Abstract
Background
Psoriasis and atopic dermatitis are common among older adults (≥65 years old), but clinical trials often exclude that population.
Objective
To synthesize evidence from observational studies on the safety of systemic therapies (conventional or biologic) for psoriasis and atopic dermatitis among older adults in a systematic review.
Methods
We searched MEDLINE and EMBASE (inception to October 31, 2019) and included observational studies reporting adverse events among older people treated with systemic therapy for psoriasis or atopic dermatitis. Outcomes were death, hospitalization, emergency department visits, infections, major cardiovascular events, renal toxicity, hepatotoxicity, and cytopenias. We assessed study quality using the Newcastle-Ottawa Scale.
Results
We included 22 studies on treatment for psoriasis and 2 for atopic dermatitis. Most studies were small and non-comparative and 20 of 24 were low quality. Studies comparing safety between medications or medication classes or between older and younger adults did not show apparent differences but had wide confidence intervals around relative effect estimates. Heterogeneity of study design and reporting precluded quantitative synthesis.
Conclusions
There is scant evidence on the safety of conventional systemic and biologic medications for older adults with psoriasis or atopic dermatitis; older adults and their clinicians should be aware of this evidence gap.
Introduction
Psoriasis has two incidence peaks, one in early adulthood and a second around 60 years of age. 1 While atopic dermatitis most commonly starts in childhood, a potential second peak in incidence among older adults has also been reported. 2 These epidemiologic patterns, together with an aging population, result in a high prevalence of these skin conditions in older adults (≥65 years old)—approximately 4% for psoriasis and 8% for atopic dermatitis. 2,3 Evidence for the safety of treatments for psoriasis and atopic dermatitis among older adults is essential, as they are at increased risk for adverse events from medications due to alterations in drug metabolism, increased comorbidity, polypharmacy, and frailty. 4
Systemic therapies for psoriasis and atopic dermatitis—both conventional and biologic—are associated with risk for serious adverse events, including effects on different organ systems and immunosuppression. 5 Despite the potential for increased risk among older adults, there is limited data available on the safety of systemic treatment in that population. 6,7 While randomized controlled trials are an important source of safety data, systematic reviews have found that older adults are often excluded from studies of treatments for psoriasis and atopic dermatitis. 8,9 Additionally, randomized clinical trials often have too few participants followed over too short a duration to detect rare adverse events and adverse events with long latency periods, making observational studies an important source of safety data for inflammatory skin disease. 10 We performed a systematic review of observational studies to better understand the safety of systemic therapies in older adults treated for psoriasis and atopic dermatitis.
Methods
We adhered to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and registered a protocol prior to conducting our review (PROSPERO: CRD42020151765). 11
Search Strategy and Selection Criteria
We searched MEDLINE and EMBASE from inception until October 31, 2019 (search strategy in Supplemental Table 1) and manually searched reference lists of relevant studies. We included studies based on the following eligibility criteria:
Population
We included of older adults (≥65 years old) treated with systemic medications for psoriasis or atopic dermatitis. We did not apply sex restrictions. Studies on patients with psoriatic arthritis were included if the patients also had psoriasis.
Intervention
We included studies of conventional systemic therapies (methotrexate, acitretin, cyclosporine, fumaric acid esters, apremilast) and biologics (etanercept, adalimumab, infliximab, certolizumab pegol, ustekinumab, guselkumab, tildrakizumab, risankizumab, secukinumab, ixekizumab, brodalumab) for psoriasis and conventional systemic therapies (corticosteroids, cyclosporine, methotrexate, azathioprine, mycophenolate) and biologics (dupilumab) for atopic dermatitis.
Comparator
We included studies with any comparator and non-comparative studies where incidence of adverse events with any of the interventions is provided.
Outcomes
We assessed eight adverse events: death, hospitalization, emergency department visits, infections, major cardiovascular events (stroke, myocardial infarction, hospitalization due to a cardiovascular cause), renal toxicity, hepatotoxicity, and cytopenias. In the protocol we planned to assess serious infections (defined as an infection requiring hospitalization or defined as serious by the authors); after protocol registration, we decided to include both serious infections and any infection.
Study Type
We included cohort and case-control studies with at least 10 participants aged ≥65 years old.
Screening, Abstraction, and Evidence Synthesis
We screened titles, abstracts, and full texts independently in duplicate (ET, TM, ML). Any citation identified as potentially relevant by any reviewer at the title and abstract stage was advanced to full-text review. Discrepancies at the full text stage were resolved by consensus amongst all three reviewers, with any remaining discrepancies resolved by the senior author (AMD). We subsequently abstracted all information about the study type, population, comparator, and outcomes (number, rates, and relative effect estimates) in duplicate.
