Abstract
Introduction
Human herpesvirus-6 (HHV-6) is a ubiquitous lymphotropic betaherpesvirus that can reactivate in drug rash with eosinophilia and systemic symptoms (DRESS). Despite recent publications advancing our understanding of HHV-6 in DRESS, the exact role of HHV-6 in disease pathogenesis remains unclear.
Methods
A scoping review with the PubMed query “(HHV 6 AND (drug OR DRESS OR DIHS)) OR (HHV6 AND (drug OR DRESS OR DIHS))” was conducted in accordance with PRISMA guidelines. Articles containing original data on at least one DRESS patient with HHV-6 testing were included.
Results
Our search returned a total of 373 publications, of which 89 met eligibility criteria. HHV-6 reactivation occurred in 63% of DRESS patients (n = 748), which was significantly more often than other herpesviruses. HHV-6 reactivation was associated with worse outcomes and greater severity in controlled studies. Case reports have demonstrated sometimes fatal HHV-6-related multi-organ involvement. Temporally, HHV-6 reactivation typically occurs 2 to 4 weeks after DRESS onset and has been linked to markers of immunologic signaling, such as OX40 (CD134), an HHV-6 entry receptor. Efficacy of antiviral or immunoglobulin treatment has only been demonstrated anecdotally, and steroid use may affect HHV-6 reactivation.
Conclusion
HHV-6 is implicated in DRESS more than in any other dermatologic condition. It is still unclear whether HHV-6 reactivation is cause or consequence of DRESS dysregulation. Similar pathogenic mechanisms precipitated by HHV-6 in other contexts may be relevant in DRESS. Future randomized controlled studies to assess effects of viral suppression on clinical outcomes is needed.
Keywords
Introduction
Drug-induced hypersensitivity syndrome (DIHS)/drug rash with eosinophilia and systemic symptoms (DRESS) is a drug-induced delayed hypersensitivity reaction that occurs 2 to 6 weeks after drug initiation. 1 Along with Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and acute generalized exanthematous pustulosis, it is considered a severe cutaneous adverse reaction (SCAR).
Currently, there are 2 sets of criteria for DIHS/DRESS. The Registry of Severe Cutaneous Adverse Reaction (RegiSCAR) scoring system for DRESS includes fever, lymphadenopathy, hypereosinophilia, atypical lymphocytes, skin involvement, internal organ involvement, time to resolution, and laboratory evaluation for other causes. A final score of <2 indicates no case; 2 to 3, possible case; 4 to 5, probable case; and >5, definite case. 2 The Japanese consensus group criteria for DIHS include development of a maculopapular rash >3 weeks after starting therapy with a limited number of drugs, prolonged clinical symptoms 2 weeks after discontinuation of the causative drug, fever (>38 °C), liver abnormalities (ALT >100 U/L) or other organ involvement, leukocyte abnormalities, lymphadenopathy, and human herpesvirus-6 (HHV-6) reactivation. Typical DIHS requires satisfaction of all 7 criteria, while atypical DIHS may be diagnosed without the latter two. 3 Despite these minor distinctions, DIHS and DRESS can generally be considered one condition and will be referred to as DRESS in the sections below.
HHV-6 is a lymphotropic betaherpesvirus that typically infects CD4 +T cells. It is generally acquired between ages 6 and 15 months, causing exanthem subitum (roseola infantum). 4 After primary infection, HHV-6 becomes latent but can undergo reactivation in vulnerable states, such as immunosuppression. 4 HHV-6 reactivation has been associated with DRESS. 5 While some posit that HHV-6 plays the role of an innocent bystander, others propose that DRESS and its various sequelae are a consequence of severe herpesvirus reactivation. 6 The exact role of HHV-6 remains unclear, and from a clinical standpoint, addressing HHV-6 reactivation in the context of DRESS is not often considered. To better understand the effect of HHV-6 on the pathogenesis of DRESS and its potential implications on treatment, we reviewed the literature from the past decade. This scoping review focuses particularly on the rate of HHV-6 reactivation compared to other herpesviruses; the relationship between HHV-6 reactivation and disease severity; the temporality of HHV-6 reactivation; the relationship between HHV-6 reactivation, immunologic parameters, and treatment; and the possibility of a rarer form of HHV-6 infection characterized by chromosomal integration.
