Abstract
Chilblain-like lesions (CLL) coinciding with Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) infection have been described in the literature. Available reviews of the literature suggest that CLL are associated with younger age, an equal sex ratio, negative testing for SARS-CoV-2, and mild to no extracutaneous manifestations (ECM) associated with COVID-19 infection. This systematic review aims to provide a summary of reports of CLL associated with the early SARS-CoV-2 pandemic in children to clarify the prevalence, clinical characteristics, and resolution outcomes of these skin findings. Sixty-nine studies, published between May 2020 and January 2022, met inclusion criteria and were summarized in this review, representing 1,119 cases of CLL. Available data showed a slight male predominance (591/1002, 59%). Mean age was 13 years, ranging from 0 to 18 years. Most cases had no ECM (682/978, 70%). Overall, 70/507 (14%) of patients tested positive for COVID-19 using PCR and/or serology. In the majority the clinical course was benign with 355/415 (86%) of cases resolving, and 97/269 (36%) resolving without any treatment. This comprehensive summary of pediatric CLL suggests these lesions are rarely associated with COVID-19 symptoms or test positivity.
Introduction
The outbreak of coronavirus disease 2019 (COVID-19), caused by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), was declared a global pandemic on March 11, 2020. Since then, there has been a surge in the literature reporting chilblain-like lesions (CLL) also known as “COVID toes.” 1 CLL morphologically resemble chilblains: erythematous to violaceous acral macules and plaques, which may evolve to become vesicobullous or crusted lesions. 2,3 Like chilblains, they may be associated with pruritus and/or pain. A systematic review by Mirza et al. (2021) including over 2,500 patients found that CLL represent the most common cutaneous manifestation associated with COVID-19 (52%). 4 CLL have also been associated with COVID-19 vaccination. 5
Several reviews have summarized reports of CLL in the pediatric population during the COVID-19 pandemic. A review by Molaee et al. (2022) 3 included 433 pediatric patients with mean age 13 ± 2 years; of those 54% were male. Similarly, Andina et al. (2021) 2 reported that CLL usually occurred in children over ten years of age, with an approximately equal male-female ratio. 2 CLL predominantly affected the feet and occasionally the hands, 2,3,6,7 and sometimes were associated with symptoms of pruritus, tenderness, pain, a burning sensation, tingling, or coldness. 7 Rates of positive COVID testing were low across all reviews with 92% negative in Shah et al. 6 and negative in almost all cases in Andina et al. and Molaee et al. 2,3 Andina et al. reported that most cases were not associated with extracutaneous manifestations (ECM) of COVID-19 infection, 2 whereas other reviews reported respiratory or gastrointestinal symptoms and fever. 6,7 All cases in Andina et al. had spontaneous regression of CLL without complications, with resolution times ranging from 12 days to over 8 weeks 2 ; similarly Khalili et al. reported that CLL usually resolved without treatment after 7 to 10 days. 7
Despite available data on CLL in pediatric patients, a comprehensive summary of CLL during the early COVID-19 pandemic is lacking. This paper aims to summarize reports of CLL in the pediatric population to clarify the clinical relevance of these skin findings.
Methods
This systematic review was conducted in adherence to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines.
Study Inclusion Criteria
Eligibility criteria for this review were established using the population, intervention, comparator, outcomes, and study design (PICOS) framework. Studies were included if they met the predetermined PICOS criteria:
Literature Search and Screening
A literature search was conducted using the MedLine, Embase, and Scopus databases on January 8, 2022 using the search terms “COVID-19” OR “coronavirus” OR “2019-nCoV” OR “SARS-CoV-2” AND “chilblain-like” OR “covid toes” OR “acral.” The references of included studies were reviewed to ensure relevant studies were not missed. Title, abstract and full-text screening was conducted in duplicate by two reviewers (S.S. and I.M.) using Covidence online systematic review software (www.covidence.org). At the full-text screening stage, studies were excluded if they did not meet components of the PICOS eligibility criteria. Any conflicts between reviewers were resolved by discussion until a consensus was reached. Our database searches did not include any language or geographical restrictions. Level of evidence for included articles was determined using the Oxford Centre for Evidence-Based Medicine 2011 Levels of Evidence. 8
Data Extraction
Data extraction was performed by one reviewer (S.S.) using a standardized extraction form which included study characteristics (publication year, country, study design, and sample size), patient characteristics (age and sex of participants), SARS-CoV-2 information (ECM, SARS-CoV-2 test result, type of testing), CLL clinical features (location and morphology of lesions), associated cutaneous symptoms (e.g., pruritus, pain, swelling), temporal association of ECM and onset of CLL), and outcomes (treatment, response outcome, response time, recurrence). For studies reporting data on the same patient population via different publications, the data from only one study (unique patient population) was included in our analysis in order to avoid ‘double counting’ cases.
