Abstract
Background
Upper limb motor deficits are common after stroke and often persist despite rehabilitation. While clinical assessments emphasize movement quality, they do not capture the underlying neuromuscular impairments, particularly in individuals with mild deficits. This limits the development of individualized treatment approaches.
Objectives
This study aimed to characterize stroke-related changes in muscle recruitment during reaching by separating tonic (gravity-compensating) and phasic (intersegmental dynamics-related) components of EMG activity.
Design
Cross-sectional observational study with a comparative design.
Methods
We recorded surface EMG from 12 upper limb muscles during goal-directed reaching in 8 individuals with unilateral ischemic stroke, 5 age-matched and 9 young individuals. Using principal component analysis, we extracted tonic and phasic components and compared their amplitude, directional tuning, and coactivation patterns across groups. Group differences were evaluated with generalized linear mixed-effects models, regression, and correlation analyses.
Results
Even individuals with mild stroke exhibited abnormal muscle recruitment. Proximal muscles were over-recruited in directions that typically require less activation, indicating altered directional tuning. Phasic activation of distal muscles was significantly reduced and worsened with time post-stroke (R2 = 0.52, P = 0.002). Tonic overactivation of proximal muscles was present across all stroke participants. Muscle coactivation patterns were hemisphere-specific: right-hemisphere stroke reduced tonic coactivation in contralateral arms, whereas left-hemisphere stroke increased it. Abnormal phasic coactivation between proximal and distal muscles correlated with impaired intersegmental dynamics compensation (R2 = 0.67, P = 0.013). Tonic and phasic impairments were often correlated, suggesting shared disruption of corticospinal and reticulospinal pathways.
Conclusion
These findings reveal distinct yet interacting deficits in tonic and phasic muscle recruitment following stroke, including persistent overactivation of proximal muscles for antigravity support and reduced phasic activation of distal muscles. These results suggest that individualized rehabilitation strategies should address both inefficient gravitational support and impaired intersegmental coordination to reduce post-stroke muscle overactivation and improve movement efficiency.
Plain Language Summary
After a stroke, many people have trouble moving their arms, and these problems can last even after therapy. Doctors usually assess movement by watching how smooth or accurate it looks, but this doesn’t show what is really going wrong in the muscles and nerves—especially for people with milder symptoms. In this study, we looked at how stroke changes the way muscles are used during reaching.
We focused on two types of muscle activity: Tonic activity, which helps hold up the arm against gravity; Phasic activity, which helps to propel the hand toward the goal.
We recorded muscle activity in the arms of people with and without stroke while they reached for a target. We then analyzed how much each muscle was used and how they worked together. We found that even people with mild stroke had unusual muscle use. Muscles near the shoulder (proximal muscles) were used too much; while muscles farther down the arm (distal muscles) didn’t activate as strongly as they should.
The patterns of muscle coordination changed depending on which neural pathway was damaged by the stroke. These patterns suggest that stroke disrupts the activity of multiple muscles and that causes movement impairment. The results show clear, specific changes in how muscles are controlled after a stroke, and point toward new ways to design better, more personalized rehabilitation tools that target the real problem inside the nervous system—not just the way movement looks on the outside.
Keywords
Introduction
Stroke is the third leading cause of combined mortality and disability worldwide, 1 with upper limb motor impairment affecting 80% of survivors 2 only half of whom regain useful upper limb function. 3 Despite advances in rehabilitation, many individuals continue to experience poor arm control even years after stroke onset. Clinical assessments often focus on observable movement quality or joint angles, which cannot assess fatigue and may overlook the underlying neuromuscular coordination strategies that shape recovery. 4 Moreover, spasticity, defined as a velocity-dependent increase in muscle tone, is a common consequence of stroke and contributes significantly to motor impairment. 5 It cannot be directly observed through movement alone and requires additional clinical or instrumented testing for accurate assessment. Understanding how stroke alters the recruitment of specific muscle groups, especially in response to biomechanical demands such as antigravity support and intersegmental dynamics, is essential for the underlying mechanisms of motor deficits. This knowledge can, in turn, guide the development of more targeted and individualized rehabilitation strategies.6,7
Clinical assessments rely on low-resolution scoring and rater training to reduce variability, but this limits responsiveness and predictive validity—especially in patients with mild deficits—due to ceiling effects.8,9 These tools also cannot distinguish impairments from passive mechanics vs active muscle coordination, leading to inconsistent treatment outcomes in trials.10-12 We previously showed that motion capture–based measures of muscle torque are more sensitive than joint angles in detecting post-stroke motor deficits. 4 This is especially valuable for patients with minimal visible impairment who still report difficulty. The current study builds on this by analyzing muscle recruitment during goal-directed reaching, aiming to identify objective physiological markers that improve on current clinical assessments.