Methods
We conducted a PubMed search of articles from January 1, 2012 to January 11,2023 using the query “(HHV 6 AND (drug OR DRESS OR DIHS)) OR (HHV6 AND (drug OR DRESS OR DIHS)).” We included all articles with original data that tested for HHV-6 in at least 1 human DIHS/DRESS patient and excluded non-English articles. This query returned 373 articles, of which 240 were excluded by title and abstract screening. The full text of 130 articles were retrieved, and 41 were excluded based on inclusion and exclusion criteria screening, yielding 89 total articles for our review. A PRISMA flowchart summarizing this can be found in Figure S1. Screening was performed by both authors. This protocol was not prospectively registered.
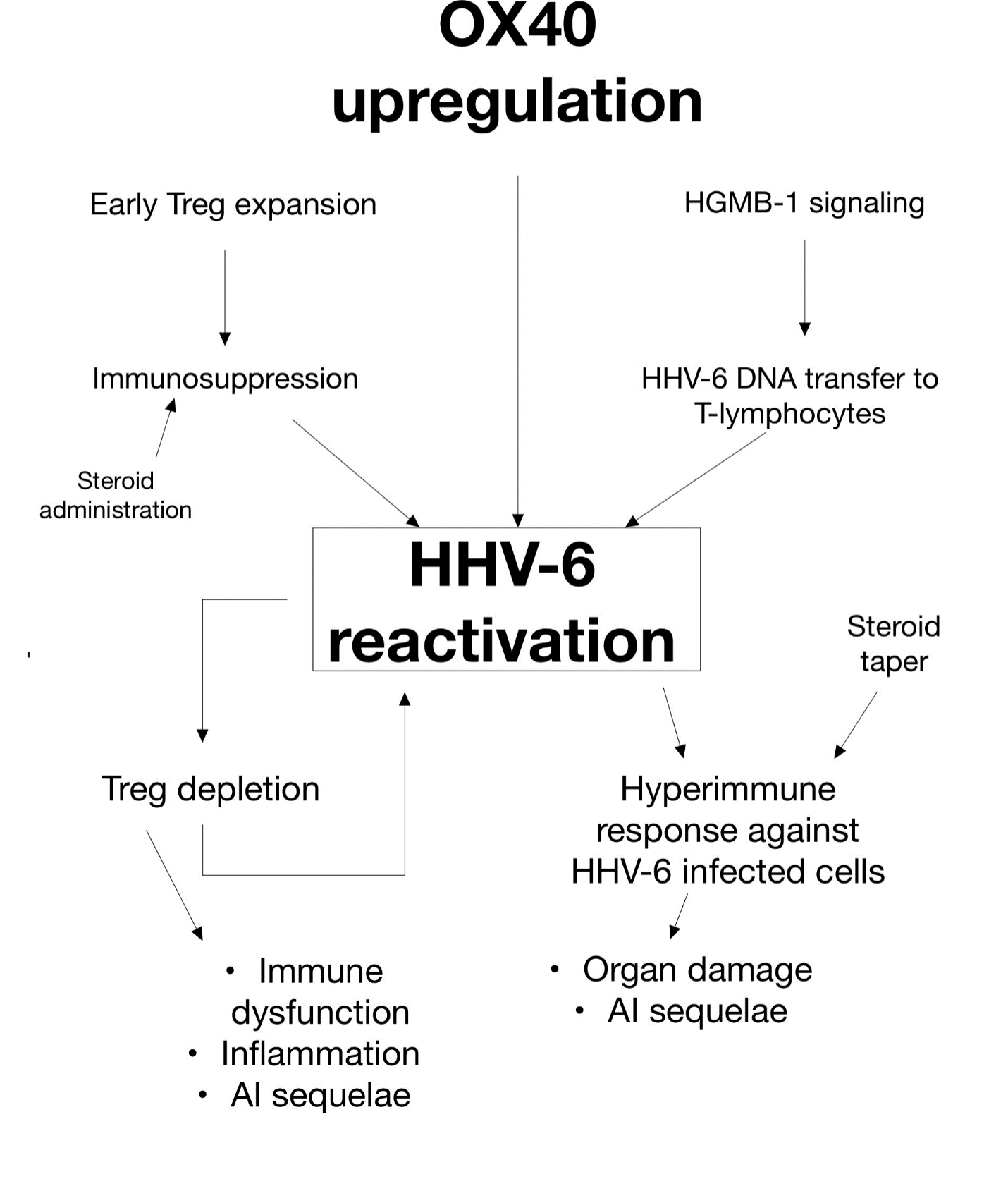
Potential precipitating and downstream events of HHV-6 reactivation in DRESS. OX40 upregulation, Treg expansion, and HGMB-1 signaling observed in DRESS may be responsible for preferential HHV-6 reactivation. HHV-6-induced Treg depletion and subsequent immune response could then lead to further adverse events. †AI – autoimmune, DIHS – drug-induced hypersensitivity syndrome, DRESS – drug rash with eosinophilia and systemic symptoms, HGMB-1 – high mobility group box-1, HHV-6 – human herpesvirus-6, Treg – regulatory T-cell.
Results
Of the 89 total articles, 56 were case reports and 33 cohort studies. Altogether, 748 DRESS patients were tested for HHV-6. Of the 748, 470 (62.8%, 95% CI 0.59-0.66) were found to have HHV-6 reactivation. Clinical data for HHV-6 positive cases can be found in Supplemental Table 1 (n =51) and HHV-6 negative case reports can be found in Supplemental Table 2 (n=5). Studies involving greater than one case are summarized in Supplemental Table 3. Interestingly, among these 748 patients, cytomegalovirus (CMV), Epstein-Barr virus (EBV), and HHV-7 was detected in 74/297 (24.7%, 95% CI 0.20-0.30), 80/264 (30.4%, 95% CI 0.25-0.36), and 25/156 (16.0%, 95% CI 0.11-0.23), respectively. These findings suggest that HHV-6 reactivation occurs at significantly higher rates than other herpesviruses in DRESS.