At the protocol stage, we anticipated that data would be insufficient to statistically pool results, so we planned for narrative synthesis only. After data extraction and assessing the overall evidence base, we proceeded with that plan. For each outcome, we describe the results of larger studies in the text, with smaller studies described only in the accompanying tables.
We planned to present results stratified by sex, but there was insufficient data in the included studies.
Study Quality and Risk of Bias Assessment
We assessed the quality of included studies independently in duplicate (ET, TM, and ML) using the Newcastle-Ottawa scale. 12 Scores below 7 indicate low quality while scores 7 and above indicate high quality. 13 Because we did not conduct any quantitative synthesis, we did not perform any statistical assessment of risk of bias across studies (eg, publication bias).
Results
We identified 8447 records in the initial search and 3 records from the reference list of relevant studies. Following removal of duplicates, we screened 8436 titles and abstracts, 815 of which were advanced to full-text review (Figure 1). We ultimately included 24 cohort studies meeting our eligibility criteria, 22 with data on treatment of psoriasis 14 -35 and 2 with data on treatment of atopic dermatitis. 36,37 We did not identify any case-control studies that met our eligibility criteria. Some studies presented data for categories of conventional systemic (n = 4) and biologic therapy (n = 7) rather than for individual medications. We found no medication-specific results for treatment with fumaric acid esters, corticosteroids, azathioprine, certolizumab pegol, guselkumab, tildrakizumab, risankizumab, ixekizumab, brodalumab, and dupilumab. No studies presented results stratified by sex. We found most the studies (20/24, 83%) to be of low quality (Supplemental Figure 1).
