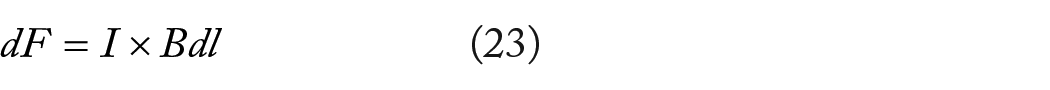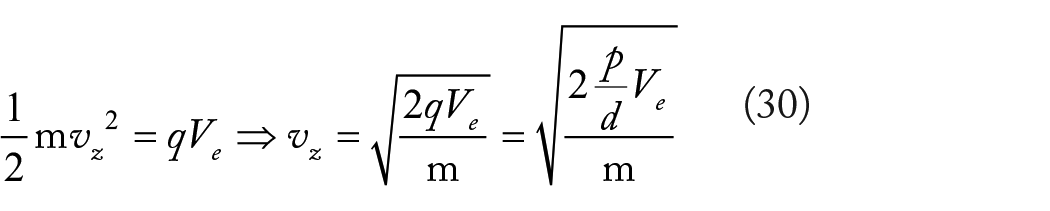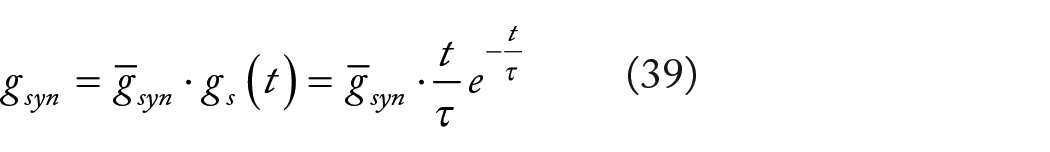Abstract
A temporal neural code reliant on the pattern of spike times rather than spike rates offers a feasible mechanism for encoding information from weak periodic external stimuli, such as static or extremely low-frequency electromagnetic fields. Our model focuses on the influence of magnetic fields on neurotransmitter dynamics near the neuron membrane. Neurotransmitter binding to specific receptor sites on membrane proteins can regulate biochemical reactions. The duration a neurotransmitter spends in the bonded state serves as a metric for the magnetic field’s capacity as a chemical regulator. By initiating a physical analysis of ligand-receptor binding, utilizing the alpha function for synaptic conductance, and employing a modified version of Bell’s law, we quantified the impact of magnetic fields on the bond half-life time and, consequently, on postsynaptic spike timing.
Keywords
Introduction
The existence of biological effects resulting from non-thermal exposure to static or extremely low frequency (ELF) magnetic fields is undeniable; however, it remains a topic of ongoing debate. Investigating the impact of electromagnetic (EM) fields on neuronal activity poses significant challenges. The biological effects triggered by electric or magnetic fields can be examined independently or in conjunction by applying an EM field. Numerous variables come into play, including the broad spectrum of frequencies, varying field strengths, signal waveform, signal duration, field application duration and the shape of the signal. Additionally, the development of experimental models presents considerable diversity, encompassing in vitro or in vivo studies involving animals or human volunteers. This diversity significantly complicates efforts to consolidate results into one or a few unified models.
Experimental data have progressively converged to identify the cell membrane as the primary target structure for the interaction of ELF electromagnetic fields with living organisms.1 -4 The cell membrane’s capacity to amplify biological signals has been elucidated through various physical models incorporating non-linear mechanisms.5 -8
Moggia et al 9 present experimental studies aimed at establishing that among the various explanatory mechanisms involved in the interaction between biological membranes and external EM fields, ligand-receptor binding stands out as the pivotal process. While the Langevin-Lorentz model remains prevalent in the study of cell receptors,10,11 there has been ongoing development and refinement of this model over the last two decades.
