Abstract
Objectives
Low- and lower-middle-income countries (LMICs) bear a disproportionately high burden of neurosurgical disease while facing severe shortages of trained neurosurgeons and anesthesiologists. Access to advanced technologies such as intraoperative neuromonitoring (IONM), crucial for complex neurosurgical procedures, is virtually nonexistent due to infrastructural, educational, and financial barriers. This study describes the development and implementation of Uganda’s first IONM program at Cure Children’s Hospital of Uganda (CCHU), using the well validated Exploration, Preparation, Implementation, and Sustainment (EPIS) framework.
Methods
In collaboration with the University of Colorado, a standardized non-traditional training pathway was created, combining remote and in-person instruction in neurophysiology, technical IONM applications, and anesthetic considerations. Over a 3-year period, the program enabled CCHU to safely perform more than 80 complex pediatric neurosurgical cases across 2 years, incorporating modalities such as somatosensory evoked potentials (SSEPs), motor evoked potentials (MEPs), and electromyography (EMG). Surgical and patient outcomes were not tracked for the purposes of this implementation study.
Results
Between March 2022 and October 2024, 80 surgeries were performed at CCHU that made use of IONM technologies. The vast majority of the surgeries were intracranial and included one awake tumor resection with direct cortical mapping (DCS). A smaller subsect of patients underwent spine surgery for either tumor removal or correction of spinal cord tethering. Due to the difficulty in patient outreach many patients were lost to follow-up, however, efforts are being made to track patients and examine their clinical outcomes.
Conclusion
Despite resource limitations, this initiative demonstrated that IONM can be effectively integrated into LMIC healthcare settings, improving surgical safety and outcomes while contributing to local workforce development. The success of this program underscores the value of international partnerships and implementation science in bridging global neurosurgical disparities and suggests a scalable model for future adoption across similar contexts.
Background
Neurosurgical Burden of Disease in LMICs
Improving access to safe and cost-effective medical care is a long sought-after goal of the public health and medical community, particularly in low-income and lower-middle-income countries (LMICs). 1 However, with limited worldwide resources, it is imperative that efforts and assets be aimed at cost effective, sustainable projects that meet a significant, recognized and specific need.
A clear opportunity for intervention exists in improving access to care for neurological disorders, as they are recognized as the leading cause of disability worldwide. It has been demonstrated that LMICs carry a disproportionately large burden of the global need for neurosurgical care, with a parallel lack of available providers. The continent of Africa specifically carries one of the greatest disparities between need for neurosurgical care and availability of care, as 15% of the global neurosurgical burden is in Africa while less than 1% of neurosurgeons live in Africa. 2 Concomitantly, the need for specialty-trained anesthesia providers is crucial to safely care for this large cohort of neurosurgical patients. While there is a paucity of specific data documenting the exact density of anesthesia providers in LMICs, the available data suggests that there is a shortage of anesthesia providers like the shortage of neurosurgical providers. In Uganda, for example, there is an estimated <1 anesthesiologist per 100 000 persons as compared to an anesthesia provider density of approximately 18 per 100 000 in the United States.3,4 Increasing access to safe and effective neurosurgical care may improve patient outcomes in addition to providing economic benefits to the region by reducing overall disability. Neurosurgical cases that require intervention but persist unaddressed may lead to increased rates of disability and death for patients. Unmet neurosurgical care in these under-resourced regions is estimated to result in GDP losses greater than $3 trillion. 5 This illustrates how cumulative disability years may not only lead to a significant impact on populations with respect to both quantity and quality of life but also further propagates the vicious cycle in which LMICs are limited in their ability to achieve their economic developmental goals.
Ongoing efforts to address the unmet need of neurosurgical care in LMICs include broadening trainee diversity as well as the development of intracontinental and international partnerships to establish training pipelines. Additionally, providing access to more advanced training like intraoperative neuromonitoring (IONM) can help improve patient safety and surgical outcomes in complex neurosurgical care. Here we describe the implementation of a novel IONM program in Mbale, Uganda that not only satisfied the objectives mentioned above but successfully delivered safe and quality care to pediatric patients whose cases were deemed too complex to perform without IONM. To our knowledge this is the first IONM program established in Uganda.
