Abstract
Coronavirus disease 2019 (COVID-19) has raised serious concerns worldwide due to its great impact on human health and forced scientists racing to find effective therapies to control the infection and a vaccine for the virus. To this end, intense research efforts have focused on understanding the viral biology of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), responsible for COVID-19. The ever-expanding list of cases, reporting clinical neurological complications in COVID-19 patients, strongly suggests the possibility of the virus invading the nervous system. The pathophysiological processes responsible for the neurological impact of COVID-19 are not fully understood. Some neurodegenerative disorders sometimes take more than a decade to manifest, so the long-term pathophysiological outcomes of SARS-CoV-2 neurotropism should be regarded as a challenge for researchers in this field. There is no documentation on the long-term impact of SARS-CoV-2 on the human central nervous system (CNS). Most of the data relating to neurological damage during SARS-CoV-2 infection have yet to be established experimentally. The purpose of this review is to describe the knowledge gained, from experimental models, to date, on the mechanisms of neuronal invasion and the effects produced by infection. The hope is that, once the processes are understood, therapies can be implemented to limit the damage produced. Long-term monitoring and the use of appropriate and effective therapies could reduce the severity of symptoms and improve quality of life of the most severely affected patients, with a special focus on those have required hospital care and assisted respiration.
Introduction
The World Health Organization (WHO) reported in December 2019 an outbreak of the atypical acute respiratory disease in central China’s Hubei province. 1
The disease, called “novel coronavirus disease” (COVID-19), 2 rapidly turned into a pandemic, with threateningly high infection rates, far exceeding those of SARS (Severe Acute Respiratory Syndrome) and MERS (Middle Eastern Respiratory Syndrome) in 2002 and 2012, respectively. 3
The new coronavirus named SARS-CoV-2 (Severe Acute Respiratory Syndrome Coronavirus type 2), previously named 2019-nCoV, belongs to the same family of the virus as SARS-CoV-1, the etiological agent of SARS.
Severe Acute Respiratory Syndrome Coronavirus type 2 is a new strain of coronavirus that has not previously been identified in humans. It’s transmitted by its natural hosts, the bats. 1
There are differences in the estimated COVID-19 case/fatality ratio. It is clear that severity is stratified by: (i) age group, with older individuals at higher risk of COVID-19 (except for a few new variants which seem to affect young people 4 ), (ii) pre-existing comorbidity such as high blood pressure, heart disease, diabetes, chronic respiratory disease, cancer and immunosuppressed patients (due to congenital or acquired disease, transplanted, or in treatment with immunosuppressive drugs) are more likely to develop severe forms of the disease. Furthermore, (iii) men in these groups also appear to be at a slightly higher risk than women, and (iv) living in the community is also a predisposing factor to infection and disease severity. 5
Gender could be an interesting topic for further investigations on the mechanism of infection and pathogenesis of SARS-CoV-2. 6
The incubation period (the time between exposure to the virus and the onset of symptoms) for COVID-19 is around one and 14 days. 7
Severe Acute Respiratory Syndrome Coronavirus type 2 is mainly transmitted by droplets and aerosols from an infected person that sneezes, coughs, talks, breaths or is in proximity to other people. Droplets may be inhaled or rest on surfaces, whereas people come in contact with and then got infected after touching their nose, mouth, or eyes.
Several copies of viral RNA have also been isolated from the faeces of infected patients, indicating that faecal-oral transmission could also be an effective route of infection.8-10 Severe Acute Respiratory Syndrome Coronavirus type 2 viral RNA persisted in the faeces for a long time.11-13
Further studies are needed to check whether there is shedding of infectious viruses in addition to the viral genome. 9
Digestive symptoms have been observed in the presence/absence 14 of respiratory illness in many cohort studies and meta-analyses with fluctuating values in per cent. Early observational studies of gastrointestinal effects, such as abdominal pain, diarrhoea, nausea, or vomiting, have been described in 2% to 10% of the infected Wuhan patients. 15 From a meta-analysis of East Asian patients, it was discovered that up to 20% suffered from gastrointestinal pain, and viral RNA was detected in the feces in approximately 50% of them.16,17
The gastrointestinal symptoms were more diffused in hospitalized patients with severe disease, 18 and they even persisted after the respiratory symptoms have disappeared.16,19
In COVID-19 patients, fever, dry cough, and fatigue are the main symptoms; sore throat, loss of appetite, abdominal/back pain, diarrhoea, vomiting, and conjunctivitis are also observed in some patients. 20
Other symptoms are myalgia, dyspnea, chest tightness, sputum production, hemoptysis, headache, pneumonia with potentially progressive respiratory failure with alveolar damage and more severe multiorgan. 21
Pneumonia is more frequently observed in elderly patients. This more severe respiratory condition is called ARDS (Acute Respiratory Distress Syndrome), and includes cytokine release syndrome (cytokine storm) 22 and is the leading cause of hospitalization.
The inflammation due to the cytokine storm in turn leads to coagulopathy associated with COVID-19 15 or thrombosis.21,23,24
Most experimental research has focused on understanding the viral biology of SARS-CoV-2 to improve antiviral therapy and vaccination strategies.
In this respect, a great deal of information has been obtained from experimental studies on cellular and animal models, aimed at clarifying the mechanisms of infection and virus entry into the various target organs, but above all those underlying pathogenesis and responsible for the serious and often fatal effects of the disease.
This paper aims to review the results of the main studies on the SARS-COV-2 virus carried out in experimental models “in vitro”, “in vivo”, and “ex vivo”, with particular emphasis on the damaging effects triggered by the virus in the central nervous system (CNS) in the short and long term (Long Covid).
A discrete number of articles and reviews were selected by PUBMED with keywords “animal models” AND “CNS” AND “SARS-CoV-2”. A few excellent reviews on the subject were published in the last 2 years and referred to in this paper.