Abnormal muscle recruitment patterns after stroke lead to inappropriate force generation that underlie motor impairment. During reaching, muscle forces serve two primary functions: (1) supporting the arm against gravity—primarily mediated by proximal muscles that span the shoulder and elbow, and (2) compensating for complex limb dynamics—including inertial, Coriolis, and centrifugal forces—primarily mediated by distal muscles that span the elbow and wrist. Muscle forces also generate stiffness and viscosity that help stabilize joints and respond to unexpected perturbations—features that are not fully captured by muscle torques alone. Muscle forces result from muscle contractions caused by motoneuron firing, which is observable with EMG. In our previous work, we demonstrated that the tonic component of EMG reflects muscle forces related to gravity compensation during reaching, while the phasic component reflects the remainder forces related to intersegmental dynamics. 13 After stroke, efforts to actively support the arm against gravity or to load the shoulder often result in coupled elbow moments, which reduce the reachable workspace and restrict movement range.14-17 This pattern suggests that neural mechanisms for gravity compensation—likely mediated by the reticulospinal system—may become abnormally engaged after stroke.18-20 In addition, stroke disrupts complex intralimb coordination and grasping,7,15,21 functions that may depend on plasticity within corticospinal projections.22-24 The reticulospinal and corticospinal systems are hierarchically organized and anatomically coupled,25-27 further complicating efforts to develop targeted interventions. In this cross-sectional feasibility study, we investigate how stroke affects these systems by testing two hypotheses: (1) stroke affects the recruitment of distal muscles that move the wrist and hand more than proximal and biarticular muscles that move the shoulder and elbow; (2) stroke affects the phasic component of muscle recruitment responsible for intersegmental dynamics compensation more than the tonic component of muscle recruitment responsible for gravity. Testing these hypotheses will result in clinically meaningful insights into post-stroke recovery and provide a foundation for individualized motor rehabilitation approaches.
Methods
Data Collection
Characteristics of Individuals With Stroke

MCA, middle cerebral artery; BS, brainstem; BG, basal ganglia; hemi is short for hemisphere.
Participants were asked to reach in virtual reality (VR) shown in 3D environment created with Vizard (Worldviz) and shown via a head-mounted display (Oculus Rift), detailed in earlier publications (Figure 1A).4,13 During reaching movements, motion capture data was recorded at 480 Hz and surface electromyography (EMG) was recorded at 2000 Hz using MA400-28 (MotionLab Systems); both datasets were synchronized offline. EMG of twelve muscles was recorded including the clavicular head of pectoralis (Pec), teres major (TerM), anterior deltoid (ADel), posterior deltoid (PDel), the long and lateral heads of triceps (TriLo and TriLa), the short and long heads of biceps (BiS and BiL), brachioradialis (Br), flexor carpi radialis (FCR), flexor carpi ulnaris (FCU), and extensor carpi radialis (ECR, Figure 1A). EMG data was analyzed in MATLAB (MathWorks Inc). The EMG data recorded from the Control group was reported by Korol and Gritsenko, 2025.