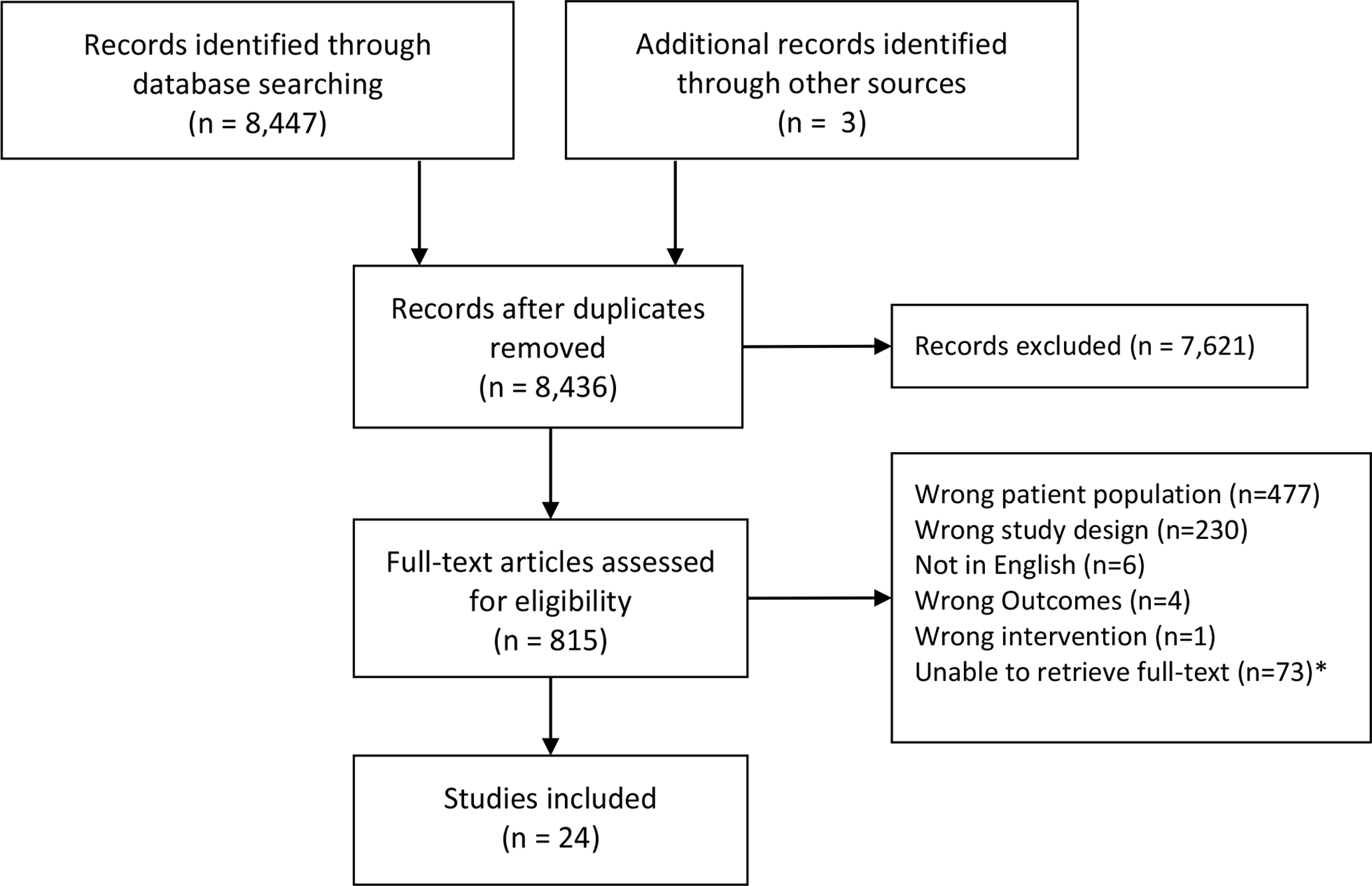
PRISMA flow diagram of included studies. * We were unable to access full-text for these studies despite utilizing both RACER and RAPID interlibrary loan systems. The 73 studies span 30 different journals and 34 had abstracts (additional details described in Supplemental Table 2).
Death
Death was studied as an adverse event in 9 studies (Table 1). Medina et al. conducted a prospective cohort study of 1793 adults treated for psoriasis. 2/204 (1.0%) older adults (≥65 years old) on conventional systemic therapy and 2/170 (1.2%) older adults on biologics had fatal adverse events. In a retrospective cohort study of 2511 adults treated with etanercept for psoriasis and followed for 3 years, 2/213 (0.9%) participants aged 65 and older died during follow-up from causes related to etanercept use. 29 A prospective study of 173 older adults (>65 years old) treated with methotrexate (n = 29), cyclosporine (n = 14), acitretin (n = 16), etanercept (n = 25), infliximab (n = 2), adalimumab (n = 24), and ustekinumab (n = 9) for psoriasis and followed for at least a year reported no deaths. 22
Summary of Studies Reporting Mortality Data Associated With Systemic Treatments in Older Adults.
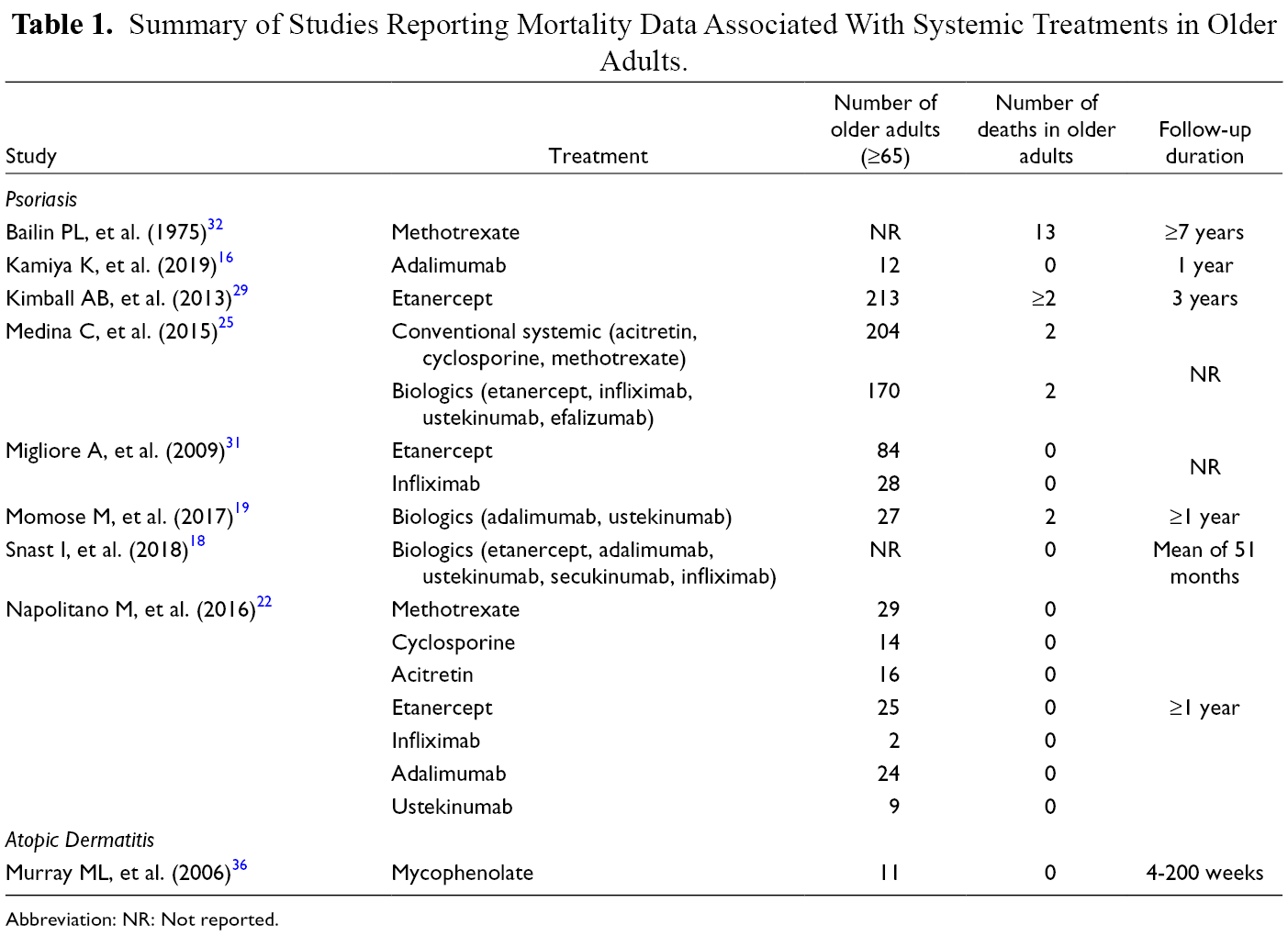
Abbreviation: NR: Not reported.
Hospitalizations