Virus Entry Into the Host Cells
Severe Acute Respiratory Syndrome Coronavirus type 2 expresses 4 essential structural proteins: Spike glycoprotein (S), Envelope protein (E), trans-Membrane glycoprotein (M) and Nucleocapsid protein (N). 25
Glycoprotein S mediates entry into the host cell, following binding to its receptor, the angiotensin-converting enzyme 2 (ACE2).1,25-28
Following binding to the ACE2 receptor, S-glycoprotein is cleaved at the S1/S2 polybasic site by a furin-like protease, the transmembrane protease serine 2 (TMPRSS2), exposing the S2 fusion polypeptide 26 and allowing the fusion of SARS-CoV-2 with the host cell.
S1 is highly variable. It includes the receptor-binding domain (RBD) 25 and determines the cell tropism. It was shown that S1 protein mutations prevent its interaction with ACE226,29 in a species- and tissue-specific manner.
Colocalization of TMPRSS2 with the ACE2 receptor on the surface of airway epithelial cells makes the lungs particularly susceptible to infection. 30
However, TMPRSS2 is not expressed in the human brain, and it is not clear what impact can this absence have on neurotropism. 31
In the CNS, the S1/S2 site of SARS-CoV-2 undergoes an effective cleavage by the furin endoprotease.26,29,32
Neuropilin-1 (NRP1) represents another potentiation factor of ACE2 in significantly promoting SARS-CoV-2 infectivity, especially in the olfactory tubercles and para-olfactory gyri of the human brain.33,34
This could clearly explain the loss of olfactory function (anosmia) in COVID-19 patients. 35
Severe Acute Respiratory Syndrome Coronavirus type 2 can infect also host cells through the cathepsins endosomal pathway,26,36 or it can use other host proteases, such as trypsin for S protein activation. 36
Therefore, SARS-CoV-2 is an opportunistic virus that can utilize multiple host cell entries and infection pathways.
Severe Acute Respiratory Syndrome Coronavirus type 2 could use other routes of entry into the host cell, such as integrins 37 and cell sialic acid receptors in the upper airways. 38
Although to date the functional importance of integrins or sialic acid receptors in mediating the entry of the SARS-CoV-2 S protein needs to be determined, they could potentially increase cell tropism, viral pathogenicity, and SARS-CoV-2 transmission.
Viral Entry Into the CNS
ACE2 receptor expression is very low in the brain; some autopsy studies have previously demonstrated the presence of SARS-CoV particles in the brain tissue. 39
Brain areas express ACE2, such as both excitatory and inhibitory neurons of the substantia nigra and cerebral ventricles. 40
The pattern of ACE2 expression in brain regions is in agreement with reported symptoms such as confusion, headache, seizures, brain enlargement, diarrhoea, nausea and vomiting.10,15,21
The high expression of the ACE2 receptor in the glial cells and the cerebral ventricles suggests the existence of 2 new potential entry pathways of the virus into the cerebrospinal fluid (CSF) and for its diffusion into the brain. 40
Several mechanisms, both direct and indirect, have been proposed for the access of SARS-CoV-2 into the CNS of patients with COVID-19.
Direct entry routes
Severe Acute Respiratory Syndrome Coronavirus type 2 could reach the CNS through neuronal and haematogenic pathways.
Neuronal invasion occurs through a mechanism of retrograde trans-synaptic viral transfer or from peripheral nerves 41 or the cribriform plate and olfactory bulb. 42
Moreover, the hyposmia was observed in some patients and the expression of ACE2 in the nasal mucosa. 43 The presence of the virus in nasal swabs cannot exclude the retrograde neuronal dissemination which involves the olfactory epithelium> olfactory bulb> olfactory nucleus in the piriform cortex. 44
In the hematogenous route, the integrity of the blood-brain barrier (BBB) is of great importance.
During an infection in severely injured patients, SARS-CoV-2 can reach the choroid plexus and circumventricular organs, devoid of BBB, and subsequently, enter the brain parenchyma.
Severe Acute Respiratory Syndrome Coronavirus type 2 could infect the brain endothelial cells expressing ACE2 receptor and subsequently perivascular astrocytes/macrophages, increasing the permeability of the BBB.
Leukocyte diapedesis through the BBB could facilitate the transfer of SARS-CoV-2 from the bloodstream to the brain parenchyma. 45
A lot of studies demonstrate the neurotropism of SARS-CoV-2.21,46,47
Recently, neuro-invasion of SARS-CoV-2 has also emerged through intestinal vagal afferents, 48 along with the lung-intestine-brain axis. 49
When the virus spreads in the brainstem, the involvement of the solitary tract and ambiguous nuclei, which receive afferent signals from pulmonary mechano-chemo-receptors, may eventually be responsible for respiratory failure and even death. 50
In some cases, SARS-CoV-2 viral particles have been highlighted in the CNS and the CSF.51-53
Researchers have also found viral particles in endothelial cells of the frontal cerebral cortex lobe during a COVID-19 patient authopsy. 54
Although there is no data on the presence of SARS-CoV-2 in astrocytes, an increase in the level of the plasma-soluble-glial-fibrillary-acidic-protein (GFAP) has been reported 55 and there is a large body of evidence to support the hypothesis that astrocytes are targets. 56
Some authors propose that SARS-CoV-2-induced pro-inflammatory microglial phenotype could contribute to the development of neurodegenerative disorders. 57
Histopathological examination of brain samples from COVID-19 patients revealed a neuronal loss in the cerebral cortex, hippocampus and cerebellar Purkinje cell layer. 58 These findings further support the hypothesis that COVID-19 could induce direct neuronal damage and they also suggest a possible explanation for some clinical neurological manifestations referred by COVID-19 patients.
