Abstract
Background
SARS-CoV-2 infection affects multiple systems, including musculoskeletal, neurological, and respiratory systems. Changes associated with physical inactivity due to prolonged hospitalization can affect the functional capacity of individuals with long coronavirus disease 2019 (COVID-19) or post-COVID-19 condition and may cause changes in some postural control functions, such as verticality.
Objectives
This study aimed to evaluate the perception of verticality in individuals with long COVID.
Design
Cross-sectional study
Methods
This study included 60 participants with post-COVID-19 condition divided into 2 groups: hospitalized group (n = 24), those hospitalized owing to SARS-CoV-2 infection; and non-hospitalized group (n = 36), those infected with SARS-CoV-2 but not hospitalized. All participants were examined using a post-COVID-19 functional status (PCFS), sit-to-stand test, grip strength assessment, painful and tactile sensory assessments, visual acuity assessment, and vestibular assessment. Verticality perception was evaluated using the subjective visual vertical (SVV) and subjective haptic vertical (SHV) tests. In both tests, the absolute values (positive values only) and true values (positive and negative values) were considered. To verify potential confounders that could influence the verticality of the results, logistic regression models were used for categorical variables and multiple linear regressions were used for continuous variables. For analysis between groups, the independent samples test (Mann–Whitney U test) was used.
Results
There were no confounders between clinical variables and verticality in either group. There was a significant increase in absolute SVV (mean deviation [MD]: 2.83; P < .0001) and true SVV (MD: −4.18; P = .005) in the hospitalized group compared to the non-hospitalized group. Furthermore, there was a significant increase in the true SHV (MD: −3.6; P = .026) in the hospitalized group compared to that in the non-hospitalized group.
Conclusion
Less accurate visual and haptic verticality perception task performance was observed in hospitalized patients with post-COVID-19 condition.
Introduction
Corona virus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), can progress to SARS, resulting in physical and functional complications in adults and elderly, following the acute phase.1,2 COVID-19 symptoms range from respiratory 3 to neurological manifestations4-6 and have been studied increasingly. Furthermore, individuals with COVID-19 may develop secondary and chronic conditions 7 that may persist for weeks or months,8-10 which is referred to as long COVID. 11
Owing to the multisystem impact of COVID-19, individuals who have the most severe form of the disease and are hospitalized may develop chronic cognitive, 12 sensory, 13 and motor14-16 impairments that can affect both short- and long-term postural control. 17 One of the determining components of postural control is postural orientation and perception of verticality. 18 The perception of verticality represents the notion of vertical and horizontal spatial orientation based on internal models and the integration of the visual, vestibular, and somatosensory systems.18,19 In addition, postural control can be influenced by body stabilization mechanisms, which are dependent on the integration of cognitive, sensory, and motor systems. 20 All of these mechanisms can be affected by COVID-19 and long periods of hospitalization. 21
Based on the aforementioned context, hospitalized patients with post-COVID-19 condition may present with changes in postural control owing to primary sensory, motor, or cognitive alterations. De Sousa et al. (2022) 22 found that patients with post-COVID-19 condition (long COVID) had worse postural control performance than healthy individuals; however, no long-term analysis of the perception of verticality has been conducted in these individuals. Verticality perception may be an important marker for understanding postural control and determining targeted treatments for individuals with post-COVID-19 condition. Therefore, this study aimed to analyze the perception of verticality in individuals with post-COVID-19 condition. The main hypothesis of this study is that hospitalized patients with post-COVID-19 condition may have less accurate verticality perception task performance compared to non-hospitalized patients with post-COVID-19 condition.
Methods
This study followed the Strengthening the Reporting of Observational Studies in Epidemiology guidelines (STROBE) (https://www.strobe-statement.org/checklists/).
Study Design and Setting
This cross-sectional study included 2 cohorts (hospitalized and non-hospitalized, ie, those without a history of hospitalization) of patients with post-COVID-19 condition. Data were collected from 3 centers: Fundação Hospital, Dr Moisés Magalhães Freire, Pirapora, MG; FisioVida Physiotherapy Clinic, Pirapora, MG; and São Cristovão Regional Health Unit, Uberaba, MG, from January 2021 to December 2022.
