Abstract
Acneiform eruptions occur frequently and early in patients on epidermal growth factor receptor inhibitors (EGFRi). Identification of baseline patient risk factors would prompt earlier referral to dermatology to optimize prevention and management. The primary objective of this retrospective study is to determine the association between clinical and demographic characteristics and the development of acneiform eruptions. A retrospective chart review was conducted on patients diagnosed with colon and head and neck cancers who started EGFRi between January 2017 and December 2021. Patients were followed until death or September 2022. Baseline demographic and clinical parameters were documented and patients were followed from the time of diagnosis to most recent visit for the development and management of an acneiform eruption. Regression analyses were performed to determine the association between baseline characteristics and the development of acneiform eruptions. A total of 66 patients were treated with cetuximab or panitumumab between 2017-2021 were included in the analysis. Forty-seven of the sixty-six patients developed an acneiform eruption while on EGFRi therapy (71.2%). Combination cancer therapy with another chemotherapeutic agent was associated with a lower risk of acneiform eruption (OR 0.03, P = .027). No other baseline features were statistically associated with a lower risk of acneiform eruption. Acneiform eruptions are a common cutaneous adverse event of EGFRi therapy. Ongoing research is required to elucidate risk factors for the development of acneiform eruptions, to improve the quality of life of oncology patients.
Keywords
Introduction
The use of targeted therapy against epidermal growth factor receptors (EGFR), a tyrosine kinase receptor, has become the standard of care for the treatment of many cancers including colon and head and neck cancers. Although these agents are associated with a lower incidence of systemic side effects than traditional chemotherapy, there is a higher frequency of mucocutaneous side effects, in 45 to 100% of patients, that severely impact the quality of life and may lead to discontinuation of life-saving treatment. 1,2
EGFR is expressed in the skin and adnexal structures and is shown to be important in keratinocyte proliferation and differentiation. 1 Although the precise mechanism of action underlying epidermal growth factor receptor inhibitors (EGFRi) triggered acneiform eruption is yet to be completely elucidated, the current consensus is that EGFRi’s affect keratinocyte proliferation in the epidermis by upregulation of p27, which leads to hyperkeratinisation and abnormal desquamation, ultimately resulting in follicular plugging. 3 Cutaneous adverse events include papulopustular/acneiform eruptions, xerosis and hair and nail changes. 4 This acneiform eruption is more common in monoclonal antibody EGFRi compared to tyrosine kinase EGFRi. 5
Evidence suggests that an acneiform eruption is associated with a favorable response to cancer therapy. 6 -9 However, it can significantly impact the quality of life of oncology patients, even when mild. In a minority of cases, the eruption is severe enough to interrupt lifesaving cancer therapy. 10,11
Limited data is available on patient risk factors for the development of acneiform eruptions. 12 -14 Given that the onset of the eruption is soon after initiating EGFR therapy, identifying high-risk groups and starting prophylactic therapy early may prevent severe cutaneous adverse reactions. Further, risk factors may be conserved amongst those who react to other targeted oncologic therapies, and therefore these data may be more broadly used by oncologists to refer early to dermatology for concurrent management.
Methods
Objectives
The primary objective of the study was to determine baseline demographic and clinical risk factors for the development of acneiform eruptions in this cohort. Secondly, we aimed to describe the association between development of acneiform eruptions and overall survival. As a tertiary endpoint, we aimed to perform an exploratory analysis of the relationship between the use of minocycline prophylaxis for EGFRi induced acneiform eruptions and overall survival.
Study Design
This is an observational, retrospective cohort study of an EGFR mutant metastatic colorectal and head and neck cancers patient population which aims to assess risk factors for the development of acneiform eruptions as well as real-world incidence of acneiform eruptions. All patients were seen and treated at the Jewish General Hospital Department of Medical Oncology in Montreal, Quebec, Canada. This study was approved by the institutional Research Ethics Board. (Protocol #2022-3122).