28
The kinematic and force data from the Stroke and Control groups was reported by Thomas et al 2021.
4
(A) Experimental Task Side View. Each Reaching Movement was Between the Central Target (Open Circle) and One of the Peripheral Targets (Outward and Inward Reaches, Green Circle Shows the Exemplar Reaching Direction), Target Locations Not to Scale. Muscle Activity was Recorded From Proximal Muscles (Purple Labels): Pec, TerM, ADel, and PDel, Biarticular Muscles (Black Labels): TriLo, TriLa, BiS, and BiL, and Distal Muscles (Orange Labels): Brd, FCR, FCU, and ECR, Abbreviations are as Described in Methods. Bright Red Muscles Support the Arm Against Gravity in This Task; Darker-Shaded Muscles Produce Force in the Direction of Gravity. (B) Examples of Filtered and Normalized Muscle Activity Profiles for Three Muscles, Abbreviated as in A, During Outward Reach to the Green Target in A. The Gray Shaded Area Shows the Control Variance, and Colors Show the Mean (Line) and Standard Error (Shaded Area) for Individual Participants From the Stroke Group. (C) Plots Show Examples of Tonic (Top Left) and Phasic (Bottom Right) Hemiparetic Score Differences (Color Lines) Relative to Control Group (Black Lines) per Reaching Direction. The Data Were Used to Calculate the Areas of the Illustrated Ellipses Termed Score Tunings. (D) Plots Show Tonic vs Phasic Hemiparetic Score Tunings Calculated From Data Shown in C per Individual With Stroke. The Rounded Rectangle Outlines Data for Subacute Participants. Squares Indicate Outward and Circles Indicate Inward Reaches; the Color Indicates Values Summed Across Proximal, Biarticular, or Distal Muscles. Lines Show Significant (P < 0.05) Linear Regressions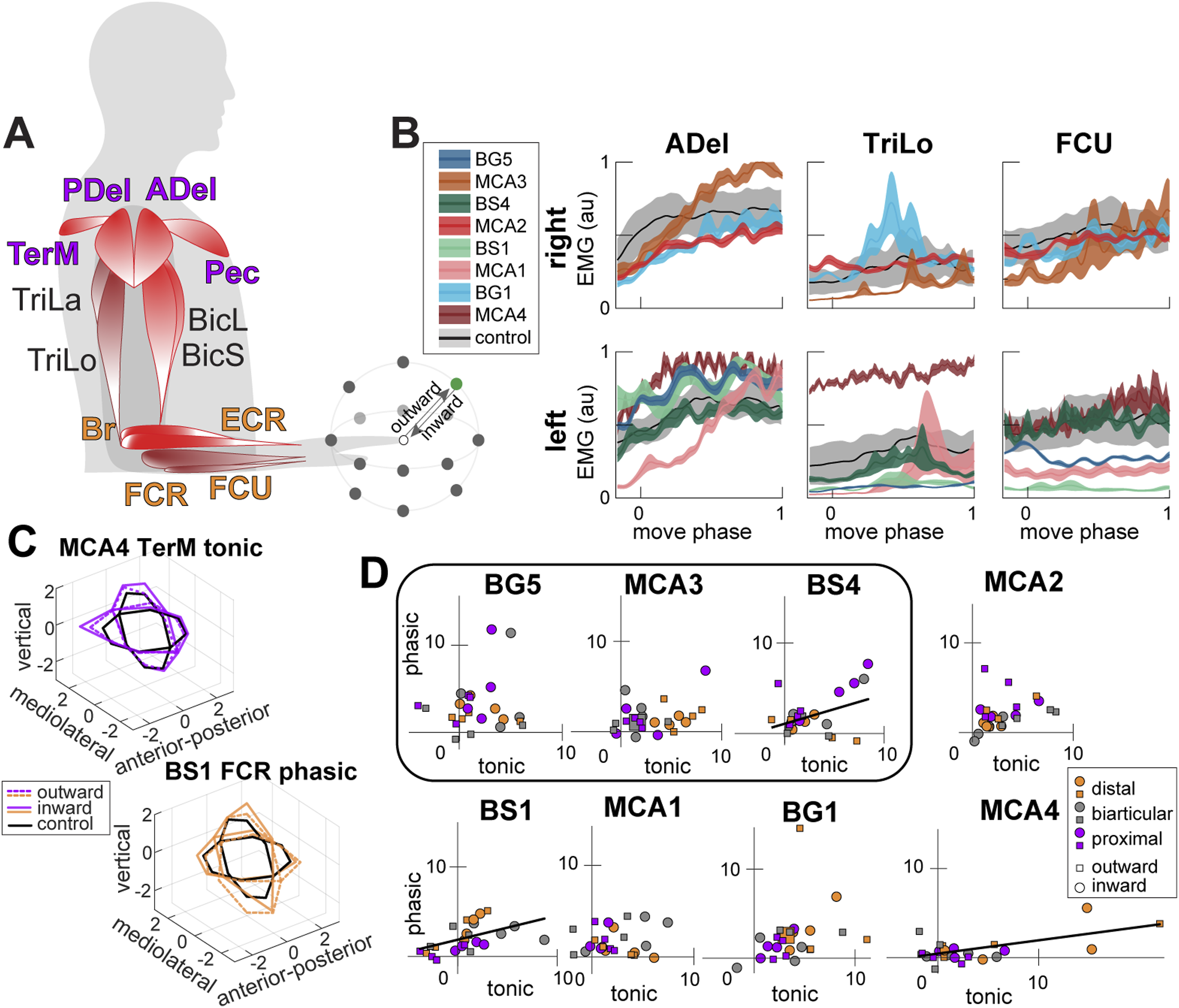
Theory
The rotational motion of a joint with a single degree of freedom (DOF), driven by muscle forces, is governed by:
Here, 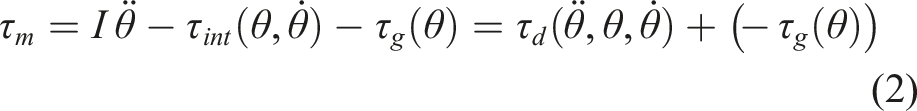
This decomposition separates the dynamic (
Muscle torque about the ith DOF is generated by the sum of forces from all M muscles crossing the joint, each scaled by their moment arm 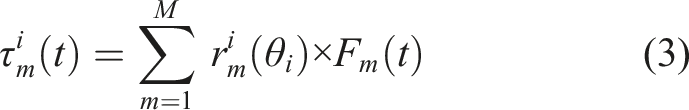
Here, the moment arms (
When neural commands embed compensation for gravity (

Thus, EMG-derived muscle recruitment can reflect biomechanical demands like gravity compensation and intersegmental coordination. We previously showed that tonic EMG aligns with
Data Analysis
EMG recordings were filtered (low-pass at 500 Hz, high-pass at 20 Hz, bandpass at 59-61 Hz), rectified, and further low-pass filtered at 10 Hz following SENIAM guidelines. EMG envelopes were normalized to movement duration, downsampled to 100 samples, and averaged across 15 repetitions per reaching direction for each muscle (total of 336 profiles). The sample size of 15 repetitions was derived empirically, it resulted in inter-trial variability in joint angle measurements of about 5°. 31 EMG amplitudes were scaled using maximum contraction values obtained across all reaching directions (Figure 1B). The inter-subject variability of muscle recruitment was calculated as the standard error of the mean of averaged normalized EMG profiles across subjects in a given group. All values in Results are reported as means ± standard deviation across subjects.