This study was approved by the Research Ethics Committee of the Federal University of Triângulo Mineiro (UFTM; approval code CAAE:30684820.4.0000.5154). All participants provided oral and written informed consent. This study was conducted in accordance with the tenets of the Declaration of Helsinki.
Participants
Study participants were recruited using convenience sampling. Inclusion criteria included individuals aged over 18 years with post-COVID condition that were either hospitalized or not and those that had the ability to complete the test independently. Post-COVID-19 condition (long COVID) was defined as “the continuation or development of new symptoms consistent with COVID-19 infection for more than 12 weeks, which could not be explained by an alternative diagnosis.” 11 The diagnosis of post-COVID-19 was based on medical evaluation, evaluation of current symptoms, and complementary examinations. Hospitalized individuals must have remained admitted to the hospital for more than 48 hours. 23 The exclusion criteria were as follows: patients with previous neurological or musculoskeletal alterations, those that RT-PCR positivity for SARS-CoV-2 during study period, those that had uncontrolled systemic arterial hypertension, and those that had visual and vestibular alterations before the period of acute infection as determined from face-to-face interviews.
Data Collection
The participants were divided into 2 groups: hospitalized patients with post-COVID-19 (hospitalized group) and non-hospitalized patients with post-COVID-19 (non-hospitalized group). They were assessed using the sit-to-stand test,24-26 grip strength,27-30 painful and tactile sensory assessment, 31 visual acuity, 32 vestibular assessment,33-35 verticality perception by subjective visual vertical (SVV),36-39 and subjective haptic vertical (SHV) tests.38-40 All clinical tests were performed by an authorized and trained researcher.
Data Sources and Measurement
Patient Demographics
Participants’ demographic data (age, sex, and education), clinical data (comorbidities and actual symptoms), vaccination regimens for COVID-19, and information on the treatment received were collected.
Post-COVID-19 Functional Status (PCFS) Scale
This scale was used to assess the level of impairment in the functional status of patients with COVID-19. The PCFS stratification comprises 5 grades: grade 0 (no functional limitations), grade 1 (negligible functional limitations), grade 2 (slight functional limitations), grade 3 (moderate functional limitations), and grade 4 (severe functional limitations). 24
Five Times Sit-to-Stand Test (5XSST)
The 5XSST assesses strength, transitional movements, balance, and fall risk by documenting the time required for a person to come to a complete stand from a sitting position, 5 times. The patients sat with their arms folded across their chest and with their backs against a standard-height chair (43–45 cm). The following instruction was given: “I want you to stand up and sit down 5 times as quickly as you can when I say ‘go’”. A practical trial was performed before the measurements. The 5XSST has demonstrated good reliability and validity.25,26
Grip Strength Evaluation
To assess grip strength, the manual grip force was measured using a manual analog dynamometer (Crown AT, Oswaldo Filizola, São Paulo/SP, Brasil). Each individual was positioned in a chair without support, with the hips and knees flexed at 90° and the feet resting on the floor. The upper limb to be evaluated was positioned with the shoulder in the adducted position, the elbow flexed to 90°, the forearm in the neutral position between 0° and 30° of extension, and the wrist at 0°-15° of adduction. The untested limb was placed on the ipsilateral thigh. The participant was asked to grip the hand with maximum force for 3 seconds, and a rest interval of 30 seconds was provided between the tests; the average values from the 3 tests were calculated.27,28 Handgrip strength was evaluated using the dominant hand as defined by the Edinburgh Inventory. 29 Test-retest reliability has been reported to be very good, and the concurrent validity of the functional tests to be high. 30
Painful and Tactile Sensory Assessment
Pain sensitivity was assessed using non-perforating, round-tipped, pointed fibers at the distal and proximal ends of the upper and lower limbs. Tactile sensitivity was assessed by touching the same region with cotton. Any abnormal sensation (hyperesthesia, hypoesthesia, or dysesthesia) in the evaluated regions compared to the face (neutral region) was considered to be “altered” in the test. 31
Visual Acuity
Visual acuity was assessed in a standardized manner using a Snellen chart placed 6 m away from the individual, who was then asked to read the letters arranged on the chart. One eye at a time was evaluated, and the eye was characterized as having normal visual acuity if the individual could see letters arranged at level 8 (20/20). 32