Relationship Between HHV-6 Reactivation and DRESS Severity
Compared to HHV-6 negative patients, HHV-6 reactivation in DRESS was found to be significantly associated with more long-term sequelae, 7 longer hospital stays, 8 increased fever likelihood and duration, 8,9 and more flares. 9 Those with HHV-6 reactivation also had significantly higher severity scores, 9 which correlated with greater HHV-6 microRNA (miRNA) expression. 10 Compared to those without autoimmune sequelae, those who developed autoimmune sequelae also had significantly higher rates of HHV-6 reactivation. 11
Multi-organ system involvement have been reported with evidence of HHV-6 reactivation within specific organ parenchyma or in serum, sometimes leading to fatal sequelae related to the liver, 12 -19 kidney, 7,12,20 -23 gastrointestinal tract, 24,25 bone marrow, 12,26 -28 lymph nodes, 12,29,30 peripheral and central nervous system, 7,31 -34 heart, 13,35 and lung. 36,37 There have been many reports of new-onset autoimmune disease, as well. 7,9,26,38,39 However, association of HHV-6 with severe, organ-specific sequelae are mostly limited to case reports, which precludes the formation of strong conclusions. Research directly comparing the extent of HHV-6 reactivation in patients with severe, organ-specific sequelae to those with less severe disease courses is needed. Measuring HHV-6 reactivation in organ parenchyma would help assess the role of HHV-6 in organ-specific disease pathogenesis (e.g., fulminant kidney failure). Additional information regarding complications associated with HHV-6 reactivation can be found in Supplemental Tables 4 and 5.