In recent years, numerous studies have utilized magnetic fields to modulate cell activity by targeting cell-surface receptors. An applied magnetic field has been shown to modify Notch receptors, initiating receptor signaling. 12 For E-cadherin, low magnetic forces have been observed to cluster the molecule, thus initiating F-actin assembly. 13 A similar mechanism has been employed to mechanically deflect and activate inner ear hair cells by targeting membrane glycoproteins. 14 Magnetic field displacement of the hair bundle has been found to increase intracellular calcium, indicating the activation of the hair cells. 15 Magnetic activation of endogenous receptors has also been utilized to enhance stem cell differentiation. 16 Other studies have employed magnetic fields to investigate the role of mechanical force on neurons. Tay et al 17 cultured cortical neurons and discovered that, in the presence of a magnetic field, a mechanical force in the piconewton range was exerted on the neurons, increasing intracellular calcium by targeting membrane calcium channels. Magnetic fields have also been employed to regulate apoptosis. Cancer cells, expressing high levels of the DR4 death receptor, have shown clustering upon application of magnetic fields, subsequently activating caspase-8 and caspase-3, ultimately leading to cell death. 17
The synapse, also known as the synaptic cleft, refers to the space containing the extracellular fluid situated between the pre- and postsynaptic neurons. Its diminutive size, approximately 20 nm wide, facilitates effective neurotransmitter-receptor binding. Fast responses at these synapses are mediated by ligand-gated ion channels (LGICs). 18
The duration, amplitude, and frequency of synaptic responses are determined by the kinetics of channel opening, closing, and desensitization. Binding to LGICs entails a specific and reversible reaction, as it typically involves weak interactions (such as ionic bonds, Van der Waals forces, and hydrogen bonds) between the receptor and ligand.2,19
Naundorf et al discovered that key features of spike initiation dynamics -such as the spike-time pattern- cannot be explained by the original Hodgkin-Huxley (HH) model. They analyzed the action potential dynamics in cortical neurons by in vivo, in vitro, and computational models 20 ; and found that the spike onset was 10 times greater than the one predicted by the original HH model. Unlike the original HH model, in which the channel opening and closing are independent processes, they proposed a model based on the cooperative action of sodium channels.
While the Naundorf et al model elucidates spike onset characteristics not accounted for in the original HH model, more comprehensive explanations have emerged in recent years. In 2009, Colwell et al introduced a HH-type model incorporating a stochastic term attributed to background synaptic activity. They utilized the path integral formalism to derive an analytical formula that correlates onset speed with onset potential. 21
In the Colwell HH model, the dynamics of neuron membrane potential
where the input current is implicitly included in equation (1) as an initial condition of the differential equation.
Background synaptic activity is modeled by assuming that synaptic conductance is stochastic, comprising excitatory conductance (
The voltage threshold is defined as the membrane potential value at which
In this paper, we propose a modified HH model based on Colwell’s advancements. In our model, the noise term arises not only from a stochastic component but also from a deterministic one, which encompasses the synapse-induced magnetic field. We adjust the Bell equation, expressing the bond half-life time, to account for the dynamics of neurotransmitters. The paper is structured as follows: firstly, we introduce a physical framework to examine the influence of magnetic fields on biological dipoles under physiological conditions. Secondly, we establish a connection between ligand-receptor binding and the synapse-induced magnetic field, deriving the explicit solution for synaptic conductance. Finally, we analyze the variation in postsynaptic spike timing as a function of magnetic fields and the stability analysis of the solutions.
Methods
Ionic conductance properties and parameters
The ionic conductances, excluding the leakage current
where
After selecting rate parameters
All HH’s conductance data were sourced from Marom and Abbott.
22
Given the absence of computers in 1952 for conducting numerical solutions, HH’s numerical algorithm was designed for manual calculations using the tools available at the time. The numerical method employed in this study is known as Euler’s method.
23
All simulations using Euler’s method were performed using a contemporary implementation in MATLAB,
24
equipped with metadata tags to facilitate adjustment of the
When fitting the limited six-parameter form of the model (equations (4), (6), and (7)), the rate parameters
Membrane potential properties and parameters
All membrane potential data were sourced from Marom and Abbott.