Head Impulse Test, Nystagmus, Test of Skew (HINTS)
Head Impulse test (HIT) is based on performing horizontal head movements, and it is expected that the eye movement is at the same speed but in the opposite position to the head movement. 33 To assess nystagmus (N), the patient was instructed to focus their gaze on the examiner’s finger as it moved slowly from left to right. 34 In the Test of Skew (TS), the individual was instructed to look forward, and the evaluator covered and uncovered each eye with a patch, which was considered abnormal (positive) if there was vertical deviation (ocular misalignment). 34 HINTS examination has demonstrated a high degree of accuracy in the diagnosis of central vertigo, with a sensitivity and specificity of 100%. 35
Verticality Tests
SVV
The SVV test was performed using a bucket test consisting of an opaque plastic bucket with an opening larger than 25 cm and a red tape positioned in a vertical line at the bottom of the bucket, which was aligned at 0° using a digital inclinometer (Clinometer App, Tue Nguyen Minh). The inclinometer was positioned outside the bucket and both were aligned with the true vertical of the Earth. The participants were instructed to place their faces in the bucket and look at the red tape. To measure the SVV, the examiner randomly rotated the bucket (avoiding any tactile cues to the patient) in the right or left direction to a final position of approximately 30° and then slowly rotated it toward the 0° position. The participants were asked to indicate when the red line reached a vertical position by asking the examiner to stop moving the bucket at that point. The participants performed 6 repetitions of this procedure: 3 toward the right and 3 toward the left. The mean perceived vertical position in the right and left directions was used as the final value.36,37 The SVV was classified as absolute or true based on the deviations from the gravitational vertical. Only the absolute values of SVV measurements without positive or negative arithmetic signs were considered for the absolute SVV. Additionally, the true SVV considers both positive and negative SVV values.36,38,39
SHV
To measure SHV, we used a device comprising a box (diameter 18.5 × 18.5 × 30 cm) and a circular rod (15 cm long and 2.5 cm thick) based on Schuler et al. (2010). 40 The rod was mounted at the center of the box. 36 The height of the rod was adjusted according to the height of the participant so that the center of the rod was approximately at the elbow level. The participants were blindfolded with a mask and allowed to reach the rod in a manner that they felt most comfortable. Moreover, the lights were turned off during testing. In each trial, the examiner randomly rotated the rod 30° to the right or left. The SHV adjustments were recorded in 6 trials, including 3 directed toward the right and 3 toward the left. The participants were then asked to confirm that they had finished adjusting the position of the rod such that it was perfectly aligned with the vertical orientation of the target. Furthermore, we assessed the accuracy of the SHV by calculating the difference between the final rod position (in degrees) and the true vertical. 40 SHV evaluation was performed using the dominant hand as defined by the Edinburgh inventory. 29 SHV was classified as absolute or true, based on the deviations of the rod related to the vertical line (0°). Only the absolute values of the SHV measurements without arithmetic signs were considered for absolute SHV. However, the true SHV considers both positive and negative SHV values.36,38,39
Bias
Selection bias was minimized by attempting to contact those who could not be contacted during the survey and by visiting the residences of those who did not possess a personal phone. To minimize recall bias, all information provided by the patient was verified using a trusted third-party and medical records.
Statistical Analysis
Data distributions were assessed for assumptions of normality using the Shapiro–Wilk test. For sample characterization and data distribution, descriptive statistics, means, and standard deviations were used for parametric variables and percentages were used for categorical variables. To verify potential confounders that could influence the verticality of the results, logistic regression models were used for categorical variables, and multiple linear regressions were used for continuous variables. For analysis between groups, the independent samples test (Mann–Whitney U test) was used. Statistical significance was set at P ≤ .05. Data were analyzed using GraphPad Prism version 8.0.0 for Macintosh (GraphPad Software, San Diego, California, USA, www.graphpad.com).