IONM Utilization in Resource-Limited Areas
LMICs can face unique challenges when incorporating emerging or established technologies into day-to-day practice, one such example is the use of IONM as an adjunct in surgical practice. The range of barriers that LIMCs often encounter can include limited access to financial resources, material support, and the necessary training to acquire and maintain skills. Additionally, the maintenance of equipment can be problematic due to the lack of proper infrastructure and the unsuitability of power grids even in the most developed areas. Issues like lack of proper electrical grounding, which is necessary for quality signal acquisition in IONM, also pose significant hurdles. The scarcity of well-maintained systems, along with geographical disparities to healthcare access, further exacerbates these challenges. These factors can make the routine incorporation of IONM in neurosurgical practice quite difficult in these settings. 6
Improving access to IONM might seem like a superfluous goal in the face of already limited access to safe and effective neurosurgical care, however there is substantial evidence to support that IONM improves patient safety and outcomes for surgical procedures where neural structures are at risk of injury.7,8 It is particularly vital during complex spine, brain, and vascular surgeries. A combination of studies, including somatosensory evoked potentials (SSEPs), motor evoked potentials (MEPs), electroencephalography (EEG), and electromyography (EMG), can be used to identify and prevent permanent neurologic injury by providing real-time feedback about the functional integrity of the somatic nervous system. In countries such as the United States, the use of IONM has increased dramatically over the past two decades, particularly in teaching hospitals; however, it is far from ubiquitous in its use and availability. 9
As previously discussed, the limitations to access neurosurgical care in LIMCs include a shortage of neurosurgeons and lack of access to facilities able to provide intensive neurosurgical care. Similarly, IONM access is limited by the number of individuals who are trained to provide high quality and reliable services. The American Board of Registration of Electroencephalographic and Evoked Potential Technologists (ABRET) is the largest organization known to provide internationally recognized accreditation to technicians trained in IONM. Traditionally, to obtain the certification for intraoperative neuromonitoring (CINM) an individual with a bachelor’s degree must undertake rigorous didactic training, participate in 100-150 cases and pass a certification exam, according to the requirements published by ABRET. This can be a lengthy, time-consuming and expensive process. Given these requirements it is not unusual to note that, according to an inquiry made to the ABRET staff database at the time of the inception of this project, only 6 individuals in the entirety of Africa claim certification through this organization. Admittedly, ABRET is not the only organization capable of providing certification, however it is the largest and most well-known. Individuals with training in neurophysiology necessary to interpret IONM signals are even more scarce worldwide. This is likely due to a lack of knowledge and awareness that leads to a lack of utilization of IONM which in turn leads to limited opportunities for individuals to train and later implement their skill set. Without a clear path to a stable practice, it is reasonable that individuals would choose to not undertake such lengthy training and certification.
In contexts like Uganda or other parts of Sub-Saharan Africa, these barriers are particularly pronounced. To date there are no ABRET CNIM technicians in the whole of Uganda, though there are individuals who provide IONM services without certification or oversight. This leaves neurosurgeons interested in operating on patients with complex intracranial or spine tumors without access to a technology which could help prevent a catastrophic surgical outcome. Many surgeons will not attempt the most complex resections for fear of injuring patients or will perform subtotal resections which are likely to be only palliative in nature thus leading to poorer patient outcomes. However, as global health initiatives continue to grow and technology becomes more accessible, there is growing interest in addressing these gaps in access to technology to improve neurosurgical outcomes. We believe that through increased awareness, training, and technological advancements IONM could see broader implementation over time. One such example is access to telemedicine and remote IONM, which would allow experts from other countries to assist surgeons in real-time via remote monitoring services. This could reduce the need for in-house experts in the short term, allowing African countries to benefit from access to IONM while local expertise is under development.