Temporal Aspects of HHV-6 Reactivation in DRESS
Recent data suggest that HHV-6 reactivation typically occurs 2 to 4 weeks after DRESS symptom onset, 9,10,40 -43 and in the case reports reviewed, HHV-6 reactivation occurred, on average, 21.6 (95% CI 17-26.2) days after DRESS onset. HHV-6 miRNA are not readily detected during the acute stages of DRESS but may appear before HHV-6 DNA viremia. 10 Milani-Nejad et al. intentionally tested for HHV-6 DNA on day 1 after DRESS symptom onset to demonstrate lack of positivity (0/24), 44 and in skin biopsies, Hashizume et al. found a sharp increase in the expression of HHV-6 antigen from day 7 to day 14 after DRESS onset. 45 As such, reports of negative HHV-6 assessed only once immediately or shortly after DRESS onset may have missed later reactivation. 8,11,44,46 -50 While there are many reports of delayed seroconversion or reactivation, 9,15,33,37,51 -54 sometimes with initially undetectable viral loads, 21,27,55,56 HHV-6 reactivation at the onset of DRESS is possible, as well. 8,28,32,57,58 It should be noted that HHV-6 reactivation has not been reported before DRESS onset, which suggests that HHV-6 reactivation likely does not cause DRESS. However, it is possible that reactivation exacerbates downstream sequelae.
HHV-6 and Immunologic Signaling
Data pertaining to relevant immunologic findings of specific studies can be found in Supplemental Table 6. Recent studies have linked specific immunologic upregulation to HHV-6 reactivation and is relevant to understanding the mechanisms behind HHV-6 reactivation and its downstream sequelae. This is summarized in Figure 1.
Th2 Cytokines
Th2 cytokines, such as thymus and activation regulation chemokine (TARC)/CCL17 has been positively associated with DRESS score and severity, 22,59 as well as HHV-6 DNA levels and reactivation. 22,59,60 Compared to those with other SCARs, patients with DRESS have been found to have significantly higher levels of TARC, 59 which tends to be elevated early in the clinical course, even before HHV-6 reactivation. 21,24,61
Upregulation of C-C motif chemokine receptor 4 (CCR4), a TARC receptor that leads to Th2 activation, 62 may also be involved in HHV-6 reactivation. 63 Circulating CCR4+ monomyeloid precursors harboring HHV-6 DNA have been elevated in early DRESS (<21 days) but not other SCARs. 45 These HHV-6 DNA-harboring monomyeloid precursors potentiate the transmission of HHV-6 DNA to CD4 +T cells for active replication. 45 HHV-6 can then persist in T-cells at stable levels, 55,57 perhaps lowering the threshold for future reactivation. High mobility group box-1 (HMGB-1), a chemoattractant for CCR4+ monomyeloid precursors, has also been significantly elevated in DRESS but not SJS patients, 45 an example of chemokinetic signaling that may promote HHV-6 reactivation (Figure 1). Interleukin (IL)−10 and IL-5 have also been significantly associated with higher TARC or HHV-6 DNA levels. 22,64,65
OX40 – HHV-6B Entry Receptor
HHV-6 can be further divided into two viral strains, HHV-6B and HHV-6A. The former is most associated with disease pathogenesis and DRESS. 66 The entry receptor for HHV-6B is the CD4 +T cell receptor OX40 (CD134). 67 OX40 has significantly correlated with increased TARC, HHV-6 viral load, and DRESS severity. 43,64,68 Like CCR4, OX40 has been significantly upregulated before peak HHV-6 reactivation in patients with DRESS compared to controls and patients with other SCARs. 43,64,69 In skin biopsies, the proportion of OX40+ CD4 T-cells is also significantly increased in DRESS compared to erythema multiforme, further supporting a unique role of OX40 in DRESS. 68 Since OX40 levels peak just before HHV-6 viral loads peak, 64 OX40 elevation may be a crucial precipitating event for HHV-6 reactivation (Figure 1).