Indirect entry routes
Regarding the indirect mechanisms, many factors including drugs, thrombotic complications, inflammatory consequences, 59 systemic inflammation, could potentially cause cerebrovascular events. 60
For example, the macrophage activation syndrome, or “cytokine storm” associated with COVID-19, could cause neuroinflammation and injury to brain tissue. 39
Neurological Clinical Manifestations
Neurological manifestations of SARS-CoV-2 infection started to be systematically observed and reported, with several initiatives to consolidate and record clinical outcomes.61,62
Neurological clinical manifestations related to COVID-19 usually may either occur soon after the infection, either late in the course of the disease, or after recovery. 63
In some patients, neurological symptoms were observed even in the absence of respiratory symptoms. 7
Therefore, understanding the impairments caused by SARS-CoV-2 is of the utmost importance, so that the therapy for patients with atypical symptoms can be adapted accordingly and as soon as possible.
The neurological manifestations of COVID-19 vary in severity.
The mildest are nausea, vomiting, 44 loss of appetite, anosmia, and ageusia, 50 dizziness, headache15,41 up to diplopia and ophthalmoplegia.64-66
On the other hand, critical illness encephalitis, myopathy, and neuromyopathy were observed among COVID-19 patients,50,67 and more diffuse signs of the corticospinal tract with enhanced tendon reflexes and ankle clonus and dysexecutive syndrome.
In some COVID-19 cases, neurological movement disorders have also been reported, such as Guillain-Barré syndrome (GBS).62,68
Guillain-Barré syndrome neuromuscular dysfunction could be a consequence of respiratory failure observed in patients 69 with CNS involvement.50,70
Viral antigens were detected in the nuclei of the brain stem,50,71 including the respiratory control center, probably reached through the vagus nerve and surrounding sites, which can enhance primary respiratory dysfunction.
Furthermore, symptoms such as anosmia and ageusia are interesting, because hyposmia is a clinical manifestation of early Parkinson’s disease, and the accumulation of α-synuclein in the olfactory system is often observed in the prodrome phase of the disease.
Some experimental studies in the mouse have shown that the ACE2 and TMPRSS2 expressions tend to increase with age. 72 If this occurs in humans, the olfactory epithelium could be more sensitive to the accumulation of SARS-CoV-2 in the elderly.
The molecular analogy between the SARS-CoV-2 spike protein and the glycolipids present on the surface of peripheral nerves could explain the observed effects. 73
Seizures, 74 and necrotizing hemorrhagic encephalitis 53 are other neurological disorders that are beginning to be observed.
A Brain MRI study showed hyperintensity in the right lateral ventricle and the hippocampus with slight atrophy in the latter. These findings indicated right lateral ventriculitis and encephalitis mainly on the right mesial temporal lobe and hippocampus. 53 The presence of such manifestations in the brain of mainly asymptomatic patients with negative RT-PCR tests for SARS-CoV-2 shows that these should also be considered clinical signs of disease as well as respiratory symptoms.
Another common neurological symptom is characterized by acute confusion 75 (cerebral fog).
The proinflammatory microglia activation induced by the direct SARS-CoV-2 infection, or by the peripheral cytokine storm, could explain the development of neuropsychiatric symptoms. 76 Delirium could be an atypical first symptom of COVID-19, particularly in the elderly. 56 Neurological damage can lead to serious consequences for the cognitive sphere. 44 Severe Acute Respiratory Syndrome Coronavirus type 2 infection is considered a traumatic experience, this increases anxiety and significant mental disorders. COVID-19 patients experience general distress, mainly attributed to the need for quarantine. 77
Recently, non-specific neurological manifestations have also been reported in the more severe pediatric multisystem inflammatory syndrome 78 associated with COVID-19.
COVID-19-related stroke is becoming a cause of growing anxiety among physicians, particularly for patients with cardiovascular comorbidities,15,44,79 obesity, 15 diabetes mellitus, 80 chronic lung disease and cancer, chronic kidney failure, Parkinson’s disease, restless legs syndrome, cerebrovascular disease, 44 and polyneuropathy.
Although a causal relationship between COVID-19 and ischemic stroke could not be confirmed due to concurrent conditions,6,44,81,82 SARS-CoV-2 infection may be related to the prothrombotic state responsible for the stroke. 83
However, it is still under investigation if the strokes are directly involved in the viral infection of the cerebrovascular system. 84
There is an urgent need for further investigation of the mechanisms underlying the pathogenesis of SARS-CoV-2 and associated vascular and CNS pathology.
Objectives of Experimental Models in the Study of SNC Involvement in SARS-COV-2 Infection
While multiple clinical trials are currently underway, preclinical research is also needed in parallel, to understand the virus and test the therapeutic agent’s safety and efficacy.
