Abstract
Background
Cladribine, a selective immune reconstitution therapy, is approved for the treatment of adult patients with highly active multiple sclerosis (MS).
Objectives
Provide experience with cladribine therapy in a real-world setting.
Methods
This is a registry-based retrospective observational cohort study. First, using data from the Czech nationwide registry ReMuS, we analysed patients who initiated cladribine from September 1, 2018 to December 31, 2021. Second, we analysed a subgroup of patients who initiated cladribine between September 1, 2018 to June 30, 2020, thus possessing a follow-up period of at least 2 years. We evaluated demographic and MS characteristics including disease-modifying therapies (DMTs) before and after cladribine administration, relapses, Expanded Disability Status Scale (EDSS), and adherence.
Results
In total, 617 patients (335 with follow-up of at least 2 years) started cladribine therapy in the study period (mean age 37.0, mean disease duration 8.4 years, 74.1% females). In most cases, cladribine was administered as a second-line drug, a total of 80.7% had been escalated from a platform DMT. During 2 years before cladribine initiation, the average annualised relapse rate (ARR) was .67. Following cladribine initiation, the ARR decreased to .28 in the first year and .22 in the second year. Overall, across the entire two-year treatment period, 69.0% of patients were relapse-free and the average ARR was .25. As for EDSS development, the median baseline EDSS was 2.5 and remained stable even after 24 months. The adherence to treatment ranged of around 90%.
Conclusion
This nationwide study confirms the efficacy of cladribine in real-world settings, especially in patients who are not treatment-naïve. In addition, the study shows an exceptionally high adherence rate, a finding that underscores the invaluable role of cladribine, but also the value of registry-based studies in capturing real-world clinical practice.
Keywords
Introduction
Cladribine, a purine nucleoside analogue initially utilised for intravenous treatment of hairy cell leukaemia, has become a pivotal therapeutic option for multiple sclerosis (MS). The oral formulation of cladribine has been approved since 2017 for managing highly active relapsing-remitting multiple sclerosis (RRMS) in the European Union, 1 with an expanded indication for an active secondary progressive form of the disease in the United States. 2 This treatment’s long-term efficacy and safety have been substantiated in notable studies such as CLARITY, CLARITY Extension, ORACLE MS, and within the PREMIERE registry.3-6 In the Czech Republic, since November 2018, cladribine has been reimbursed as an escalation treatment for patients diagnosed with RRMS. The place of oral cladribine within therapeutic protocols is heavily influenced by reimbursement conditions. In the period of this study, cladribine was reimbursed in patients with RRMS with signs of an unfavourable prognosis of their disease, experiencing at least one moderate to severe relapse despite treatment with at least one first-line disease-modifying therapy (DMT). Oral cladribine has also found utility in lateral switching from other high-efficacy DMTs (HE-DMTs) in suboptimal tolerability, safety, or efficacy. 7
One of the most salient characteristics of cladribine is its administration protocol, which consists of repeated pulses that produce a persistent long-term effect, making it a valuable selective immune reconstitution therapy (SIRT). SIRT has a reduction or depletion phase, followed by a repopulation phase and then by a reconstitution phase, during which the immune system recovers normal effector function. Oral cladribine results in the peripheral depletion of lymphocytes that is gradual, occurring over several weeks, has a greater impact on B cells than T cells, and is followed by gradual reconstitution of the peripheral lymphocyte counts over several months. However, the reconstituted immune system is both qualitatively and quantitatively different to that before treatment, which explains sustained efficacy and/or prolonged remission. 8 The cumulative dose is 3.5 mg/kg of body weight, delivered in 2 yearly pulses, each involving 2 treatment weeks. 9 Long-term clinical efficacy is expected after full dosing. However, despite these well-defined protocols, there is a dearth of long-term data on how to proceed in subsequent years, particularly regarding which patients may require additional pulses and how to identify non-responders. This paper aims to bridge this knowledge gap by presenting an analysis of clinical experiences with cladribine therapy derived from the ReMuS, the Czech national registry of patients with MS. We investigate adherence to this treatment and evaluate its therapeutic efficacy in a subgroup of patients who have undergone at least 2 years of follow-up. This real-world evidence can enhance understanding of cladribine’s role in managing RRMS and improve future therapeutic strategies.