Principal component analysis (PCA) was used to separate tonic and phasic EMG components. Normalized, demeaned EMG profiles (12 muscles × 14 directions × inward/outward) were arranged into matrices (An × m; n = 100, m = 336). PCA assumptions for normality (Shapiro-Wilk test) and orthogonality (dot product calculation) were met, while homogeneity (Levene’s test) was not, indicating unequal variance across muscles and directions.
PCA utilized MATLAB’s singular value decomposition, extracting eigenvectors (principal components), variance accounted for (VAF), and scores. PCA was applied separately to data from Control and Stroke groups’ left and right arms (Control: left n = 9, right n = 9; Stroke: left paretic n = 5, right paretic n = 3, left non-paretic n = 3, right non-paretic n = 5). The PCA data is shared in a Figshare repository. 32
Previous studies established that the first eigenvector represents tonic (gravity-related) EMG activity, while the second eigenvector captures phasic (intersegmental dynamic-related) EMG activity, 13 both potentially altered by stroke. PCA scores quantify representation differences of EMG features across reaching directions and muscles. Due to minimal differences, 28 Control scores from left and right arms were combined to enhance normative variability estimate. Paretic scores were then adjusted by subtracting bilateral control scores, measuring stroke-related deficits. To control for the effects of age, paretic scores were also adjusted using the data from the Age-matched group and reported in supplementary figures.
Score differences across reaching directions indicate feature recruitment variability dictated by task demands. The elliptical areas defined by these score differences, relative to Control group, were calculated (Figure 1C), providing quantitative measures of muscle over- or under-recruitment in stroke participants for tonic and phasic components.
Statistics and Reproducibility
To determine the impact of stroke on the temporal evolution of the tonic and phasic biomechanical features in EMG, the first 2 eigenvectors and their VAF were compared between bilateral control data and the paretic and non-paretic data from Stroke group. We used the maximal coefficient of determination obtained from cross-correlation (crosscorr function in MATLAB Signal Processing Toolbox) between the mean control profile and the non-paretic or paretic individual profile for the 1st and 2nd eigenvector separately.
During reaching in certain directions, some muscles may have low tonic and phasic EMG, which would result in low PCA score values. Moreover, stroke can also cause reduced tonic and phasic EMG, which would also result in low scores. Here, we defined low scores as values below 5% of the maximal score. This threshold was informed by pilot testing and prior analyses in similar datasets,13,28 where a 5% cutoff effectively separated low-recruitment EMG signals from those with meaningful biomechanical representation in the principal components. To evaluate the effect of stroke on score amplitude, we performed 2 two-sample t-tests comparing low scores of Control and Stroke groups for tonic and phasic components separately. The familywise error adjusted alpha was equal to 0.0253 using Bonferroni-Sidak correction.
We hypothesized that (1) stroke affects the recruitment of distal muscles that move the wrist and hand more than proximal and biarticular muscles that move the shoulder and elbow; (2) stroke affects the phasic component of EMG more than the tonic component. To test these hypotheses, we fitted a non-parametric generalized linear mixed-effects (GLME) model (fitglme function in Statistics and Machine Learning Toolbox, MATLAB) with an identity link and normal error distribution to examine the effects of three continuous predictors—Component (Tonic vs Phasic), Direction (Outward vs Inward), and Muscle Group (Proximal, Biarticular, vs Distal)—on the sum of score tunings, along with their interactions. One-sample Kolmogorov-Smirnov test was performed on the GLME model residuals to check that the normality assumption is not violated (alpha = 0.05; P = 0.58). Random effects variance components were estimated for the intercept across subjects and for the residual error. The table below shows beta coefficients (β) that represent the estimated effects of predictors (fixed effects) on the outcome variable, adjusted for the random effects structure in the model and the associated standard error (SE), confidence intervals (CI), and P-values. The hypotheses will be rejected if the beta values are insignificant at alpha = 0.05.
To test if stroke affects both tonic and phasic EMG in individual participants, we regressed tonic vs phasic score tunings using linear regression analysis (regress function in MATLAB Statistics and Machine Learning Toolbox). We excluded outliers in a percentile threshold from 2.5% to 97.5% (isoutlier function in MATLAB Data Preprocessing Toolbox).
To evaluate how stroke deficits measured from joint moments in our earlier study are related to the EMG deficits, we regressed the score tunings for tonic and phasic components during outward and inward reaches against gravitational and dynamic torque performance index. We have also regressed the score tunings against the years since the stroke.