World Health Organization has assembled an international working group of ad hoc experts, known as WHO-COM, to develop cellular or animal models that can mimic symptoms and disease processes observed in SARS-CoV-2 infected patients. 85
Through the "Solidarity" trials, WHO has launched a global campaign to accelerate the testing of therapeutic agents and vaccines on an unprecedented scale. 86
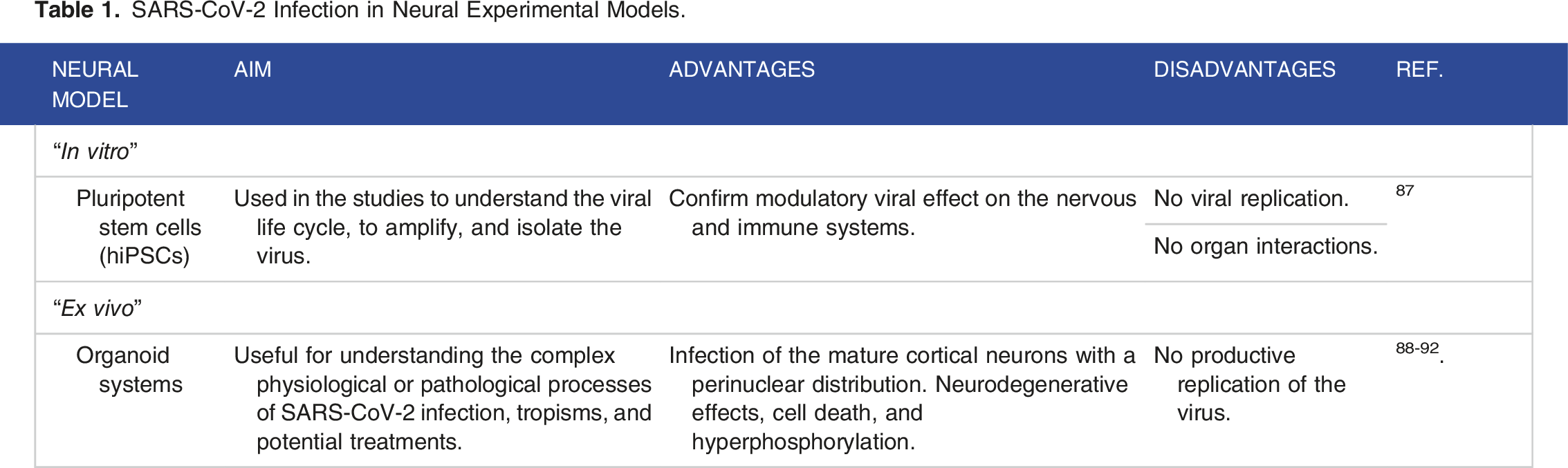
SARS-CoV-2 Infection in Neural Experimental Models.
SARS-CoV-2 Infection in Animal Experimental Models.

SARS-CoV-2 Infection in Animal Experimental Models.
“In vitro” Neural Models
Neural progenitor cells (NPCs) derive from human induced pluripotent stem cells (hiPSCs) and have been used in the “in vitro” studies to understand the viral life cycle, amplify, and isolate the virus. 87 These studies have shown that the SARS-CoV-2 can infect hiPSC cells in culture suggesting the possibility that the virus exerts a modulatory effect on the nervous and immune systems, but it is unable to produce copies of itself. Although many cellular models for studying SARS-CoV-2 infection have been developed, so far none of them allows us to understand the evolution of the neuronal damage observed in patients.
Animal Models
The poor understanding of the pathogenetic mechanisms of SARS-CoV-2 and the difficulty to obtain promising information from “in vitro” studies were the impetus for developing animal models of the disease.
Many animal species have been studied, but among those that have demonstrated susceptibility to the viral infection only a few - mice, ferrets, hamsters, and non-human primates – proved to be useful and easy to handle under laboratory conditions. 99
To date there are no results of SARS-CoV-2 infection from experiments carried out in rats, despite the Wistar rat (Rattus norvegicus domesticus) model has been extensively utilized for studying neurodegeneration’s pathological mechanisms. It has been shown that the rat expresses the ACE2 receptor for the spike protein in different brain areas, in cerebral vascular tissue, in the components of the BBB and the astrocytes.139,140
In the future, it will be useful to conduct studies with the viral agent of COVID-19 in the humanized rat.
Mouse (Mus musculus) model
A mouse (Mus musculus) model would have desirable characteristics of treatability, ease to use and availability. It would be cost-effective, and it would allow mechanistic studies 93 to identify the attributes of severe COVID-19 outcomes in some infected people. However, wild-type and inbred mouse strains cannot be used for SARS-CoV-2 experimental research due to the low affinity between the viral S protein and the murine ACE2 receptor (mACE2), a necessary link for the initiation of viral infection. 94
At present, no mouse model summarizes all aspects of COVID-19 in humans, especially unusual features such as pulmonary vascular disease and hyper-inflammatory syndromes observed in adults and children, respectively. 95
However, continuous improvement of models could close them to the aspects of human disease as well.
One strategy to modify the SARS-CoV-2 spike protein affinity and achieve effective binding to mouse ACE2 is the sequential passage of SARS-CoV-2 into mouse lung tissue. 96
Studies have recently been conducted with BALB/c and C57BL/6 mice 97 infected with modified SARS-CoV-2 strains, such as SARS-CoV-2MASCp6, 96 capable of binding efficiently to the murine ACE2 receptor in young and old mice, causing disease similar to human COVID-19.
In addition to their use in studying pathogenesis, these models have allowed researchers to experiment with vaccines and treatments for the disease. Viruses that contain spike protein mutations with increased binding affinity to mouse ACE2 are expected to be selected.
Mice sensitized to the infection develop only very mild disease. 97
Considerable efforts are underway to develop mouse models for studying the infection and pathogenesis of SARS-CoV-2 and involving humanized engineering mice or/and Collaborative Cross mice (CC). 99
The genetic diversity of CC mice models enhances viral susceptibility, but infection depends, anyway, on a functional receptor that allows the virus to enter the cell. It is probably CC studies could help scientists to find SARS-CoV-2 different phenotypes that better reproduce human infection, overall unusual features (pulmonary vascular disease and hyperinflammatory syndromes) often fatal.95,98
In the future, humanized mice could become a unique tool for gaining insight into our strategies for vaccine development against SARS-CoV-2, early intervention, and antiviral therapy.