Development and Implementation of an IONM Program at CCHU
Cure Children’s Hospital of Uganda (CCHU) is a specialized Pediatric Neurosurgical hospital located in Mbale City in eastern Uganda. The hospital offers both inpatient and outpatient neurosurgical care for children from all over the country and neighboring countries as well. CCHU performs approximately 2000 pediatric neurosurgical procedures annually as of 2024. Most of the disease burden is hydrocephalus 65-75%, spina bifida 15-20%, brain and spine tumors and other congenital anomalies account for the rest. The number of children presenting with complex neurosurgical diseases like spinal lipomyelomeningocele, spinal tumors, posterior fossa brain tumors and supratentorial tumors has continued to rise over the years. For some complex neuro surgical conditions, performing surgery without IONM poses a great risk for injury to neural tissues potentially leading to worse neurological outcomes post-operatively. After identifying the great need of this patient population and meeting the motivated individuals at CCHU a partnership was born. In this narrative we will describe the experiences and challenges of implementing a de novo IONM program in Uganda using the established and validated Exploration, Preparation, Implementation and Sustainment (EPIS) framework.
Methods
Through a pre-existing and ongoing partnership between University of Colorado Department of Anesthesiology (CU) and CCHU it was identified that implementing an IONM program would be advantageous to the region’s populace. High-level goal-oriented conversations were initiated in 2018, and stakeholders, needs and potential challenges were identified. Teaching protocols were developed and implemented by experienced technicians as well as fellowship trained Neuroanesthesiologists with IONM certification. The didactic component focused on basic neuroanatomy, physiology, the technical aspects of IONM as well as clinical effects and interactions of anesthetics with IONM signals. The practical component of the course began with virtual case simulations using Cadwell Inc’s proprietary simulation software, Cascade, and progressed to proctored patient care administered by experienced technicians. The CNIM candidate underwent a non-traditional path to obtain certification though was subject to the same measures of quality and safety as well as formal testing to complete the program. The anesthesiologist who is now responsible for oversight of the IONM program at CCHU was trained with the same rigorous didactic and in-person training.
Anesthetic protocols were developed and tailored to the needs of the patient population and the availability of drugs and equipment at CCHU. The technical components of signal acquisition were standardized, and protocols were developed to suit the procedures being performed at CCHU.
Our efforts spanned over 4 years from the discovery phase to the implementation and maintenance phases. This was purely an implementation and quality improvement study and was vetted by an internal ethics committee at CCHU comprised of local stakeholders. Patient demographic information was not formally tracked outside of CCHU’s internal medical record keeping. Patients were individually evaluated and selected to receive IONM based on the anatomy and planned surgical approach in attempts to minimize the risk of injury to functional neural structures, this was done by the responsible neurosurgeon.
Anesthesia Protocols and Monitoring Modalities
Anesthesia Protocols
Each patient scheduled for surgery underwent a thorough preoperative assessment, including a medical history review, physical examination, and review of relevant laboratory tests and pertinent imaging. Any potential contraindications or risks associated with the surgery were identified and every attempt made to address them preoperatively.
Based on the patient’s medical history, surgical procedure, and individual considerations, an appropriate anesthesia plan was formulated. A range of techniques including general anesthesia, sedation, regional anesthesia, or a combination were explored for each patient as appropriate. Maintenance of anesthesia was planned to be synergistic with the appropriate IONM modalities whenever possible. Propofol TIVA was preferred for any case involving SSEPs and MEPs to prevent signal decrements due to volatile anesthesia. 10 Non-depolarizing neuromuscular blockers were contraindicated in cases involving MEPs and EMG monitoring. There were no contraindications to succinylcholine, narcotics or ketamine. Dexmedetomidine, lidocaine and magnesium infusions were avoided as they can affect the quality of IONM signals to varied degrees. 11
Throughout the surgical procedure, the patient’s vital signs, including heart rate, blood pressure, oxygen saturation, and end-tidal carbon dioxide, were continuously monitored to meet safety standards. Additionally, depth of anesthesia monitoring, such as bispectral index (BIS), was utilized when available to ensure appropriate anesthetic depth.