Methods
Data Collection, Standard Protocol Approvals and Patient Consents
Data for this retrospective observational cohort study was acquired from the ReMuS registry as of June 30, 2022. Established in 2013, ReMuS aggregates data from all 15 MS centres in the Czech Republic, monitoring virtually all patients receiving DMTs. As of the specified date, a total of 19,805 patients with MS were included in the ReMuS registry. The registry systematically collects and analyses various parameters: demographic, MS-related, treatment-related, socio-economic, and MRI examinations. However, MRI activity data was not considered. Data collection utilises standardised software iMed, and each MS centre exports its data biannually. 10 The data then undergoes a multi-level quality control process (over 100 pre-programmed checks). Quality reports highlighting suspicious, invalid, or missing information are then circulated to the respective centres. 11
Population of Interest
This study delineates 2 patient groups. Group A includes patients who initiated cladribine therapy from September 1, 2018 to December 31, 2021. Group B, a subgroup of group A, consists of patients who started their cladribine therapy between September 1, 2018 to June 30 2020, thus possessing a longer guaranteed follow-up period of at least 2 years.
Variables assessed
The study monitored a comprehensive set of variables. Beyond the basic demographic factors of sex and age, various clinical parameters were assessed. These included the history of DMTs, the progression of disability as tracked by the expanded disability status scale (EDSS), the occurrence and severity of relapses, the annualised relapse rate (ARR), and treatment adherence.
DMT use was categorised as follows: no DMT, if the patient was DMT-naïve before oral cladribine; platform DMT (P-DMT), including beta-interferons, glatiramer acetate, teriflunomide, and dimethyl fumarate; and HE-DMT, including monoclonal antibodies, modulators of sphingosine-phosphate receptors, and cladribine. The duration of DMT before initiating oral cladribine, the number of DMTs used before cladribine, and reasons for discontinuing the DMT preceding cladribine were also monitored. Moreover, we tracked patients who switched from cladribine to other DMTs during the observation period, noting the specific DMTs. Considering different mechanism of action of the analysed DMTs, the following rules were applied to calculate the time: (1) short-term treatment interruptions up to 90 days are not taken into account and are thus part of the time on treatment; (2) DMTs ocrelizumab and rituximab are considered active for 2 years after the last infusion or until another DMT is administered (whichever comes first); (3) alemtuzumab is considered active from the first administration until any switch of the DMT.
The number of relapses was evaluated 12 and 24 months prior to the initiation of cladribine and subsequently in 2 12-month periods as well as a cumulative 24-month period after the first dose, including the percentage of their severity determined according to the Czech reimbursement criteria. A mild relapse was defined as an EDSS score increase by .5 point, or an increase by 1 point in 1-3 functional systems (except the bowel, bladder, and cerebral/mental). A moderate relapse was defined as an EDSS score increase by 1-2 or increase by 2 in 1 or 2 EDSS functional systems or increase by 1 in 4 and more EDSS functional systems. A severe relapse was defined by exceeding the moderate relapse criteria.
The ARR was calculated at the individual patient level, using the number of relapses during each respective follow-up period and the length of that period in years. To evaluate the ARR before the initiation of cladribine, a 24-month period was selected. This extended timeframe was chosen to mitigate potential biases associated with increased disease activity immediately before escalating therapy, providing a more comprehensive overview due to the ineffectiveness of previous treatments. During cladribine treatment, the full therapeutic effect may not be immediately observable, and residual disease activity from prior treatments could still be present. Therefore, and given the limited duration of available follow-up data, we analysed the ARRs at 2 key intervals: the initial 12 months and the subsequent 12 to 24 months post-treatment initiation. This approach was chosen to illustrate the emerging effectiveness of cladribine over time. To provide a more comprehensive view, we also included an aggregated ARR for the entire two-year period following the initiation of therapy. In addition, for comparability with the CLARITY study, 3 we also evaluated ARR at 96 weeks after cladribine initiation. It is crucial to note that our analysis is descriptive and does not include statistical comparisons between individual intervals.