A more effective approach to increasing the binding affinity between mACE2 and spike protein is to engineer mice to express human ACE2. In fact, several transgenic models have been developed, in which hACE2 is under the expression of a total or tissue-specific promoters. 99
Typical replication was observed in the lungs of infected engineering mice, which are often interested in body weight loss. Histopathological examination demonstrated the presence of severe interstitial pneumonia with involvement of macrophages and lymphocytes both in the interstitial and in the alveolar cavity, while no one of these phenomena was found in wild type. 100 In K18-hACE-2 [B6. Cg-Tg (K18-ACE2)2Prlmn/J] transgenic mice under the control of the human cytokeratin 18 promoter, susceptibility was highly associated with local and systemic “chemokine storm”, especially in the brain, and elevated levels of λ-interferon (IFN-λ) in the lungs. A mild to moderate tissue pathology including vasculitis was shown in the brain, accompanied by the presence of SARS-CoV-2 nucleocapsid protein antigen (NP) and hACE2 expression.101,102 Lung and brain involvement, together with perivascular inflammation observed in K18-hACE-2 transgenic mice, shared many features with severe SARS-CoV-2 infection in human. 103
Some recent studies have confirmed the neurotropism of SARS-CoV-2 in K18-hACE-2 transgenic mice. 104 An increasing viral titre in the brain was observed after intranasal administration of virus. 105 A high density of infected cells was detected in the forebrain, while a low amount was observed in the dentate gyrus, the globus pallidus, and in the cortex, restricted to layer 4. Evidence of infected cells has not been revealed in the cerebellum. These results could suggest that the neurological symptoms observed in COVID-19 patients are the consequences of CNS invasion by the SARS-CoV-2 106 and the CNS itself could represent a site for the virus replication. 105
Different scenarios have been proposed on the mechanisms of virus entry into the CNS. According to several authors, in K18-hACE-2 transgenic mice, the virus enters the brain through the nerve connections with the olfactory bulb, which would explain its absence in the nuclei of the brainstem not anatomically connected to it. In the olfactory bulb, virus entry could be possible by ACE2 and TMPRSS2 proteins in sustentacular cells of the olfactory epithelium, which are, according to some authors, the first could be invaded. 141
Once established in the brain, the virus could also reach other areas through non-neuronal pathways (blood or Virchow-Robin spaces). 142
Other authors are agreeing with the existence of alternative routes of the virus entry into the brain, supporting the observation that, in some cases, the infected olfactory neurons are very immature cells and lack the axonal projection to permit the virus to reach the brain. 143
High levels of spike protein receptors were observed in the lungs, liver, kidney, gastrointestinal system, and brain of HFH4-hACE2/C3B6 transgenic mice. 107 Symptoms developed by the infected animals reflect a pathological picture seen in humans. The infection of HFH4-hACE2 mice with SARS-CoV-2 has been developed as a severe pathological picture that often led to the death of the animal, more often observed in males. 107
The viral titre in the lung tissues was comparable to that measured in the humans. The reinfection of the surviving animals only caused mild pneumonia, in the contrast to the naive-infected mice which developed a severe form of interstitial pneumonia and have high viral RNA copy numbers in the brain and lungs. 107
By using CRISPR/Cas9 knock-in technology, it has been created a mouse hACE2 model by utilizing young and old specimens. Viral replication was shown in the brain of both animals, after intranasal infection by SARS-CoV-2. Alveolar inflammatory infiltrates and vascular alterations were most widespread in elderly animals if compared with wild-type C57BL/6 mice, as to the pathological lesions observed in older patients with COVID-19. 96
The TMPRSS2 −/− knockout mouse could be another important tool for COVID-19 research that has been extensively studied to clarify the TMPRSS2’s “in vivo” role during coronavirus infection. In murine models of SARS-CoV and MERS-CoV infection, the TMPRSS2-deficient strains of mice showed a lower loss of body weight and virus replication in the lungs. 108 The TMPRSS2 inhibition by serine protease inhibitors inoculated intranasally before SARS-CoV-2 infection of the K18-hACE-2 reduced body weight loss, viral burden, and lethal effects. 109
This finding bodes well for the use of TMPRSS2 inhibitors in the prevention and treatment of SARS-CoV-2 infection and disease pathogenesis.
Therefore, the evaluation of therapies and antivirals must also leverage transgenic mouse models of the lethal end-stage COVID-19 disease, characterized by ARDS.
Syrian hamster (Mesocricetus auratus) model
Molecular docking studies were performed on the link between ACE2 of various mammals and the SARS-CoV-2 protein S, with the discovery that the Syrian hamster (Mesocricetus auratus) might be suitable. 111
This animal model has previously been used in the study of SARS-CoV 112 because it expresses an ACE2 receptor homologous to the human receptor. Intranasal inoculation of animals with the β–CoV/Hong Kong/VM20001061/2020 strain corroborated the presence of viral antigen in the epithelial cells of the nasal and bronchial mucosa with progression to pneumocytes. 113
Mononuclear cell infiltrates and apoptosis, because of sustained inflammatory processes, were observed in the lungs and in the superior airways, where the expression of viral antigens was remarkable. The inflammation was sustained by the production of IFN-γ, macrophage, and pro-inflammatory cytokines in the invaded tissues, very early after infection (2 to 3 days). 110 A progressive body weight loss was often observed. 99 The severity of the pathological picture can lead to death in 5% of cases. 111 Other clinical signs were lethargy, ruffled fur and hunched posture which last for about 15 days after intranasal administration.