Hospitalizations were studied as an adverse event in 4 studies (Table 2). In a retrospective cohort study of 187 older adults (>65 years old) treated with methotrexate (n = 74), cyclosporine (n = 36), acitretin (n = 62), etanercept (n = 83), infliximab (n = 16), adalimumab (n = 18), and ustekinumab (n = 4) for psoriasis reported 2/74 (2.7%) hospitalizations for methotrexate, 5/83 (6.0%) hospitalizations for etanercept, and 3/16 (18.8%) hospitalizations for infliximab; with no hospitalizations reported for the remaining treatments. 26 In a retrospective cohort study of 2511 adults treated with etanercept for psoriasis followed for 3 years, hospitalization was reported in 1/213 (0.5%) older adults (≥65 years old) . 29 A retrospective study on older adults (≥65 years old) treated with etanercept (n = 84) and infliximab (n = 28) for psoriasis reported no hospitalizations. 31
Summary of Studies Reporting Hospitalization Data Associated With Systemic Treatments in Older Adults.

Abbreviation: NR: Not reported.
Emergency Department Visits
No studies reported the incidence of emergency department visits as an adverse event.
Infection
Infections were the most commonly reported outcome (18 studies; Table 3).
Summary of Studies Reporting Infection Data Associated With Systemic Therapies in Older Adults.
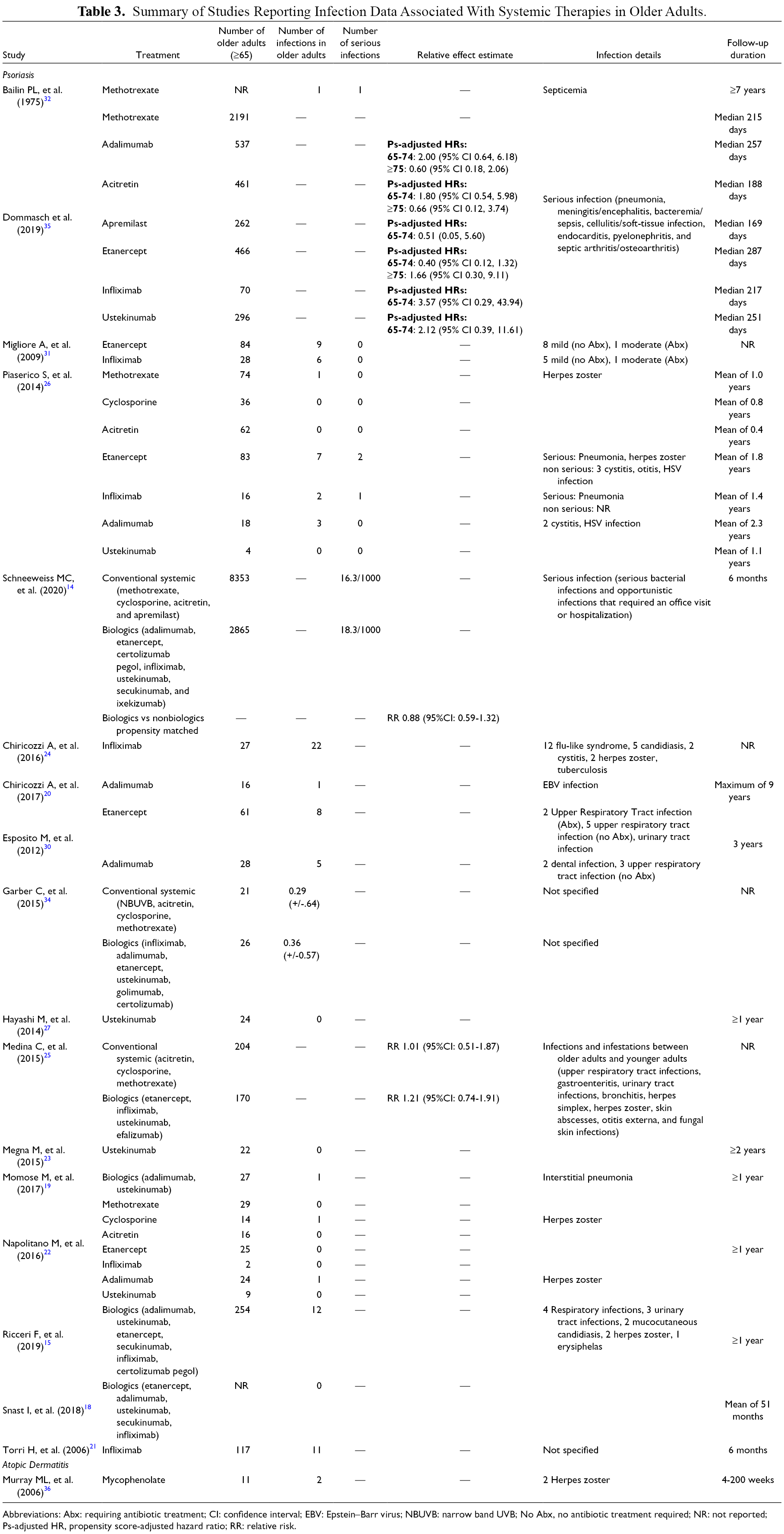
Abbreviations: Abx: requiring antibiotic treatment; CI: confidence interval; EBV: Epstein–Barr virus; NBUVB: narrow band UVB; No Abx, no antibiotic treatment required; NR: not reported; Ps-adjusted HR, propensity score-adjusted hazard ratio; RR: relative risk.