The hemiparetic score values change across reaching directions in proportion to how much a given muscle is over- or under-recruited for a given movement. To determine whether these abnormal recruitment patterns are similar across muscles, we regressed the hemiparetic score differences for 28 reaching directions between each muscle pair for tonic and phasic EMG components separately. We excluded outliers that is more than three median absolute deviations from the median (isoutlier function in MATLAB Data Preprocessing Toolbox). This was achieved with linear regression analysis for Control and paretic arm Stroke Groups with adjusted alpha = 0.0008 (Bonferroni-Sidak correction). Linear regression analysis was performed using regress function in MATLAB Statistics and Machine Learning Toolbox. Each matrix, C
n×m
, consisted of n = 28 reaching directions by m = 12 muscles per participant for tonic and phasic EMG components. Significant relationships were indicated by moderate and high coefficients of determination (R2) for each muscle pair. The resulting matrix of R2 values, represented as heatmaps in Figures 2 and 3, show how many muscles were over- or under-recruited together in each participant. We also preserved the sign of the regression slope to indicate whether the coupled recruitment represents co-contraction (positive sign) or antagonistic activation (negative sign) of the muscle pair. Tonic Coactivation. (A) Heatmaps Show Matrices of Significant Regressions Between Tonic Scores of Muscle Pairs Across Reaching Directions With the Left (Left Plot) and Right (Right Plot) Limb by a Young Control Participant (S5 From Korol et Al 2025) Who Showed Fewer Coactivations Between Muscles of Their Left Arm. The Colors Show the Values of the Coefficient of Determination (R2) for Regressions Between the Tonic Scores of Muscle Pairs Across Reaches in Multiple Directions. (B) Symbols Show the Number of Moderate and Strong (R2 > 0.5) Muscle Coactivations in Individuals With (Red) and Without (Black) Stroke. (C) Heatmaps of Tonic Muscle Coactivations for Participants With Stroke During Reaching With Their Paretic Arm. Colors are as in A Phasic Coactivation. (A) Heatmaps Show Matrices of Significant Regressions Between Phasic Scores of Muscle Pairs Across Reaching Directions With the Left (Left Plot) and Right (Right Plot) Limb by a Young Control Participant (S5 From Korol et Al 2025). The Colors Show the Value of the Coefficient of Determination (R2) for Regressions Between the Phasic Scores of Muscle Pairs Across Reaches in Multiple Directions. (B) Symbols Show the Number of Moderate and Strong (R2 > 0.5) Muscle Coactivations in Individuals With (Red) and Without (Black) Stroke. (C). Heatmaps of Phasic Muscle Coactivations for Participants With Stroke During Reaching With Their Paretic Arm. Colors are as in A

Sensitivity analysis: to evaluate the effect of differences between dominant and non-dominant motor control on the estimate of stroke deficits, the same analyses were applied to hemiparetic scores normalized to matched data from left- or -right arm of control participants. The results were the same.
Results
Variability in Muscle Activation Profiles
Participants exhibited consistent motion and joint moment profiles, as reported previously.4,13 In contrast, muscle activity profiles were more variable across repetitions of the same trials and across participants (Figure 1B). Our prior studies demonstrated that the primary features within these noisy EMG signals can be effectively captured by two principal components, one representing tonic activity associated with antigravity support, and the other representing phasic activity related to dynamic movement toward the target.13,28 To understand how muscle recruitment is altered after a stroke, we first compared the features extracted by PCA from the EMG data of the Control, Age-matched, and Stroke groups. Specifically, we examined whether the principal components (ie, eigenvectors) were similarly oriented across groups. We found that the temporal patterns of both primary features of the Stroke group closely resembled those in the Control and Age-matched groups (Supplemental Figure S1A) and accounted for a comparable proportion of variance in EMG (Supplemental Figure S1B). In the paretic limbs of stroke participants, the first (tonic) and second (phasic) components explained 53% ± 13% and 17% ± 5% of the variance, respectively. These values were similar to those observed in the Control group (52% ± 7% and 16% ± 5%), Age-matched group (55% ± 8% and 15% ± 5%), and in the non-paretic limbs of the Stroke group (54% ± 10% and 19% ± 7%). These findings suggest that the structure of EMG variability remains largely intact after stroke, with consistent patterns of variance across individuals. This aligns with clinical observations that EMG patterns in stroke survivors may appear qualitatively normal, even when substantial motor deficits are present.
Post-stroke Changes in Tonic and Phasic Components of Muscle Activity
Next, we analyzed the principal component scores, where the amplitude reflects how strongly a specific biomechanical feature — related to gravity or intersegmental dynamics — is expressed in a muscle’s EMG activity during movement. Low scores indicate that the feature accounts for little variance in the EMG signal for certain muscles and reaching directions, suggesting abnormal recruitment patterns in the hemiparetic arm after stroke. Our data show that, in control participants, 18% ± 7% (mean ± SD across participants) of tonic scores were low, or below 5% of the maximum, compared to 23% ± 9% for the paretic arm of participants with stroke. A two-sample t-test showed no significant difference between groups in the proportion of low tonic scores, t(24) = −1.31, P = 0.2013. However, the percentage of low tonic hemiparetic scores across individuals increased with time since stroke (Pearson correlation coefficient R = 0.85, P = 0.0068). Moreover, a significantly higher proportion of phasic hemiparetic scores were low (30% ± 6%) compared to Control group (24% ± 6%) (two-sample t-test, t(24) = 2.69, P = 0.0127). There was no linear relationship between the percentage of low phasic hemiparetic scores and time since stroke (R = 0.01, P = 0.9812). This suggests that stroke reduces both components of EMG activity, though to varying degrees depending on individual pathophysiology; the phasic component associated with intersegmental dynamics is most affected.