22
The voltage differential between the cell’s interior and the synapse influences both the excitatory synaptic response (EPSP) and spike generation. Under resting conditions, a neuron typically maintains a membrane potential of approximately
The membrane potential of the neuron is contingent upon the disparate concentrations of
Potassium ions (
Here,
Conversely,
The neuron expends metabolic energy to uphold the membrane voltage at a less depolarized level of
At the threshold level (
The negative, inward total current not only governs the rapid rise phase of the spike but also contributes to the synaptic current. This current initiates depolarization of the membrane potential, reaching and often surpassing
The sole distinction, from an electrical standpoint, between synaptic response and spike generation lies in the driving force behind the synaptic current. Unlike spike generation, where the driving force stems from the disparity between membrane voltage and the ion-specific equilibrium potential, excitatory receptors (primarily AMPA receptors) constitute nonspecific ionic channels. Consequently, the net current is determined by the synaptic conductance (dependent on neurotransmitter-receptor binding) and a nonspecific synaptic equilibrium potential, typically around
Physical Derivation of the Model
Electric dipole moment of neurotransmitters under physiological conditions
Finite electric dipoles typically consist of two-point charges of equal magnitude: a negative charge (
The point dipole moment is obtained in the limit as
here,
Sivasankar et al
27
determined the value of
Dynamics of electric dipoles in magnetic fields
A current element
The current
When a charged particle encounters a static field at right angles, it undergoes a force known as the Lorentz force, which alters the particle’s motion into uniform circular motion around the magnetic field lines. According to Newton’s second law, the radial force acting on the particle is

(a) Trajectories of charged particles in a magnetic field and (b) a charged particle with a velocity component parallel to a magnetic field and another perpendicular to it, moves in a helical path around the field lines.
The energy principle can be applied to derive force distributions. To develop a simple model for force density distribution, we require the expression for the force on an electric dipole in polarizable media, which can be derived from the Lorentz force law. The force on a stationary electric charge is governed by the Lorentz law with a frequency of zero. Considering two charges
where
Unless the electric field at the location
The first and last terms cancel each other out. In a more concise notation, this expression is thus represented as,
where we have identified the dipole moment
Time-scale hierarchy in complex systems
In dynamics involving far-from-equilibrium irreversible processes, the relax integrability function tends to converge toward stable asymptotic configurations. The relaxation time is contingent upon the dissipative process parameters operating at each level. Neuronal time scales exhibit dynamism, with a succession of relaxation times discernible, each succeeding level characterized by an increase in magnitude33,34:
Microsecond scale, associated with the rearrangement of ionic concentrations and reactive phenomena in tissue electrolytes.
Hundreds of microseconds scale, associated with the activation of sodium channels and subsequently with calcium channels involved in membrane excitation.
Millisecond scale, associated with the membrane time constant, pulse and stimulation duration, and electrochemical double layer polarization at the membrane-electrolyte interface.
Hundreds of milliseconds scale, associated with the mechanisms of activation and inactivation of membrane channels related to recovery phenomena.
Minute scale, involved in membrane adaptation phenomena (slow excitability variations dependent on ionic accumulation in regions where transport processes are disturbed).
The differences in the numerical order of characteristic relaxation times among hierarchical scales in dissipative systems often enable simplification of their description by applying 2 connecting scale principles. First, on a particular time scale, the variables of the higher time scale can be treated as constant parameters (as their relaxation times are significantly greater). Second, the variables of the lower time scale can be disregarded (as their relaxation times are much smaller). 35
Suppose we have an electric dipole initially at rest within a static vertical magnetic field. The movement of the dipole depends on the EM field configuration, specifically on the electric potential
While the magnetic field may not be conservative, the EM field as a whole is conservative. Therefore, applying the principle of conservation of energy to a dipole positioned in a region of potential
Taking
Therefore, the electrical potential associated with extremely weak static magnetic fields induces temporal variations in neurotransmitter dynamics at a much lower time scale, from a hierarchical perspective, compared to what is typical in classical studies of chemical synapses. This suggests that the very weak synapse-induced magnetic fields may serve as fine control mechanisms in shaping the overall process of postsynaptic spike-time patterns.
Dynamics of hybrid multiscale systems: Combining deterministic and stochastic elements
Deterministic and stochastic hybrid systems no longer evolve with a deterministic trajectory in the parameter space, but rather with a certain probability distribution defined within that same space. A general formulation for these systems is based on nonlinear differential equations incorporating both a stochastic noise term, which models fluctuations, and a deterministic term.39,40 The statistical properties of the noise completely define the system through its stochastic differential equation (SDE). This additive noise, resulting from fluctuation timescales and system response time, is typically assumed to have zero correlation time (white noise). Additionally, there may be an additional source of fluctuations arising from environmental influences, known as external noise.