Results
Clinical and Demographic Data of Both Groups.

PCFS, Post-COVID-19 functional status.
HG, hospitalized group; NHG, non-hospitalized group; HINTS, head impulse nystagmus test of skew; *Stroke in the posterior circulation.
aMann–Whitney test
bX2 test
cFisher’s exact test
Nystagmus was observed with the naked eye in 12 patients, including 10 patients in the hospitalized group (41.7%). In addition, only 2 patients in the hospitalized group (8.3%) had an abnormal HIT. Eight patients (33.3%) had right nystagmus with a normal HIT. There was a greater presence of dizziness (P = .004), nystagmus to the right during the HINTS (P < .01), and greater palmar grip in the hospitalized group than in the non-hospitalized group (P = .04). In addition, there was an increase in the level of impairment in the functional status (PCFS) of the hospitalized group compared with that of the non-hospitalized group (P = .003).
Regression Models to Identify Potential Confounders in the Analysis of Verticality in the Non-Hospitalized Group.

Absolute SVV: Goodness of Fit R2 = .58; and F change = 2.435.
True SVV: Goodness of Fit R2 = .61; and F change = 2.747.
Absolute SHV: Goodness of Fit R2 = .59; and F change = 2.202.
True SHV: Goodness of Fit R2 = .64; and F change = 2.809.
*Persistent dizziness was not included in the model because it was linearly dependent on the HINTS.
aMultiple linear regression
bmultiple logistic regression models.
Regression models to identify potential confounders in the analysis of verticality in the hospitalized group.
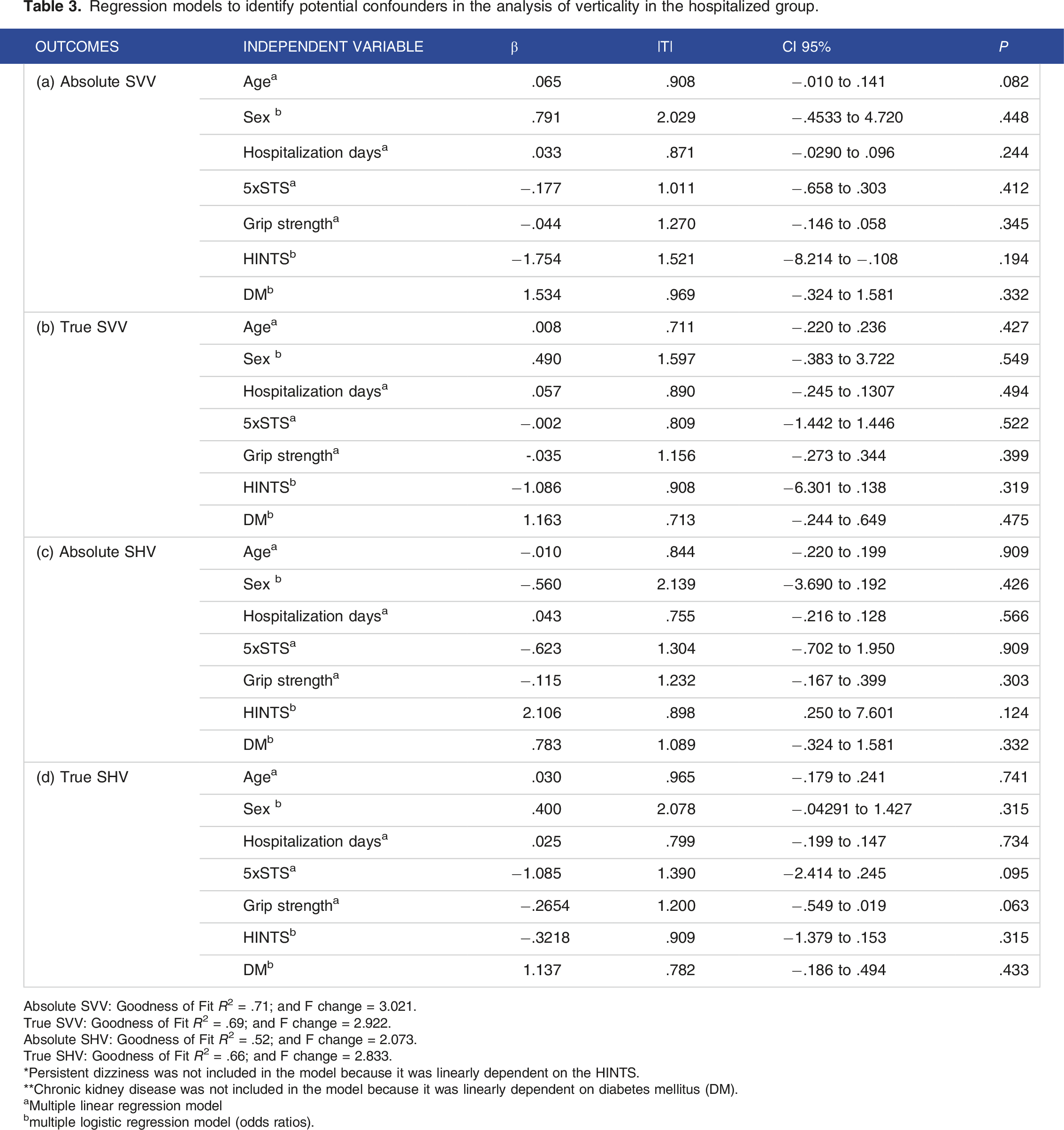
Absolute SVV: Goodness of Fit R2 = .71; and F change = 3.021.
True SVV: Goodness of Fit R2 = .69; and F change = 2.922.
Absolute SHV: Goodness of Fit R2 = .52; and F change = 2.073.
True SHV: Goodness of Fit R2 = .66; and F change = 2.833.