Modalities for Intraoperative Monitoring
Monitoring modalities were chosen based on the location of surgery and the structures at risk and included Somatosensory Evoked Potentials (SSEPs), Motor Evoked Potentials (MEPs) and Electromyography (EMG). SSEP monitoring involves stimulating peripheral nerves and recording the electrical responses over the scalp covering the sensory cortex region. 12 These signals provide valuable information about the integrity of the somatosensory pathways and are particularly useful in procedures involving the primary sensory cortex, spinal cord or peripheral nerves. MEP monitoring assesses the integrity of motor pathways by stimulating the motor cortex and recording responses at target muscles. 12 MEPs are commonly used during procedures that pose a risk to the motor system, such as spinal cord surgeries or surgeries near motor pathways. EMG monitoring involves the placement of electrodes on specific muscles to monitor their electrical activity. It helps assess nerve and muscle function and detect changes that may indicate nerve injury or abnormal muscle activity during surgery. 12 Triggered EMG can be used to identify peripheral nerves by recording electrical activity over the target muscle.
Monitoring was performed by qualified personnel who were trained remotely for over a year including rigorous online didactic courses, virtual lectures and workshops and finally an in-person training. The recorded data was analyzed in real-time and communicated to the surgical team and remote interpreting physician for prompt intervention if any abnormalities or significant changes were detected. In-person and virtual consultation services were provided by trained Neuroanesthesiologists credentialed in the interpretation of IONM at CU. At CCHU the project lead anesthesiologist, Dr Nantongo, visited CU and received in-person training in complex neuroanesthesia cases and IONM signal acquisition and interpretation for a month. Her focus was on cases with complex spine pathology and intracranial tumors.
Results
Summary of Intracranial and Spine Surgical Procedures Performed at CCHU Between March 2022 and October 2024
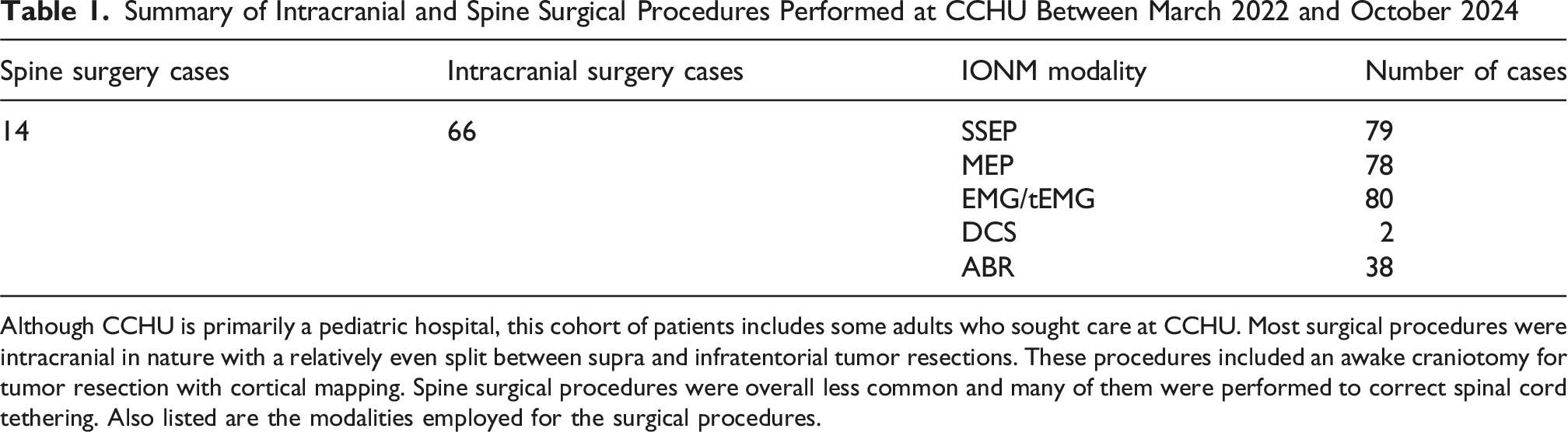
Although CCHU is primarily a pediatric hospital, this cohort of patients includes some adults who sought care at CCHU. Most surgical procedures were intracranial in nature with a relatively even split between supra and infratentorial tumor resections. These procedures included an awake craniotomy for tumor resection with cortical mapping. Spine surgical procedures were overall less common and many of them were performed to correct spinal cord tethering. Also listed are the modalities employed for the surgical procedures.