The most common assumption regarding the stochastic term present in the SDE is that it follows a normal distribution, meaning it is Gaussian. In many systems, noise represents the influence of random factors whose cumulative effect, according to the central limit theorem, converges to a Gaussian distribution. White noise intensity is constant in the frequency-domain. Its correlation function is defined as follows, 41
Here, the parameter
In a region of local instabilities in phase space induced by external stochastic influences, such as thermal motions or fluctuations in boundary conditions, a particle can be significantly affected by a deterministic force field. The existence of deterministic chaos is not a prerequisite for this phenomenon. If a deterministic force field is sufficiently strong and persistent, deviations from a stochastic trajectory can be profound, leading to predictable, even deterministic trajectories that are asymptotically stable. The outcome is noisy behavior that would not be observable without the influence of deterministic dynamics but cannot be sustained by deterministic dynamics alone.
The probability distribution
The parameters
Extended kinetics of the ligand-receptor bell model incorporating synaptic magnetic fields
The temporal evolution of the reversible chemical reaction between 2 reactants A and
Here,
In this equation,
In our study, we introduce a theoretical model rooted in mechanics force fields theory to simulate the impact of magnetic forces on molecular interactions, particularly chemical bonds. When an external force is exerted, it alters the activation energy of the chemical reaction. The Bell model establishes a relationship between the magnitude of the force and the dissociation constant of the reaction, 44
In the equation,
where
Here,
Neuron models that integrate alpha function conductance-based synapses
Synaptic noise acts as the primary origin of membrane potential fluctuations in neurons and plays a significant role in shaping their integrative behaviors, including synaptic conductance. Destexche and Rudolph-Lilith 46 conducted a review of experimental synaptic noise measurements and formulated a model grounded in stochastic noise processes.
A common approach to estimating the alteration in conductance within the postsynaptic neuron is through the alpha function model. In numerous synapses, the progression of synaptic conductance undergoes a finite-duration phase. The alpha function delineates this phase of conductance progression as not infinitely rapid, implying it possesses a finite rise time,
It is worth noting the similarity between equations (32) and (38), which validates the application of stochastic-deterministic noise theory to conductance-based synapses. Consequently, we can express the kinetics of synaptic conductance as follows,
Hence, we eventually deduce a membrane potential dynamics equation rooted in the Colwell HH model, integrating the synapse-induced magnetic field,