Regulatory T-Cells (Tregs)
Regulatory T-cells (Tregs) are derived from CD4+ T cells and are naturally responsible for suppressing the immune response. 70 However, overactivity may impair immune responses to pathogens. 70 Thus, dysfunction of Tregs play a large role in DRESS and viral reactivation. Expansion of Tregs in the acute stages of DRESS, but not SJS/TEN, may explain reduced lesion severity in DRESS and concomitant suppression of the body’s innate antiviral capabilities, 71 resulting in an increased likelihood of HHV-6 reactivation. HHV-6 reactivation may then lead to infection and depletion of CD4+ Tregs (Figure 1), a mechanism which has been proposed in HHV-6-related acute graft-versus-host disease (aGVHD). 72 In the acute stages of DRESS (2-15 days after onset), roughly 10% of OX40-expressing CD4+ cells have been found to be Tregs, 69 potentially facilitating HHV-6 infection and subsequent downregulation. A fatal case of HHV-6 infection in bone marrow Tregs has also been reported. 12 Additionally, HHV-6-induced Treg dysfunction may contribute to significant positive correlations between HHV-6 reactivation and inflammatory markers, such as lactate hydrogenase (LDH) and C-reactive protein (CRP), in DRESS. 60,73
Th1 Cytokines
Dysregulation of Th1 chemokines has also been seen with HHV-6 reactivation. Increased tumor necrosis factor-α (TNF-α) has been shown to precede HHV-6 reactivation. 65,73 In occupational trichloroethylene hypersensitivity syndrome (OTHS), a delayed hypersensitivity syndrome clinically similar to DRESS, HHV-6 reactivation has significantly correlated with increased interferon-γ (IFN-γ). 65 Increased C-X-C motif chemokine ligand 10 (CXCL10)/IP-10 expression has been associated with both HHV-6 reactivation and long-term sequelae in DRESS, 7,9 as has increased expression of C-X-C motific chemokine receptor 3 (CXCR3), the receptor for CXCL10/IP-10. 7 Soluble IL-2 receptor (sIL-2R) has been elevated in HHV-6 reactivation 29 and has correlated with increased HHV-6 viral load. 41 An increase in IL-6 has also been detected before peak HHV-6 reactivation. 9,65,73
Interestingly, Chen et al. demonstrated that patients who eventually underwent HHV-6 reactivation had decreased levels of various chemokines (e.g., IFN-γ, IL-1Rα, IL-1β, IL-2, IL-6, macrophage inflammatory protein 1α, and TNF-α) during early DRESS (<10 days from onset) compared to those who did not. 9 With the exception of CXCL10/IP-10, which exhibited significant early elevation, these chemokines rose significantly after HHV-6 reactivation but gradually decreased in patients without HHV-6 reactivation. 9 Initial downregulation of these proinflammatory chemokines may be associated with decreased antiviral activity via an unclear mechanism, perhaps related to Treg upregulation in early stages, 71 thus predisposing to later HHV-6 reactivation. 9
Treatment of HHV-6 Reactivation in DRESS Patients
Discontinuation of the offending drug is of utmost importance in managing DRESS. Initiation of systemic steroids is also a core part of treatment. 74 Oral prednisolone 40 to 60 mg daily is acceptable, but some opt for 1.0 to 1.5 mg/kg intravenous methylprednisolone, depending on severity. 74 Topical corticosteroids may be sufficient for mild disease. 74 However, steroids may increase susceptibility to infection. Ishida et al. demonstrated that systemic steroids increased the duration and degree of HHV-6 reactivation. 40 Interestingly, Tohyama et al. found that administration of high-dose steroids (>1.0 mg/kg/day) within 1 week of DRESS onset decreased the likelihood and degree of HHV-6 reactivation, as well as sIL-2R and TARC levels; however, no steroids, low-dose steroids given at any time, or high-dose steroids given a week after DRESS onset resulted in elevated HHV-6 viral load. 41 Chen et al. reported no effect on the likelihood of HHV-6 reactivation with the administration of high-dose systemic steroids within the first week of hospitalization. 9 Larger, prospective studies are needed to validate the efficacy of early steroid administration in the prevention of HHV-6 reactivation in DRESS patients.