The EDSS was evaluated at multiple points in the treatment timeline, including at baseline and at 6, 12, 18, and 24 months post-initiation of cladribine. The EDSS scores were considered valid only if taken at least 30 days after a relapse, with relapse-related scores not included in the analysis.
Finally, the analysis focuses on the adherence to the treatment administration and its timing regarding Summary of Product Characteristics, where intended weeks of cladribine administration are the 5th, 53rd and 57th week from cladribine treatment initiation. Adherence was assessed only in those treatment weeks where sufficient follow-up of a patient was available.
Statistical Analysis
Analysis was performed with R Statistical Software (v4.2.2; R Core Team 2022) and relevant packages. Nominal variables were summarized using absolute and relative frequencies. For numerical variables, means, standard deviations, and median, minimum and maximum, where appropriate, were calculated. The ARR and its 95% confidence intervals (95% CI) were calculated using the method approximating the Poisson distribution to a normal distribution.
Results
Baseline Patient Characteristics
This study evaluated data from 617 patients (group A) who initiated cladribine therapy during the specified study period from September 1, 2018 till December 31, 2021. Among this population, a subset of 335 patients (group B) began their treatment between September 1, 2018, and June 30, 2020, providing a follow-up period of at least 2 years. Group A comprised 74.1% females. The average age in this group was 37.0 years. The mean disease duration at the start of cladribine therapy was approximately 8.4 years (median 6.74 years), and the mean baseline EDSS 2.9 (median 2.5). The characteristics in group B were relatively similar. The mean duration of the disease was slightly lower at 8.2 years (median 6.6 years), but the mean and median baseline EDSS at the initiation of cladribine therapy mirrored those in group A, standing at 2.9 and 2.5, respectively.
During the 24 months prior to cladribine initiation, data was available for 522 patients from group A. In these patients, 698 relapses were reported, culminating in an average ARR of .67 (95% CI [.62, .72]). A majority (62.8%) of relapses were of moderate intensity. A total of 33.9% of these patients experienced at least 2 relapses within the 24-month window before beginning cladribine treatment. In group B, complete data from 289 patients was available for analysis. The average ARR in this group was also .70 (95% CI [.63, .77]), maintaining parity with group A. Here too, relapses were predominantly moderate in severity (62.9%), indicating a consistent pattern between the 2 groups.
Baseline Characteristics Before Cladribine Initialization.

DMT = disease-modifying therapy, P-DMT = platform disease-modifying therapy, HE-DMT = high-efficacy disease-modifying therapy, EDSS = Expanded disability status scale, ARR = annualized relapse rate.
aNormal approximation of the Poisson distribution.
Previous Disease-Modifying Therapy
Regarding group A, the administration of cladribine was preceded mainly by treatment with one DMT (54.6%, n = 337; more in Table 1). Analysis of the DMT given immediately before cladribine indicated that most patients (80.7%, n = 498) had been escalated from a P-DMT, some (15.7%, n = 97) had a lateral switch, and a few (3.6%, n = 22) were treatment-naïve. The latter were either participants in cladribine clinical trials or had previously received another study drug, including placebos. Interferon beta was the most common preceding therapy (n = 222), being the only active substance in 205 of these patients (92.3%). Glatiramer acetate and fingolimod followed, with the former being the second most common preceding therapy (n = 140) and the latter being the most used HE-DMT (n = 65) (Figure 1). The mean duration of all previous DMTs before cladribine was 57.7 months for P-DMT and 6.7 months for HE-DMT. The discontinuation of the previous DMT was primarily due to a lack of efficacy (69.7%) or poor tolerance (10.3%). No reason was provided in 12.4% of patients. In group B, the administration of cladribine was preceded by treatment with one DMT in 186 patients (55.5%), two in 84 (35.1%), and more than two in 46 patients (13.7 %). Cladribine was primarily administered as the second DMT, less often as the third, with a mean of 1.7 previous DMTs (median 1) in both groups. The maximum number of DMTs preceding cladribine was 6. Disease modifying therapy prior to oral cladribine, group A (n = 617) HE-DMT = high-efficacy disease-modifying therapy, P-DMT = platform disease-modifying therapy, IFN = interferons, GA = glatiramer acetate, DMF = dimethyl fumarate.
