Abstract
Background:
Patients with immune-mediated diseases on immunosuppressive therapies have more infectious episodes than healthy individuals, yet vaccination practices by physicians for this patient population remain suboptimal.
Objectives:
To evaluate the safety and efficacy of vaccines in individuals exposed to immunosuppressive therapies and provide evidence-based clinical practice recommendations.
Methods:
A literature search for vaccination safety and efficacy in patients on immunosuppressive therapies (2009-2017) was conducted. Results were assessed using the Grading of Recommendation, Assessment, Development, and Evaluation system.
Results:
Several immunosuppressive therapies attenuate vaccine response. Thus, vaccines should be administered before treatment whenever feasible. Inactivated vaccines can be administered without treatment discontinuation. Similarly, evidence suggests that the live zoster vaccine is safe and effective while on select immunosuppressive therapy, although use of the subunit vaccine is preferred. Caution regarding other live vaccines is warranted. Drug pharmacokinetics, duration of vaccine-induced viremia, and immune response kinetics should be considered to determine appropriate timing of vaccination and treatment (re)initiation. Infants exposed to immunosuppressive therapies through breastmilk can usually be immunized according to local guidelines. Intrauterine exposure to immunosuppressive agents is not a contraindication for inactivated vaccines. Live attenuated vaccines scheduled for infants and children ⩾12 months of age, including measles, mumps, rubella, and varicella, can be safely administered as sufficient time has elapsed for drug clearance.
Conclusions:
Immunosuppressive agents may attenuate vaccine responses, but protective benefit is generally maintained. While these recommendations are evidence based, they do not replace clinical judgment, and decisions regarding vaccination must carefully assess the risks, benefits, and circumstances of individual patients.
Immunosuppressive treatments dysregulate immunity and increase the risk of infections and associated morbidity and mortality.1-4 While immunization significantly mitigates these risks, vaccination rates in patients with immune-mediated diseases (IMDs) treated with immunosuppressants remain suboptimal, primarily due to the absence of physician recommendations.5-7 The reticence of physicians to vaccinate these patients may reflect the challenges of weighing the quality of protection achieved while on immunosuppressive treatment with the perceived risks of disease aggravation and vaccine-induced adverse events (AEs). This underscores the importance of improving physician awareness and the need for comprehensive guidelines for the management of this patient population. This publication aims to critically evaluate evidence regarding the safety and efficacy of vaccinating individuals exposed to immunosuppressive therapies and provide evidence-based clinical practice recommendations.
Current knowledge of the safety and efficacy of vaccination in this setting is mainly derived from nonrandomized trials and observational studies using postimmunization antibody titres to vaccine antigens as surrogate markers of protection. Although it may be argued that this measure imperfectly reflects the magnitude and quality of the immune response, it is the best-characterized and most routinely used correlate of protective immunity for commercially available vaccines.8-14 As most vaccines mediate protection through humoral responses that block infection, viremia, or bacteremia, 15 postimmunization serological titres and functional antibody characteristics are valuable indications of vaccine efficacy.
Methods
A multidisciplinary committee comprising gastroenterologists (J.K.M., A.B., B.B., A.H.S.), dermatologists (K.A.P., M.G., R.B., V.H.), rheumatologists (B.H., J.E.P., J.W., S.J.), and infectious disease specialists with expertise in vaccinology (D.K., D.C.V.) was assembled to develop guidelines on the practical management of patients considering vaccination while on immunosuppressive therapies. Synapse Medical Communications performed literature searches in accordance to the Grading of Recommendation, Assessment, Development, and Evaluation (GRADE) system across multiple databases (Embase, MEDLINE, PubMed). 16 Major search terms included vaccination, rheumatoid arthritis (RA), inflammatory bowel disease (IBD), psoriatic arthritis (PsA), psoriasis (PsO), autoimmune disease, and concepts of interest such as specific vaccines, disease-modifying antirheumatic drugs (DMARDs), glucocorticoids, and biologic agents. The literature search identified clinical trials, meta-analyses, systematic reviews, observational studies, case series, and existing guidelines published from 2009 to 2017. Reference lists were manually searched to identify relevant articles and included based on the committee’s discretion. Published studies were reviewed by the committee and assessed for content and GRADE evidence levels. 16 The quality of evidence was rated as “high” (indicating that further research is unlikely to change the confidence in the estimate of effect), “moderate” (implying that further research is likely to have an impact on the confidence in the estimate of effect), “low” (suggesting that further research is likely to have a strong impact on the confidence in the estimate of effect), or “very low” (meaning that any estimate of effect is very uncertain).
The Steering Committee (K.A.P. [chair], J.K.M., D.K., B.H.) developed the initial statements, which underwent 2 rounds of revisions according to feedback received from all authors. All 14 members voted on a web-based platform to determine the level of agreement for each statement using a 5-point scale (strongly agree, agree, neutral, disagree, strongly disagree). Statements achieving ⩾75% agreement were included in the guidelines. Of the 15 statements considered, 2 statements were rejected (see Appendix 1, available online).
The strength of a recommendation was evaluated according to GRADE and rated as “strong” when desirable consequences clearly outweighed undesirable consequences, “conditional” when desirable consequences probably outweighed undesirable consequences, or “weak” when the balance between desirable and undesirable consequences was closely balanced or uncertain. Clinical practice recommendations are listed in Table 1.
Recommendation Statements.

Abbreviation: MMR, measles, mumps, rubella.
Good Clinical Practice Statement
GRADE: Strong recommendation; moderate-level evidence
Vote: 64.3% strongly agree, 28.6% agree, 7.1% neutral
Evidence Summary
Studies in patients with type 1 diabetes, celiac disease, and IBD showed that individuals with these IMDs who are not treated with immunosuppressive agents generally have comparable serologic responses to vaccination as healthy individuals.17-20 As several DMARDs and biologic agents interfere with the immune response to vaccines (Figure 1, Figure 2, Table 2, and Table 3), it is imperative to assess the patient’s immunization history at diagnosis and administer age- and disease-appropriate vaccines according to local guidelines prior to treatment initiation to optimize efficacy. Concerns regarding the risk of disease exacerbation are unfounded as studies have shown that immunization did not generally cause clinically significant worsening of underlying IMDs (Tables 2 and 3). Indeed, a meta-analysis evaluating the impact of influenza and pneumococcal vaccination in systemic lupus erythematosus (SLE) demonstrated that immunization had no significant effect on the SLE disease activity index (SLEDAI) score. 21 While some studies reported increased joint pain following the administration of pneumococcal or influenza vaccines to patients with RA, spondyloarthritis (SpA), or PsA,22,23 these symptoms were transient and self-resolving.
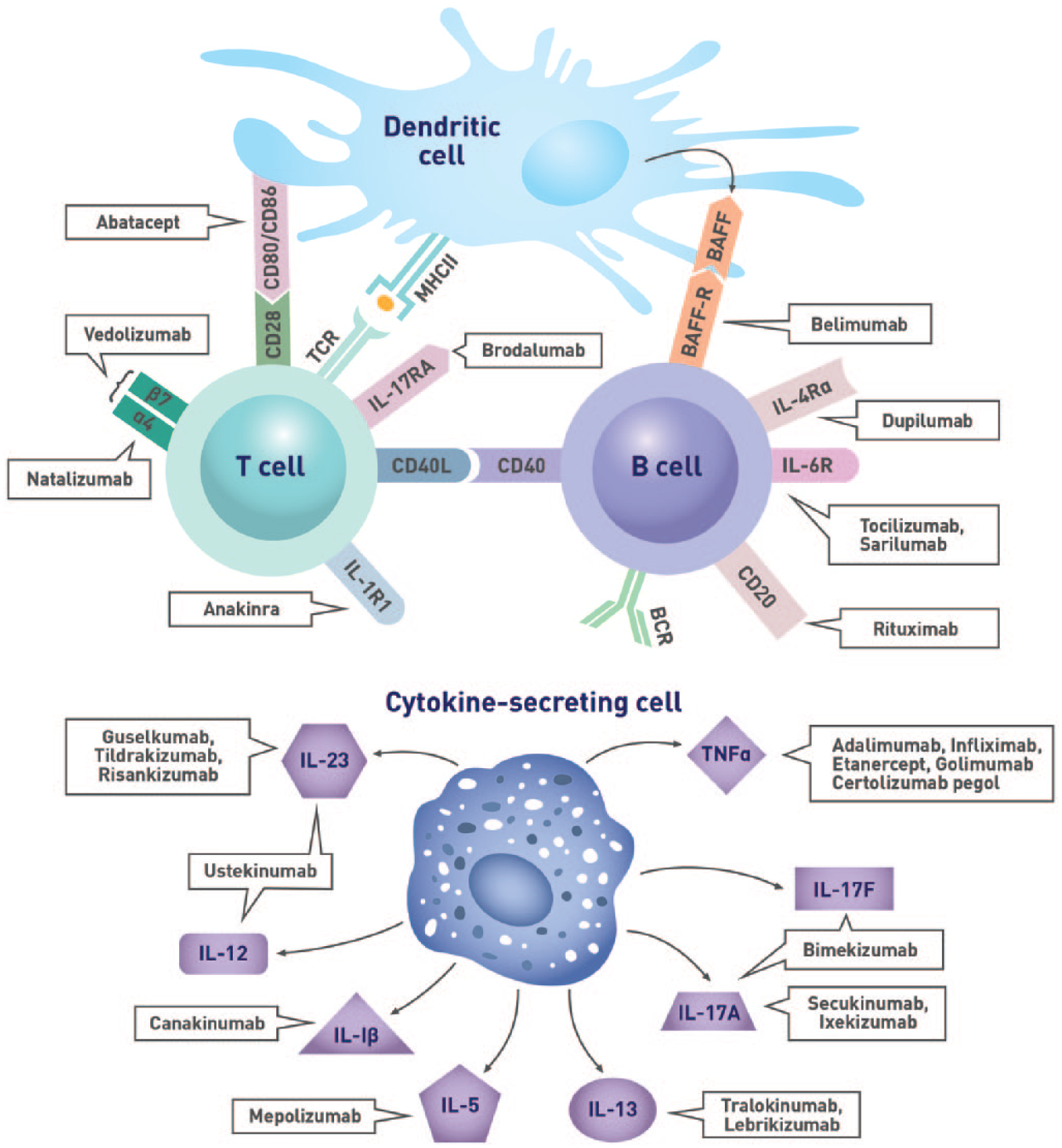
Immunological targets of biologic agents.