Antivirals, such as ganciclovir or foscarnet, have demonstrated efficacy anecdotally. 15,17,18,20,31,36,56,75 Complete resolution of DRESS has been reported in transplant patients on systemic steroids, who were given ganciclovir or valganciclovir. 58,75 Kim et al. successfully treated a steroid-refractory case with tofacitinib, a Janus kinase inhibitor, and valganciclovir. 63 Additionally, agents with anti-HHV-6 activity have reduced drug-induced T-cell proliferation in a dose-dependent manner. 63,76 -78
A few reports have demonstrated neutral to positive outcomes with intravenous immunoglobin (IVIg) administration, 26,31,33,35,54,79 while a small study of 6 patients reported no clinical efficacy or impact on viral reactivation. 80
Chromosomally Integrated HHV-6
Approximately 1% of the population has a full HHV-6 genome integrated into the telomere of each chromosome, resulting in chromosomally integrated HHV-6 (ciHHV-6). 81 These individuals consistently have a high whole blood HHV-6 viral load (>5.5log10 copies/mL), which can be mistaken for active infection. 81 ciHHV-6 individuals will additionally exhibit HHV-6 DNA positivity in somatic cells, such as hair follicles. 81
Only 1 DRESS patient with ciHHV-6, who unsurprisingly exhibited HHV-6 viremia throughout the entire clinical course, has been reported. 73 ciHHV-6 was ruled out in 3 patients, 24,57,82 but not in others with suspiciously high HHV-6 viral loads (5log10 to 6log10 copies/mL). 13,15,19,83 Thus, the true incidence of DRESS with ciHHV-6 may be underestimated. DRESS patients with high viral loads may benefit from ciHHV-6 testing to avoid misdiagnosis of HHV-6 reactivation and subsequent mistreatment. 81 Additionally, certain drugs, such as histone deacetylase inhibitors, are known to induce ciHHV-6 replication in vitro. 81 Although no studies have linked ciHHV-6 to greater odds of DRESS, testing for ciHHV-6 may provide insight on whether ciHHV-6 is a DRESS risk factor.
Discussion
HHV-6 reactivation outside of DRESS has been associated with severe and significant complications, including multi-organ system involvement and autoimmune sequelae. 5,84 -88 However, testing for HHV-6 infection can be nuanced. As >90% of individuals are seropositive for latent IgG HHV-6 antibodies, serology taken at 1 point in time is not an ideal determinant of active infection. 89 Reactivation is best supported by serology when there is a rapid, >4 fold increase in IgG or IgM antibodies between 2 points in time (i.e., seroconversion) or evidence of active IgM antibodies. 89,90 Alternatively, detection of viremia by quantitative polymerase chain reaction is typically considered evidence of active infection. 89,90 Whole blood remains the most reliable specimen for assessing viremia, but ciHHV-6 individuals will have consistently high whole blood levels, regardless of active infection status. PBMC viral load is also valid, as HHV-6 is lymphotropic. 89 Testing too early may miss reactivation, as the average time to HHV-6 reactivation is reported to be 2 to 4 weeks after DRESS onset. Miyagawa et al. also reported that HHV-6 DNA could not be detected in the serum of patients with DRESS until viral DNA load had peaked in PBMCs. 55 Furthermore, HHV-6 may reactivate within specific organs, such as the liver 47 or heart, 91 and elude detection in the blood.