Serious infections were studied as an adverse event in 5 studies. Schneeweiss et al. conducted a retrospective study on older adults (≥65 years old) treated with conventional systemic therapy (n = 8,353) and biologics (n = 2,865) followed for 6 months and reported the rate of serious infection to be 16.3 and 18.3 per 1000 patients respectively. 14 They used propensity score matching to compare the risk of serious infections for biologics vs non-biologics and found a relative risk of 0.88 (95% CI: 0.59-1.32). Dommasch et al. compared the risk of serious infection with acitretin (n = 461), apremilast (n = 262), etanercept (n = 466), adalimumab (n = 537), infliximab (n = 70), and ustekinumab (n = 296) relative to methotrexate (n = 2191) in older adults (65, 74, and ≥75). In their propensity score-adjusted models, confidence intervals were too wide to make conclusions regarding the relative safety of medications among older adults. 35 A retrospective study by Piaserico et al. of 187 older adults (>65 years old) treated with methotrexate (n = 74), cyclosporine (n = 36), acitretin (n = 62), etanercept (n = 83), infliximab (n = 16), adalimumab (n = 18), and ustekinumab (n = 4) for psoriasis reported serious infections in 2/83 (2.4%) people treated with etanercept and 1/16 (6.3%) treated with infliximab. 26 Non-serious infections were also reported in 1/74 (1.4%) people treated with methotrexate, 5/83 (6.0%) treated with etanercept, 1/16 (6.3%) treated with infliximab, and 3/18 (16.7%) treated with adalimumab; with no infections reported for the remaining treatments. 26 Migliore et al. conducted a retrospective study on older adults (≥65 years old) treated with etanercept (n = 84) and infliximab (n = 28) for psoriasis reported no serious infections for both treatment groups. Non-serious infections were reported in 9/84 (10.7%) older adults treated with etanercept and 6/28 (21.4%) treated with infliximab.