Disease Characteristics During Cladribine Treatment
Relapses From the Initiation of Cladribine Therapy (the Data was Exported as of 30 June 2022).
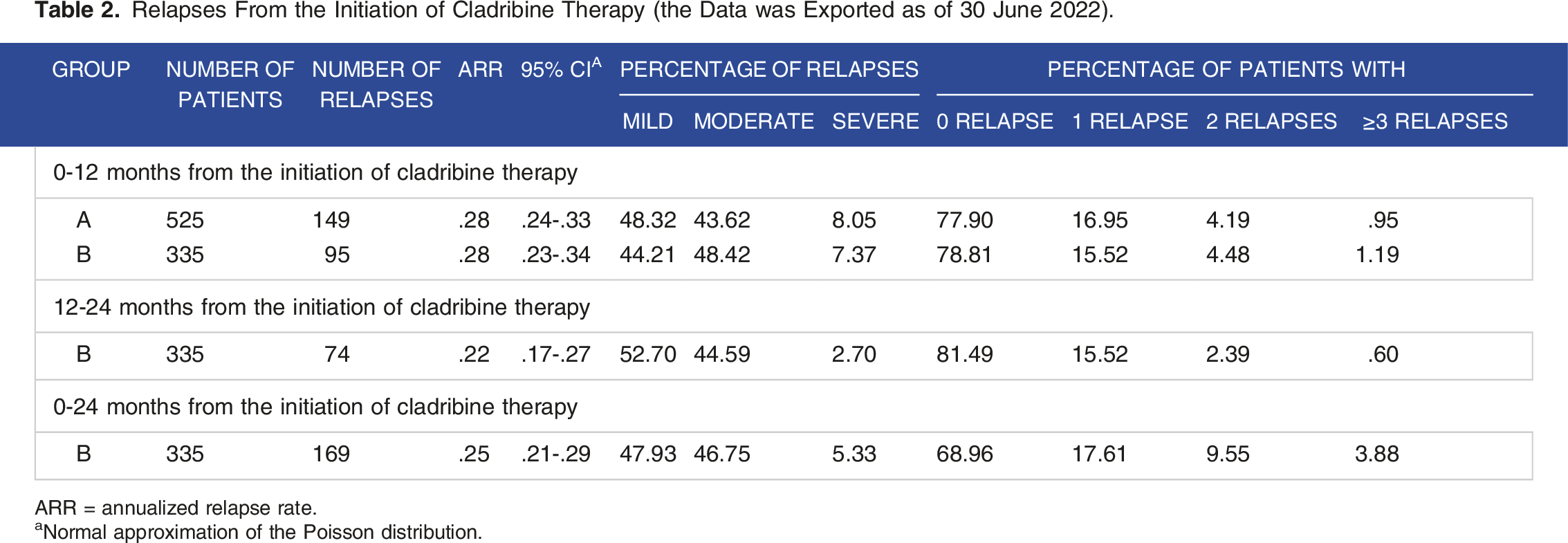
ARR = annualized relapse rate.
aNormal approximation of the Poisson distribution.
Progression of Expanded Disability Status Scale During Oral Cladribine Treatment in Group B.

EDSS = Expanded disability status scale.
Description in Patients Who Switched From Cladribine to Other Disease-Modifying Therapy.

DMT = disease-modifying therapy, EDSS = Expanded disability status scale.
Adherence to Oral Cladribine by Scheduled Treatment Weeks.