Detection of small non-coding RNA (sncRNA) has recently emerged as a more sensitive method of detecting early HHV-6 reactivation in DRESS compared to traditional measurements of viral DNA load. 92 miRNA may also be a more sensitive marker for early HHV-6 detection. 10 With the recent development of better HHV-6 detection methods, prophylactic HHV-6 screening for patients at higher risk of DRESS may have clinical benefit. Wang et al. demonstrated that HHV-6 detection is strongly predictive of OTHS, an HHV-6 related hypersensitivity syndrome (OR = 5.71, P = .02). When paired with high-risk human leukocyte antigen (HLA) typing, the positive predictive value increased drastically (OR = 91.80, P < .001). 93 OTHS is almost identical to DRESS and likely arises via similar mechanisms, though it is due to hypersensitivity to occupational trichloroethylene and has a longer latency period. 94 Upregulation of chemokines such as IFN-γ are seen in both DRESS and OTHS. 95
The potential of HHV-6-induced Treg dysregulation precipitating aGVHD 72 may partly explain the clinical similarity between DRESS and aGVHD. 75 In the setting of hematopoietic stem cell transplantation (HSCT), HHV-6 was found to be an independent significant risk factor (HR 2.65 p < .001) for aGVHD in a 2018 meta-analysis. 96 When the donor source in HSCT is from umbilical cord blood, HHV-6 is preferentially reactivated, and similarly to DRESS, there are significant correlations between viral load and OX40 levels. 97,98 In umbilical cord blood transplantation, HHV-6 reactivation was also associated with the development of aGVHD (HR 3.00 P = .004). 99
aGVHD and DRESS are both type IV hypersensitivity reactions with similarities in presentation (e.g., fever, morbilliform eruption, lymphadenopathy, multiorgan involvement, and abnormal laboratory findings). The possibility of HHV-6 playing an exacerbating role in both conditions has not been ruled out, as demonstrated by a case of DRESS complicated by GVHD enterocolitis. 25
Anti-OX40 antibodies have led to reduced aGVHD severity in mice and reduction of T-cell activity. 100,101 Decreasing HHV-6 infection via downregulation of its main entry receptor may contribute to positive therapeutic outcomes in DRESS, as well. As reviewed above, upregulation of the HMGB-1/CCR4 or OX40 axes have been directly implicated in HHV-6 reactivation (Figure 1) and could represent future therapeutic targets. Improved outcomes with decreased HHV-6 replication as a result of immunologic targeting would support an active role of HHV-6 reactivation in exacerbation of disease.
Because OX40 activation can exacerbate states of immune dysfunction via T-cell expansion and downregulation of Tregs, 102 it may potentially serve as a biomarker for DRESS severity and/or HHV-6 reactivation. Data on immunomodulation in DRESS is scarce. Aside from Janus kinase inhibitors and valganciclovir, 63 TNFα inhibitors have been used with success. 76,77 As HHV-6 is known to induce expression of TNFα, 78 potential treatments could downregulate such HHV-6-associated inflammation.
As in other settings with HHV-6 reactivation, cytokine-induced immune responses, such as increased natural killer cell activity against HHV-6 infected cells, 87,103 could precipitate autoimmune disease and organ damage in DRESS patients. Furthermore, as systemic steroids are tapered, flares may arise due to an increased immune response against reactivated HHV-6. This is reflected in reports of subacute thyroiditis or inflammatory bowel disease (IBD) during steroid taper 16,51 and may be analogous to the immune reconstitution inflammatory syndrome seen in HIV patients. 71 HHV-6 is implicated in many proinflammatory disease states, including inflammatory cardiomyopathy and multiple sclerosis, 104,105 and could have similar pro-autoimmune effects in DRESS.