A prospective cohort study of 1793 adults treated for psoriasis with 204 older adults (≥65 years old) on conventional systemic therapy and 170 on biologics reported the relative risk of any infections between older adults and younger adults to be 1.01 (95% CI: 0.51-1.87) for conventional systemic therapy and 1.21 (95% CI: 0.74-1.91) for biologics. 25 A retrospective study on 254 older adults (>65 years old) treated with biologics for psoriasis and followed for at least a year reported 12/254 (4.7%) cases of infection. 15 A prospective study of 173 older adults (>65 years old) treated with methotrexate (n = 29), cyclosporine (n = 14), acitretin (n = 16), etanercept (n = 25), infliximab (n = 2), adalimumab (n = 24), and ustekinumab (n = 9) for psoriasis and followed for at least a year reported infection in 1/14 (7.1%) patients treated with cyclosporine and 1/24 (4.2%) treated with adalimumab therapy; with no infections reported for the remaining treatments. 22 A multi-center retrospective study of older adults (≥65 years old) treated with etanercept (n = 61) and adalimumab (n = 28) for psoriasis and observed for 3 years reported infections in 8/61 (13.1%) people treated with etanercept and 5/28 (17.9%) treated with adalimumab. 30 Similarly, a prospective study of 768 adults treated with infliximab for psoriasis and followed for 6 months reported infection in 11/117 (9.4%) participants aged 65 and older. 21
Major Cardiovascular Events
Major cardiovascular events were studied as an adverse event in 7 studies (Table 4). A prospective cohort study by Medina et al. on 1793 adults with psoriasis (204 older adults [≥65 years old] on conventional systemic therapy and 170 older adults on biologics) reported the relative risk of cardiac disorders between older adults and younger adults treated with conventional systemic therapy to be 1.11 (95% CI: 0.36-3.12) and with biologics to be 0.50 (95% CI: 0.11-1.73). 25 A retrospective cohort study on 254 older adults (>65 years old) treated with biologics for psoriasis and followed for at least a year reported one cardiovascular event (0.4%) and one cerebrovascular event (0.4%). 15 A retrospective study on older adults (≥65 years old) treated with etanercept (n = 84) and infliximab (n = 28) for psoriasis reported 1/84 (1.2%) case of myocardial infarction with etanercept treatment and no cases with infliximab. 31
Summary of Studies Reporting Major Cardiovascular Event Data Associated With Systemic Treatments in Older Adults.
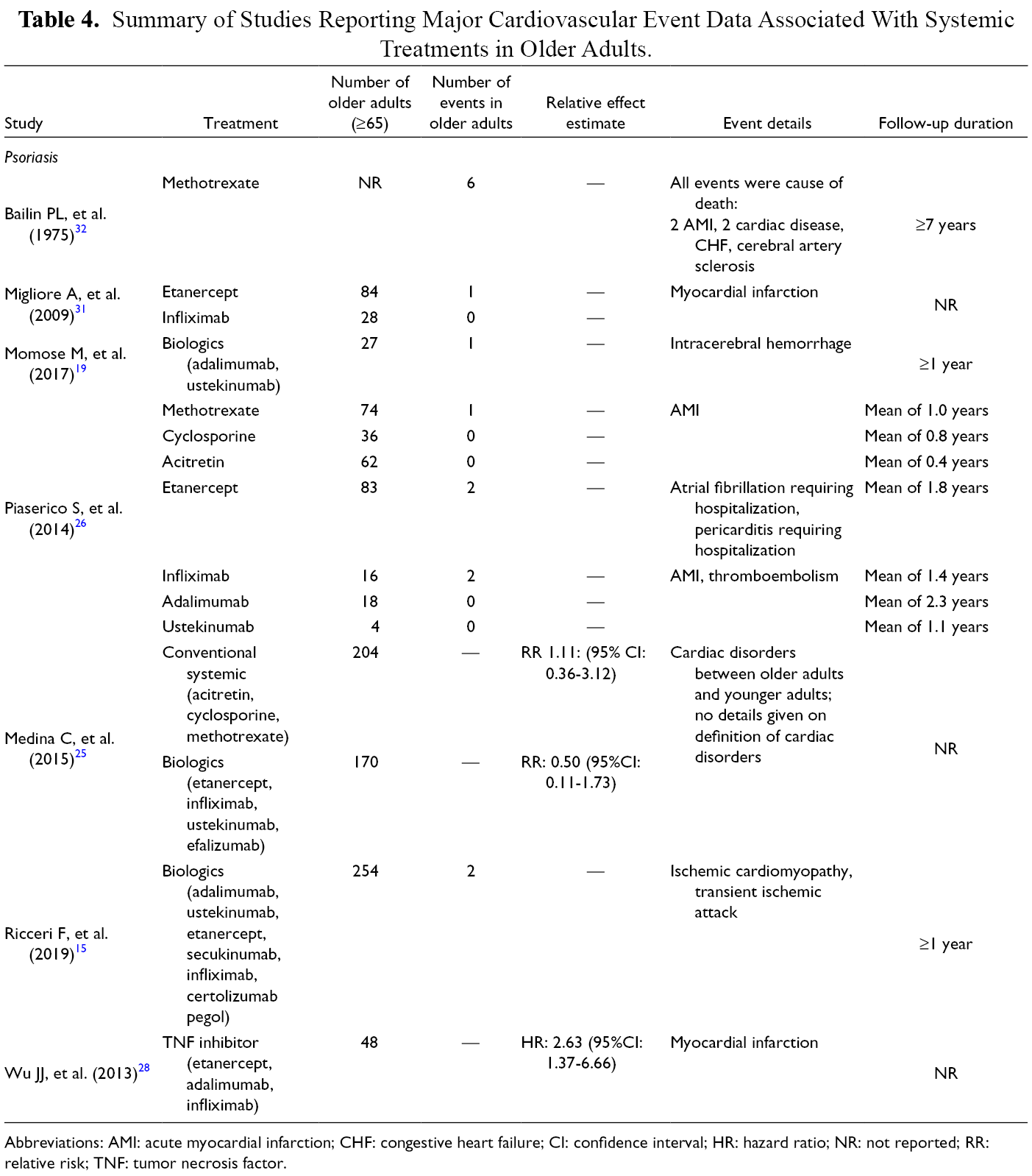
Abbreviations: AMI: acute myocardial infarction; CHF: congestive heart failure; CI: confidence interval; HR: hazard ratio; NR: not reported; RR: relative risk; TNF: tumor necrosis factor.
Renal Toxicity