Here,
Figure 2 depicts the temporal progression of synaptic conductance (
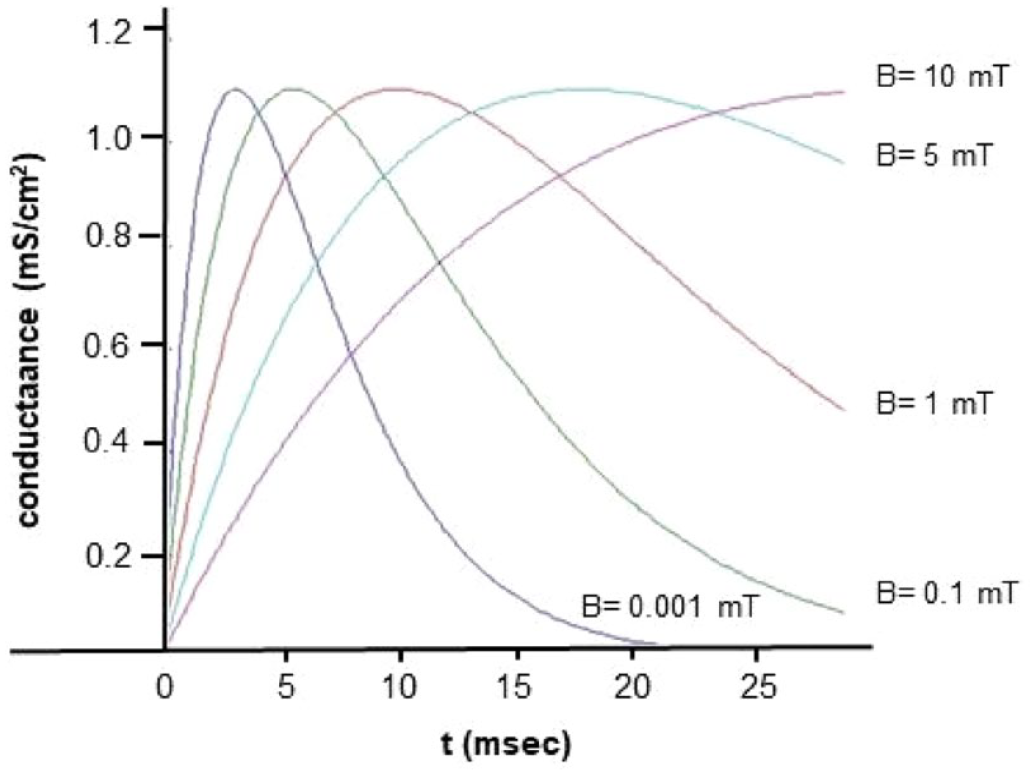
Synaptic conductance analytic solutions using the alpha-function model at different synapse magnetic field values (
Results and Discussion
Cell membrane proteins can be arranged as a kind of electric amplifiers, such as is used in electronics. Every periodic current can be analyzed using Fourier series as a sum of a constant component (DC) and alternating components. These are entirely transformed by the transformer, all in the same ratio, while the DC component induces no voltage across the secondary. Consequently, the transformed voltage is, except for the transformation factor, identical to the initial voltage, minus its mean value—(Figure 3a)—(the transformed voltage has a zero-mean value).

In (a), we can observe the decomposition of a rectified alternating current (primary current) into a direct current (untransformed) and a rectified alternating current with zero average offset. In (b), the phase shift of the intensity of a sinusoidal alternating current due to an external disturbance is presented, in this case, a magnetic field.
The phase shift
From the perspective of alternating current circuit theory, it’s feasible to manipulate the phase of the resulting electric current (the initiation timing) by adjusting the amplitude of the sinusoidal potential at the moment
A single excitatory synapse, such as a glutamatergic synapse, which accounts for nearly 90% of synapses in the cortex, generates a current ranging from 5 to 110 pA, with an average of 25 pA, and a maximal conductance of 700 to 1000 pS (ranging from 0 to 1000 pS). The reliability of a synapse to elicit or influence spikes depends not only on its conductance but also on its proximity to the spike initiation point, typically located near the soma where the axon emerges, and often distant from the synapse itself. At most, a single synapse produces 100 pA at the origin, with a local membrane potential variation of less than 3 to 5 mV, insufficient to surpass the threshold for firing. As the potential wave travels from the origin to the soma, where the spike is generated, these values decrease due to dendritic cable properties. Typically, a synaptic event occurring in the dendritic tree induces less than 0.5 mV in membrane potential, which, when added to the resting level, fails to reach the threshold. These observations lead to a significant conclusion: firing a spike necessitates the temporal integration of currents arriving from multiple synapses.
A post-synaptic current intensity of
In a recent study, 47 we explored the induced magnetic fields within living tissues. Our findings suggest that if we acknowledge the presence of off-diagonal terms in the synapse conductivity tensor, an exogenous EM field can indeed trigger a synapse magnetic field, thereby altering neurotransmitter dynamics.