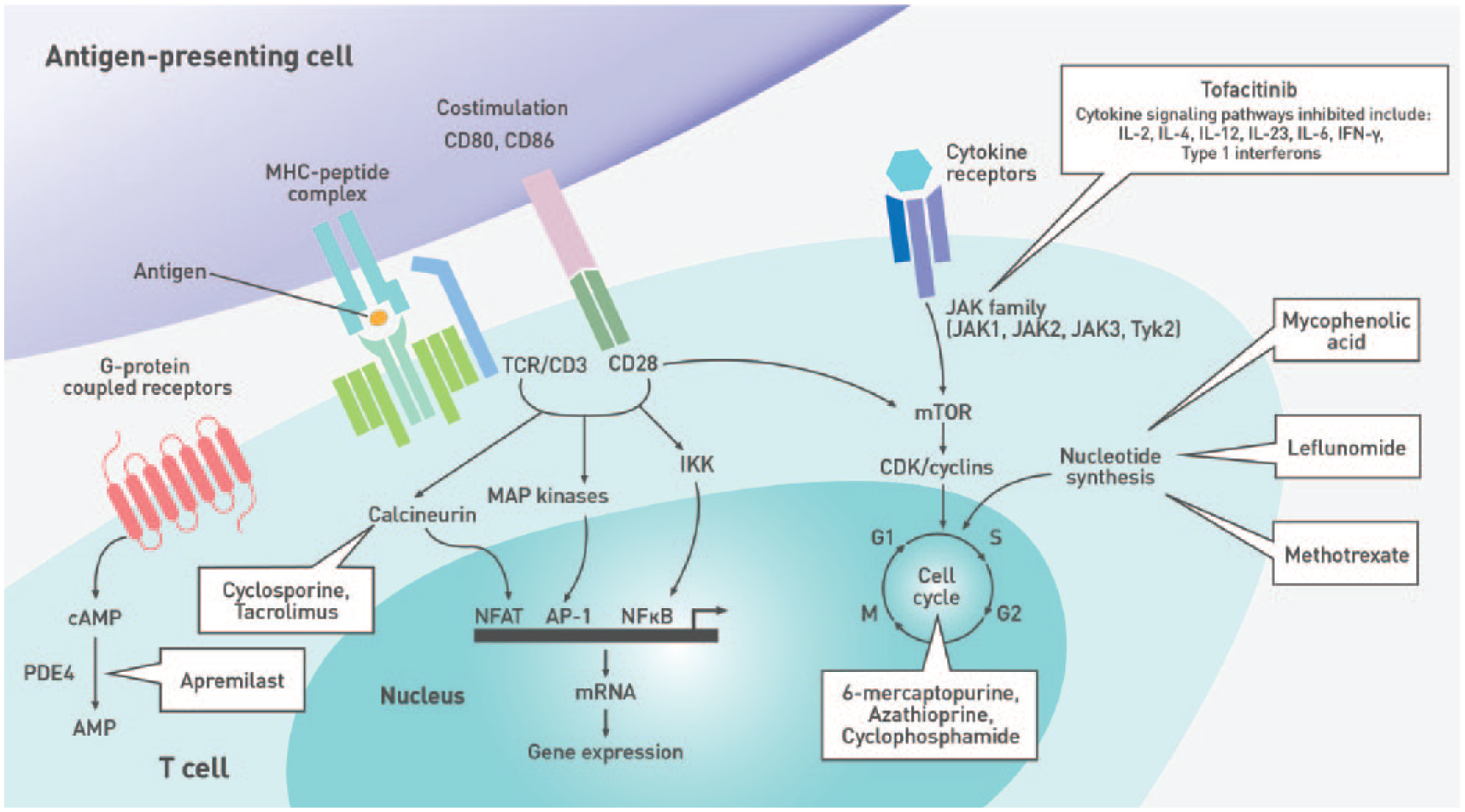
Immune pathways targeted by nonbiologic disease-modifying antirheumatic drugs.
Safety and Efficacy of Vaccination in Patients on Biologic Agents.
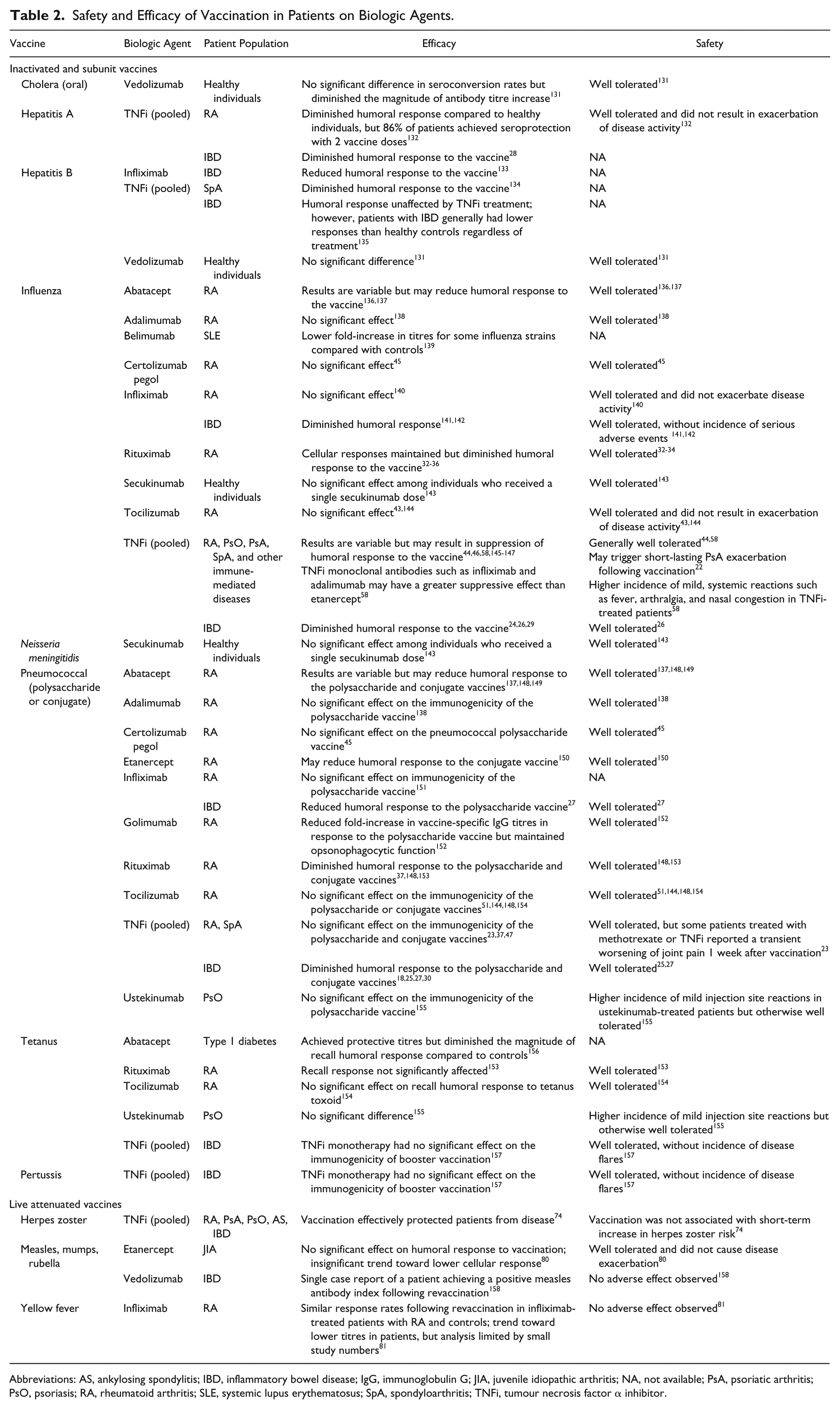
Abbreviations: AS, ankylosing spondylitis; IBD, inflammatory bowel disease; IgG, immunoglobulin G; JIA, juvenile idiopathic arthritis; NA, not available; PsA, psoriatic arthritis; PsO, psoriasis; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SpA, spondyloarthritis; TNFi, tumour necrosis factor α inhibitor.
Safety and Efficacy of Vaccination in Patients on Nonbiologic DMARDs and Corticosteroids.