Renal toxicity was studied as an adverse event in 3 studies (Supplemental Table 4). A prospective cohort study by Medina et al. on 1793 adults with psoriasis (204 older adults [≥65 years old] on conventional systemic therapy and 170 older adults on biologics) reported the relative risk of renal and urological disorders between older adults and younger adults treated with conventional systemic therapy to be 0.18 (95% CI: 0.00-1.49) and with biologics to be 1.12 (95% CI: 0.13-4.69). 25 A retrospective study by Piaserico et al. on older adults (>65 years old) treated with methotrexate (n = 74), cyclosporine (n = 36), acitretin (n = 62), etanercept (n = 83), infliximab (n = 16), adalimumab (n = 18), and ustekinumab (n = 4) for psoriasis reported renal insufficiency in 5/36 (13.9%) people treated with cyclosporine and no cases in the other treatment groups.
Hepatotoxicity
Hepatotoxicity was studied as an adverse event in 6 studies (Supplemental Table 5). A prospective cohort study by Medina et al. on 1793 adults with psoriasis (204 older adults [≥65 years old] on conventional systemic therapy and 170 older adults on biologics) reported the relative risk of hepatobiliary disorders between older adults and younger adults treated with conventional systemic therapy to be 0.70 (95% CI: 0.24-1.72) and with biologics to be 1.12 (95% CI: 0.30-3.04). 25 A prospective cohort study on 59 older adults (>65 years old) treated with conventional systemic therapy and 60 treated with biologics for psoriasis for 6 months reported elevated liver enzymes in 6/59 (10.2%) people treated with conventional agents and 3/60 (5%) treated with biologics. 22
Cytopenias
Cytopenia was studied as an adverse event in 2 studies (Supplemental Table 6). One retrospective cohort study reported anemia in 3/74 (4.1%) older adults (>65 years old) treated with methotrexate (n = 74) for psoriasis; with no other cytopenias observed in other treatment groups (cyclosporine [n = 36], acitretin [n = 62], etanercept [n = 83], infliximab [n = 16], adalimumab [n = 18], and ustekinumab [n = 4]). 26 A retrospective cohort study of 15 older adults (>70 years old) treated with methotrexate for atopic dermatitis and followed for a mean of 12 months, lymphopenia was reported in 1/15 (6.7%). 37
Discussion
In this systematic review of observational studies on the safety of conventional systemic therapies and biologics for older adults with psoriasis or atopic dermatitis, we found little evidence to guide treatment decisions for this important patient population. We identified 24 mostly small and low-quality observational studies, 22 of which were on psoriasis. For atopic dermatitis, which is more common, we found only 2 studies. We did not find any studies that fit the eligibility criteria for nine interventions and no studies reported differences by sex. Most studies were small and non-comparative. Those that did provide comparisons as relative effect estimates, either between medications or classes of medications or between older adults and a younger reference population, were limited by imprecision due sample sizes being too small resulting in very wide confidence intervals.