Hence, the following question emerges: Could an exogenous EM field, generating a weak synapse magnetic field (
When applying a continuous input of 1

Numerical simulation of the model with input driving current of 1
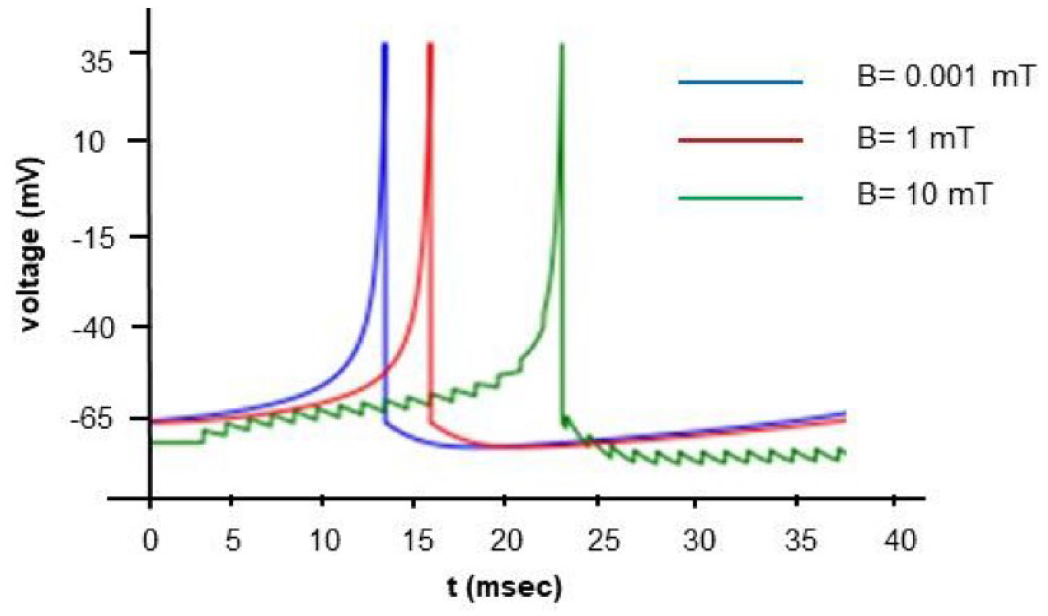
Numerical simulation of the model with input driving current of 5
Anastassiou et al51,52 investigated the impact of spatially inhomogeneous extracellular electromagnetic fields on neurons, specifically focusing on alterations in spike timing. They observed that extracellular magnetic fields, within the same order of magnitude as those utilized in our study, induced changes in somatic membrane potential of less than 0.5 mV under subthreshold conditions. Furthermore, they conducted simultaneous recordings from up to 4 patched neurons located proximally to each other. Their findings suggest that despite their small magnitude, these magnetic fields could significantly influence spike timing, aligning with the theoretical outcomes of our study.
We can analyze the stability of disturbances within the framework of stable oscillator theory. Figure 6 illustrates the phase shift among identical oscillators with matching periods, initially synchronized, upon application of a continuous current input of 100

Numerical simulation of the model with input driving current of 100
Stability analysis of the spike-timing model based on bi-dimensional dynamic systems
Our modified HH model is a dynamic system consisting of the state variables
The system state is a 2-dimensional vector
First, we need to establish the equilibrium points to study the nullclines of the state variables. A nullcline is the set of locations in the phase plane where the state variable is at rest. In this system, it would correspond to the conditions
The intersections of these nullclines will be the points where none of the state variables are changing, thus indicating membrane equilibrium. In Figure 7, we observe the cubic-shaped nullcline for

Representation of the nullclines of the model with
Unlike 1-dimensional systems, stability cannot be determined solely by the slope of the
Now we will analyze the stability of the equilibrium point of
The corresponding value of
The trace of the matrix is
Next, in Figure 8, we can observe trajectories in the phase plane with different initial values of continuous current inputs (1 and 10
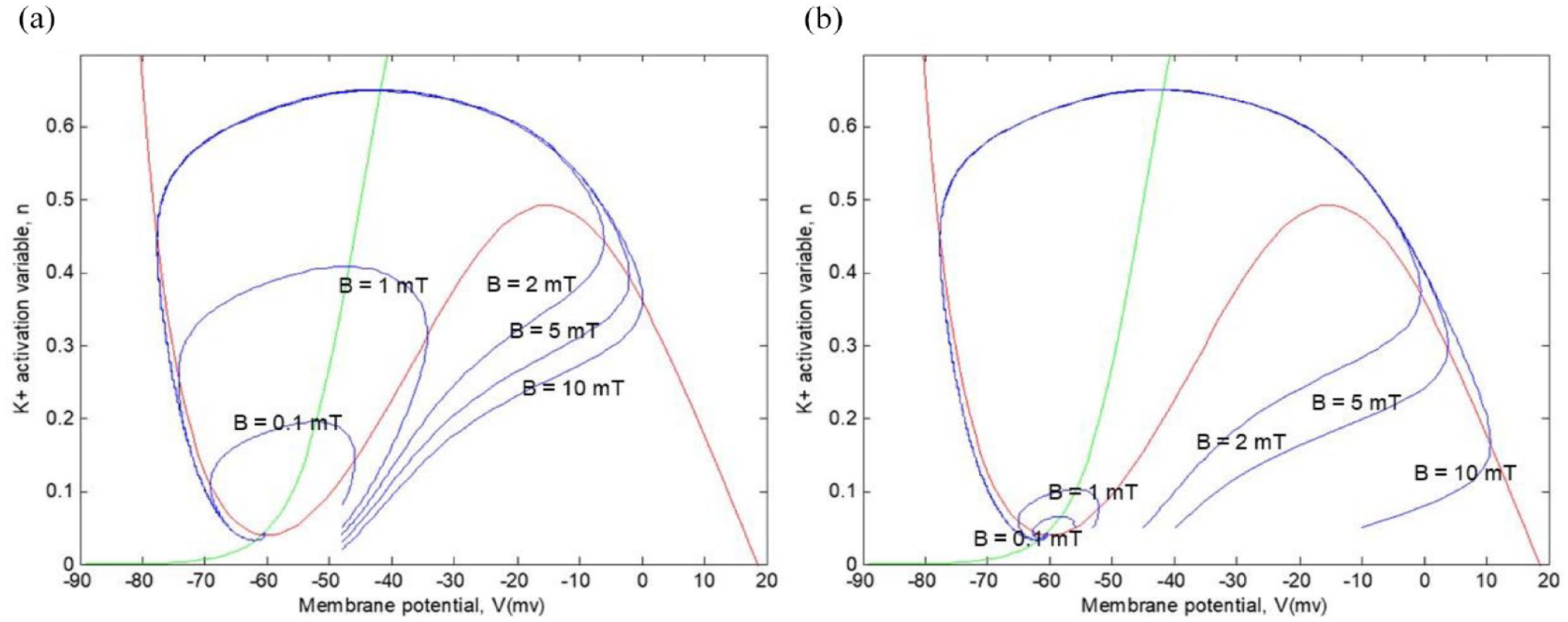
Representation of various action potential trajectories with different continuous current inputs and different
Figure 9 shows the emergence of the limit cycle between the values of

Representation of various action potential trajectories with different continuous current inputs and