*Persistent dizziness was not included in the model because it was linearly dependent on the HINTS.
**Chronic kidney disease was not included in the model because it was linearly dependent on diabetes mellitus (DM).
aMultiple linear regression model
bmultiple logistic regression model (odds ratios).
In the visual verticality perception analysis, a significant increase in absolute SVV deviations from the gravitational vertical (MD: 2.83; P < .0001) and true SVV deviations from the gravitational vertical (MD: −4.18; P = .005) was observed in the hospitalized group compared with that in the non-hospitalized group (Figure 1). In the visual haptic perception analysis, a significant increase in true SHV deviations from the vertical line (MD: −3.6; P = .026) was observed in the hospitalized group compared with that in the non-hospitalized group. There were no significant differences in the absolute SHV deviations from the vertical line between the groups (MD = .67; P = .385) (Figure 2). Absolute and true SVV in hospitalized and non-hospitalized patients with long COVID. Abbreviations: COVID, coronavirus disease 2019; NH, Non-hospitalized; H, hospitalized; SVV, subjective visual vertical. Absolute and True SHV in hospitalized and non-hospitalized patients with long COVID. Abbreviations: COVID, coronavirus disease 2019; NH, Non-hospitalized; H, hospitalized; SHV, subjective haptic vertical.

Figure 3 shows the SVV and SHV of both groups and the minimum and maximum values of the true vertical. Hospitalized group was observed to have greater variability in subjective visual and haptic values than the non-hospitalized group. True SVV and SHV (minimum and maximum values) of hospitalized and non-hospitalized patients with long COVID. Abbreviations: COVID, coronavirus disease 2019; NH, Non-hospitalized (dark gray); H, hospitalized (light gray); SVV, subjective visual vertical; SHV, subjective haptic vertical.