SARS-CoV-2 infection in Syrian hamsters overlaps with the characteristics observed in humans with aged/male hamsters showed severe clinical manifestations concerning young/females.111,114-116 ACE2 in these animals shows a marked analogy with that in humans, with only 4 different amino acid residues out of a total of 29 amino acids. 99
Virus replication and RNA have been observed in the respiratory tract including the nose, trachea, lung, and small intestine.111,117
Inflammation of lamina propria and necrosis of intestinal epithelial cells have been detected. 118
An immune response with high titres of neutralizing antibodies has been observed in some cases. 99
On the contrary hamster’s infection after the oral inoculation of the virus was less severe with a weak infiltration of the oral cavity and stomach, while a more sustained inflammation was detected in the intestinal villi. 119
Virus transmission has occurred among intra-species individuals making this model useful for understanding the transmission dynamics of this novel coronavirus. 111
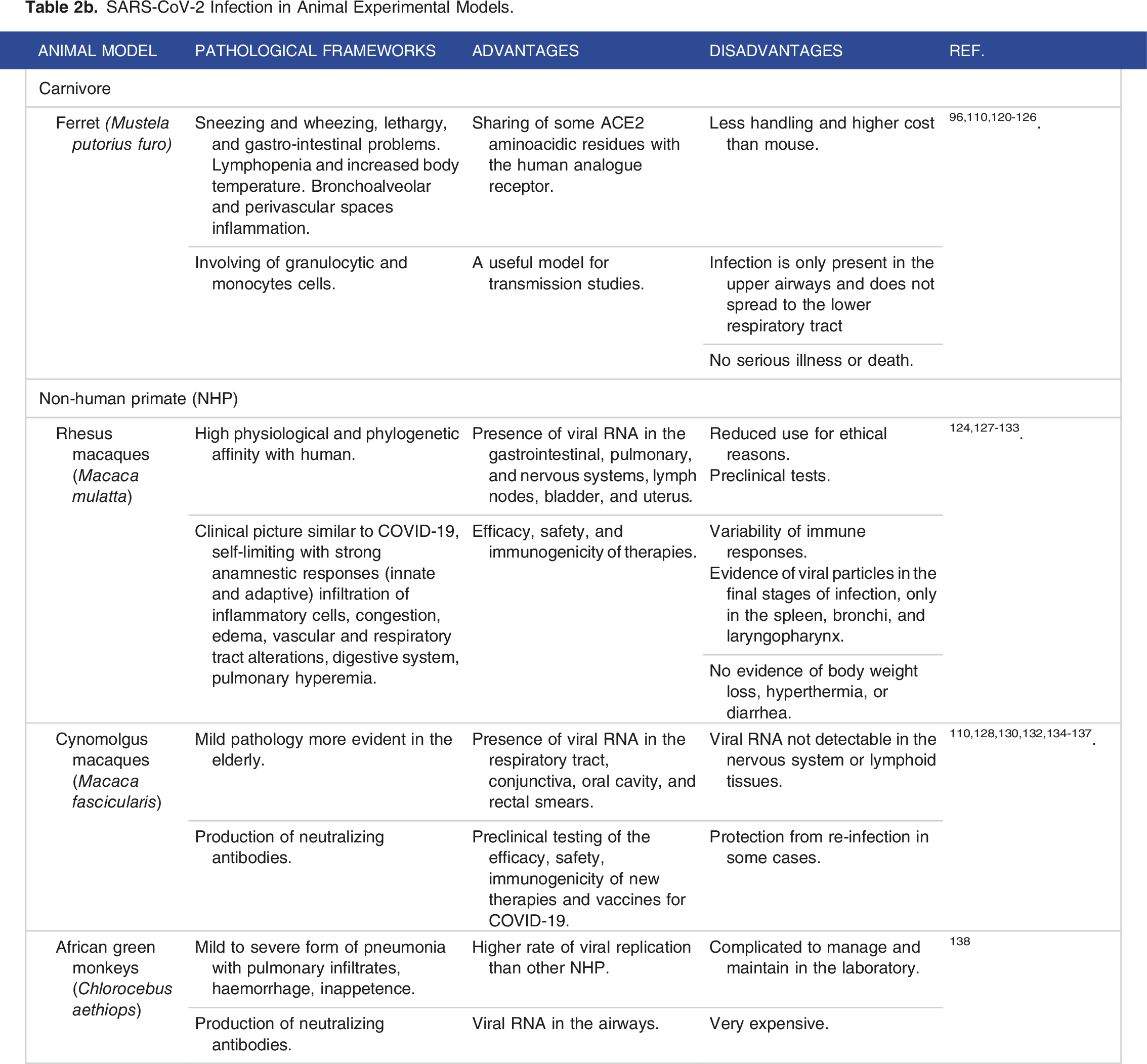
Ferret (Mustela putorius furo) model
The ferret (Mustela putorius furo) was in the past a useful model to study the pathogenesis of syncytial respiratory virus 120 and other coronaviruses 121 because of the similarity of lung morphology to that of humans. Susceptibility to SARS-CoV-2 has also been demonstrated. 96
A validated ferret model was developed in the animals infected by mucosal or aerosol exposure to SARS-CoV-2. 122 In both cases, they showed mild clinical signs, including sneezing and wheezing, probably due to the nose and oropharynx mucus buildup, together with lethargy and gastro-intestinal symptoms. 123 Lymphopenia and neutrophilia have also been described. A rise in body temperature was reported only in a few experimental ferret models.
Virus shedding and replication, seroconversion, production of neutralizing antibodies and development of cellular immunity have been detected in the upper respiratory tract a few days after exposure and for up to 8/15 days post-infection.124,125 The reason why viral replication has not been shown in the lower respiratory tract is not fully understood yet.
From the results so far, it can be concluded that SARS-CoV-2 in the ferret is responsible, similarly to what happens in man, for the respiratory tract infection with bronchoalveolar and perivascular spaces inflammation that involving of predominantly granulocytic and monocytes cells. This is probably due to the sharing of some ACE2 amino acid residues with the human analogue receptor that in the ferret is expressed by type II pneumocytes and epithelial cells of the tracheobronchial submucosal glands. 121
For all these reasons, the ferret model is confirmed as a suitable model for testing the effectiveness of therapies aimed at preventing the spread of upper respiratory tract infections including the etiological agent of SARS-CoV-2 in humans.