Data Synthesis
After data collection, it was determined that quantitative evidence synthesis was not feasible due to heterogeneity in study design and data reporting. Instead, results are presented in narrative form with descriptive analysis of available data.
Results
After title and abstract screening of 354 studies and full text review of 184 studies, ultimately 69 studies met inclusion criteria as outlined by the PICOS criteria and were summarized in this review, representing 1,119 cases of CLL (for PRISMA diagram and reasons for exclusion, please see Supplemental Figure 1). Thirty studies (43%) were based in Italy, 17 (25%) in Spain, six (9%) in the United States, five (7%) in France, and one each (2% each) in Australia, Canada, Belgium, mixed population Brazil/USA, the Czech Republic, Germany, Iran, Morocco, Switzerland and the United Kingdom. Additionally, one study 9 (2%) included data from the United States, Canada, UK, Ireland, Argentina, and Kuwait. Studies were published between May 2020 and January 2022. The vast majority of studies, 61/66 (92%) reported cases from the first wave of the pandemic (spring and summer 2020), 10,11 while 5/66 (~8%) reported cases from the second wave (fall and winter 2020-2021) and 1/66 (~2%) from the third wave (mid-February 2021 to June 2021). These numbers total over 100% because one study reported data from the first and second waves. 9 Timing of cases could not be determined in three studies. Seventeen studies were case reports, 49 were case series, and three were cohort studies. The majority of studies were level of evidence 4 (49/69) and 5 (17/69) with the remaining three having evidence level of 3 (3/69).
Patient Characteristics
Available data showed 591/1002 (59%) of cases were male (Table 1). Mean age was 13 years, ranging from 0 to 18 years.
Clinical Characteristics, COVID-19 Testing Results and Outcomes of Pediatric Cases of Chilblain-Like Lesions (CLL) Coinciding With the COVID-19 Pandemic.
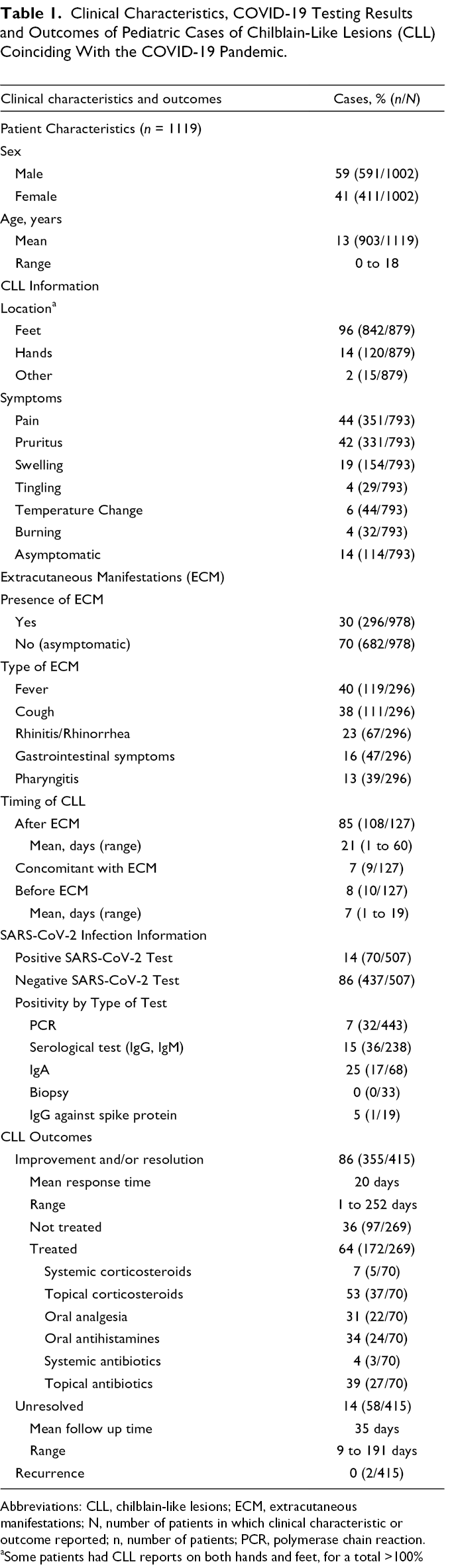
Abbreviations: CLL, chilblain-like lesions; ECM, extracutaneous manifestations; N, number of patients in which clinical characteristic or outcome reported; n, number of patients; PCR, polymerase chain reaction.