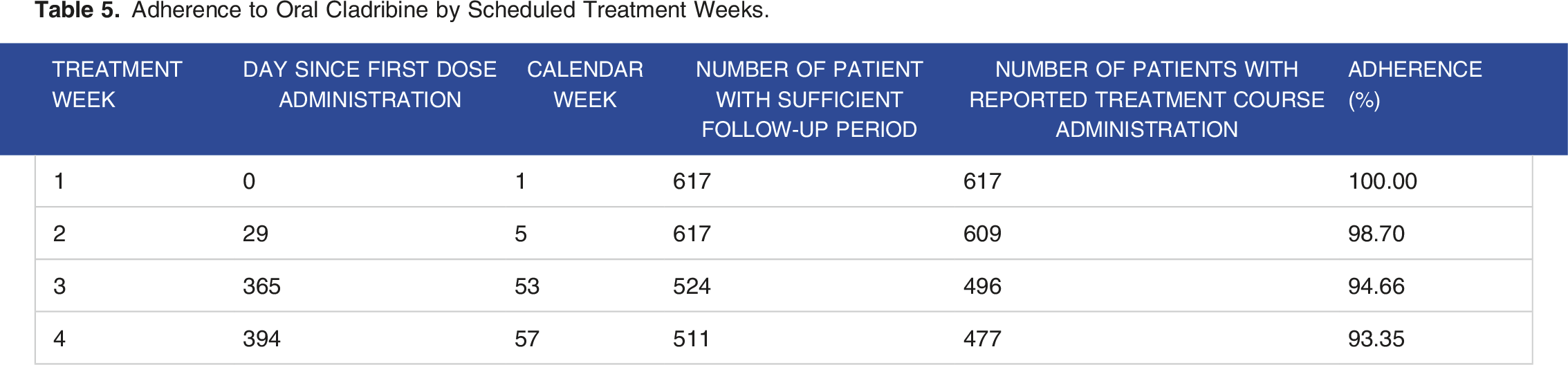

Adherence plot.
Discussion
This comprehensive view from the national ReMuS registry offers crucial insights into the use of cladribine in real-world settings where reimbursement criteria, among other factors, need to be taken into account. These limited the availability of cladribine as a first-line treatment in the Czech Republic during the study period, so our patient cohort was not treatment-naïve, with only a few exceptions (3.6%). In most cases, cladribine was administered as a second-line drug, a total of 80.7% had been escalated from a P-DMT. During 2 years before cladribine initiation, the average ARR was .67. Following cladribine initiation, the ARR decreased to .28 in the first year and .22 in the second year. Overall, across the entire two-year treatment period, 69.0% of patients were relapse-free and the average ARR was .25. As for EDSS development, the median baseline EDSS was 2.5 and remained stable even after 24 months. Another important aspect of this study is the evaluation of treatment adherence, which appears to be excellent in real-world practice. Specifically, 92.9% of patients received the second course within a one-week range of the prescribed date, and the recommended second course of treatment scheduled for week 5 was adhered to by 88.2% of patients in week 5 or 6.