STDP, or Spike Timing-Dependent Plasticity, is a phenomenon whereby the precise timing of neuronal spikes influences both the direction and magnitude of synaptic strength alterations.53,54 Often regarded as the fundamental learning rule governing synapses, STDP has undergone successive refinements to address predictive limitations. In proposing an alternative fundamental principle, we suggest that exogenous or endogenous EM fields induce changes in crucial biochemical intermediates, such as neurotransmitters, serving as more direct triggers of plasticity mechanisms. The modulation of chemical synapses by STDP is dictated by the temporal discrepancy between pre- and postsynaptic neuronal firing, essentially the synchronization between neurons. 55 Concurrently, the magnetic coupling among neurons can enhance signal exchange and foster global synchronization within the neural network.56,57
We have observed that the mechanism underlying the temporal information processing of a neuron is determined by a non-linear current-voltage relationship associated with the interactions between the exogenous EM field and certain proteins within the excitable membrane. Therefore, the variability of the spike timing of a neuron would be strongly influenced by extracellular EM fields in close proximity to a bifurcation point. However, we have demonstrated that within the range of
Conclusions
Cognitive neuroscience has revealed a direct correlation between structural connections throughout the brain’s temporal hierarchical levels. Considering that all cognitive functions exhibit temporal characteristics, it is conceivable that the accurate estimation of transient alterations within specific temporal hierarchies is crucial for interpreting information from weak periodic endogenous or exogenous stimuli, such as static or extremely low-frequency magnetic fields. Thus, investigating the effects of magnetic fields on spike-time dynamics and their influence on other core cognitive functions may shed light on fundamental processes in typical cognition and their dysregulation in neuropsychological and mental disorders.
The EM field theory enables the determination of fields at the mesoscopic scale based on the morphological properties and bioelectrical tissue parameters at the microscopic scale. In this study, structural and functional intricacies are simplified by delineating 2 domains (internal and external neuron spaces), characterized by a set of interconnected fields governing the movement of charged particles within a magnetic field.
Estimating synaptic conductance for any noise correlation time remains an unresolved issue. While we outline a physical framework with simplifying assumptions to aid in understanding or predicting phenomena, obtaining data from experimental or quasi-experimental studies would be valuable and insightful.