If HHV-6 plays a role in the development of disease sequelae, then directly targeting viral replication with antiviral treatment may be of clinical benefit. HHV-6 antivirals also address the reactivation of other non-trivial herpesviruses, such as CMV. Prospective monitoring and treatment of active herpesvirus infection in DRESS have been increasingly proposed. 6,17,26,74 However, treatment for viral reactivation is often not considered in the clinical setting, and data for such intervention is scarce outside of anecdotal reports. Further studies are needed to assess the efficacy of antivirals. In HSCT patients, ganciclovir and foscarnet have demonstrated efficacy and are currently recommended for treatment of severe HHV-6 sequelae (i.e., HHV-6 encephalitis). 88 While clinical efficacy is unconfirmed, cidofovir and its derivatives have also shown in vitro activity against HHV-6. 106
IVIG treatment is pooled antibody replacement therapy primarily indicated for immunodeficient states, as in bone marrow transplantation or HIV infection. 107 Immunodeficiency may be induced in DRESS, contributing to HHV-6 reactivation. Hypogammaglobulinemia and decreased peripheral B-cells have been associated with both DRESS and HHV-6 reactivation. 7,108 Thus, neutralizing antibodies in IVIG may have antiviral effects against HHV-6 reactivation. However, evidence for its use is scarce. Treatment with IVIG has resulted in modest outcomes, with anecdotal success, 26,31,33,35,54,79,80,104 but treatment may be more effective in patients with hypogammaglobulinemia or more severe immune deficiency. However, another small cohort demonstrated that 18/18 patients improved after 72 hr after administration of IVIG, with minimal adverse reactions. 109 There are currently no randomized controlled trials for antiviral or IVIG treatment in DRESS, and their effects on HHV-6 reactivation and clinical outcomes requires further research.
Limitations
Limitations of this scoping review include lack of data to provide pooled quantitative data comparing HHV-6 positive and HHV-6 negative patients. Heterogeneity between measures and outcomes also made direct comparison difficult. Nonetheless, we were able to report cumulative data for figures such as overall reactivation rate and average time point of reactivation. Case studies are prone to publication bias as well, potentially explaining the low number of HHV-6 negative cases (n = 5). Additionally, relevant non-PubMed indexed studies may have been missed in our search. However, the studies included in this review are likely representative, as cohorts came from diverse origins.
With a virus as ubiquitous as HHV-6, demonstrating a causative role in any event is difficult, but given the recent advances in assessing active versus latent infection, it is possible to correlate events of interest with the degree of active infection, such as through viral load or DNA expression measurements. We encourage future researchers to consider these points regarding HHV-6 testing in order to increase standardization for potential pooled analysis in the future.
Conclusion
HHV-6 reactivation is more closely associated with DRESS than any other dermatologic condition. Preliminary cohort studies have associated HHV-6 reactivation with worse outcomes and severity. Although recent studies have advanced our understanding, it is not entirely clear whether HHV-6 is a cause of consequence of the dysregulation seen in DRESS. However, given the mechanisms by which HHV-6 exacerbates sequelae in other settings, it is plausible that similar mechanisms could be at play in DRESS. Randomized controlled trials to assess the effects of viral suppression and further research correlating HHV-6 reactivation to clinical outcomes will shed light on the true role of HHV-6 reactivation in DRESS.
Supplemental Material
Figure S1 - Supplemental material for Immunopathogenic Insights on Preferential Human Herpesvirus-6 Reactivation in Drug Rash With Eosinophilia and Systemic Symptoms: A Scoping Review
Supplemental material, Figure S1, for Immunopathogenic Insights on Preferential Human Herpesvirus-6 Reactivation in Drug Rash With Eosinophilia and Systemic Symptoms: A Scoping Review by Harrison Zhu and Vicky Ren in Journal of Cutaneous Medicine and Surgery
Supplemental Material
Tables - Supplemental material for Immunopathogenic Insights on Preferential Human Herpesvirus-6 Reactivation in Drug Rash With Eosinophilia and Systemic Symptoms: A Scoping Review
Supplemental material, Tables, for Immunopathogenic Insights on Preferential Human Herpesvirus-6 Reactivation in Drug Rash With Eosinophilia and Systemic Symptoms: A Scoping Review by Harrison Zhu and Vicky Ren in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