Results of the Non-parametric Generalized Linear Mixed-Effects Model

Lower bound adjustment for the main effect, bold values are significant. Based on a linear model with 96 observations. Both Muscle Group and Component were treated as continuous variables. β - beta coefficients representing the estimated effects of predictors on the hemiparetic score area.
SE, standard error; t, student’s t distribution value.
All P-values are two-tailed.
Temporal Progression of Muscle Recruitment Deficits
Deficits in tonic muscle recruitment varied with the time since stroke. However, the deficits in tonic recruitment of proximal muscles were markedly different for outward and inward movements (Figure 4A), indicating different neural control mechanisms for outward reaching and returning retrieval movements. In chronic stroke participants, tonic overactivation of hemiparetic biarticular muscles during outward reaches decreased as the number of years since stroke increased (R
2
= 0.90, P = 0.019; Figure 4A). This trend was especially evident in the short head of triceps (R
2
= 0.30, P = 0.027). Furthermore, the difference in tonic activation between proximal and distal muscles increased with time post-stroke (R
2
= 0.45, P = 0.019) with differences between outward and inward reaches diminishing (Figure 4B). This was driven by increasing tonic overactivation of proximal muscles and decreasing tonic activation of distal muscles, but each of these regressions with years since stroke were not significant (chronic proximal: R
2
= 0.16, P = 0.245; chronic distal: R
2
= 0.35, P = 0.070). In subacute participants, tonic muscle activation showed marked differences between outward and inward reaches. Outward reaches were dominated by proximal over-recruitment, with summed proximal muscle score tunings of 6.70, 11.56, and 25.09 and corresponding distal muscle differences of −2.4, 2.90, and 6.04 for subjects BG5, BS4, and MCA3, respectively. In contrast, inward reaches exhibited more variable deficits in proximal and distal muscle recruitment, with summed score tunings of 9.29, 6.69, and 20.72 for proximal muscles, and 9.59, 22.66, and 12.36 for distal muscles in the same subacute individuals. Similar patterns were observed when scores were calculated relative to the Age-matched group (Supplemental Figure S4). Taken together, the data support clinical observations that subacute stroke phase is distinct from the chronic phase. As more time passes after stroke, the abnormal tonic recruitment of proximal muscle tends to increase or stay the same while the abnormal tonic recruitment of biarticular and distal muscles tends to decline. Deficits in Muscle Recruitment. (A) Plot Shows Tonic Score Tunings for the Biarticular Muscles Plotted Against the Number of Years Post-stroke. Squares Indicate Outward and Circles Indicate Inward Reaches for Individual Participants; Color Coded as in Figure 1. The Dashed Line Shows a Significant (P < 0.05) Linear Regression for the Outward Reaches by Chronic Participants. (B) Plot Shows Tonic Score Tunings Calculated Between Proximal and Distal Muscles Plotted Against the Number of Years Post-stroke, Styled as in A. The Line Shows a Significant Linear Regression Across all Reaching Directions by Chronic Participants. (C) Plot Shows Phasic Score Tunings for the Distal Muscles Plotted Against the Number of Years Post-stroke, Styled as in A. The Line Shows a Significant Linear Regression Across all Reaching Directions and Participants. (D) Plots Show Phasic Score Tunings Calculated Between Proximal and Distal (Left Plot) and Biarticular and Distal (Right Plot) Muscles Plotted Against the Number of Years Post-stroke, Styled as in A. Lines Show Significant Linear Regressions Across all Reaching Directions and Participants. (E) Plot Shows Phasic Hemiparetic Score Tunings for the Biarticular Muscles Plotted Against the Dynamic Performance Index, Styled as in A The Dashed Line Shows a Significant Linear Regression for the Outward Reaches by Chronic Participants. (F) Plot Shows Phasic Hemiparetic Score Tunings Calculated Between Biarticular and Distal Muscles Plotted Against the Dynamic Performance Index, Styled as in A. The Line Shows a Significant Linear Regression Across all Reaching Directions and Participants
Deficits in phasic muscle recruitment also varied with the time since stroke. The phasic over-activation of distal muscles decreased across all participants at different times since stroke (R 2 = 0.52, P = 0.002; Figure 4C; Supplemental Figure S4C). These trends were present in flexor carpi radialis (R 2 = 0.40, P = 0.009) and flexor carpi ulnaris (R 2 = 0.36, P = 0.015). Moreover, the difference in phasic over-activation between proximal and distal muscles (R 2 = 0.44, P = 0.005) and between biarticular and distal muscles (R 2 = 0.46, P = 0.004) also increased with time post-stroke (Figure 4D; Supplemental Figure S4D). These trends were primarily driven by the progressive decrease in abornimal phasic recruitment of distal muscles in individuals further removed from the time of stroke onset, consistent with the results shown in Figure 4C and Supplemental Figure S4C. Moreover, the differences between proximal and distal muscles in phasic and tonic activation were correlated across all participants (R 2 = 0.38, P = 0.011), indicating a similar underlying neural mechanism of decreasing abnormal tonic and phasic recruitment of distal muscles. Overall, these results suggest that adaptation over time since stroke is most evident in the reduced activation of distal muscles compare to the activation of proximal and biarticular muscles, especially in the phasic component of muscle recruitment.