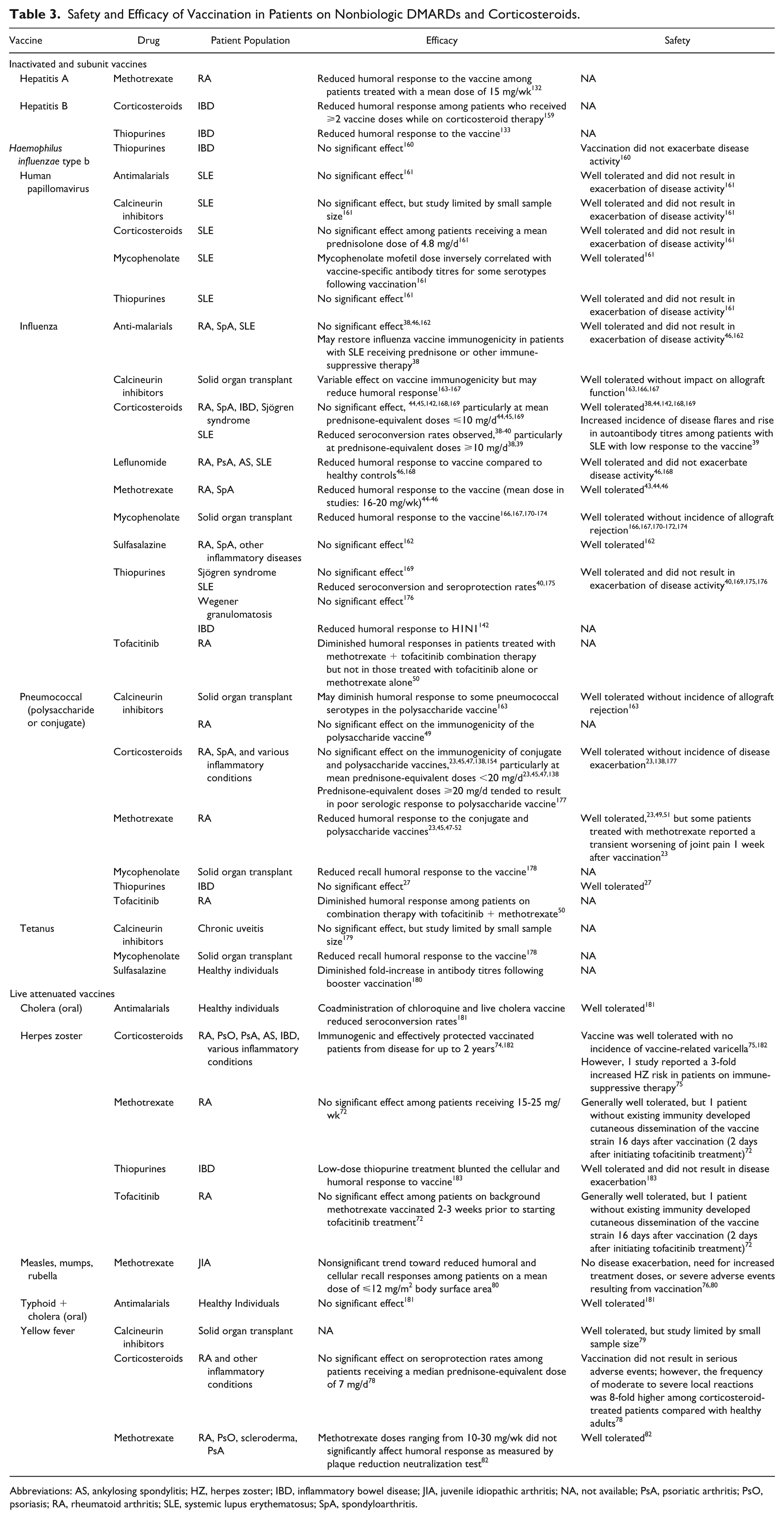
Abbreviations: AS, ankylosing spondylitis; HZ, herpes zoster; IBD, inflammatory bowel disease; JIA, juvenile idiopathic arthritis; NA, not available; PsA, psoriatic arthritis; PsO, psoriasis; RA, rheumatoid arthritis; SLE, systemic lupus erythematosus; SpA, spondyloarthritis.
Biologics
Biologics may impede optimal immune responses to certain vaccines (Table 2). For example, patients with IBD receiving tumour necrosis factor α inhibitors (TNFi) tended to have attenuated humoral responses to influenza, pneumococcal, and hepatitis A virus (HAV) vaccines.18,24-30 A meta-analysis of 9 studies (n = 728) revealed that patients with IBD on TNFi monotherapy had a significantly lower probability of achieving adequate immune responses to routine vaccinations, including those against hepatitis B virus (HBV), HAV, influenza, and pneumococcus, compared with untreated patients (odds ratio [OR], 0.32; 95% confidence interval [CI], 0.21-0.49; P < .001). 31
Rituximab (RTX)–based B-cell depletion therapy also lowered antibody titres and seroprotection rates in response to influenza vaccines compared with DMARDs and/or prednisone.32-35 In addition, RTX treatment was often associated with failure to attain protective antibody titres against all influenza strains contained within the vaccine.32,34,36 Similarly, a meta-analysis demonstrated the negative impact of RTX on pneumococcal vaccine response rates, with RTX-treated patients with RA (n = 88) having significantly poorer responses to both the 6B (OR, 0.25; 95% CI, 0.11-0.58; P = .001) and 23F (OR, 0.21; 95% CI, 0.04-1.05; P = .06) serotypes compared with controls. 37
Nonbiologic Agents
Corticosteroids and many DMARDs negatively affect vaccine immunogenicity (Table 3). For example, treatment with prednisone-equivalent doses ⩾10 mg/d diminished humoral responses to influenza vaccines in patients with SLE.38,39 A meta-analysis of 15 studies demonstrated that, compared with healthy individuals, corticosteroid treatment lowered the probability of seroconversion in patients with SLE, with relative risk ratios (RRs) of 0.66 (95% CI, 0.53-0.82), 0.49 (95% CI, 0.26-0.91), and 0.51 (95% CI, 0.24-1.09) for influenza H1N1, H3N2, and B, respectively. 40 Similarly, methotrexate (MTX) suppressed humoral responses to both influenza41-46 and pneumococcal vaccines.23,45,47-52 In 1 study, patients with RA without preexisting influenza or Streptococcus pneumoniae immunity receiving either placebo (n = 36) or placebo + MTX (n = 78; mean MTX dose of 17.2 mg/wk) were immunized with the influenza and 23-valent pneumococcal polysaccharide (PPSV23) vaccines. 45 Four weeks after vaccination, the placebo group achieved an influenza vaccine response rate of 84.6% (95% CI, 70.7%-98.5%) vs 50.9% (95% CI, 37.9%-63.9%) for the placebo + MTX group. Likewise, 89.3% (95% CI, 77.8%-100.0%) of placebo-treated patients achieved ⩾2-fold antibody titre increases to ⩾3 of 6 pneumococcal antigens tested compared with only 50.0% (95% CI, 37.3%-62.7%) of MTX-treated patients. The suppressive effect of MTX was further exemplified in studies by Park et al,41,42 which showed that patients with RA on MTX had a poorer vaccine response to the seasonal influenza vaccine than those in whom treatment was withheld for 2 weeks before and after vaccination and those who discontinued treatment for 2 to 4 weeks after vaccination.
Inactivated Vaccines
GRADE: Conditional recommendation; moderate-level evidence
Vote: 71.4% strongly agree, 28.6% agree
Evidence Summary
To optimize efficacy, the length of time required to develop robust immune responses to administered vaccines should be considered (Figures 3 and 4). Booster immunizations reactivate immune memory and induce high vaccine-specific IgG titres as quickly as 7 days after vaccination.53-55 In contrast, humoral responses to primary vaccination are characterized by the initial production of antigen-specific, low-affinity immunoglobulin M (IgM) antibodies after a lag phase lasting up to a week postimmunization.55,56 Higher affinity and avidity immunoglobulin G (IgG) antibodies eventually become detectable in the blood within 10 to 14 days 57 and may take up to 4 to 6 weeks to achieve maximum levels.53,56 It is therefore ideal to defer immunosuppressive treatment for ⩾2 weeks after vaccination to allow sufficient time to develop a robust and protective humoral response.
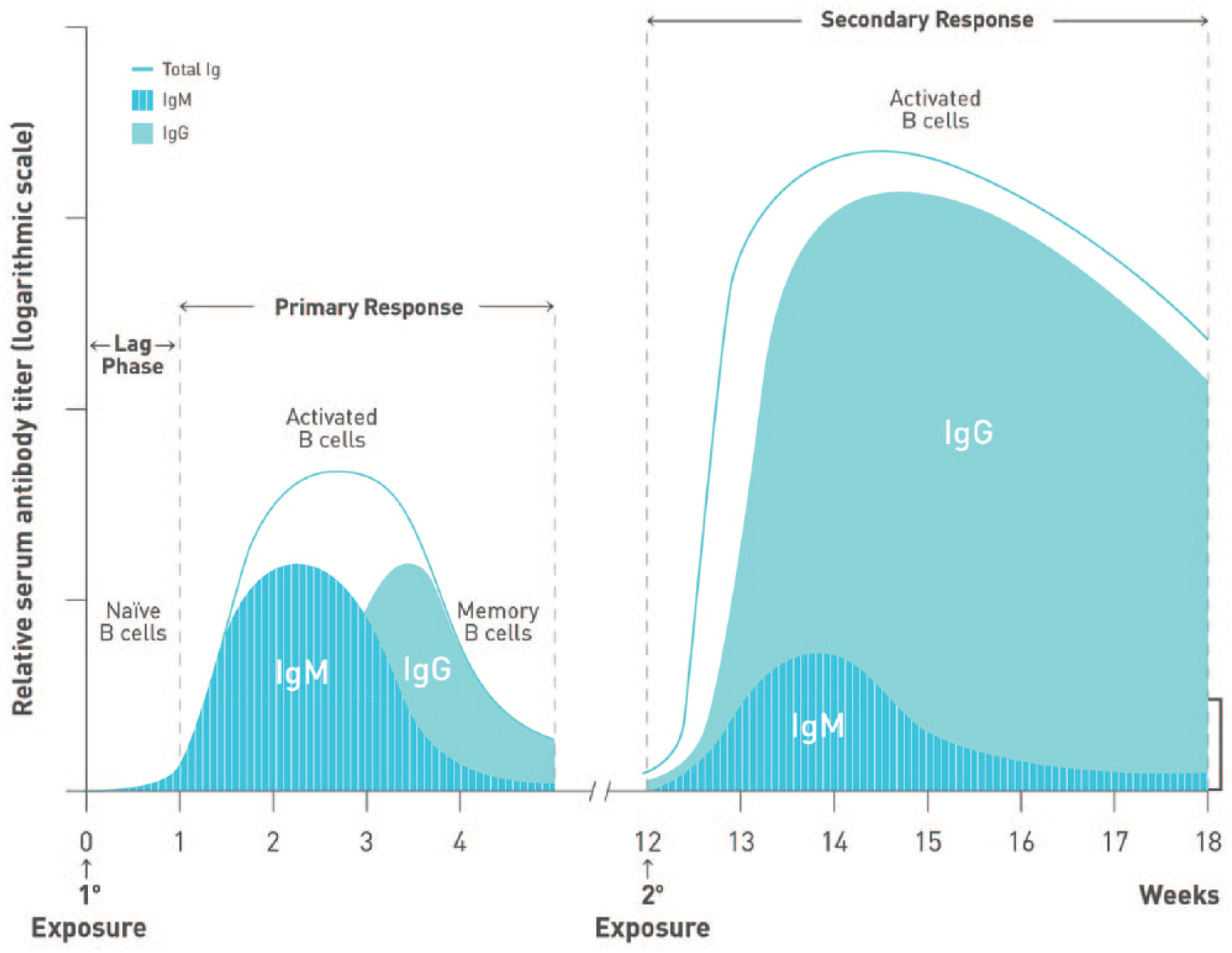
Kinetics of antibody response following immunization. The kinetics of B-cell activation and antibody (immunoglobulin M [IgM], immunoglobulin G [IgG]) production during primary and secondary responses to antigen are depicted. The primary response is characterized by a short lag phase lasting approximately 1 week, followed by the production of low-affinity IgM. IgG becomes detectable within 10 to 14 days after antigen exposure. Conversely, secondary responses reactivate memory B cells, resulting in quicker responses and higher IgG titres than those observed during a primary response.