All patients who were started on monoclonal antibody EGFRi between January 2017 and December 2021 that were treated with first-line monoclonal antibody EGFRi (panitumumab and cetuximab) at our institution were included in this study. The following data were extracted from medical records: patient characteristics including diagnosis date, stage at time of initial diagnosis, sex, age at time of diagnosis, Eastern Cooperative Oncology Group performance status (ECOG-PS), smoking history and current smoking status; past medical history, medication history, tumor characteristics and other laboratory values including baseline blood tests including WBC, CRP, ALT, Cr, LDH, albumin; cancer treatment characteristics including name of EGFR inhibitor, start and stop dates of therapy, number of cycles and reason for treatment discontinuation. Duration of EGFRi treatment was calculated as the time (in months) elapsed between the start and end dates of the treatment. Response to treatment was defined by the treating physician as per RECIST 1.1 criteria and was based on radiographic imaging (CT/PET) and categorized for analysis purposes as an objective response [complete response (CR) +partial response (PR), stable disease (SD) and progressive disease (PD)]. 15
Acneiform eruption outcome data were obtained by reviewing scanned paper charts hosted on a computer and electronic charts. The following information regarding acneiform eruptions was obtained from clinical charts: date of the first documented acneiform eruption, timing of eruption relative to start of EGFRi, Common Terminology Criteria for Adverse Events (CTCAE) V5 grading of acneiform eruption, prevention and/or management of acneiform eruption by oncology, patient’s response to oncology’s therapy, referral to dermatology, management of acneiform eruption by dermatology, response to dermatologic therapy. Prevention or management of acneiform eruptions was characterized by: conservative measures (topical emollient or observation), topical steroids, benzoyl peroxide (BPO), topical antibiotics and oral antibiotics. If CTCAE grade was not reported, one was assigned for mild, two for moderate and three for severe acneiform eruptions.
Baseline laboratory values were defined as blood tests available on the hospital network within a timeframe of 1 month prior to beginning the EGFRi. Baseline past medical history was defined as any documented past medical history at a physician’s note prior to the diagnosis of metastatic colorectal or head and neck cancer. Baseline laboratory results were grouped as normal or abnormal using standard laboratory reference ranges. 16 If a difference between sexes for a given range existed, the more conservative cut-off value was selected.
Index date was defined as the date of starting EGFRi therapy. Database was locked on September 1, 2022 and patients were followed up until death or September 1, 2022, whichever came first. Patients referred to the Jewish General Hospital Department of Medical Oncology only for a second opinion were excluded due to lack of follow-up information. Patients who were prescribed panitumumab and cetuximab but did not complete any cycles were excluded from the analysis.
Statistical Analysis
Demographics, clinical characteristics, and treatment patterns are described using frequencies and proportions for categorical data and using means with standard deviation or medians for continuous data.
We defined an acneiform eruption as an “acne-like, papules and/or pustules” distributed on the head, neck and upper trunk. Acneiform eruptions were included in the analysis if they occurred while the patient was on EGFRi, or within 1 month of EGFRi discontinuation.
Multivariate logistic regression analysis was performed to identify the significant risk factors for the onsets of acneiform eruptions, the predictors’ effects on the outcome are presented as odds ratios (ORs) with 95% confidence intervals. The McFadden
In survival analysis, Kaplan-Meier curves were used to describe the survival time (time to death) of patients of groups stratified by the onset of acneiform eruptions and the use of minocycline, respectively. The P value of the log-rank test for the difference in survival between the groups was reported. To adjust for the impact of other observed characteristics of patients (age, sex, smoking, results of various blood tests, treatment received, etc.) on prognosis, Cox proportional hazards regression analysis with multiple covariates was performed to assess the joint effects of risk factors on survival. The estimates are presented as hazard ratios (HRs) with 95% confidence intervals.
In all analyses, the contributions of individual predictors were assessed by examining the significance of the Wald statistics; coefficients with P values < .05 were considered statistically significant. The analyses were conducted using R base function glm() (version 4.2.0) and R packages survival (version 3.5-0) and survminer (version 0.4.9). 17,18
Results
In total, sixty-six patients who started on cetuximab or panitumumab between 2017 and 2020 were included in the analysis. The majority of patients received panitumumab (n = 62, 94.0%) and (n = 4, 6.0%) received cetuximab. Sixty-two patients had a diagnosis of colorectal cancer (n = 61, 92.4%), four had squamous cell carcinoma of the head and neck (6.0%) and one (1.5%) had undifferentiated carcinoma.
The average age of the cohort was 57.8, There were twenty-seven were females (40.9%). All of the patients with recorded ECOG status had an ECOG status of zero or one at time of diagnosis. Sixty patients (91.0%) had medical comorbidities before the diagnosis of cancer. Nine patients had a known diagnosis of another primary malignancy at the time of EGFRi initiation. Fourteen patients had diabetes (21.2%), 27 had hypertension (40.9%), and five cardiovascular disease (7.6%).