aSome patients had CLL reports on both hands and feet, for a total >100%
COVID-19 Testing Results
Overall, 70/507 (14%) of patients tested positive for COVID-19 using either: polymerase chain reaction (PCR) of nasopharyngeal swab, serology (IgM or IgG), or PCR of biopsy specimen (Table 1). PCR test was positive in 32/443 cases tested (7%). Serological antibody testing (IgG and IgM) was positive in 36/238 cases tested (15%). IgA serology testing was positive in 17/68 (25%) cases. IgG against COVID spike protein and PCR of skin biopsy were negative in 1/19 (5%) and all cases respectively.
Extracutaneous Manifestations
According to available data, 296/978 (30%) of CLL cases had extracutaneous manifestations (ECM) consistent with infection (i.e., fever, pharyngitis, or rhinorrhea) (Table 1). Available data showed that 108/127 (85%) cases experienced ECM prior to the onset of CLL (mean: 21 days prior; range 1 to 60 days); 9/127 (7%) concomitantly with CLL; and 10/127 (8%) after the onset of CLL (mean: 7 days after, range 1 to 19 days). The most commonly reported ECM included fever in 119/296 (40%), cough in 111/296 (38%), rhinitis/rhinorrhea in 67/296 (23%), gastrointestinal symptoms in 47/296 (16%) and pharyngitis in 39/296 (13%) (Table 1). More rarely ECM included malaise, fatigue, weakness, myalgia, arthralgia, chills/dysthermia, irritability, coryza, dyspnea, anosmia, dysgeusia, conjunctivitis, chest pain, and headache.
A subgroup analysis of patients shown to be COVID-19 positive by PCR, serology, or biopsy revealed that ECM were reported in 17/40 (43%) of cases (Table 2). ECM occurred before CLL in 14/17 (82%) of cases (mean: 18 days prior; range: 2 to 42 days); concomitantly in 1/17 (6%) and after in 2/17 (12%) (Table 2).
Clinical Characteristics and Outcomes of Pediatric Cases of Chilblain-Like Lesions (CLL) Coinciding With the COVID-19 Pandemic in Cases With Positive COVID-19 Testing.
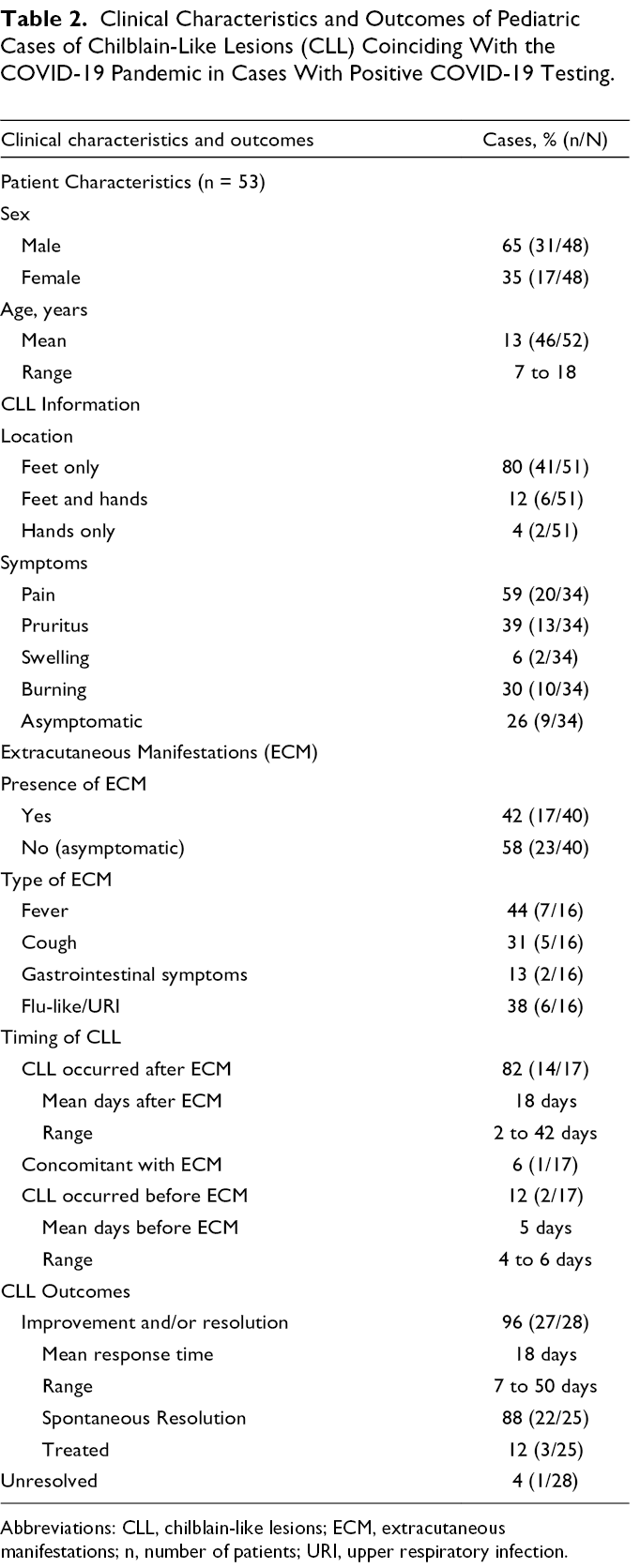
Abbreviations: CLL, chilblain-like lesions; ECM, extracutaneous manifestations; n, number of patients; URI, upper respiratory infection.
Characteristics of CLL
CLL were located on the feet in 842/879 (96%) cases and on the hands in 120/879 (14%). In 15/879 (2%) cases, CLL were located elsewhere: on the face in two cases, and the exact locations were not reported in the other cases. We did not report number of cases with CLL both on the hands and feet as 434 cases did not report feet only, hands only, and feet and hands together as separate entities. 9 CLL were generally described as erythematous to violaceous papules, macules, and/or plaques (Supplemental Table 1). Uncommon morphological features included erosions, vesicles, and bullae (Supplemental Table 2a & Supplemental Table 2b). CLL were painful in 351/793 (44%) cases, pruritic in 331/793 (42%), and associated with swelling in 154/793 (19%). CLL were asymptomatic in 114/791 (14%) cases (Table 1).
