Abstract

To the Editor,
Rosacea is a neuroimmune-mediated dermatosis characterized by erythema, telangiectasias, and phymatous change of the face. 1 Subtypes for disease categorization exist but have largely been replaced by a phenotype approach using patients’ primary clinical features to guide management. 2 -4 Monitoring patients’ response to treatment may be difficult since objective, quantitative measures for rosacea are not widely used, and patient perception of erythema change may not match assessments performed by dermatologists. 5 Facial analysis systems designed to provide quantitative data on patients’ facial skin characteristics may be of value. 3 We tested whether patients’ change in facial redness could be measured using facial artificial intelligence (AI) technology.
The study was approved by the Wake Forest University Health Sciences IRB. Individuals ≥ 18 years of age receiving Nd:YAG 1064 nm laser therapy (vascular platform) for rosacea treatment were asked to participate. Patients receiving botulinum toxin-A injections were used as a negative control. Images were captured using a portable facial imaging device (AI Skincare, Perfect Corporation) and a computer facial imaging device (VISIA® Complexion Analysis Model Generation 7, Canfield Scientific, Inc), 1 hr before and 6 weeks after a patient’s procedure. Pre- and post-procedure scores for “redness” were recorded as percentiles and their differences calculated by subtracting the pre-procedure score from the post-procedure score, where a positive number indicates improvement. A Wilcoxon Two sample Test was performed to assess change in redness between procedures, and a Pearson correlation coefficient was calculated to determine the strength of the association between the devices. Statistical Analysis System (version 9.4, Cary, NC, USA) was used for data analytics.
Twenty-seven patients were enrolled, 23 (85%) completed the study of whom 12 had vascular laser therapy and 11 had botulinum toxin-A injection. The mean (SD) of pre- and post-procedure change in redness was −3.3 (6.5) and −11.6 (22.4) for patients receiving vascular laser and 1.9 (4) and 21 (11) for patients receiving botulinum toxin-A injection, using the portable and computer facial imaging devices, respectively (p = .014). The correlation between scores from the two devices was strongly positive (r2 = .58, p = .003, Figure 1), particularly in the patients who received laser therapy (r2 = .66, p = .019).
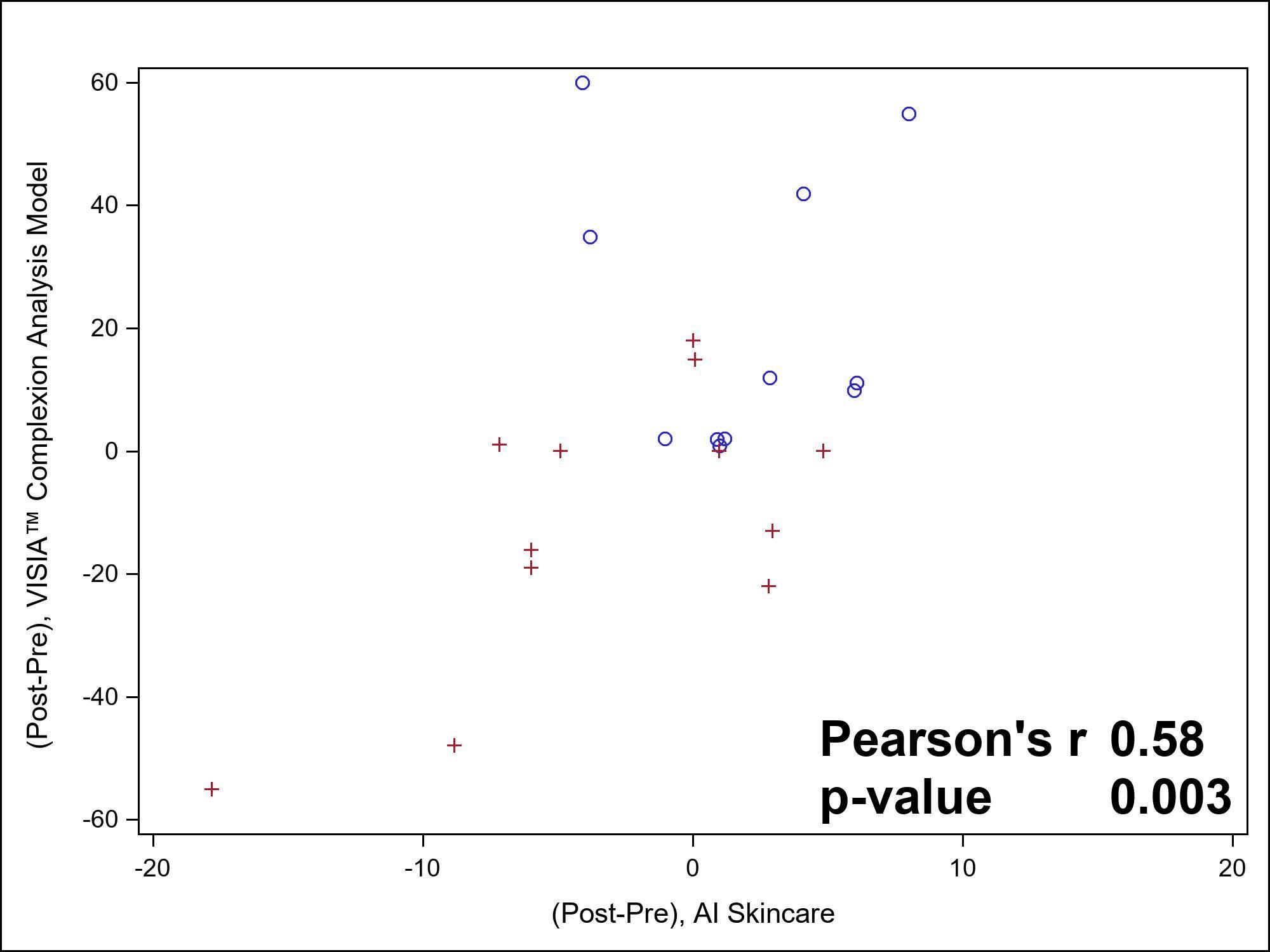
Assessing change in facial redness using a skin analysis device. The VISIA® Complexion Analysis Model Generation 7 (Canfield Scientific, Inc) and AI Skincare (Perfect Corporation) were used to assess patients’ facial redness at baseline and 6 weeks post-treatment. Mean difference in scores was compared using Pearson correlation coefficient. Each point represents an individual patient. Patients who received laser therapy are represented with a red cross and botulinum toxin-A injections with a blue circle.