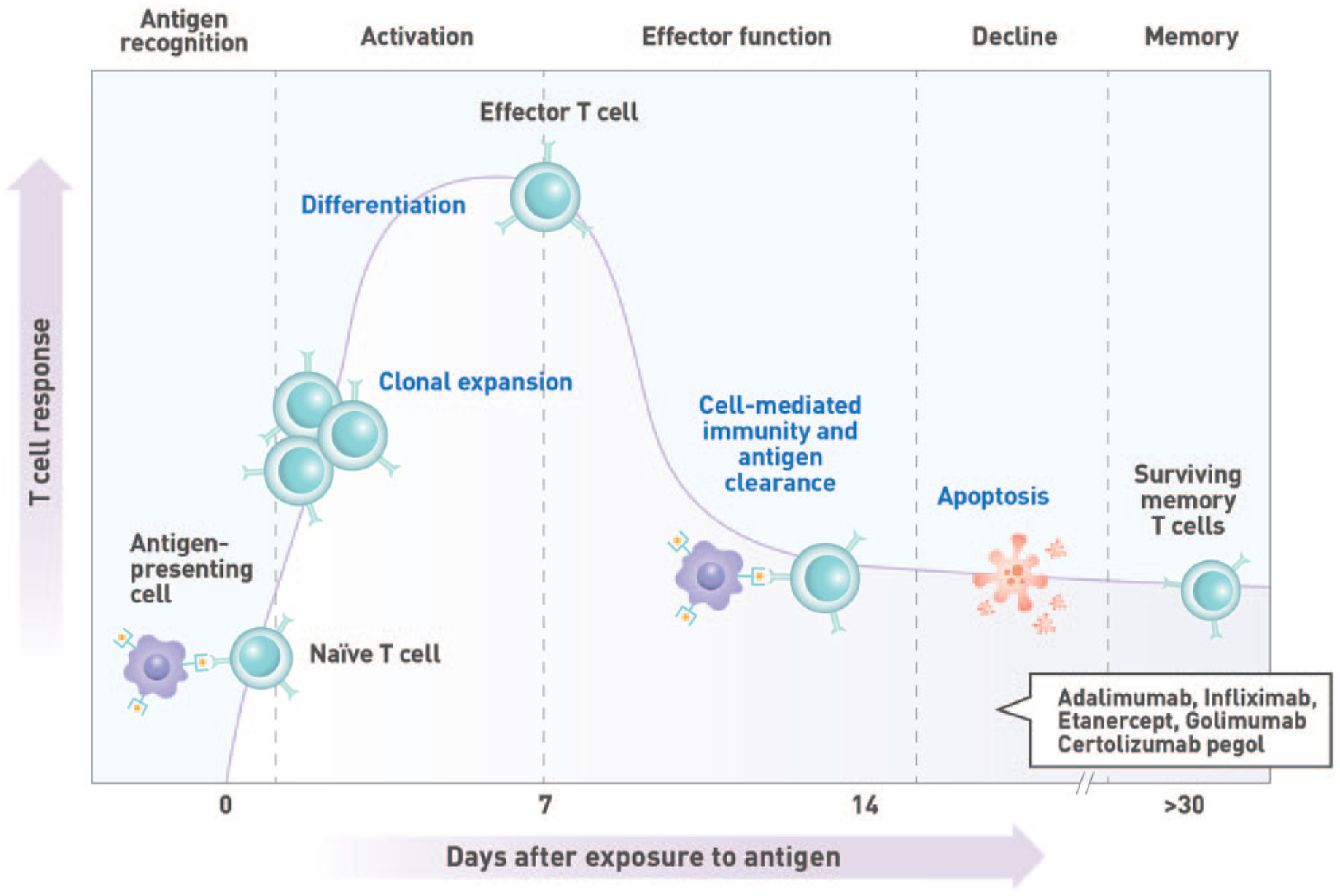
Kinetics of T-cell response. T-cell activation is initiated by the recognition of antigenic peptides displayed on the surface of antigen-presenting cells in the draining lymph nodes. This results in the clonal expansion of the activated T cell and the acquisition of effector cell function within approximately 1 week of antigen exposure. Cellular immunity plays a vital role in the clearance of intracellular pathogens and the development of robust T-cell–dependent humoral immune responses.
One study demonstrated the benefit of deferring treatment following vaccination. In this study, humoral response to the seasonal influenza vaccine was compared between MTX-treated patients with RA who stayed on treatment and those who withheld treatment for 2 weeks postvaccination. 41 Patients who discontinued treatment for 2 weeks postvaccination had a significantly higher rate of satisfactory response to all 4 influenza antigens contained within the vaccine compared with those who stayed on MTX (46% vs 22%; P < .001). Similarly, another study compared the humoral response of patients with RA who continued MTX or withheld treatment for 2 weeks before and after vaccination or 4 weeks after vaccination. 42 Compared to patients who remained on treatment, those who discontinued MTX for 2 weeks before and after vaccination (31.5% vs 51%; P = .044) or 4 weeks postvaccination (31.5% vs 46.2%; P = .121) tended to have greater response rates to all 3 influenza vaccine antigens.
GRADE: Strong recommendation; moderate-level evidence
Vote: 28.6% strongly agree, 64.3% agree, 7.1% neutral
Evidence Summary
Treatment with immunosuppressive agents should not affect the decision to administer inactivated vaccines to patients with IMDs. While vaccine antigenicity may be attenuated compared with healthy individuals, humoral response is not abolished and significant increases in antigen-specific antibody titres, often reaching protective levels, are generally achieved. In a study by Adler et al, 44 DMARD-treated patients (n = 28) with RA, SpA, vasculitis, or connective tissue disease (CTD) achieved lower fold-increases in vaccine-specific antibody titres than healthy controls (n = 40) after receiving the influenza A/H1N1 vaccine (7.7 vs 13.3). Despite this, 79% of patients still achieved seroprotective titres ⩾1:40. While the seroprotection rate in DMARD-treated patients is reduced compared with healthy individuals (98%), the majority still benefitted from vaccination despite the negative effect of immunosuppressive treatment on vaccine immunogenicity.
Several studies established the safety and tolerability of inactivated vaccines in patients with IMDs on immunosuppressive therapies (Tables 2 and 3). No serious vaccine-related AEs have been reported, although differences in the incidence of mild AEs have been observed. One such study noted higher rates of mild systemic reactions such as fever (8.3% vs 0.9%; P = .01), arthralgia (12.5% vs 4.3%; P = .03), and nasal congestion (13.3% vs 4.3%; P = .014) among patients treated with TNFi compared with healthy controls after receiving the A/H1N1 vaccine. 58
In patients receiving intermittent treatment in whom optimal vaccine immunogenicity is desired and the clinical situation allows, vaccination can be given at the nadir of immunosuppression. For immunosuppressive agents given as a bolus at intervals exceeding 4 weeks, such as infliximab (IFX) or cyclophosphamide, it is suggested that the vaccine be administered midcycle or 2 weeks prior to the next dose. Alternatively, immunosuppressive therapy may be temporarily discontinued prior to vaccination. The length of treatment discontinuation should take drug pharmacokinetics (Tables 4 and 5) and dosage into consideration.
Pharmacokinetic Half-Lives of Biologic Agents.