Discussion
This study demonstrated that individuals with post-COVID-19 condition that were hospitalized showed less accurate performance for SVV and SHV than non-hospitalized individuals. Studies have evaluated the perception of verticality in individuals with acquired 36 and degenerative 31 diseases of the central nervous system (CNS). However, no previous studies have examined this variable in individuals with long-term COVID. Possible explanations for these findings include changes in the central and peripheral nervous systems, in addition to deficits in postural control due to long-term motor, sensory, or cognitive changes.18,41
Giardini et al. (2022) 14 observed poor dynamic balance and increased sway during quiet stance in patients with severe disease in the acute phase of COVID‐19. Guzik et al. (2022) 22 observed that COVID-19 may lead to balance impairment in young adults. The findings revealed significant differences in postural stability, mainly with eyes closed, between individuals who recovered from COVID-19 and healthy controls. Similarly, Gervasoni et al. (2022) 15 reported that the hospitalized group in their study had worse postural control on an unstable platform with eyes closed than the non-hospitalized group. Both studies16,22 showed greater balance impairments in trials with eyes closed, suggesting an alteration in the interaction between the vestibular and proprioceptive pathways. This finding may explain the changes in the visual and haptic verticality observed in our study. In addition, hospitalized individuals with long-term COVID may have more pronounced chronic systemic symptoms and myopathic changes. 22 These alterations may be a pathological substrate for the worsening of postural control, justifying the misinterpretation of verticality perception.
Another factor that may explain the results of this research is the neurotropism process; this is because the COVID-19 virus binds to an angiotensin-2 converting enzyme (ACE2) to infect human cells, causing the virus to deposit genetic material in human cells in various regions of the central nervous system, causing acute and long-term degenerative brain changes.42-45 In addition, more severely ill patients undergoing hospitalization have a greater storm of pro-inflammatory cytokines in the acute phase, with an increased risk of developing a neurodegenerative condition.46,47 Neurodegenerative processes can alter long-term verticality, 31 however, the underlying mechanisms are not yet well established. Day et al. (2021) 47 demonstrated an altered perception of verticality in individuals with neurodegenerative conditions, suggesting that these individuals may have abnormal graviceptive information perception due to the reduced activity of somatosensory cortical areas.
It can also be inferred that changes in verticality observed in this study may have been caused by changes in the peripheral nervous system. Several authors have identified that changes in balance and symptoms of dizziness in individuals with COVID-19 can occur through a change in the function of the vestibular system innervated by the vestibulocochlear nerve, which is more susceptible to hypoxia-mediated vascular ischemia in this region, causing alterations in balance,22,37,48-50 which may affect their verticality. These alterations may be physiological substrates for alterations in individuals’ perceptions of verticality, as primary alterations in balance can lead to abnormal sensory judgments in the visual and proprioceptive systems in the long term. However, the absolute SHV deviations were similar between the groups. The absolute measures of verticality indicated a deviation from the center; however, the magnitude of this deviation was observed in the true measures, which were considered negative and positive deviations. Because hospitalized patients presented deviations with high variability, true verticality values can be used in clinical practice, as they provide accurate information on performance in each test.
In our regression model, no potential confounders were identified between clinical variables and verticality in either group. Sociodemographic factors such as age and sex did not influence the perception of verticality in our study. The patients included in our study were young adults; Silva et al. (2022) 51 recently showed that adults, independent of sex, had better verticality perceptions and precision in visual and proprioceptive verticalities than children and older adults. It was also observed that strength did not influence the perception of verticality, reinforcing the idea of its involvement in sensory pathways.16,22 Although HINTS did not influence the perception of verticality in the regression model, nystagmus was present in 41.7% of hospitalized patients, and 8 patients (33.3%) had nystagmus with normal HIT. According to the HINTS battery, nystagmus combined with a normal HIT suggests the presence of a central structural lesion.33-35 According to hospital records and magnetic resonance imaging, of the 8 patients with a suspected central lesion, 3 had uremic encephalopathy, 3 had a history of migraine, and 2 had a minor stroke in the territory of the posterior circulation. Acute vertigo was observed in 2 patients with nystagmus and abnormal HIT, and 8 patients with nystagmus and normal HIT had persistent symptoms of dizziness. Viola et al. (2020) 52 showed that 18.4% of individuals had balance disorders after COVID-19, 94.1% reported dizziness, and 5.9% reported acute vertigo attacks. Pazdro-Zastawny et al. (2022) 53 observed that patients diagnosed with COVID-19 were more likely to suffer from vertigo/dizziness and to recover more slowly. Finally, chronic conditions such as diabetes mellitus had a higher incidence in the hospitalized group. Patients with diabetes and poor postural control could have alterations in the vestibular-visual interaction, 54 however our findings did not show an influence on verticality tests.