Of the 53 confirmed COVID-19 positive cases, location of CLL was reported in 51 cases. Of these, 41/51 (80%) involved the feet only, while 6/51 (12%) involved both the hands and feet, and 2/51 (4%) involved the hands only. According to available data, 9/34 (26%) had no cutaneous symptoms of CLL. CLL were painful in 20/34 (59%) cases and pruritic in 13/34 (39%) (Table 2).
Reported Treatments
Treatment status was reported in 627/1119 (56%) of cases. Overall, 252/627 (40%) of CLL cases received treatment. The most commonly reported treatments for CLL were topical steroids (202), followed by warming (95), oral analgesics (63), and oral antihistamines (24) (for other treatments used, see Supplemental Table 3). As the strength of topical steroids was inconsistently reported, all topical steroids are grouped together.
Resolution
Resolution outcomes were reported in minority of cases (415/1119, 37%) (Table 1). CLL resolved in 355/415 (86%) cases in a mean of 22 days (range 1 to 252 days). Among cases with resolved CLL, treatment data were available in 269 cases; 172/269 (64%) received some form of therapy. In resolved cases that received therapy, available data showed that topical steroids were used in 37/70 (53%), topical antibiotics in 27/70 (40%), oral antihistamines in 24/70 (35%), oral analgesia in 22/70 (32%), and systemic steroids in 5/70 (7%). Type of treatment was not reported in the other 102 cases that resolved with treatment. CLL were unresolved in 58/415 (14%). Follow up ranged from 9 to 191 days with mean 35 days. CLL recurred in2/415 cases.
Of 53 confirmed SARS-CoV-2 positive cases, 28 had resolution outcomes reported, and all but one resolved (Table 2).
Discussion
This systematic review summarizes sixty-nine studies (1,119 cases) of CLL in the pediatric population. Consistent with prior reviews in the pediatric population, the vast majority of cases included in this review were older children and adolescents. 2,3 Our review found a slightly higher percentage of males than females affected with CLL. In contrast, other pediatric reviews have found a roughly equal sex ratio, 2 while reviews in the general population have found an equal 12,13 or female predominance. 14 In line with previous reviews, 6,7,13 the vast majority of CLL cases have foot involvement (96%), with the hands as the second most common site (14%).