Both devices captured change in facial redness in rosacea patients receiving vascular laser therapy 6 weeks post procedure. Rosacea patients require, on average, ten laser therapy treatments and response to treatment between laser therapy varies, which may have limited the results. The variance of scores was greater using the computer imaging device, which suggests greater sensitivity to change compared to the portable device; however, the cost, bulky device size, and time required to capture and analyze images using computer facial analysis systems make that device less practical compared to the portable facial imaging system. Using facial imaging software on an iPhone or iPad may be more convenient for physician offices and patients; the strong positive correlation between the portable and computer imaging device supports the use of both. Using portable facial detection software to obtain patient images at baseline and interval patient-appointments may be valuable to personalize care and enhance patient satisfaction for improved health-related outcomes.
Footnotes
Ethics Approval
Approved Institutional Review Board of Wake Forest University (IRB#00085220).
Consent for Publication
Consent for the publication of de-identifiable material was obtained by the authors.
Acknowledgments
We would like to acknowledge the Biostatistics, Epidemiology, and Research Design unit of the Wake Forest Clinical and Translational Science Institute, which is supported by the National Center for Advancing Translational Sciences, National Institutes of Health through Grant Award Number UL1TR001420.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Feldman has received research, speaking and/or consulting support from AbbVie, Accordant, Almirall, Alvotech, Amgen, Arcutis, Arena, Argenx, Biocon, Boehringer Ingelheim, Bristol-Myers Squibb, Dermavant, Eli Lilly and Company, Eurofins, Forte, Galderma, Helsinn, Janssen, Leo Pharma, Micreos, Mylan, Novartis, Ono, Ortho Dermatology, Pfizer, Regeneron, Samsung, Sanofi, Sun Pharma, UCB, Verrica, Voluntis, and vTv Therapeutics. He is founder and part owner of Causa Research and holds stock in Sensal Health. McMichael has received research, speaking, and/or consulting support from: AbbVie, Almirall, Arcutis, Bristol Myers Squibb, Eli Lilly and Co, Galderma, Janssen, Johnson & Johnson, Leo, L’oreal, Ortho Dermatology, Pfizer, Procter and Gamble, Sanofi-Regeneron, UCB. Zaino, Pixley, Kontzias, and Taylor have no conflicts to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by a starter grant from Perfect Corporation.