SVV deviation was associated with COVID-19 severity (for both hospitalized and non-hospitalized individuals); however, the regression model showed no association with hospitalization duration (longer hospitalization resulted in greater SVV deviation). In our sample, we included patients with moderate impairment (PFCS 2-3) who had hospital stays similar to those with severe impairment (PFCS 4). In addition, we did not assess the quality of hospital care (amount of mobility training, number of times the patient left the bed with a nurse or other health professional, dose of rehabilitation, or medications administered). These confounding variables may have positively interfered so that patients, even those with a longer hospital stay, did not experience worsening of the perception of verticality.
Limitations and Directions for Further Research
This study has certain limitations. First, the small sample size may have limited the power of our results. Second, visual or vestibular alterations were not evaluated using objective tests, such as fundoscopy, campimetry, videonystagmography, or objective analysis of postural control. Furthermore, almost 30% of the individuals had chronic diseases (ie, diabetes mellitus and chronic kidney disease). Although the statistical model did not show an association between diabetes mellitus and verticality, these results should be considered in future studies with more representative samples. It is necessary to replicate this study with a larger sample encompassing more age groups, including older individuals, children, and adolescents. Furthermore, the participants were followed up only once; however, the results suggest the need for a follow-up study to investigate the long-term effects of COVID-19 on verticality perception.
However, the findings of the present study may provide important information regarding the aspects inherent in postural control that can be modified by systemic SARS-CoV-2 infection. Moreover, these findings may support the functional status of patients with long COVID and provoke interest in conducting future research on this topic in specific populations. Changes in visual and haptic vertical perception in individuals with long COVID can indicate a deterioration in postural control. Therefore, individuals with post-COVID-19, complaining of imbalance, should be systematically evaluated to determine the best therapeutic strategy to alleviate the postural problems.
Conclusions
This study observed a worsening of visual and haptic verticality perception in hospitalized individuals with post-COVID condition compared with those who were not hospitalized. Our results demonstrate the importance of verticality evaluation in patients with post-COVID condition, mainly to trace visual or haptic postural control alignment strategies to improve verticality perception, minimize the risk of falls, and help prevent adverse health-related effects. Further research should consider more age groups with long-term follow-up.
Footnotes
Author contributions
Bruno Fonseca: Conceptualization, Investigation, Methodology, Writing – original draft, Pedro de Andrade: Investigation, Methodology, Writing – original draft, Maria Eduarda Henrique: Investigation, Methodology, Jussara Baggio: Conceptualization, Methodology, Supervision, Writing – review & editing, Rodrigo Bazan: Conceptualization, Supervision, Writing – review & editing, Luciane de Souza: Conceptualization, Investigation, Methodology, Project administration, Writing – original draft, Writing – review & editing, Gustavo Luvizutto: Conceptualization, Formal analysis, Formal analysis, Methodology, Project administration, Writing – original draft, Writing – review & editing
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants were received from the National Council for Scientific and Technological Development (CNPq), the Ministry of Science, Technology, Innovations, and Communications (MCTIC), and CNPq (Process 401192/2020-2).
Ethics Approval:
This study was approved by the Research Ethics Committee of the Federal University of Triângulo Mineiro (UFTM) (CAAE:30684820.4.0000.5154).
Consent to Participate:
Participants signed an informed consent form to participate in the study.
Data Availability Statement
The data given this article are Data supporting the findings of this study are available from the corresponding author, GJL, upon reasonable request. All data used to write up this manuscript is available for review upon a reasonable request.