Pain (44%) and pruritus (42%) were the most commonly reported cutaneous symptoms associated with CLL, and 14% of patients with CLL had no associated cutaneous symptoms, consistent with prior work. 2 From the limited data available on resolution outcomes, the majority of CLL (86%) resolved, a lower resolution rate than previous reviews, possibly explained by shorter follow up duration. 2,7 The majority of patients whose CLL resolved received treatment (64%), which most commonly included topical steroids, and antihistamines and oral analgesics for symptom management. CLL resolved in a mean of 20 days (range 1 to 252 days), longer than has been reported prior (1 to 2 weeks). 6,7,12 Only two cases were reported as recurrent, likely an underestimate due to lack of follow up in many cases. Other studies estimate the CLL recurrence rate is 55% to 63% in the general population. 15 -17 Longer follow up and rigorous reporting of responses to treatment will be helpful to better elucidate the natural history and treatment outcomes of CLL.
More than two thirds of cases in this review had no extracutaneous manifestations of COVID-19 infection, which is greater than that seen in reviews of the general population (<50%). 12,13 This discrepancy could be explained by increased care-seeking by asymptomatic individuals due to increased media attention to “COVID toes” and milder COVID-19 symptoms in the pediatric population generally. It has been proposed that CLL represent a virally-induced interferonopathy, 18 -20 and individuals with CLL have a robust immune reaction characterized by a T-cell and interferon-rich microenvironment, leading to a mild course of illness. 20 In 85% of cases, ECM preceded CLL, consistent with previous observations that CLL tend to arise late in the course or in the post-acute phase of COVID-19. 7,21,22
Based on limited available testing data, the SARS-CoV-2 positivity rate was 14%, consistent with rates observed in systematic reviews and meta-analyses in the general population (15%) 12,23 but higher than seen in prior pediatric reviews. 6 The highest positivity rate was obtained with IgA testing (25%), followed by serologic (IgG and IgM) tests (15%), and PCR tests (7%). Serologic and PCR positivity results are slightly higher than seen in a meta-analysis of the general population (12% and 6% respectively). 23 Positive rates may still be underestimated for several reasons. Low proportion of IgG and IgM testing results may be because the viral load was too low to induce antibody formation and/or the patient developed an early and robust interferon type I response, muting early viral replication and not permitting the development of detectable IgM and IgG. 24 For example, in one study, there were households where all household members were seropositive except the individual with chilblains, 25 suggesting exposure to COVID-19 without antibody generation. Low rates of positive PCR tests may be due to rapid clearance of virus by the innate immune system 7 and tests occurring outside the time window of viral replication and shedding in the nasopharynx. 18,26,27 Generalizability of IgA results is limited due to the small sample size (68). The high positivity rate with IgA testing may be explained by pediatric patients with SARS-CoV-2 producing more IgA than IgG. 28 Further, with IgA being the most abundant antibody at mucosal sites, strong local protection may prevent viral spread and damage to the respiratory tract, explaining lack of symptoms and ‘immune memory’ with few patients presenting with ‘memory’ IgG. 14,29 However, IgA testing results must be interpreted with caution due to the high sensitivity of this test resulting in false positives. 23 Our results also demonstrated that PCR on skin biopsy specimens and IgG against COVID-19 spike protein reported a 0% and 5% positivity rate, respectively. Though the sample sizes were small, these results suggest that it is not useful to obtain a biopsy of a child’s CLL for the purposes of COVID-19 PCR testing.