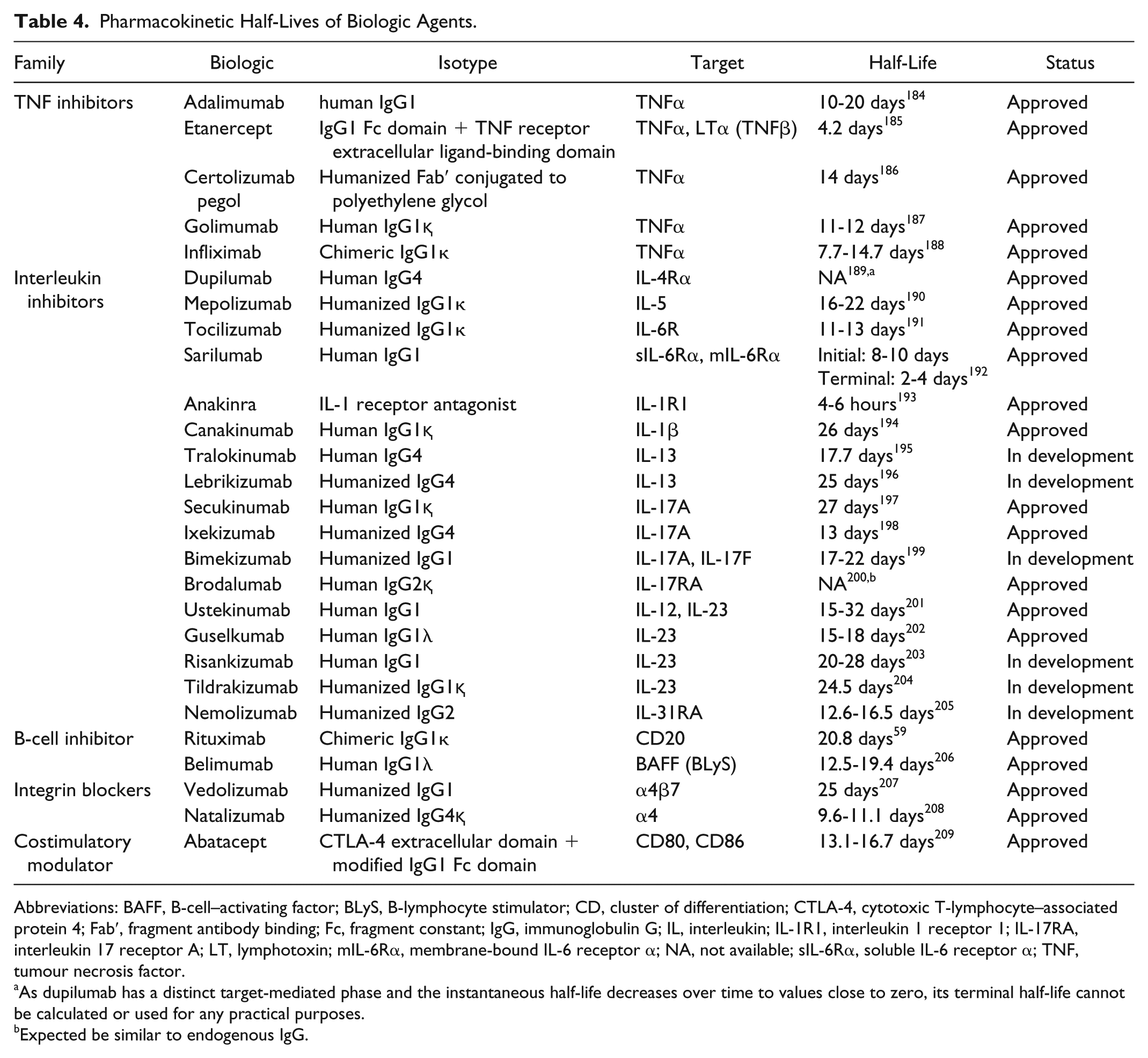
Abbreviations: BAFF, B-cell–activating factor; BLyS, B-lymphocyte stimulator; CD, cluster of differentiation; CTLA-4, cytotoxic T-lymphocyte–associated protein 4; Fab′, fragment antibody binding; Fc, fragment constant; IgG, immunoglobulin G; IL, interleukin; IL-1R1, interleukin 1 receptor 1; IL-17RA, interleukin 17 receptor A; LT, lymphotoxin; mIL-6Rα, membrane-bound IL-6 receptor α; NA, not available; sIL-6Rα, soluble IL-6 receptor α; TNF, tumour necrosis factor.
As dupilumab has a distinct target-mediated phase and the instantaneous half-life decreases over time to values close to zero, its terminal half-life cannot be calculated or used for any practical purposes.
Expected be similar to endogenous IgG.
Pharmacokinetic Half-Lives of Nonbiologic DMARDs.
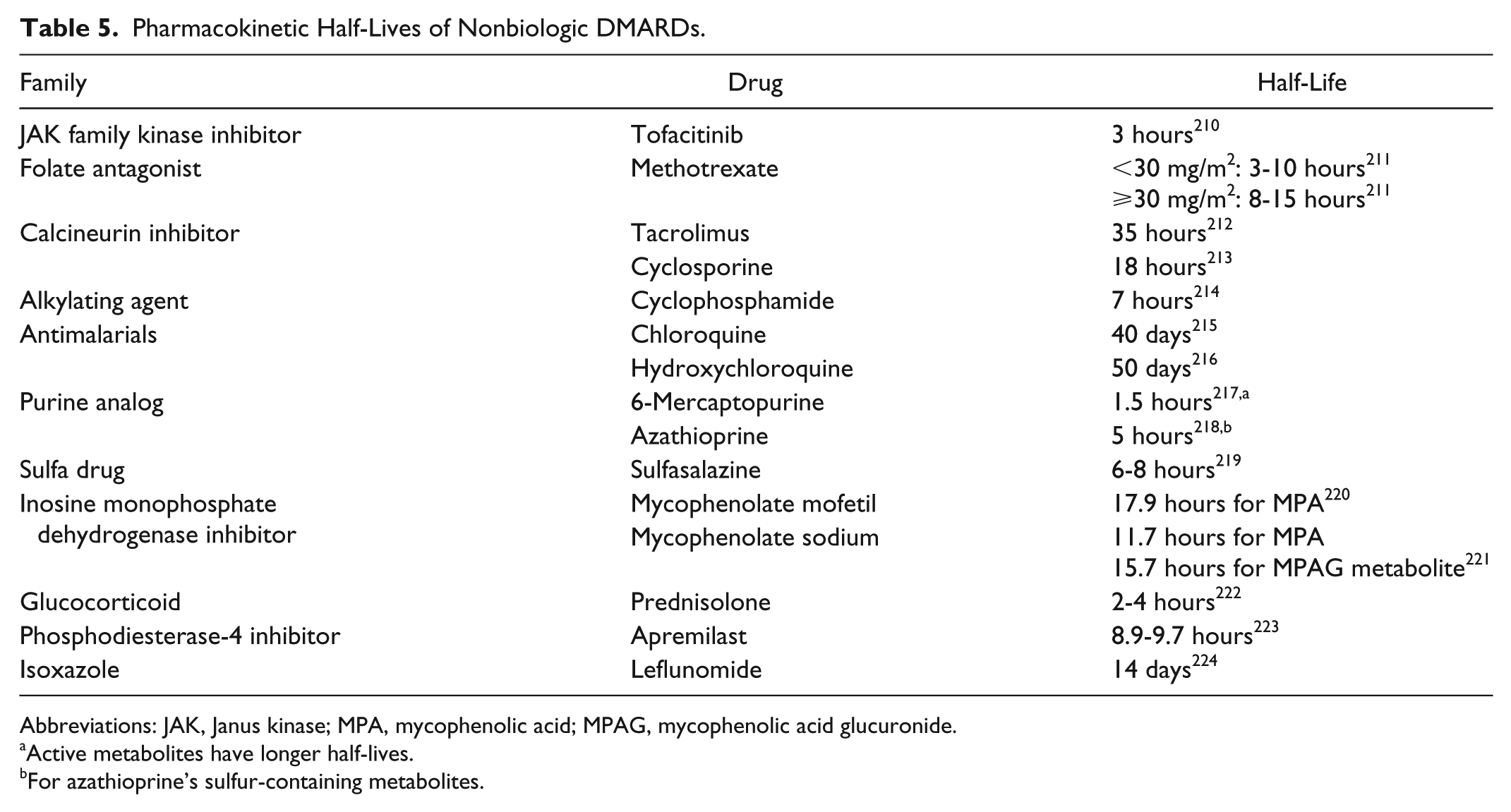
Abbreviations: JAK, Janus kinase; MPA, mycophenolic acid; MPAG, mycophenolic acid glucuronide.
Active metabolites have longer half-lives.
For azathioprine’s sulfur-containing metabolites.
Studies in RA have demonstrated that discontinuing MTX treatment until 2 weeks after immunization with the seasonal influenza vaccine increases vaccine response without increasing the risk of flares compared to those who stayed on treatment. 41 However, flares tended to be more common among patients who received a 4-week MTX treatment break around the time of vaccination. 42 Therefore, while a washout period before vaccine administration may improve the patient’s immune response to the vaccine, clinicians should use their judgement to evaluate the risks vs benefits of treatment disruption.
GRADE: Strong recommendation; low-level evidence
Vote: 14.3% strongly agree, 78.6% agree, 7.1% disagree
Evidence Summary
RTX pharmacokinetics does not directly correlate with B-cell reconstitution following treatment. Despite having a half-life of approximately 21 days, 59 1 study showed that B-cell reconstitution occurred after a mean of 8 months from the last RTX dose (n = 24; range of 5-13 months). 60 In line with this finding, patients immunized with the trivalent influenza vaccine >5 months after RTX infusion (n = 13) exhibited greater fold-increases in antibody titres for influenza H1N1 (2.1 vs 1.1; P = .02), H3N2 (1.7 vs 1.3; P = .23), and B (3.6 vs 1.6; P = .01) than patients vaccinated ⩽5 months (n = 16) after their last RTX dose. 34 Similarly, another study reported that patients with RA vaccinated 6 to 10 months after their last RTX infusion (n = 12) exhibited modestly restored IgG responses and significant increases in antibody titres for the H3N2 and H1N1 vaccine strains compared with those vaccinated 4 to 8 weeks after RTX treatment (n = 11).33,35
For most IMDs, RTX is typically administered at most every 6 months, unless clinical evaluation indicates a need for alternative dosing (in RA, RTX is administered every 6 to >12 months, depending on response duration). 61 Therefore, to allow for a degree of B-cell reconstitution, as well as provide sufficient time to mount an adaptive immune response to the vaccine, it is recommended that inactivated vaccines be given at least 5 months following the last RTX dose and approximately 1 month prior to subsequent B-cell depleting therapy. When feasible, titres assessing seroconversion should be considered.
Live Attenuated Herpes Zoster Vaccine
The live attenuated herpes zoster (HZ) vaccine is indicated for adults ⩾50 years of age to prevent HZ and postherpetic neuralgia. It contains the Oka varicella-zoster virus strain (VZV), which is present at higher viral titres (19 400 plaque-forming units [pfu]/dose) than that used for the primary prevention of chickenpox (1350 pfu/dose).62,63 Among those with previous exposure and immunologic memory to VZV, the vaccine mitigates disease risk by boosting the anti-VZV adaptive immune responses.