Viral transmission from infected ferrets was observed in naive direct and indirect contact.125,126 It makes the animal an ideal model for transmissibility studies that will support the control of the spread of the virus in communities. 110
Non-human primate models
Large animal models are preferable to faithfully reproduce COVID-19 in humans although they are not able to reproduce all the human pathological manifestations. 144
Several groups analyzed different patterns of SARS-CoV-2 and COVID-19 infection in non-human primates (NHP): rhesus macaques (Macaca mulatta), cynomolgus macaques (Macaca fascicularis), and African green monkeys (Chlorocebus aethiops).127,128
All models confirmed the presence of the virus in the upper respiratory tract and lung. 110 African green monkeys, cynomolgus, and rhesus macaques showed mild disease and neutralizing antibody production.116,124
Different experimental procedures for viral inoculation were tested in NHP, by intranasal, ocular, oral, and intratracheal administration routes. 127
Reports indicate that rhesus macaques tend to develop the self-limiting disease with strong anamnestic responses (innate and adaptive). 129 Viral RNA was detected in many organs, including the brain, very early after a first infection, while after a second re-infection any viral replication has been detected by quantitative reverse transcription polymerase chain reaction (qRT-PCR). 130
The intragastric SARS-CoV-2 administration in these species has shown similar clinical symptoms to those due to intranasal inoculation. 8 After intranasal inoculation, viral particles were described both in lung and intestinal tissues. No body weight loss, no hyperthermia, or diarrhoea were observed.
Pulmonary hyperemia occurred a few days after an intragastric administration.
Histopathological observation of monkeys in both intranasal/intragastric infection routes showed the presence of inflammatory cells infiltrating, congestion, oedema, and vascular alterations in the respiratory tract. Changes have also been observed in the stomach and intestine epithelial cells. Thus, confirming that SARS-CoV-2 can produce pathogenic effects, as observed in human patients. 8 Macaca mulatta showed serious symptoms of the infection whose severity depends on age, such as demonstrated in a study in which Macaca mulatta has been compared to Macaca fascicularis and to a New World monkey, Callithrix jacchus. Aged monkeys developed a major form of the disease with delayed immune responses and severe cytokine storm.124,131 In the initial stages of infection, SARS-CoV-2 RNA was detected in different body areas, such as the gastrointestinal system, lymph node stations, bladder, and uterus. In the final stages, RNA was limited to the spleen, bronchi, and laryngopharynx. Macaca fascicularis developed a humoral response with increased production of neutralising antibodies.134,135 In some cases, the immune response was able to protect from reinfection. 130 In this species, the orofecal transmission was also observed, with viral RNA detected in rectal smears.110,136 The presence of RNA has not been detected in lymph nodal and nervous systems. Seroconversion was observed 2 weeks after infection. The pathogenicity, less severe concerning Macaca mulatta, was still present for up to 4 weeks.
This primate represents the closest human model in pathophysiology that can be used for COVID-19 studies. 128
Rhesus and cynomolgus macaques were considered useful models for preclinical testing of the efficacy, safety, immunogenicity of new therapies and vaccines for COVID-19.132,137
Rhesus macaques used in these COVID-19 studies 133 confirmed the therapeutic effects of the adenovirus-vector vaccine, 132 DNA vaccine candidates expressing protein S139, and remdesivir treatment. 132
African green monkeys showed mild to severe forms of pneumonia with pulmonary infiltrates, haemorrhage, inappetence, and the presence of viral RNA in the airways.
The efficacy of high-titre plasma derived from the convalescent subject was tested on specimens of Chlorocebus aethiops infected by intratracheal and nasal routes with SARS-CoV-2 particles 138 with a marked reduction of inflammatory lung impairment and coagulopathies.
Are the “in vivo” Animal Models Clarifying the SARS-CoV-2 Neuro-Infection?
The results of WHO-COM members are convincing on the animal susceptibility to SARS-CoV-2 infection. Clinical manifestations in animal models ranging from mild to moderate and severe mainly affect the respiratory tract, but could reach also other organs including the CNS. 145
Several studies have demonstrated the brain invasiveness of the SARS-CoV-2 and its closest relatives SARS-CoV-1 and MERS as well as others Betacoronaviruses. 145
Severe Acute Respiratory Syndrome Coronavirus type 2 is very similar structurally to other coronaviruses, with which it shares more than 70% of the amino acid and genome sequences.146,147
The common link between the new SARS-CoV-2 with other coronaviruses is the key molecule ACE2, which allows these to enter the cell and is expressed in several organs including the CNS and the endothelial cells. Protein S1 of the SARS-CoV-2 binds ACE2 receptors with higher affinity concerning other SARS-CoV species. This aspect are suggesting that SARS-CoV-2 could have a higher neuroinvasive potential. 145
Among experimental models used to investigate SARS-CoV-2 pathogenesis, the primate species Macaca mulatta was a more susceptible species to SARS-CoV-2 infection than Macaca fascicularis and Callithrix jacchus. 134 The reason is the genetics of the spike binding site. The Macaca mulatta’s ACE2 receptor has an amino acid sequence homologous to the human one, while that of Callithrix jacchus differs by 4 amino acids that are essential for the binding. 134 However, despite symptoms in infected animals reflecting the multi-organ clinical pathology observed in COVID-19 patients, unfortunately, no significant viral particles were detected in different areas of the brain including the frontal lobe, brainstem, and cerebellum. 145
The results of these experiments, apart from confirming the presence of the virus or RNA in certain brain areas, and suggesting mechanisms responsible for neurological damage, do not explain neurotropism.
More knowledge about nervous system involvement and immune-mediated CNS pathology comes from data obtained with previous coronaviruses, such as SARS/MERS.142,148
Two main routes of neuroinvasion have been proposed: (i) the neuronal route, and (ii) the haematogenous route. However, scientists believe that there are other routes of transmission from the periphery to the CNS that need to be investigated. 145
To date, only 2 mouse models, K18-hACE2 and HFH4-hACE2-C3B6 have been possible to evaluate the neuroinvasive potential of SARS-CoV-2.105,107
According to experiments in these animal models, SARS-CoV-2 inoculated intranasally reaches the brain through the olfactory nerves. 104 Some authors are convincing that the same route is used by the virus in human. 149 The hypothesis is now has been confirmed in a COVID-19 patient in which the virus enters the brain via the transmucosal olfactory pathway. 150
Besides, ACE2 is also expressed in the mouse brain’s choroid plexus of the lateral ventricle as it does in humans. 149 Ventricles and glial substance could be another pathway for SARS-CoV-2 invasion of liquor and brain areas thus confirming the results of experiments conducted on human brain organoids.151,152
More humanized animal models and new studies are needed to trace the pathway of the virus from its entry to the various organs of the body, to demonstrate the link between its presence in the SNC, and the appearance of symptoms such as headache, anosmia/hyposmia, dysgeusia/hypogeusia, nausea, vomiting, neurodegeneration, haemorrhagic necrosis and occasionally death 153 seen in COVID-19 patients.