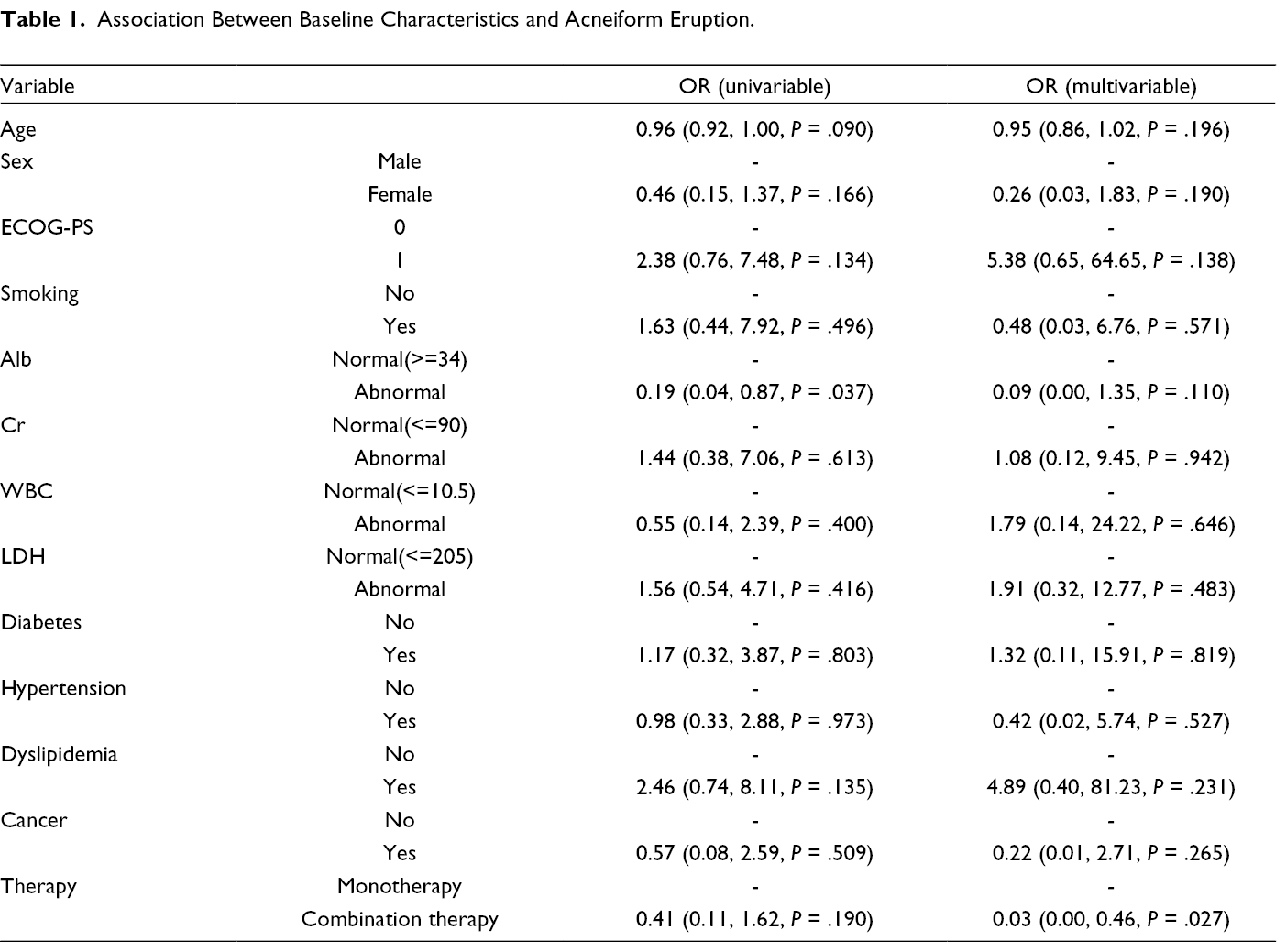
Forty-seven of the sixty-six patients included in the analysis developed an acneiform eruption while on EGFRi therapy (71.26%). Of those with recorded eruption severity, twenty-one were mild (36.2%), defined by CTCAE 1, eight were moderate (13.7%), CTCAE 2, and five severe (8.6%), CTCAE 3. The only factor associated with an increased risk of acneiform eruption included EGFRi combined with another chemotherapeutic agent with an odds ratio of 0.03 (P = .027). No other baseline factors were associated with a statistically significant association with acneiform eruption. (Table 1)
Association Between Baseline Characteristics and Acneiform Eruption.