Post-stroke Altered Muscle Recruitment Underlies Motor Deficits
Stroke-induced motor deficits during reaching are reflected in muscle torques, which represent the forces required to move the limb. These torques can be further decomposed into components corresponding to the forces necessary for arm support against gravity and those required to propel the limb during reaching. Given that muscle torques arise from the combined actions of all muscle groups, our EMG analysis enabled quantification of the contributions of specific muscle groups to post-stroke motor impairments. We have shown in an earlier study that the performance index derived from the dynamic component of muscle torques, which is similar to the phasic component of EMG, 13 is a sensitive indicator of the severity of post-stroke motor deficits. 4 Here too we have found that the severity of motor deficits measured with the performance index derived from muscle torques was linearly related to the number of years post-stroke (gravitational performance index vs years: R 2 = 0.27, P = 0.039; dynamic performance index vs years: R 2 = 0.32, P = 0.023). Moreover, the phasic over-recruitment of biarticular muscles during outward reaches exhibited a strong linear relationship with the summed performance index derived from the dynamic component of shoulder and elbow muscle torques (R 2 = 0.55, P = 0.036; Figure 4E; Supplemental Figure S4E). This association was particularly notable in the short head of the biceps (R 2 = 0.28, P = 0.034) and showed an inverse relationship in the flexor carpi radialis (R 2 = 0.29, P = 0.031) across both outward and inward reaches. Furthermore, phasic score differences between biarticular and distal muscles were linearly related to the dynamic performance index (R 2 = 0.27, P = 0.038; Figure 4F; Supplemental Figure S4F). Collectively, these findings show that stroke-related alterations in muscle recruitment directly impair the ability to generate propulsive forces and coordinate multi-joint movement during reaching.
The results above show that post-stroke muscle activation deficits affect entire muscle groups rather than individual muscles. To explore this further, we examined muscle coactivation—that is, the simultaneous recruitment of multiple muscles to perform a given task. The coactivation between antagonistic muscles also contributes to joint or limb stiffness, an important parameter controlled by the central nervous system. In previous work, we showed that in control participants, tonic activity in biarticular and distal muscles, especially in the left arm, was broadly correlated across reaching directions, reflecting their coactivation to support the arm against gravity. 28 However, there was substantial variability across individuals, with two control participants showing few co-activating muscle pairs in the left arm. After stroke, the number of tonically co-activating muscle pairs in left-paretic participants was near the lower end of the control range, whereas in right-paretic participants, it was near the upper end (Figure 2). In contrast, control participants showed minimal phasic coactivation in both arms 28 (Figure 3A), and this pattern remained largely unchanged after stroke, with few phasically co-activating muscle pairs observed (Figure 3B and C). Notably, we found a significant inverse linear relationship between the number of phasically co-activating proximal and distal muscles and the dynamic performance index (R 2 = 0.67, P = 0.013), indicating that greater impairments in generating propulsive forces are associated with more abnormal coactivation between proximal and distal muscles.
Discussion
Our results show that stroke disrupts muscle recruitment during reaching, even among individuals with mild impairments. Distal muscle impairment severity increases over time post-stroke, but proximal muscles are also abnormally over-recruited during movements that usually require less activation, indicating disrupted directional tuning. Additionally, paretic muscle coactivation between proximal and distal muscles is abnormal. These findings reject the hypothesis that stroke primarily impacts distal muscles more severely than proximal or biarticular muscles, as both groups are variably affected.
Our results further reject the hypothesis that stroke disproportionately affects the phasic component of EMG compared to the tonic component. Instead, deficits in phasic and tonic recruitment correlate linearly in some participants, and phasic and tonic differences between proximal and distal recruitment correlate across participants and align with overall reaching impairments. This suggests that stroke prompts widespread muscle over-activation, particularly for supporting the limb against gravity, resulting in inefficient movements and potential fatigue.
These findings are consistent with the idea that stroke-induced motor impairments reflect differential damage to descending motor pathways that are reflected in the abnormal reruitment of muscles. The musculoskeletal anatomy organizes muscles into proximal and distal groups. 33 This organization is reflected at the spinal 34 and cortical levels. 35 The persistent overactivation of proximal muscles, particularly in tonic EMG components associated with gravity compensation, suggests increased reliance on the reticulospinal system—a phylogenetically older pathway involved in posture, proximal control, and muscle tone regulation.20,25,26 Conversely, the reduced phasic activation of distal muscles and impaired intersegmental coordination are likely consequences of corticospinal tract dysfunction, which is essential for dynamic, fractionated, and goal-directed control of distal musculature.22-24,36 This interpretation is supported by evidence that the corticospinal and reticulospinal systems are anatomically coupled yet functionally distinct, with the latter often compensating for lost corticospinal input following stroke.14,17-19,37,38 By separating tonic and phasic muscle activity, our study provides a physiological window into the functional status of these parallel motor pathways, offering a potential biomarker framework for pathway-specific deficits that could inform targeted rehabilitation strategies.