Discussion
The implementation of IONM technology in a resource limited country has allowed for enhanced surgical care of technically and clinically complex neurosurgical pathologies. The surgical population at CCHU is made up mostly of intracranial pathologies and all cases performed prior to this implementation were done without the benefit of IONM. Starting in 2021, and over a period of 3 years, more than 80 surgeries have been performed with IONM at CCHU, patient age ranged between 6 months to 18 years. Regarding the case load, the predominant pathologies were intracranial tumors with posterior fossa tumors accounting for over 80% and spine pathologies accounted for only 10% of the total number of cases. For all these cases SSEPs, MEPs and EMG modalities were performed where appropriate.
Additionally, through other multidisciplinary collaborations in the last 3 years CCHU is now able to provide pediatric epilepsy surgery, some patients with lesional epilepsy may also benefit from the already implemented IONM services through advanced cortical mapping techniques.
From the inception of the relationship between CCHU and CU there was a desire to develop IONM to enhance surgical capabilities and improve patient outcomes. Prior work in implementation science has established the need for a validated framework to better ensure project success. Validated frameworks are crucial for successful technological implementations within clinical care. These frameworks help in identifying factors facilitating adoption, assessing organizational readiness, and guiding implementation strategies. 13 The EPIS framework is a comprehensive, multiphasic approach for guiding the implementation of technology and practice paradigms in clinical settings. 14 This versatile and utilitarian framework encompasses four distinct phases and has been applied in various settings. Our ability to undertake a successful project was bolstered by the sound methodology and prior history of evidence-based interventions within this framework. 15
Exploration Phase
The exploration phase involves identifying needs and barriers through research, expert consultation, and scoping activities. 16 This phase is crucial for adapting interventions to local contexts and priorities. The established relationship between CCHU and CU had resulted in multiple high-level conversations related to patient quality and safety, standards of care, and needs assessments. Aspirations to treat even more complex surgical patients and to improve outcomes led to the discussion involving moving forward with IONM as a conduit to achieve these goals. During this exploration phase, it was determined that patient demographics, surgeon desire to prioritize the technology, a suitable partner available to train and guide implementation, and the ability to recruit and train future technologists all existed and could work towards the goal of IONM implementation.
Preparation Phase
Within this phase, key considerations include assessing the suitability of work environments and planning for workflow adjustments. 17 Effective strategies in this phase involve providing on-site assistance and training, specific program roll-out planning, and identifying staff with adequate time to engage in the program. Within this phase, ongoing discussions with the hospital Executive Director and Chief of neurosurgery, lead to the defining of roles of an IONM supervising physician (anesthesiologist) and future technologists. These individuals were targeted for their desire to advance skills and dedication to complete training. Online training in IONM technology began approximately 3-month prior to in-person implementation. Education consisted of live, online lectures, textbook readings, training modules, and structured examinations. Training courses followed ABRET training standards for technologists. Because of the technical complexity of IONM and the time needed to place electrodes, acquire signals, and troubleshoot any concerns, it was determined that education for the entirety of surgical personnel would be needed. Additionally, patience and perseverance were stressed, as all new technologies have a learning curve associated with efficiency and efficacy.
Implementation
During implementation organizational vision, leadership support, and personnel resilience are key components of success. Teams and communication within these teams along with preemptive strategies to maximize communication between teams are essential. 16 During this phase, as well as during preparation, pre-operative multidisciplinary meetings were held with neurosurgery, anesthesiology, nursing, and IONM, to review patient images, comorbidities, discuss surgical and anesthesia plans, and discuss new IONM workflow. Software technology provided by Cadwell Inc. allowed for operative simulations to be carried out. Multiple cases were simulated and allowed for trainees to prepare and troubleshoot technologies prior to implementation with actual patients. At the completion of online training and education, plans were made for in-person implementation. Surgical cases were booked, travel to CCHU was planned for members of CU and equipment and consumables were prepared for transportation.