In our cohort, rash was associated with response to therapy (OR 4.48 P = .009). In the survival analysis, there was a trend toward an association between rash and improved overall survival. (Figure 1) No association was found between tetracycline antibiotic prophylaxis and overall survival. (Figure 1)
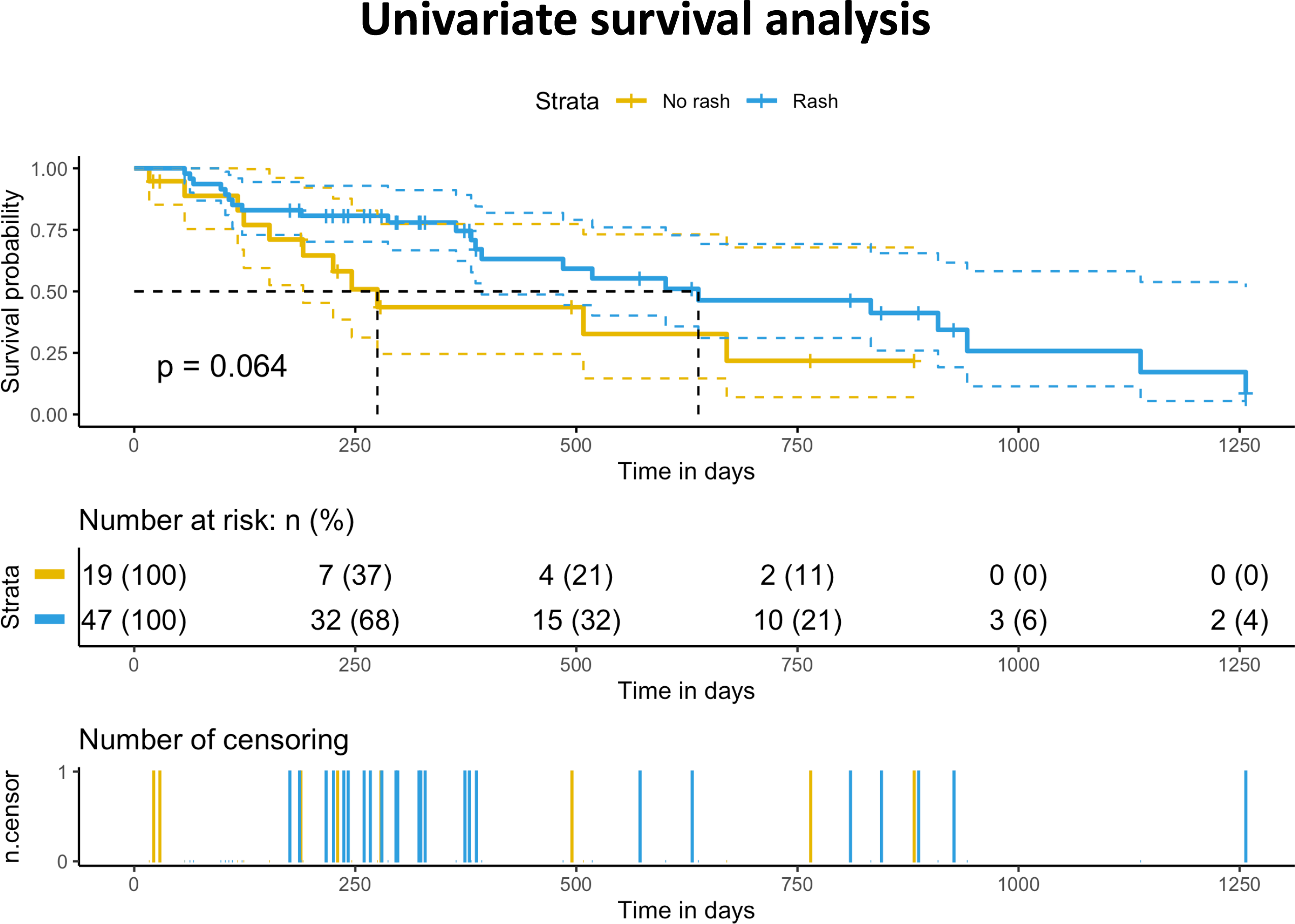
Association between overall survival and acneiform eruption.
Discussion
More than half of the patients in our cohort developed an acneiform eruption while on EGFRi therapy. Combination cancer therapy was the only baseline feature associated with a lower risk of acneiform eruptions. As a secondary endpoint, we found an association between the development of acneiform eruption and overall survival.
In our cohort, EGFRi monotherapy, in contrast to combination therapy, was associated with an increased risk of acneiform eruption. Eleven patients (16.4%) in our cohort were treated with EGFRi combination therapy, which included combinations with cytotoxic therapy (n = 9), BRAF inhibitors (n = 1) or MEK inhibitors (n = 1). This differs from what is previously reported in the literature. In a study conducted by Balagula et al., the development of an acneiform eruption was more common when patients received EGFRi, in combination with cytotoxic chemotherapy. In their paper, the group hypothesized that with multiple agents acting at the same time, there are higher rates of EGFR dysfunction, leading to the clinical findings of an acneiform eruption. 19 Surprisingly, our analyses demonstrate the contrary. We postulate that this difference may be due to pooled results of heterogeneous combination treatments with EGFRi in our cohort and our small sample size.
