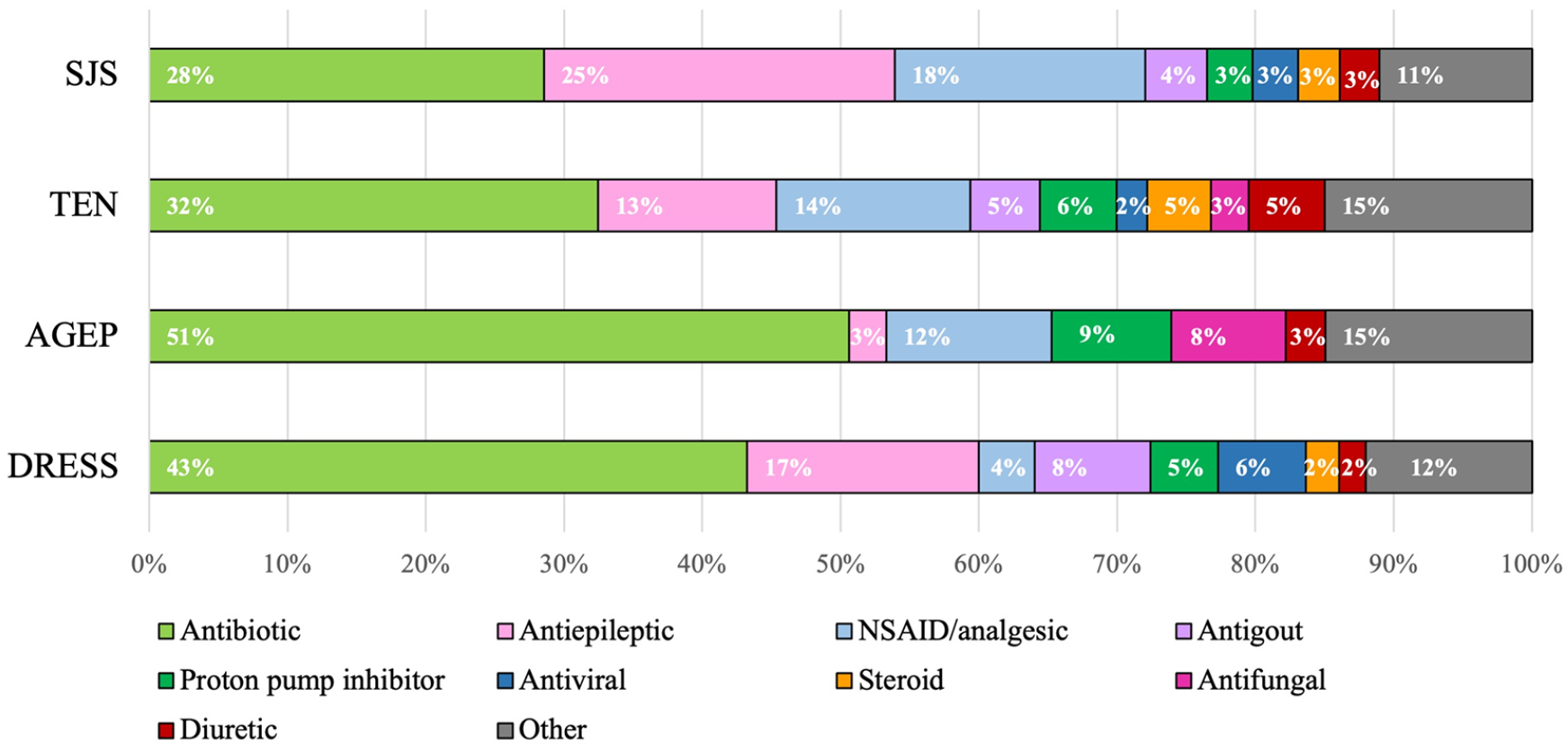
The proportion of SJS, TEN, AGEP, and DRESS cases for each drug class. Drug classes making up less than 5% of total cases across all categories are pooled together as “Other.”
Case/Non-Case Analysis
The ROR values for drugs with at least 125 cases of SJS, TEN, AGEP, and DRESS reported in FAERS are listed and stratified in Supplementary Figure S1 for those that were found to have an ROR value greater than 5.00 (the expanded list is included as Supplementary Table 1).
Discussion
Prior Pharmacovigilance and Case-Control Studies on SCARs
The typical causal agents in cases of SCARs have been increasingly recognized over the last few decades. However, no analysis using data from the United States FAERS database nor one analyzing all 4 categories of SCARs has been completed to date.