Typically, live vaccines are more immunogenic than their inactivated counterparts due to their ability to mimic natural infection and effectively activate the humoral and cellular arms of the immune system. 53 However, the HZ subunit vaccine (HZ/su) recently approved in Canada and the United States may actually provide a more effective alternative to the live attenuated vaccine. In 2 phase 3 clinical trials (ZOE-50, ZOE-70), the HZ/su vaccine efficacy over a mean follow-up of 3.7 years was 97.2% (95% CI, 93.7-99.0; P < .001) in individuals aged ⩾50 years and was 91.3% (95% CI, 86.8-94.5; P < .001) in those aged ⩾70 years.64,65 These rates were greater than the 51.3% reduction in HZ incidence observed in individuals ⩾60 years immunized with the live attenuated vaccine in the Shingles Prevention Study. 66 Furthermore, the subunit vaccine had an efficacy of 88.8% (95% CI, 68.7-97.1; P < .001) against postherpetic neuralgia in individuals ⩾70 years old. In the Shingles Prevention Study, efficacy against postherpetic neuralgia was 66.8% in the same age group vaccinated with the live attenuated vaccine.64,66 To date, however, there are no studies directly comparing the 2 vaccines. There are also no published efficacy data on the use of the subunit vaccine in cohorts with IMDs.
As the adjuvant, AS01B, is a component of the HZ/su vaccine, there has been some anecdotal concern regarding the risk of autoimmune/inflammatory syndrome induced by adjuvants (ASIA). However, pooled data from the clinical trials have shown no significant differences in the incidence of IMDs up to a year postvaccination between individuals receiving HZ/su (90/14 645; 0.6%) and those receiving placebo (105/14 660; 0.7%). 67 In addition, a subgroup analysis of patients with a history of IMD showed comparable incidences of possible postvaccination disease exacerbation in the HZ/su (27/983; 2.8%) and placebo (27/960; 2.8%) groups, suggesting that the vaccine is likely safe in this patient population. 67
Due to its safety and efficacy, the Centers for Disease Control and Prevention (CDC) now recommends the use of the HZ/su vaccine over the live attenuated version. 68 However, should the live vaccine be considered, the following statements aim to address concerns regarding its use in patients with IMDs on immunosuppressive therapies.
GRADE: Conditional recommendation; moderate-level evidence
Vote: 21.4% strongly agree, 78.6% agree
Evidence Summary
The live attenuated HZ vaccine should ideally be administered prior to initiation of immunosuppressive therapy. Some guidelines recommend HZ vaccination 3 to 4 weeks prior to starting therapy,69,70 likely due to evidence demonstrating the presence of the vaccine virus strain up to 4 weeks after vaccination. 71 Further prospectively designed studies are needed; however, evidence from 1 study suggests that HZ vaccination is safe and effective when administered as early as 2 weeks before treatment initiation. 72 HZ vaccine immunogenicity was assessed in patients with RA on MTX (15-25 mg/wk) who received tofacitinib (n = 55) or placebo (n = 57) 2 to 3 weeks postvaccination. 72 Six weeks after immunization, the fold-increase in VZV-specific IgG titres (2.11 vs 1.74) and number of VZV-specific T cells (1.50 vs 1.29) were comparable between the tofacitinib and placebo groups, respectively, suggesting that tofacitinib did not interfere with humoral or cellular immune responses to the vaccine. Immunization was generally safe, with only a single case of cutaneous dissemination of the vaccine strain observed in a patient without preexisting VZV immunity. 72
GRADE: Strong recommendation; moderate-level evidence
Vote: 64.3% strongly agree, 21.4% agree, 14.3% neutral
Evidence Summary
According to the CDC, treatment with low-dose MTX (⩽0.4 mg/kg/wk), azathioprine (⩽3.0 mg/kg/d), 6-mercaptopurine (⩽1.5 mg/kg/d), short-term steroid therapy lasting <2 weeks, or prednisone-equivalent doses <20 mg/d is not sufficiently immunosuppressive to preclude the use of live vaccines. 73 Conversely, treatment discontinuation lasting 1 or 3 months before live vaccine administration is recommended for patients on high-dose systemic steroid therapy lasting >2 weeks or immunomodulatory biologics, respectively. 73 Data, like those expected from the VERVE trial (NCT02538757) assessing the outcomes of HZ vaccination in TNFi users, are required to accurately assess the risks in specific patient populations. In the interim, retrospective analyses provided insights into the risks and benefits of HZ vaccination in patients with IMDs receiving immunosuppressive therapies.
A retrospective cohort study demonstrated the safety and efficacy of the HZ vaccine in patients ⩾60 years with RA, PsA, ankylosing spondylitis (AS), PsO, or IBD treated with biologics (TNFi, abatacept, or RTX), DMARDs, and/or oral glucocorticoids (n = 463 541). 74 During the 42-day window in which infection risk with the vaccine strain is greatest, none of the patients exposed to biologics (n = 633) developed varicella or HZ. Furthermore, vaccination lowered the HZ incidence over a 2-year median follow-up in biologic-treated, vaccinated patients (incidence ratio [IR], 8.5; 95% CI, 5.1-14.4) vs unvaccinated patients (IR, 16.0; 95% CI, 15.2-16.8). Among the 551 patients on TNFi, the IR of HZ in vaccinated patients was 8.5 (95% CI, 4.8-15.0) vs 15.9 (95% CI, 15.1-16.8) for those unvaccinated. Similarly, among immunized DMARD- and glucocorticoid-treated patients, the IRs were 7.0 (95% CI, 4.7-10.3) and 10.3 (95% CI, 6.7-15.8), respectively, vs 13.6 (95% CI, 13.1-14.2) and 17.2 (95% CI, 16.5-17.9) for their respective unvaccinated counterparts. 74
Notably, 1 study reported that patients with IMDs on immunosuppressive therapy at the time of HZ vaccination (n = 4826) were at greater risk of HZ 42 days postvaccination (OR, 2.99; 95% CI, 1.58-5.70) than those who discontinued immunosuppressive treatment >30 days before vaccination (n = 9728). 75 However, it is important to note that no cases of disseminated VZV from the vaccine strain were observed, and latent zoster virus reactivation was speculated to be the probable cause.
Taken together, studies on the use of the live attenuated HZ vaccine on patients with IMDs on immunosuppressive therapies suggest that it is generally safe and well tolerated. It is important to note that most patients receiving the HZ vaccine would have had previous VZV exposure and immunity, which decreases the risk of disseminated disease from the vaccine strain even in individuals on immunosuppressive treatment. However, caution may be warranted in patients without preexisting immunity to the virus. Serologic testing for VZV prior to immunization with the live attenuated vaccine should be considered.
Other Live Vaccines
GRADE: Strong recommendation; very low-level evidence
Vote: 21.4% strongly agree, 71.4% agree, 7.1% neutral
GRADE: Strong recommendation; very low-level evidence
Vote: 21.4% strongly agree, 57.1% agree, 21.4% neutral
Evidence Summary
While there is currently no evidence regarding the optimal time to initiate immunosuppressive therapies after immunization with live attenuated vaccines, it is intuitive that risks would be mitigated if treatment is initiated after vaccine-induced viremia clears, whenever the clinical situation allows. Table 6 lists data regarding the length of viremia resulting from commercially available live vaccines.
Length of Viremia Following Vaccination With Live Attenuated Vaccines.

Abbreviation: PCR, polymerase chain reaction.
GRADE: Conditional recommendation; low-level evidence
Vote: 14.3% strongly agree, 64.3% agree, 14.3% neutral, 7.1% disagree
Evidence Summary
The use of yellow fever (YF); measles, mumps, and rubella (MMR); and oral typhoid vaccines in patients on immunosuppressive therapy has not been extensively examined. While further investigation is necessary, small observational studies provided insights into the safety and efficacy of live vaccines in this patient population.76-82 Recall humoral and cellular responses to MMR in children with juvenile idiopathic arthritis (JIA) treated with etanercept + MTX (mean dose of 12 mg/m2 body surface area [BSA] once weekly [QW]) (n = 5) or low-dose MTX alone (mean dose of 9 mg/m2 BSA QW; n = 5) were not significantly different from healthy children (n = 22). 80 Moreover, the MMR vaccine was demonstrated to be safe for patients with JIA, with no reports of aggravated disease activity, increased medication use, or severe AEs following vaccination.76,80
Primary or secondary YF vaccination also induced protective serologic responses in the majority of IFX-treated patients with RA (94% [16/17]), 81 and adults with chronic inflammatory conditions receiving systemic corticosteroid therapy for a median of 10 months (100% [20/20 patients examined]; prednisone-equivalent dose: 5-20 mg/d). 78 In addition, patients with IMDs on MTX (n = 11), prednisolone (n = 1), leflunomide (n = 1), or etanercept (n = 2) achieved postvaccination neutralizing antibody titres comparable to healthy controls. 82 In all studies, the YF vaccine was administered without adverse sequelae, although patients in 1 study experienced a higher frequency of transient local reactions (RR, 8.0; 95% CI, 1.4-45.9), including erythema, tenderness, and pain compared with healthy individuals. 78
A systematic review by Croce et al 83 noted that of the 253 YF vaccine doses administered to patients with IMDs or solid organ transplants, only 1 case of vaccine-related infection was reported. The fatal case of vaccine-associated viscerotropic disease occurred in a patient with SLE and RA treated with dexamethasone and possibly MTX. 83 While the patient was given a vaccine from a lot associated with >20 times the risk of viscerotropic disease, 83 it is possible that the underlying autoimmunity and immunosuppressive treatment may have also contributed to the fatal outcome. Therefore, prudent assessment of the risks and benefits of vaccination in each individual patient is necessary to prevent potentially deleterious consequences. Whenever possible, physicians should assess the patient’s risk of YF exposure and vaccinate accordingly prior to initiation of immunosuppressive regimens.