This lack of evidence for increased risk of specific adverse events in older adults should not be misconstrued as reassuring. A cohort study by Medina et al. pooled all adverse and serious adverse events and compared them between older adults (≥65 years old) and younger adults in a multivariable model. While there was no increased risk of adverse events overall (hazard ratio (HR) 1.09, 95% CI: 0.93-1.30), older adults had a three-fold increased risk of serious adverse events (HR 3.2, 95% CI: 2.0-5.1). This is similar to evidence from other diseases treated with similar medications. For instance, a systemic review and meta-analysis on the safety of biologics in older adults with inflammatory diseases (predominantly rheumatoid arthritis and inflammatory bowel disease) identified a higher risk of infection and malignancy in older adults (>65) when compared to younger adults. 38 In addition, regulatory information for most systemic medications included in our review urges caution (Supplemental Table 7).
We could not pool the results of studies due to too few studies included for a given comparison and due to heterogeneity of reporting. For instance, many studies did not report the total number of older adults in the study population. Of these studies, we included a few in our narrative synthesis as they reported the number of older adults experiencing specific outcomes. There were also differences in the age cut-offs used by studies to define older adults (eg., >65, ≥65, >70, and ≥75 years old).
The lack of data in older adults is in part, due to the common exclusion (either explicitly or indirectly because of factors such as comorbidities and polypharmacy) of older adults from atopic dermatitis and psoriasis RCTs. 8,9 Future RCTs should eliminate explicit upper age limits and minimize exclusion criteria that preferentially exclude older adults. In the absence of new RCT data, observational studies using routinely collected data (so-called “real-world” data) can add to our knowledge on the safety of systemic medications for atopic dermatitis and psoriasis.
Our findings have implications for a large segment of the population given that psoriasis and atopic dermatitis affect approximately 4% and 8% of older adults, respectively. 2,3 Despite the lack of evidence for their safety among older adults, conventional systemic therapies and biologics are still routinely prescribed for this population. This is warranted to provide older adults with efficacious treatments to improve their quality of life, but shared decision making requires that older patients understand their theoretically higher risk 39 -41 and that there is a dearth of specific evidence on the safety of these medications for people in their age group. In addition, there is heterogeneity among the older adults, and the risk of serious adverse events is likely lower for a healthy 65 year-old than for an 87 year-old with significant comorbidities. Future observational studies in this population and expert consensus could provide guidance for improved care in older adults with psoriasis and atopic dermatitis.
Supplemental Material
Supplementary Material 1 - Supplemental material for Safety of Systemic Medications Among Older Adults With Psoriasis and Atopic Dermatitis: A Systematic Review of Observational Studies
Supplemental material, Supplementary Material 1, for Safety of Systemic Medications Among Older Adults With Psoriasis and Atopic Dermatitis: A Systematic Review of Observational Studies by Evan Tang, Talha Maqbool, Megan Lam, Gaelen P. Adam, Mina Tadrous, Paula A. Rochon and Aaron M. Drucker in Journal of Cutaneous Medicine and Surgery
Supplemental Material
Supplementary Material 2 - Supplemental material for Safety of Systemic Medications Among Older Adults With Psoriasis and Atopic Dermatitis: A Systematic Review of Observational Studies
Supplemental material, Supplementary Material 2, for Safety of Systemic Medications Among Older Adults With Psoriasis and Atopic Dermatitis: A Systematic Review of Observational Studies by Evan Tang, Talha Maqbool, Megan Lam, Gaelen P. Adam, Mina Tadrous, Paula A. Rochon and Aaron M. Drucker in Journal of Cutaneous Medicine and Surgery
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the last three years, Dr. Drucker has been a consultant for Sanofi, RTI Health Solutions, Eczema Society of Canada and Canadian Agency for Drugs and Technology in Health. He has received honoraria from Prime Inc, CME Outfitters and Eczema Society of Canada. His institution has received educational grants from Sanofi and research grants from Sanofi and Regeneron. The remaining authors declare that there are no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