The first large-scale SCARs study assessing medication risk of SJS/TEN, a case-control analysis in 1995, involved 245 case reports obtained through databases from France, Germany, Italy, and Portugal and reported the relative risks (RRs) of the identified medications. 9 This study largely established the list of medications that we now consider to be prototypically associated with SCARs; NSAIDs, antiepileptics, allopurinol, sulfonamide antibiotics, phenobarbital, and corticosteroids which were all found to have high RR values. A second European case-control study done in 2008, called the EuroSCAR study, using 379 case reports of SJS/TEN between 1997 and 2001 from Austria, France, Germany, Israel, Italy, and the Netherlands sought to update the 1995 report. 10 This study validated the classically implicated medications from the prior report, adding a strong association with nevirapine (RR: >20) and weaker associations between pantoprazole (RR: 18), sertraline (RR: 11), and tramadol (RR:20) and SJS/TEN. This group then published a follow-up study in 2010 proposing a validated algorithm, algorithm of drug causality for epidermal necrolysis (ALDEN), for determining the causality of drugs listed in cases of SJS/TEN. 11 They applied the algorithm to the 329 cases from their previous EuroSCAR study and found insignificant RRs and no probable causality for common drug exposures including angiotensin-converting enzyme (ACE) inhibitors (RR: 0.9), thiazide diuretics (RR: 0.7), furosemide (RR: 0.7), and spironolactone (RR: 1.0).
The same group performed a case-control study to determine the odds ratios (OR) of drugs associated with AGEP in 2007, using 97 validated cases in the EuroSCAR network. 12 They report the highly associated drugs with AGEP as the antibiotics: pristinamycin (OR: ∞), aminopenicillins (OR: 23), quinolones (OR: 33), and sulfonamides (OR: ∞), along with hydroxychloroquine (OR: 39), terbinafine (OR: ∞), and diltiazem (OR:15). In addition, this group performed the first major study exploring medications associated with DRESS in 2013, using 201 cases obtained from RegiSCAR (Registry of Severe Cutaneous Adverse Reactions) from hospitals in Austria, England, France, Germany, Israel, Italy, Taiwan, and the Netherlands. 8 They concluded that the drugs with the highest causality were antiepileptics (30%), allopurinol (18%), and antibiotics (23%), particularly sulfonamides and dapsone (12%).
Our SCARs Analysis
The primary goal of this analysis was to create a stratified list of the RORs for medications with >125 reported cases of SCARs for physicians to reference, including medications that have more recently been marketed to update the current literature. When stratifying the ROR values, we identified that medications belonging to commonly implicated classes; antibiotics, antiepileptics, NSAIDs, and antigout medications, ranked high in ROR values and made up a majority of the cases by class; 77%, 64%, 75%, and 72%, respectively, for SJS, TEN, AGEP, and DRESS. This validates the findings of the previous SCAR studies that established the prototypical agents. Specific findings for AGEP, such as high association with terbinafine (ROR: 65.00, 95% CI: 56.21-75.29), hydroxychloroquine (ROR: 18.46, 95% CI: 16.02-21.27), and diltiazem (ROR: 14.05, 95% CI: 11.89-16.60) also validate the previous study from the EuroSCAR group. 12 In addition to these expected results based on the prior literature, our analysis identified many non-prototypical drugs/drug classes that identified to have significant association with SCARs: acetylcysteine, anticoagulants, diuretics, immunotherapies, PPIs, antivirals, and antifungals.
Acetylcysteine
The mucolytic medication, acetylcysteine, ranked in the top 3 strongest ROR signals for SJS and TEN (ROR: 64.38 and 93.12) in our analysis. Although this appears to be a novel finding as there are no reports of acetylcysteine-associated SJS/TEN in the literature, we find that acetylcysteine is commonly used in the treatment of SJS/TEN.19,20 This suggests that perhaps its use in SJS/TEN management may be resulting in falsely elevated ROR results. When further investigated, cases with exposure to acetylcysteine did not have SJS or TEN listed under their “Reason for Use” in individual FAERS reports. While acetylcysteine may be, therefore, a confounding medication exposure administered for the treatment of SJS/TEN, this cannot be definitely ruled out with the available data.