When contrasted with the patient demographic in the CLARITY registration study, our analysis finds a similar average age (37.0 vs 37.9 years) and median baseline EDSS score (mean 2.9 vs 2.8, median 3.0 vs 2.5). Notably, our group had a slightly longer disease duration (8.4 vs 7.9 years), and considerably more patients had undergone at least one prior DMT treatment (96.4% vs 26.1%). Also, in contrast to our study, the CLARITY study did not involve patients pre-treated with HE-DMTs. These differences can be part of an explanation of a higher ARR value (.24) after 96 weeks from initiation of cladribine therapy observed in our set compared to .14 in the CLARITY study. Moreover, ReMuS records all relapses, whereas CLARITY only considered qualifying relapses. 3 As for EDSS progression, our data is comparable to those from the CLARITY and CLARITY extension studies. In those studies, an improvement in their median EDSS was observed from a baseline value of 3.0 to 2.5 after 5 years of therapy initiation.3,12
When comparing our results with another real-world study of 286 patients monitored in the Danish MS registry, we observed that the ARR a year before cladribine initiation was .67 and that the reduction of ARR after cladribine initiation was over 80% in both studies. However, this ARR reduction was slightly higher in the Danish group (ARR of .11 in two years after cladribine initiation). There are probably 2 reasons for this. First, in the Danish analysis, the researchers re-baselined the reference timeframe for ARR evaluation to 3 months later in order to eliminate relapses caused by the inefficacy of previous treatment or rebound effect and to avoid evaluating a period when the cladribine effect was still incomplete. Second, our patients had higher pre-treatment rates compared to the Danish study, in which 12.7% of patients were treatment-naïve. Despite a higher ARR following cladribine initiation in our cohort, only 5.5% (.3% caused by side effects) switched to another therapy, compared to the 11.2% (2.6% due to side effects) in the Danish study. 13 However, at least 7% of patients in our study met the criteria for residual activity (5.1% had at least two relapses, 2.1% had a severe relapse) within 12 months of the first administration of cladribine and, as non-responders, should have been considered for conversion to another HE-DMT.14,15 And last but not least, not only our study but also data from the Danish, Finnish and also Italian real-world studies confirm high EDSS stability and suggest a higher efficacy of cladribine in treatment-naïve patients.13,16,17 It is also worth mentioning that an analysis of Czech and Swedish RRMS cohorts confirmed a better prognosis for patients in Sweden, where a higher proportion of patients received HE-DMT as initial treatment (42%). 18 Fortunately, with changes in reimbursement criteria in the Czech Republic, the proportion of patients initiating HE-DMTs has increased from 2.1% in 2013 to 18.5% in 2021. 11 The benefits of cladribine use in real-world practice are also supported by a small cohort study of 90 patients from the Australian MSBase registry. In an Australian study, approximately 80% of patients were EDSS progression-free and 65% remained relapse-free after 2 years after cladribine initiation. 19
When evaluating the effectiveness of therapy, we must not forget about adherence as a key condition for the functioning of any treatment. Patient registries reflecting the conditions of real-world practice, not the artificially created controlled conditions of clinical trials, are a key source for these data. In 2009, World Health Organisation (WHO) found that only 50% of patients with chronic disease were adherent to their medications. 20 Adherence to the DMTs in MS varies widely between 41% and 93%,21,22 and patients with good adherence to DMTs have a decreased risk of relapse, a lower frequency of hospital visits, and an increased quality of life compared to non-adherent patients.23-26 One of the many factors playing a role in adherence is therapy-specific attributes. 27 In patients with MS, an adherence rate for the injectable DMTs ranging from 41 to 88% 21 was reported and around 80% for a once- or twice-daily dose of oral medications. 28 The adherence rate of around 90% is therefore unique in competition with other DMTs and is a great advantage of cladribine. This is probably mainly due to the very convenient administration method and the short administration time. This information is all the more valuable because it comes from real-world data, not from the artefactual setting of clinical trials.
However, our study has some limitations, mainly the absence of MRI data as an objective indicator of disease activity. Another limitation is the lack of targeted search for adverse events and the relatively short follow-up, which is undoubtedly needed in further studies to confirm long-term efficacy and safety. Given the relatively small number of non-responders, we also did not perform an analysis of this subgroup. To uncover the characteristics of patients who do not have a sufficient response to cladribine therapy, more extensive data and further studies are needed.
Conclusion
This nationwide study of patients treated with cladribine provides valuable insights from clinical practice where conditions differ from those in registration studies. Our study confirms the efficacy of cladribine in a real-world setting where most patients are not naïve to treatment. Finally, our study shows extremely high adherence rates, a finding that underscores the invaluable role of cladribine, but also the value of registration studies in capturing real-world clinical practice.