Identifying underlying causes of movement inefficiency and fatigue remains challenging, but assessing mechanical impedance provides a quantitative framework. 39 Impedance, comprising stiffness and viscosity, increases during unstable tasks or when precision is required.40-42 For stroke, impedance can be synonymous with spasticity, which is defined as a velocity-dependent increase in muscle tone. 5 Increased muscle tone in both agonistic and antagonistic muscles are observable as increased co-contraction that would increase both stiffness and viscosity of the joint DOFs these muscle span. Our results show that after stroke, lateralization of impedance control is disrupted: right-hemisphere strokes reduce consistent muscle coactivations typical of non-dominant hemisphere-driven gravity compensation, 28 while left-hemisphere strokes increase coactivation. Greater deficits in generating propulsive forces also correlate with abnormal coactivation patterns, underscoring that stroke-induced disruption in impedance control significantly contributes to motor inefficiency and fatigue. Quantifying such disruptions may help pinpoint their underlying neural causes and guide more targeted rehabilitation strategies.
Limitations
A primary limitation of this study is the relatively small sample size, particularly within the Stroke and Age-matched groups, which may limit statistical power and the ability to control for age and detect nuanced patterns across stroke subtypes. Power analysis (MATLAB function sampsizepwr) indicates that to detect the mean change of 0.478 ± 1.027 SD in post-stroke recruitment between muscle groups (Table 1) with 80% power, the study needs 30 participants with stroke. Future studies with this sample sizes will reveal the details of how the plasticity in the two pathways affect muscle recruitment after stroke.
Another limitation of this study is that we did not directly measure spasticity or its directional characteristics using standardized clinical scales such as the Modified Ashworth Scale or instrumented assessments of reflex excitability. Instead, we inferred aspects of abnormal muscle tone and limb stiffness through patterns of co-contraction observed in the tonic and, to a lesser degree, phasic EMG components. While elevated co-contraction reflect abnormal muscle recruitment, this indirect approach cannot quantify the resulting increase in limb or joint stiffness or viscosity. Future studies incorporating clinical assessments of spasticity or direct measurements of mechanical impedance are needed to better interpret the relationship between co-contraction and spasticity-related motor deficits after stroke.
Conclusion
This study demonstrates that stroke alters the pattern of muscle recruitment during reaching, resulting in distinct but interacting impairments in tonic and phasic EMG components. These impairments reflect disrupted control of antigravity support and intersegmental dynamics, likely due to differential damage to reticulospinal and corticospinal pathways. We observed over-recruitment of proximal muscles and under-recruitment of distal muscles, along with abnormal coactivation patterns that were hemisphere-specific and predictive of functional impairment. Importantly, both tonic and phasic deficits evolved with time since stroke, suggesting that neural compensation and maladaptation may reshape motor output over the course of recovery. These findings highlight the importance of assessing muscle recruitment patterns, rather than joint kinematics alone, to better identify residual deficits and guide targeted rehabilitation. Future work should validate these neuromechanical biomarkers in larger cohorts and investigate whether personalized interventions informed by tonic and phasic muscle recruitment profiles can improve functional outcomes after stroke.
Supplemental Material
Supplemental material - Stroke-Related Changes in Tonic and Phasic Muscle Recruitment During Reaching Reveal Pathway-Specific Motor Deficits
Supplemental material for Stroke-Related Changes in Tonic and Phasic Muscle Recruitment During Reaching Reveal Pathway-Specific Motor Deficits by Anna S. Korol, Amelia Adcock, and Valeriya Gritsenko in Journal of Central Nervous System Disease
Footnotes
Acknowledgements
The authors gratefully acknowledge the generous and inspiring contributions of the volunteers who participated in this study. Their willingness to share their time, effort, and experiences made this research possible. Thanks to their commitment, we are one step closer to unraveling the complexities of neural control of movement and advancing future medical breakthroughs in neurorehabilitation.
Ethical Considerations
The study was approved by West Virginia University’s Institutional Review Board (Protocol #1311129283) on the 4th of December, 2014. Participants were recruited from Morgantown, WV area between 2014 and 2016.
Consent to Participate
All participants provided written informed consent prior to enrolment in the study. This research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Author Contributions
ASK was responsible for data curation, analysis, and drafting of the manuscript. AA contributed to the conceptualization of the study, participant recruitment, data interpretation, and manuscript preparation. VG oversaw study conceptualization, participant recruitment, data curation and analysis, data interpretation, and contributed substantially to manuscript writing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: V.G. was supported by National Institute of General Medical Science grants No. P20GM109098 and P30GM103503. A.S.K. was supported by a fellowship from National Institute of General Medical Sciences grant No. T32AG052375. This work was supported in part by the Office of the Assistant Secretary of Defense for Health Affairs through the Congressionally Directed Medical Research Programs under Award No. W81XWH-21-1-0138. Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the Department of Defense. This material is based upon work supported by the Air Force Office of Scientific Research under award number FA9550-24-1-0208. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Neurowired provided financial support for travel of VG and ASK to scientific conferences.
Declaration of Conflicting Interests
The author declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VG is a Founder of Neurowired, a company that develops biomedical technology.
Data Availability Statement
The data is available on Figshare 32 .
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