Anticoagulants
In our analysis, the anticoagulants, enoxaparin, and heparin carried significant ROR signals for TEN, AGEP, and DRESS (ROR: 11.39, 27.37, and 16.61) and in SJS and TEN (ROR: 5.79 and 11.55), respectively. Distinguished from heparin-induced skin necrosis, rare, but known adverse effect likely due to microvasculature clotting, SJS and TEN have only rarely been reported in patients on enoxaparin or heparin.21,22 Similarly, only 3 case reports have reported enoxaparin in association with AGEP and one has associated this drug with DRESS.23-26 There has been an association between these heparins with other type IV hypersensitivity reactions including urticariform plaques and eczema-like infiltrated plaques, suggesting perhaps there is an immune component to these drugs at play that has not been fully described.27,28 However, because heparins are some of the most commonly used drugs in a hospital setting, it is important to note that this could result in falsely elevated ROR values in cases where the SCAR occurred while the patient was hospitalized. Nevertheless, its common use along with its strong association with SCARs despite its lack of prior reporting warrants further investigation.
Diuretics
Our results also demonstrate several diuretic medications with high association with SCARs: spironolactone, hydrochlorothiazide, and furosemide. Spironolactone, commonly used in the treatment of female hormonal acne, was found to have a moderate association with TEN (ROR: 14.45) differing from the finding of no significant association in the EuroSCAR study. 11 The FDA-approved medication label for spironolactone indicates SJS, TEN, and DRESS among the ADRs identified in clinical trials or post-market reports. 29 Despite this association listed on the prescribing information, few studies have reported data on spironolactone’s association with TEN.30,31
In addition, we found furosemide to be moderately associated with SJS (ROR: 9.46), TEN (ROR: 26.32), AGEP (ROR: 10.71), and DRESS (ROR: 10.45) in FAERS. In a search of the literature, there are several case reports linking furosemide to SCARs; however, this association has not been identified by larger studies, including EuroSCAR where again no significant difference was found in SJS/TEN.12,32-34 Like furosemide, we report significant ROR signals for the thiazide diuretic, hydrochlorothiazide, in cases of SJS (ROR: 9.26), TEN (ROR: 13.55), and DRESS (ROR: 9.56). This finding also differs from the lack of association found in the EuroSCAR study and is one that we could not find elsewhere in the literature.9,10
Immunotherapies
The more recently approved immunomodulating adjunctive medications for patients with advanced or unresectable malignant melanoma, the BRAF inhibitor, vemurafenib, and the programmed death-1 (PD-1) inhibitor, pembrolizumab were highly associated with and DRESS (ROR: 17.35) and TEN (ROR: 7.31), respectively. Vemurafenib has been associated with DRESS in more recent literature; however, this drug is rarely included in lists of medications implicated in DRESS despite its strong ROR value. 35 Likewise, pembrolizumab has been increasingly associated with these reactions, 36 but as it is a newer drug has yet to be fully recognized as a strongly associated medication. As immunomodulating agents, their intense immune activation likely plays a role in their association with these hypersensitivity reactions. 37 In addition, the increased vulnerability of patients with an underlying malignancy to poor outcomes for TEN specifically makes knowing the medications with an increased risk highly critical. 38
Proton pump inhibitors
PPIs, omeprazole, pantoprazole, and esomeprazole, made up the highest number of cases as a group after the 4 commonly implicated groups with 3%, 6%, 9%, and 5% of the cases for SJS, TEN, AGEP, and DRESS, respectively. A few larger-scale studies from the United Kingdom and Taiwan have reported data on the link between the PPIs and SCARs.39,40 In particular, the UK case-control study involving 489 cases from the Clinical Practice Research Datalink (1995-2013) found a significant association between omeprazole to SJS/TEN (OR 5.85, 95% CI: 2.07-16.56) and lansoprazole (OR 3.66, 95% CI: 0.98-13.72). 40 Our results validate this association with TEN for omeprazole (ROR: 7.61) and lansoprazole (ROR: 5.96) and expand on it to include AGEP (ROR: 6.57, 95% CI: 5.61-7.71) for omeprazole and DRESS (ROR: 6.44) for lansoprazole. In addition, we found that pantoprazole and esomeprazole were also significantly associated with TEN (pantoprazole only), AGEP, and DRESS. Pantoprazole was identified by the EuroSCAR study as weakly associated (RR: 18, 95% CI: 3.9-85) to SJS/TEN warranting further study, which we validate for TEN with an ROR of 9.23 (95% CI: 8.31-10.24). 10 Despite PPIs’ association with TEN, AGEP, and DRESS found in the existing literature and validated with our study, they are often excluded from or labeled as low risk among lists of medications known to cause SCARs demonstrating the importance of updating the current literature.41,42
Antivirals
Nevirapine, first reported in the EuroSCAR study and then validated by others, is a medication known to be highly associated with SJS/TEN. We too identify nevirapine as being highly associated with both SJS (ROR: 34.49) and TEN (ROR: 24.15).10,43 We additionally find that acyclovir, like nevirapine, has a significant association with these reactions (ROR: 13.54 for SJS and 26.28 for TEN). Furthermore, a novel finding of valacyclovir (ROR: 39.06) and acyclovir (ROR: 61.72) with a strong signal in AGEP was found, an association that we could not identify in existing literature. For DRESS, several antivirals used in the management of HIV displayed significant ROR signals; abacavir (ROR: 26.62), raltegravir (ROR: 23.27), ritonavir (ROR: 10.69), tenofovir (ROR: 6.58), along with acyclovir (ROR: 30.63), valacyclovir (ROR: 21.77), and telaprevir (ROR: 14.16). These antiviral drugs are not commonly reported in these reactions; therefore, this is of note.