We establish a connection between the STPD phenomenon and equations that depict the movement of neurotransmitters under an induced magnetic field. Our findings uncover the pivotal role of EM fields in governing the precise timing of pre- and postsynaptic spikes. This timing intricately shapes the outcomes of the STDP rule, while the propagation delay of neural signals significantly influences the connectivity patterns and dynamics of neural networks.58,59
The macroscopic study of complex phenomena such as STPD is based on the application of constitutive equations to a supposedly continuous model subject to given constraints. These equations arise from the incorporation into conservation relationships (mass, momentum, energy. . .) of phenomenological laws known as kinetics, which connect the various currents therein (diffusion, chemical reaction rates, stresses, heat. . .) with corresponding state variables and their spatial derivatives (Fick’s law, chemical kinetics, Fourier’s law. . .).
In the particular case of STPD, the mechanisms generating instability are the autocatalytic transport of ions across excitable membranes in nerve cells or positive or negative feedback between neurons. Metabolic oscillations result from the control of enzyme activity, while oscillations in protein synthesis result from genetic regulation. The common point of biological rhythms in our study is that they have an exogenous origin. They possess all the properties of dissipative structures, in the sense that they exhibit a phenomenon of temporal self-organization under conditions of non-equilibrium. It is the internal regulatory mechanisms of biological systems that, through the bias of nonlinear kinetics, lead to instability phenomena associated with the emergence of periodic behaviors beyond the bifurcation point. In our case, within the physiological parameter range of intensity (
Our approach to predicting timing changes in spike induced by magnetic fields can be extended to local neural networks. In fact, some authors consider extracellular EM field oscillations as a marker of neural network oscillations.60 -62 It is even suggested that information contained in the oscillations of endogenous extracellular potentials could provide particularly relevant insights into the temporal coding of network activity.63,64 This offers promising prospects regarding potential therapies for pathological neural synchronization, such as in the case of Parkinson’s disease, for example. The results may suggest that, by appropriately choosing the delay time, a mechanism of control like STPD through locally applied external EM field to a neural subpopulation could suppress global neuron synchronization. The coupling mechanisms of network activity to exogenous EM field are not an alternative to classical approaches but complementary mechanisms that take into account the spatiotemporal characteristics of the involved EM field.
A fresh perspective on the macroscopic system, arguably one of the most debated topics in neuroscience research, emerges from examining the average synaptic activity time in neural networks through the lens of path integrals. This approach offers insights into the partial understanding of asymptotic behavior on a microscopic scale. In the long run, it becomes imperative to delve into the topological equations governing neural temporal hierarchical levels, an area where analytical findings are scarce. This poses an issue of exceptional interest, as theoretical conjectures supported by experimental evidence indicate that neural networks retain information over timescales corresponding to their anatomical hierarchy.
Supplemental Material
sj-docx-1-bec-10.1177_11795972241272380 – Supplemental material for A Physical Framework to Study the Effect of Magnetic Fields on the Spike-Time Coding
Supplemental material, sj-docx-1-bec-10.1177_11795972241272380 for A Physical Framework to Study the Effect of Magnetic Fields on the Spike-Time Coding by Manuel Rivas and Marina Martinez-Garcia in Biomedical Engineering and Computational Biology
Footnotes
Author Contributions
M.R. and M.M. wrote the main manuscript text and prepared all the figures. All listed authors have contributed significantly to the manuscript and consent to their names and order of authorship on the manuscript. Co-authors will be kept informed of editorial decisions and changes made. There is no conflict of interests involving the conduction or the report of this research.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Founding grant CIGE/2022/066 from Generalitat Valenciana, Spain.
Availability of Data and Materials
Hereby, we M. R. and M. M. consciously assure that for this manuscript the following is fulfilled: (1) This material is the authors’ own original work, which has not been previously published elsewhere; (2) The paper is not currently being considered for publication elsewhere; (3) The paper reflects the authors’ own research and analysis in a truthful and complete manner; (4) The paper properly credits the meaningful contributions of co-authors and co-researchers; (5) The results are appropriately placed in the context of prior and existing research; (6) All sources used are properly disclosed (correct citation). Literally copying of text must be indicated as such by using quotation marks and giving proper reference, and (7) All authors have been personally and actively involved in substantial work leading to the paper and will take public responsibility for its content.
Ethical Approval
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.