It is possible, given that CLL are histopathologically and clinically identical to idiopathic chilblains, that some cases of CLL may be idiopathic and unrelated to SARS-CoV-2 infection. 13,20,30 CLL may be provoked by behavioral changes brought on by lockdowns and quarantines; specifically, altered peripheral circulation secondary to sedentary behaviors and inadequate coverage of hands and feet when exposed to cold interior environments. 23,31 -33 Improvements in CLL cases following thermal protection measures and the relaxation of confinement restrictions give some support for behavioral hypotheses. 23,34 -36 Additionally, caregivers and children in isolation may have had greater opportunity to observe these cutaneous changes than if normal work and educational activity were permitted. 32 Finally, the increased media attention to COVID toes may have led to increased care-seeking by patients with chilblains during the pandemic. 33
There is lack of geographic diversity in available data with 84% (58/69) of studies from Europe and 10% (7/69) from North America. More specifically, results show that almost half (43%) of studies originate from Italy and over two thirds (68%) originate from both Italy and Spain. This geographic bias is not well explained in the literature, though it may reflect an increased burden of disease, 37 stricter confinement regulations, 38 and/or a high volume of COVID-19 research publications originating in Italy generally. 39 Some speculate that this discrepancy results from a heterogeneity in reporting dermatologic findings worldwide or less reporting in warmer climates like Asia where pernio is rarely seen. 14,40
This review highlights a number of topics for continued research. First, although Fitzpatrick skin type was not reported on in this review due to limited data, there is under-representation of skin of color patients in the current literature around CLL. 41 For example, in a large case series of 318 patients with reported “COVID toes,” only 3% were Hispanic and 1% were Black. 42 In a large registry-based study, subjects were Caucasian despite ethnically diverse locations, perhaps due to difficulty in recognizing subtle erythema in darker skin and/or poorer access for skin of color populations to telemedicine during lockdown periods. 9 The diagnostic value of CLL is another area for future research; work by Visconti et al. found that the predictive value of acral lesions for positive COVID-19 swab tests had an odds ratio of 1.7(95% CI 1.3–2.28), which is greater than for fever (OR 1.48), a widely used criterion to suggest testing. 43 Another topic of interest, beyond the scope of this review, is the relationship between COVID-19 vaccination and CLL, particularly with a rising rate of vaccination. We did not extract vaccination data for this review as the majority of cases predated vaccine availability. In addition, future research may explore differences in CLL clinical characteristics between COVID-19 waves and variants; unfortunately, as the vast majority of studies included in this review were from the first wave (92%) and did not report on COVID-19 variants, we had insufficient data to draw rigorous conclusions.
There are several limitations to this review. First, without images, detailed descriptions of morphology, or reported laboratory workup to exclude other causes of CLL, we are unable to verify that all the cases included in this review truly represent CLL, potentially impacting the quality of results. Second, data on SARS-CoV-2 testing was only available in 45% of cases, in part due to limited testing of asymptomatic cases, limiting the generalizability of our results. Finally, in large national or registry-based studies, the same case may have been reported several times, leading to an overestimate in number of cases. Where possible we have made efforts to avoid double-counting cases, such as in publications by the same research groups that include a subset of cases published in their other works.
In conclusion, this review summarizes 69 papers representing 1,119 cases of CLL in the pediatric population occurring during the COVID-19 pandemic. Results found that CLL affect children with a slight male predominance and a mean age of 13. Over two-thirds of cases had no extracutaneous manifestations consistent with COVID-19 infection. Only 14% of cases were confirmed positive for SARS-CoV-2. However, this may be underreported due to a lack of testing availability, poor test validity, and test timing. CLL were described as painful and pruritic in approximately half of cases each, and were erythematous to violaceous, edematous, purpuric acral macules, plaques, and papules located mostly on the feet, and occasionally on the hands. CLL resolved in 86% of cases with available data, in a mean of 20 days, with 64% of these cases receiving treatment. The causal relationship between CLL and SARS-CoV-2 is yet to be fully explained, but the relationship may be elucidated with widespread use of molecular and serological SARS-CoV-2 testing and ongoing reporting of skin findings. To that end, we encourage physicians and health-care providers submit cases to the COVID-19 dermatology registry. 44
Supplemental Material
Figure S1 - Supplemental material for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review
Supplemental material, Figure S1, for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review by Samantha Y. Starkey, Nadia Kashetsky, Joseph M. Lam, Jan Dutz and Ilya M. Mukovozov in Journal of Cutaneous Medicine and Surgery
Supplemental Material
Table S1 - Supplemental material for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review
Supplemental material, Table S1, for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review by Samantha Y. Starkey, Nadia Kashetsky, Joseph M. Lam, Jan Dutz and Ilya M. Mukovozov in Journal of Cutaneous Medicine and Surgery
Supplemental Material
Table S2 - Supplemental material for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review
Supplemental material, Table S2, for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review by Samantha Y. Starkey, Nadia Kashetsky, Joseph M. Lam, Jan Dutz and Ilya M. Mukovozov in Journal of Cutaneous Medicine and Surgery
Supplemental Material
Table S3 - Supplemental material for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review
Supplemental material, Table S3, for Chilblain-Like Lesions (CLL) Coinciding With the SARS-CoV-2 Pandemic in Children: A Systematic Review by Samantha Y. Starkey, Nadia Kashetsky, Joseph M. Lam, Jan Dutz and Ilya M. Mukovozov in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
By reasonable request to corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