GRADE: Conditional recommendation; low-level evidence
Vote: 28.6% strongly agree, 64.3% agree, 7.1% neutral
Evidence Summary
There is evidence to suggest that live vaccines may be safely given to patients with IMDs treated with immunosuppressive agents. However, due to factors such as advanced age, disease severity, comorbidities, and potent immunosuppressive medications, some patients may be at greater risk of AEs or disseminated infection after receiving live attenuated vaccines. For instance, the risk of developing YF vaccine-associated viscerotropic disease increases with age, with elderly individuals having a 5000-fold increased incidence (0.05%) compared to children (0.00001%). 84 In situations where patient safety is of paramount concern and the decision is made to interrupt immunosuppressive therapy prior to immunization, the duration of the treatment break prior to vaccine administration should ideally take drug pharmacokinetics into consideration (Tables 4 and 5) to minimize the interference of immunosuppressive agents on vaccine response and their potential effect on risk of disseminated infection.
Vaccination of Infants With Early Exposure to Immunosuppressive Agents
GRADE: Strong recommendation; very low-level evidence
Vote: 57.1% strongly agree, 42.9% agree
Evidence Summary
Biologics
Serological responses to routine childhood vaccinations in infants exposed to TNFi up to the third trimester (final dose at 17-39 weeks’ gestation) were described in 3 observational studies and 1 case report.85-88 Adequate and protective responses to tetanus, pneumococcal, and diphtheria vaccines were generally achieved, although some variability in Haemophilus influenzae type b (Hib) vaccine immunogenicity was observed.85,88 Furthermore, routine childhood vaccinations were well tolerated, resulting in no safety concerns or severe AEs.85-91
More recently, data from the Pregnancy in Inflammatory Bowel Disease and Neonatal Outcomes (PIANO) registry comparing vaccine responses in infants born of mothers who were exposed to biologic therapies (IFX, adalimumab, vedolizumab, certolizumab pegol, golimumab, natalizumab, or ustekinumab) vs those unexposed during gestation revealed no significant differences in seroprotection rates to the Hib (21/38 [71%] vs 4/8 [50%]; P = .41) and tetanus toxoid vaccines (33/41 [80%] vs 6/8 [75%]; P = .66) between the 2 groups. 92
Three case reports examined the efficacy of routine childhood immunizations such as tetanus, diphtheria, and HBV among infants exposed to RTX up to the third trimester (last RTX dose at 30-34 weeks’ gestation).93-95 Despite all infants exhibiting low 94 or undetectable93,95 B-cell counts at birth, protective antibody levels were generally achieved following vaccination without serious AEs.
DMARDs and Glucocorticoids
Two case-control studies examined HBV vaccine immunogenicity among infants born to mothers treated with dexamethasone, thiopurines, or cyclosporine for autoimmunity or CTD during pregnancy.96,97 Despite exposure to immunosuppressive drugs in utero, these infants achieved protective antibody titres and exhibited comparable IgG serum levels, lymphocyte counts, and other immune parameters to unexposed infants. Importantly, vaccination with inactivated vaccines was demonstrated to be safe. In a study involving 30 infants exposed to thiopurines due to maternal IBD during gestation, diphtheria, tetanus, pertussis, inactivated polio, Hib, and pneumococcal conjugate vaccines were safely administered without serious AEs or complications. 98
Exposure to higher doses of immunosuppressive agents in the solid organ transplant setting did not significantly affect the immune response to childhood vaccinations. 99 Of the 17 infants born to renal transplant recipient mothers treated with thiopurines, tacrolimus, prednisone, and/or cyclosporine during pregnancy, 82% exhibited B-cell numbers lower than the 10th percentile of normal Brazilian values (280/mm3) while 29.4% exhibited abnormalities in other lymphocyte populations at birth. Despite this, primary immunization with Hib, pneumococcal, and tetanus vaccines was safely administered and resulted in protective antibody titres that were comparable to those of infants unexposed to immunosuppressive agents in utero.
GRADE: Strong recommendation; low-level evidence
Vote: 42.9% strongly agree, 57.1% agree
Evidence Summary
Live attenuated MMR and varicella vaccines are indicated for children 12 to 15 months of age.100,101 At this time, <1% of the initial biologic agent levels should be present in the infant based on the known half-life of 30 to 52.5 days of maternally derived IgG antibodies.102-104 In the case of in utero RTX exposure, continued presence of the biologic agent in the infant was shown to delay B-cell development in some cases. However, circulating CD20+ B-cell numbers were shown to normalize 3 to 6 months after birth, possibly suggesting integrity to adequately respond to vaccines given at later time points.93,94,105 In fact, studies demonstrated that infants exposed to either TNFi or RTX in utero achieved protective antibody levels without complications upon completion of the MMR vaccine regimen.85,90,93,95
Similarly, nonbiologic immunomodulatory agents with relatively long half-lives such as leflunomide (14 days) and antimalarials (40-50 days) are expected to be cleared or present in minute concentrations that are unlikely to negatively affect the immunogenicity or safety of vaccines given ⩾12 months after birth.
GRADE: Strong recommendation; very low-level evidence
Vote: 35.7% strongly agree, 57.1% agree, 7.1% neutral
Evidence Summary
Biologic Agents
Due to the inefficient transfer of IgG antibodies across the mammary epithelium, only low levels of IgG-based biologics are typically found in breastmilk.87,106-112 Any antibodies ingested by the infant are not taken up by the intact intestinal mucosa 113 and are subject to degradation by proteolytic enzymes in the stomach and intestine.113,114 Consequently, breastfeeding while being treated with Ig-based biologics poses little to no risk of infant drug exposure, as evidenced by the continuous decline in serum levels of biologics acquired through gestational exposure in breastfed infants.87,110,115 For this reason, infants breastfed by mothers on biologic therapy should receive routine immunizations without delay according to local immunization guidelines.
Of note, subtherapeutic IFX levels were observed in an infant exposed to the drug solely through breastfeeding. 112 Five days after maternal infusion with IFX, the biologic agent was detected in the infant (1700 ng/mL), despite being only partially breastfed. No AEs were observed in the child, and the level present in the infant’s serum is below the therapeutic range and not expected to interfere with the safety or efficacy of childhood immunizations. However, the mechanism of infant absorption of the biologic in this case is unclear, and further investigation may be warranted.
Nonbiologic Agents
Hydrocortisones, antimalarials, azathioprine metabolites, sulfasalazine, MTX, cyclosporine, and tacrolimus have been detected in breastmilk at concentrations lower than maternal serum levels.116-124 At these levels, the weight-adjusted exposure of the breastfed infant is estimated to be minimal (Table 7) 125 and would likely have little effect on vaccine immunogenicity and efficacy. However, live attenuated vaccines should be administered with caution to infants breastfed by mothers on cyclophosphamide as cases of neutropenia and hematopoiesis suppression were documented in infants exposed to this agent through breastmilk.126,127
Exposure of Infants to Immunosuppressive Agents Through Breastmilk.
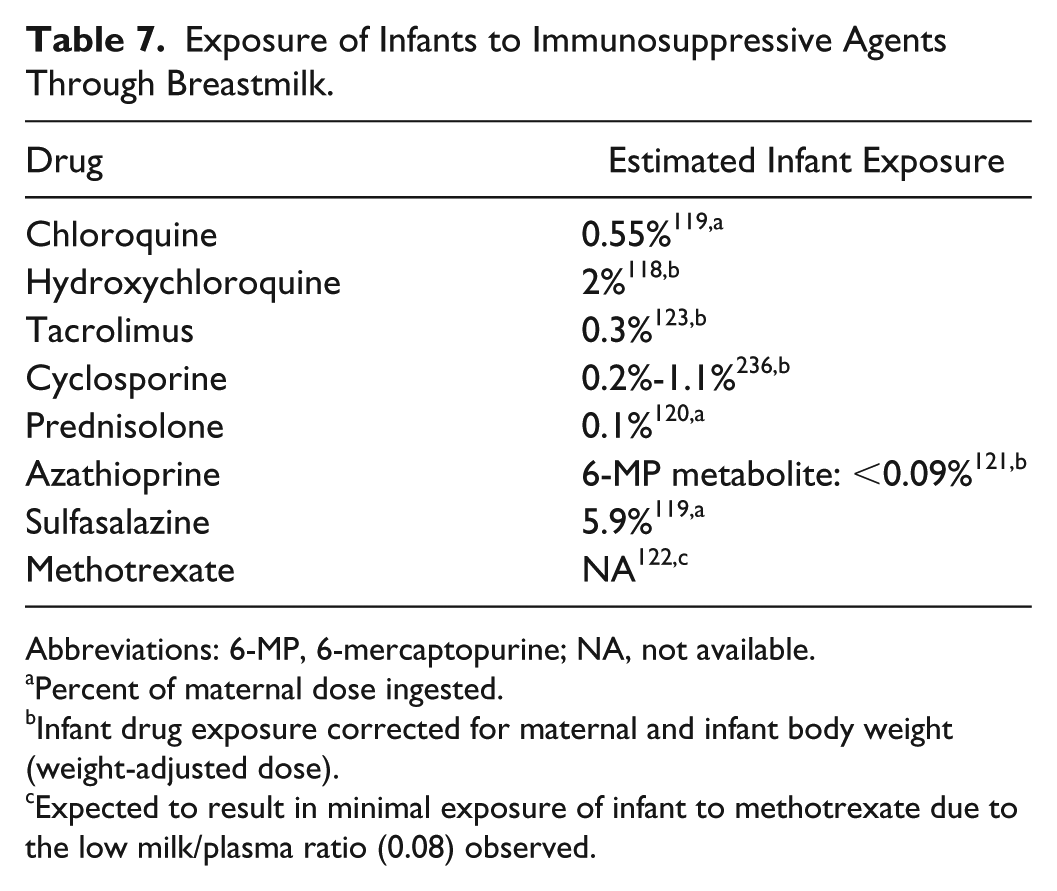
Abbreviations: 6-MP, 6-mercaptopurine; NA, not available.