Organoid Systems
Due to the greater similarity with the “in vivo” conditions, the use of organoids allows us to better understand the complex physiological or pathological processes of SARS-CoV-2 infection, tropisms, and potential treatments.88-90
Human brain organoids represent a widely used SARS-CoV-2 CNS infection model. 154
It has been shown in brain organoids that SARS-CoV-2 primarily infects mature cortical neurons with a perinuclear distribution. Neurodegenerative effects, cell death, and hyperphosphorylation, as well as Tau protein dislocation, have also been observed. However, no productive replication of the virus was observed in these cells, 91 which would support the hypothesis that SARS-CoV-2 can use the brain as a long-term reservoir, 40 potentially favouring the development of neurodegenerative diseases 92 and emphasizing the need to specifically analyze the effect of the virus on the CNS.
There are no final data on the replicative capacity of SARS-CoV-2 in neuronal cells.
Severe Acute Respiratory Syndrome Coronavirus type 2 can use the neural cell mechanism to replicate in human brain organoids, in particular in mature NPCs and cortical neurons. 28
It has also been suggested active replication and productive infection of the virus, in the neural cells. 154
Furthermore, a hypoxic environment and extensive neuronal cell death were observed in areas infected with high-density SARS-CoV-2, suggesting that virus infection could promote neighbouring cell death. 28
Finally, it was observed how IgG antibodies against SARS-CoV-2 in the CSF of a COVID-19 patient can block SARS-CoV-2 infection in brain organoids. 28
Discussion
The SARS-CoV-2 infection has represented a clinical challenge because of its high transmissibility and the wide range of severity of the disease observed in the human population: from asymptomatic to mild, until severe clinical manifestations, with often fatal results. 155
Cell cultures are a good study system due to their ease of handling and storage. There are no limits to the availability of reagents, influences due to organ interactions of the whole organism are lacking, and the experimental data are quickly and easily understood. For these reasons, they have allowed studies on SARS-CoV-2 pathogenesis.
The researchers have demonstrated the presence of the virus in the neural progenitor derived from hiPSCs, suggesting the possibility of real involvement of nervous and immune tissues during infection. In fact, although the virus is capable of infecting cells, no viral copy production has been observed. Moreover, it induces an immune response with the production of type III interferon and IL-8. 87
Several animal models of SARS-CoV-2 infection are being used to study the disease and to test candidate therapeutic compounds, including mice, hamsters, ferrets, and NHP. The results are difficult to analyze due to inter- and intra-species variability.
Although the mouse seems to be the simplest model to use, the cheapest, and easiest to obtain, the wild type is unfortunately not very susceptible to infection, and the use of engineered mice has so far not yielded encouraging results.107,108
It has been suggested that, during infection, the modulation of the ACE2 expression could be involved in the neuropathological manifestations observed in COVID-19 patients. 140
The hamster is easily manipulated and develops lung damage like humans. Nevertheless, research reagents for studying the immune response are also in short supply here.
The ferret, unlike the mouse, is susceptible to infection with SARS-CoV-2 and transmits it easily both by direct contact and through breathing. 126 It is like humans from an histo-anatomical point of view. 110 The excessive cost, the poor handling, and need specific equipment, as well as the limited availability of immunological reagents specific to this species are limiting factors to use of this animal model.
Animal models of non-human primates have a certain age-dependent variability in infection that makes them terribly like the behaviour observed in humans. For ethical reasons, their use is subject to strict rules and is often limited to a few specimens. They also show high variability in innate and adaptive immune responses within the same species, which makes their use for this type of experimentation more difficult.
Despite it’s possible to study the SARS COV-2 infection in several animal models, none of them is useful in fully understanding the mechanisms underlying viral tropism and the onset of the pathological phenomena observed in humans. The main limiting factors are in the techniques of virus inoculation, as well as in the absence of predisposing risk factors, or other factors, such as age and general health conditions that contribute to aggravating the symptoms of the disease. 124
Conclusion
The ever-expanding list of cases, reporting clinical neurological complications in COVID-19 patients, strongly suggests the possibility of the virus invading the nervous system.
However, there is no clear evidence on how the virus invades the SNC and the experimental models studied, so far, have not helped to clarify the mechanisms.
It is necessary to investigate whether the virus can use the low levels of ACE2 receptors in the brain to infect neurons.
Additionally, since the virus has been detected in the CSF of many COVID-19 patients, its ability to replicate in CSF needs to be thoroughly investigated.
It is also essential to evaluate the possible effects of the virus on different parts of the brain, spinal cord, and peripheral nervous system to understand the pathophysiology underlying the neurological symptoms observed in patients in more detail.
As the neurological complications of viral infection remain very poorly understood, there is an immediate need for experimental studies. It involves the animal models useful to investigate the possible mechanisms by which the virus can enter the brain, as well as the cause of the harmful effects observed in the nervous system of the COVID-19 patients.
Footnotes
Acknowledgments
The authors thanks Antonio Macrì for technical support.
Author’s Contributions
A.M.P. developed and wrote the draft. I.V. collaborated in writing and editing of the manuscript. M.G.M. collaborated in supervisioning of the final manuscript.
All the authors read and approved the final version.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