Antifungals
We report a significant association between fluconazole and SJS, TEN, and DRESS (ROR: 17.13, 30.21, and 14.04, respectively). Although there have been a number of case reports identifying this association, larger-scale pharmacovigilance studies have not yet reported this finding, including EuroSCAR, which had insufficient exposures to conclude the medication.44-46 We also identify amphotericin B, as having a strong ROR signal for TEN (ROR: 30.76), which we believe to be a novel association.
Other drugs
Various additional atypical drugs were identified to have an association with SCARS, these include ramipril in TEN and DRESS (ROR: 7.02 and 5.14), metoclopramide in SJS and TEN (ROR: 5.41 and 11.21), ondansetron in TEN (ROR: 10.44), filgrastim in TEN (ROR: 12.08), propofol in TEN (ROR: 11.46), amlodipine in TEN (ROR: 5.48), peginterferon alpha-2a in DRESS (ROR: 6.09), and hydroxyzine in DRESS (ROR: 15.21).
Limitations
These findings should be interpreted keeping in mind the limitations of the FAERS database and case/non-case studies. The medication of interest in particular reaction reported to the FDA does not require proof of causation by the reporter; therefore, we cannot make conclusions regarding the causation of the medications in this analysis. In addition, inherent limitations such as underreporting and selective reporting of reactions can result in underestimation or overestimation of ROR value. 47 These errors in reporting can be due to a variety of factors such as reporter experience, reaction severity, notoriety of a drug in causing a reaction, media coverage, etc. 47 Despite these potential biases, case-non-case analyses using a public database such as FAERS are particularly useful in studying rare adverse reactions and remain an effective means of identifying medication associations that warrant further study into potential causality.
Conclusion
Our study, in congruence with the prior literature, validates the strong association between SCARs and antibiotics, antiepileptics, NSAIDs, and antigout medications. Moreover, this study adds to the current knowledge of SCARs-associated medications finding significant results for atypical agents including, acetylcysteine, anticoagulants, diuretics, immunotherapies, PPIs, antivirals, and antifungals. Due to the potentially lethal nature of SCARs and the ever-growing number of new medications on the market, having a current list of medications with possible SCARs association is a useful tool for physicians and a basis for future studies in determining the causality of these agents.
Supplemental Material
sj-docx-1-cms-10.1177_12034754231220931 – Supplemental material for Medication Associations with Severe Cutaneous Adverse Reactions: A Case/Non-Case Analysis Using the FDA Adverse Event Reporting System
Supplemental material, sj-docx-1-cms-10.1177_12034754231220931 for Medication Associations with Severe Cutaneous Adverse Reactions: A Case/Non-Case Analysis Using the FDA Adverse Event Reporting System by Hannah Godfrey, Patrick Jedlowski and Rebecca Thiede in Journal of Cutaneous Medicine and Surgery
Supplemental Material
sj-tif-2-cms-10.1177_12034754231220931 – Supplemental material for Medication Associations with Severe Cutaneous Adverse Reactions: A Case/Non-Case Analysis Using the FDA Adverse Event Reporting System
Supplemental material, sj-tif-2-cms-10.1177_12034754231220931 for Medication Associations with Severe Cutaneous Adverse Reactions: A Case/Non-Case Analysis Using the FDA Adverse Event Reporting System by Hannah Godfrey, Patrick Jedlowski and Rebecca Thiede in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.