Percent of maternal dose ingested.
Infant drug exposure corrected for maternal and infant body weight (weight-adjusted dose).
Expected to result in minimal exposure of infant to methotrexate due to the low milk/plasma ratio (0.08) observed.
Discussion
Consensus Level
These guidelines present recommendations on general immunization practices for patients with IMDs treated with immunosuppressive therapies, as well as infants exposed to such agents during the third trimester of pregnancy or through breastfeeding. Consensus was reached on 13 statements, with 2 statements being rejected (Appendix 1).
Comparison With Other Guidelines
In contrast to the recommendations in this publication, several guidelines, including those from the National Advisory Committee on Immunization (NACI), Advisory Committee on Immunization Practices, American College of Gastroenterology, and European Crohn’s and Colitis Organization, have suggested waiting at least 3 to 4 weeks after immunization with live vaccines prior to commencing immunosuppressive therapy.69,70,73,128 In addition, the 2014 NACI guidelines recommended that the live HZ vaccine not be given to individuals receiving high-dose corticosteroids or immune-suppressing medications, unless treatment has been discontinued for a period of 3 days to up to 1 year prior to vaccination, depending on the medication. 128
However, in individuals with prior exposure and immunity to VZV, the live attenuated HZ vaccine acts as a booster immunization, which elicits a secondary immune response with faster kinetics than primary vaccination. Studies on tofacitinib in patients with RA demonstrated that treatment initiation within 2 weeks of vaccination did not compromise the safety or efficacy of the vaccine. 72 Furthermore, data from retrospective studies suggest that vaccination even while on immunomodulatory biologics (TNFi, abatacept, or RTX), DMARDs, and glucocorticoids is safe and effective, 74 potentially eliminating the need for long wait times between vaccination and treatment initiation. Presented with this evidence, the committee deemed the live attenuated HZ vaccine to be relatively safe for individuals with preexisting immunity to varicella, even in those on certain immunosuppressive treatments. In treatment-naive patients, deferring the initiation of immunosuppressive therapy 2 to 4 weeks postvaccination should be sufficient to mount an effective adaptive response to the vaccine.
While this manuscript was being prepared for publication, the NACI released the 2018 recommendations on the use of HZ vaccines, which state that the HZ/su vaccine, not the live attenuated vaccine, may be considered for immunocompromised adults aged ⩾50 years. 129 This echoes the CDC recommendation that the subunit vaccine be considered over the live attenuated vaccine for immunocompetent adults ⩾50 years of age or those anticipating immunosuppression. 68 Aligned with the NACI and CDC guidelines, this publication preferentially recommends considering the subunit vaccine for patients with IMDs on immunosuppressive regimens until further data are available.
With regards to vaccinating patients on RTX, the Public Health Agency of Canada recommends that treatment be discontinued 6 to 12 months prior to vaccination with live or inactivated vaccines, 130 whereas a minimum washout period of 5 months preimmunization is recommended in this publication. While longer washout periods would be ideal and allow B-cell numbers to rebound following B-cell depletion therapy, extended treatment cessation is not always feasible if disease control is to be maintained. Thus, in patients requiring RTX infusion every 6 months, the committee determined that immunizing patients 5 months following the last RTX dose and at least 4 weeks prior to the subsequent dose would allow for a degree of B-cell reconstitution and sufficient time to mount an adaptive immune response to the vaccine.
Limitations
While these guidelines were developed according to the best evidence available to date, the body of evidence regarding the safety and efficacy of vaccination among individuals receiving immunosuppressive therapy for IMDs remains incomplete. Large-scale clinical trials assessing the effectiveness of vaccines in this patient population have not been performed, and data regarding the safety and efficacy of immunization for patients on recently approved drugs and biologic agents are currently lacking. Furthermore, antibody thresholds may be an imperfect surrogate for vaccine-induced protection, and the lack of more accurate measures of vaccine efficacy may hinder our complete understanding of how immunosuppressive drugs and biological agents affect the immunological response to immunization.
Conclusions
The use of immunomodulatory biologics, DMARDs, and glucocorticoids may result in attenuation but not abolishment of the immune response to vaccines. The risks and benefits of immunization should be weighed to determine patient eligibility and appropriate timing of vaccine administration relative to immunosuppressive therapy.
While specialists endorse the importance of giving age- and disease-appropriate vaccines to patients with IMDs, primary care physicians (PCPs) are often tasked with carrying out immunizations in these patients. To ensure that necessary vaccines are given and patients receive consistent care across health care providers, communication between specialists and PCPs is imperative. Should the need arise, these guidelines may serve to inform not only specialists but also family physicians and other health care providers on issues regarding the safety and efficacy of vaccines in patients with IMDs on immunosuppressive therapies.
Supplemental Material
Appendix_1_Final – Supplemental material for Vaccination Guidelines for Patients With Immune-Mediated Disorders on Immunosuppressive Therapies
Supplemental material, Appendix_1_Final for Vaccination Guidelines for Patients With Immune-Mediated Disorders on Immunosuppressive Therapies by Kim A. Papp, Boulos Haraoui, Deepali Kumar, John K. Marshall, Robert Bissonnette, Alain Bitton, Brian Bressler, Melinda Gooderham, Vincent Ho, Shahin Jamal, Janet E. Pope, A. Hillary Steinhart, Donald C. Vinh and John Wade in Journal of Cutaneous Medicine and Surgery
Footnotes
Acknowledgements
We thank Synapse Medical Communications, Oakville, Ontario, which provided medical writing assistance in the form of drafting and revising as per authors’ directions and in accordance with the standards set out by the International Committee of Medical Journal Editors. Medical writing support was funded by the Dermatology Association of Ontario.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kim Papp is an advisory board participant, steering committee member, investigator, speaker, and/or consultant for AbbVie, Akros, Allergan, Amgen, Anacor, Astellas, AstraZeneca, Baxalta, Baxter, Boehringer Ingelheim, Bristol-Myers Squibb, CanFite, Celgene, Coherus, Dermira, Dow Pharma, Eli Lilly, Forward Pharma, Galderma, Genentech, GSK, Janssen, Kyowa Hakko Kirin, LEO Pharma, MedImmune, Meiji Seika Pharma, Merck (MSD), Merck Serono, Mitsubishi Pharma, Novartis, Pfizer, Regeneron, Roche, Sanofi Genzyme, Takeda, UCB, and Valeant. Boulos Haraoui is an advisory board participant, speaker, consultant, and/or investigator for Amgen, AbbVie, Janssen, Lilly, Merck, Pfizer, Novartis, and UCB. Deepali Kumar is an advisory board participant, speaker, investigator, and/or consultant for Astellas, GSK, Janssen, Oxford Immunotec, Pfizer, Qiagen, and Sanofi. John K. Marshall is an advisory board participant, speaker, and/or consultant for AbbVie, Allergan, Celgene, Celltrion, Ferring, Hoffman-La Roche, Hospira, Janssen, Lilly, Merck, Pfizer, Procter & Gamble, Shire, and Takeda. Robert Bissonnette is an advisory board participant, investigator, speaker, and/or consultant for AbbVie, Amgen, Boehringer Ingelheim, BMS, Celgene, Eli Lilly, Galderma, GSK Stiefel, Immune, Incyte, Janssen, Kineta, Leo Pharma, Merck, Novartis, Pfizer, and Xenoport and is a shareholder of Innovaderm Research. Alain Bitton is an advisory board participant, speaker, and/or investigator for AbbVie, Janssen, Takeda, Shire, Ferring, Pfizer, Merck, and Pharmascience. Brian Bressler is an advisor and/or speaker for AbbVie, Actavis, Allergan, Amgen, Celgene, Ferring, Genentech, Janssen, Merck, Pendopharm, Pfizer, Shire, and Takeda and received research support from AbbVie, Alvine, Amgen, Atlantic Pharmaceuticals, Boehringer Ingelheim, BMS, Celgene, Genentech, GlaxoSmithKline, Janssen, Merck, Qu Biologic, Red Hill Pharma, and Takeda. Melinda Gooderham is an advisory board participant, investigator, consultant, and/or speaker for AbbVie, Actelion Pharmaceuticals, Akros Pharma, Amgen, Arcutis, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Dermira, Eli Lilly, Galderma, GSK, Janssen, LEO Pharma, MedImmune, Novartis, Pfizer, Regeneron Pharmaceuticals, Roche, Sanofi Genzyme, Sun Pharmaceutical Industries, UCB, and Valeant Pharmaceuticals. Vincent Ho is an advisory board participant, investigator, and/or speaker for Abbvie, Lilly, Novartis, Janssen, and Sanofi. Shahin Jamal is an advisory board participant for Abbvie, Amgen, BMS, Celgene, Eli Lilly, Janssen, Merck, Novartis, Pfizer, Roche, Sandoz, Sanofi Aventis, and UCB. Janet E. Pope is a consultant for Lilly, Merck, Novartis, Pfizer, Roche, Sanofi, Sandoz, and UCB and received research grants from Merck, Pfizer, Roche, UCB, and Seattle Genetics. A. Hillary Steinhart is an advisory board participant, speaker, investigator, and/or consultant for Abbvie, Amgen, Arena Pharmaceuticals, Celgene, Ferring, Genentech, Hoffmann-La Roche, Hospira, Janssen, Merck, Pfizer, Pharmascience, Red Hill Biopharma, and Takeda. Donald C. Vinh is an advisory board participant, speaker, consultant, and/or investigator for Cidara, CSL Behring Canada, Merck Canada, and Shire Canada. John Wade is an advisory board participant and consultant for Abbvie, Amgen, BMS, Celgene, Janssen, Lilly, Novartis, Roche, Sanofi, and UCB.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Development of the vaccination guidelines was funded by the Dermatology Association of Ontario.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