Sustainment
This phase is essential for long-term success. Because of the mutual desire of partners to provide sustainable, long-term improvement in patient quality and safety, we created systems to aid in achieving this goal. Online support was provided by the team at CU, offering clinical case review, real-time technological assistance, and post-operative debriefings. In addition, follow-up in-person visits were completed at 90 days and 6-months post-implementation. Financial support from CCHU ensured that consumable resources are available to continue providing this necessary technology. Since implementing this IONM program, CCHU has served as a hub for other international partners to begin establishing their own IONM programs, notably a coalition with anesthesiologists and technologists in Ethiopia. This collaboration is ongoing and will make use of the knowledge and lessons learned while establishing IONM at CCHU with hopes of improving and optimizing the implementation process for future sites (Figure 1). Exploration, preparation, implementation and sustainment cycle for implementation of sustainable projects
Conclusion
This study demonstrates the successful implementation of a novel IONM training program at CCHU, marking a significant step toward enhancing neurosurgical care in a LMIC setting. Utilizing the EPIS framework, we established a sustainable model that trained local personnel, integrated advanced monitoring modalities and improved surgical safety for complex pediatric neurosurgical cases, such as intracranial and spinal tumors. Our project supports the establishment of future IONM programs despite initial barriers like resource scarcity and infrastructure challenges. Accessible IONM can be effectively adapted to resource-limited environments, offering real-time neural integrity feedback that enhances patient outcomes in procedures previously deemed too high-risk or inoperable. One unforeseen challenge manifested itself in record keeping and in difficulties keeping track of patient outcomes. Many patients traveled to CCHU from remote parts of Uganda and other countries in Africa, making it very difficult for them to afford to travel back to Mbale after their initial procedure. This resulted in some patients being lost to follow-up. This highlights the importance of having a robust communication system in place to keep track of patient outcomes to help guide the utilization of this technology in new and remote settings.
Furthermore, the public-private partnership underscores the potential of international collaboration to address the disproportionate neurosurgical burden in LMICs, where Africa bears 15% of the global need but hosts less than 1% of neurosurgeons. 2 Beyond the underscored clinical benefits, this initiative also supports economic development by developing local workforce capacity, reducing disability and associated GDP losses, while aligning with broader public health goals. However, limitations—including the steep learning curve for personnel, reliance on external funding for consumables, and the absence of certified ABRET CNIM technicians in Uganda—highlight the need for cautious scaling and ongoing support. Future efforts should focus on expanding local training pipelines, leveraging telemedicine for remote IONM support, and securing sustainable funding to ensure long-term viability. This pioneering program not only bridges a critical gap in Ugandan neurosurgical care but also sets a precedent for integrating advanced technologies into LMIC healthcare systems, paving the way for further innovation and improved patient safety worldwide.
Footnotes
Acknowledgements
We would like to thank the University of Colorado School of Medicine for their institutional and financial support. As well as Cadwell Inc, for their in-kind donation of IONM equipment. And lastly, we would like to thank the staff at CCHU all of whom were invested in making this project a success.
Ethical Considerations
Ethics approval was obtained from an internal ethics committee at CCHU that included local stakeholders.
Consent for Publication
No patient identifying information is included in this report.
Author Contributions
Julio Montejano MD, project implementation, training, virtual support, manuscript writing, editing and corresponding author. Betty Nantongo MD, project implementation, manuscript writing. Tessa Klumpp MD, manuscript writing. Teguo Daniel Djoyumm CNIM DrPH, project planning and implementation, virtual support, manuscript writing. Joshua, Lahiri, project implementation, training, virtual support and manuscript writing. Daniel Watiti MCO, manuscript writing. Humphrey Okechi MD, project planning and implementation, manuscript writing. Emmanuel Wegoye MD, project planning and implementation, manuscript writing. Colby Simmons DO, MBA, project inception, planning, implementation, manuscript writing and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Funding obtained through the University of Colorado School of Medicine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