Given the high frequency of acneiform eruptions in patients on EGFRi, as well as a paucity of concrete data on risk factors for the development of the eruption, clinical guidelines recommend minocycline prophylaxis. 20 Minocycline is shown to decrease the severity and may prevent the development of the acneiform eruption. 21,22 With improved understanding of the role of the gut microbiome in health and disease, there are concerns of antibiotic-induced dysbiosis affecting response to targeted colon cancer therapy. 23 Therefore, in our cohort, comprised predominantly of colorectal cancer patients, we performed an exploratory analysis assessing the effect of minocycline prophylaxis on overall survival. We were unable to find a statistically significant association between minocycline and shorter overall survival, suggesting that minocycline prophylaxis does not decrease targeted therapy efficacy or impact overall survival. (Supplemental Figure 1) This initial clinical data is to be confirmed in larger trials, where this association is explored as a defined primary or secondary endpoint.
Importantly, our results demonstrate that in the real-world setting, there is a consistent association between the development of acneiform eruption and positive response to EGFRi therapy. This relationship is reported by two large meta-analyses, which show a longer overall survival in patients with acneiform eruptions. 9,24 This finding has important clinical implications. Given that the acneiform eruption occurs usually within the first two to three weeks of EGFRi therapy, the skin acts as an early marker for response to treatment, providing important prognostic information. However, the acneiform eruption may disrupt quality of life, and if severe, lead to premature treatment discontinuation. 10,11 This highlights the second clinical takeaway from this association: there is a need for multidisciplinary care in the management of oncology patients. There is demonstrated benefit from integrated dermatologic care in this patient population. 25 Dermatologists are less likely to recommend treatment discontinuation for the management of acneiform eruptions. 26 Early access to a dermatologist decreases rates of treatment discontinuation and improves the quality of life of oncology patients. 27,28
There is a growing number of targeted therapies that may cause acneiform eruptions. MEK inhibitors were one of the first targeted therapies with well documented evidence of this cutaneous adverse event. 29 There is emerging evidence for the use of EGFRi in combination with BRAF or MEK inhibitors to delay the development of EGFRi resistance. 30 When used together, EGFRi and MEK inhibitors act synergistically to cause higher rates of severe acneiform eruptions that are dose-limiting. 31 Amivantamab, a combination EGFR and mesenchymal epithelial transition factor inhibitor, and Vendatanib, a vascular endothelial growth factor, are also associated with acneiform eruptions. 32,33 Acneiform eruption led to treatment discontinuation in a study of efortumab vedotin, a novel nectin-4-derived antibody-drug conjugate. 34 With the rapid introduction of novel targeted cancer therapy, acneiform eruptions are emerging as a cutaneous adverse event across multiple classes of medications. This further underscores the importance of research on cutaneous adverse events of cancer therapy to complement ongoing cancer therapy innovation.
There are limitations to our study. The first is the small sample size of our cohort. Secondly, this study is a single center, which may not be generalizable to other patient populations. Finally, we relied upon physician reporting of skin toxicity, not a standardized or objectively quantified method of assessment. Despite limitations, our paper does contribute to the literature by providing real-world data on the incidence of cutaneous adverse events of targeted cancer therapy, and by identifying the uncertainty that remains regarding risk factors for acneiform eruptions.
Conclusions
Acneiform eruptions are a common cutaneous adverse event of EGFRi therapy and are emerging as a cutaneous adverse event reported with the use of novel targeted therapies. In our cohort, the use of EGFRi monotherapy was associated with an increased risk of acneiform eruption. Importantly, our real-world data demonstrate an association between acneiform eruption and positive response to EGFRi therapy. Our findings highlight the importance of continued investigation into the risk factors of acneiform eruptions, with the ultimate objective of improving the quality of life of oncology patients.
Supplemental Material
Figure S1 - Supplemental material for An Analysis of Risk Factors for the Development of Acneiform Eruptions in Patients on Monoclonal Antibody Epidermal Growth Factor Receptor Inhibitors
Supplemental material, Figure S1, for An Analysis of Risk Factors for the Development of Acneiform Eruptions in Patients on Monoclonal Antibody Epidermal Growth Factor Receptor Inhibitors by Rachel Bierbrier, Kathleen D’Aguanno, Sarah Oliel, Yixiao Zeng, Khashayar Esfahani and Kevin Pehr in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