Ethical Statement
Ethics Approval
ReMuS was established by the Endowment Fund IMPULS and approved by local ethics committees in each MS centre (Faculty of Medicine and University Hospital in Pilsen, Charles University: 314/23; First Faculty of Medicine, Charles University in Prague and General University Hospital, Prague: 1701/12 S-IV; Faculty of Medicine and University Hospital Hradec Kralove, Charles University in Prague: 201308 S25P; KZ a.s., Hospital Teplice: II/13/6; Tomas Bata Hospital, Zlin: 3/16; 3rd Faculty of Medicine, Charles University in Prague and Hospital Kralovske Vinohrady, Prague: EK-VP/14/2013; Second Faculty of Medicine and Motol University Hospital, Charles University, Prague: EK-79/13; Faculty of Medicine, Palacky, University and University Hospital Olomouc: 81/13; University Hospital and Masaryk University Brno: 6/13; Thomayer Hospital, Prague: MEK – 150a/13; Hospital Pardubice: 12/13; Hospital Ceske Budejovice: 3/13; University Hospital Ostrava and Medical Faculty, Ostrava University: 238/2013; Hospital of Jihlava: 2/13; University Hospital U Svate Anny and Masaryk University Brno: 11JS/2016). ReMuS is based on informed consent, thus it is possible to use retrospective data for scientific and research purposes without requiring new approvals.
Informed Consent
An informed consent was obtained from all patients enrolled in ReMuS.
Footnotes
Acknowledgements
The authors are very grateful to all employees of MS centres participating in the data collection. Without their hard work and dedication, this study would never have been possible.
Author Contributions
Pavel Potuznik: Conceptualisation; Data curation; Investigation; Resources; Writing – original draft; Writing – review & editing; Funding acquisition. Jiri Drahota: Methodology; Software; Validation; Formal analysis; Data curation; Writing – original draft Writing – review & editing. Dana Horakova: Conceptualisation; Investigation; Resources; Writing – original draft; Writing – review & editing, Supervision. Marek Peterka: Investigation; Resources; Writing – review & editing. Aneta Mazouchova: Methodology; Software; Validation; Formal analysis; Data curation; Writing – original draft; Writing – review & editing. David Matyas: Investigation; Resources; Writing – review & editing. Zbysek Pavelek: Investigation; Resources; Writing – review & editing. Marta Vachova: Investigation; Resources; Writing – review & editing. Eva Recmanova: Investigation; Resources; Writing – review & editing. Ivana Stetkarova: Investigation; Resources; Writing – review & editing. Jana Libertinova: Investigation; Resources; Writing – review & editing. Jan Mares: Investigation; Resources; Writing – review & editing. Pavel Stourac: Investigation; Resources; Writing – review & editing. Marketa Grunermelova: Investigation; Resources; Writing – review & editing. Alena Martinkova: Investigation; Resources; Writing – review & editing. Jana Adamkova: Investigation; Resources; Writing – review & editing. Pavel Hradilek: Investigation; Resources; Writing – review & editing. Radek Ampapa: Investigation; Resources; Writing – review & editing. Michal Dufek: Investigation; Resources; Writing – review & editing. Eva Kubala Havrdova: Investigation, Resources. Dominika Stastna: Conceptualization; Investigation; Resources; Writing – original draft; Writing – review & editing.
Declaration of Conflicting Interests:
The authors declared potential conflicts of interest with respect to the research, authorship and/or publication of this article as following: PP received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme. JD has nothing to disclose. DH received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. MP received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. AM has nothing to disclose. DM has nothing to disclose. ZP received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. MV received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. ER received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. IS received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. JL received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. JM received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. PS received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. MG received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. AM received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. JA received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. PH received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. RA received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. MD received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. EKH received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag. DS received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen Cilag.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the General University Hospital in Prague project [grant number MH CZ-DRO-VFN64165], the Charles University: Cooperatio Program in Neuroscience, a Czech Ministry of Health project [grant number NU22-04-00193], a Czech Ministry of Health project - Conceptual Development of Research Organization - [Faculty Hospital in Pilsen - FNPl, 00669806] and by a National Institute for Neurological Research project funded by the European Union (Next Generation EU, Programme EXCELES, ID Project No. LX22NPO5107).
Data availability statement
Anonymised data not published within this article will be made available by request from any qualified investigator.
