Abstract
Background
Oxidative stress is associated with the pathogenesis of MS. Edaravone (EDV) has been proposed as a therapeutic resource for central nervous system diseases, and it was effective in reducing oxidative stress. However, the antioxidant mechanisms of EDV are poorly studied.
Objective
This study aimed to evaluate the effects of EDV on resting, phagocytosis, and PKC-activated granulocytes derived from MS patients and a healthy control group.
Methods
The effects of EDV on ROS production in phagocytosis (ROS production in the presence of opsonized particles) and PKC-stimulated granulocytes were evaluated in a luminol-dependent chemiluminescence method. Calphostin C was used in some experiments to compare with those of EDV.
Results
EDV inhibited ROS production in phagocytosis of opsonized particles and PKC-stimulated granulocytes from MS patients and healthy control group. In the presence of calphostin C, the inhibition of ROS production was similar to that observed with EDV.
Conclusion
These findings suggest the involvement of EDV on the ROS-PKC-NOX signaling pathways modulating oxidative stress in MS. EDV represents a promising treatment option to control oxidative innate immune response for MS.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic immune-mediated inflammatory disease of the central nervous system (CNS). Neuroinflammation, a key characteristic of MS, is orchestrated by the influx of leukocytes in the CNS and the loss of blood-brain barrier (BBB) integrity causing oxidative injury and inflammation.1-5 The role of the innate immune system appears to be relevant in chronic degenerative diseases, such as MS. Oxidative stress, a state from an imbalance between oxidizing species and antioxidant response, is associated with the pathogenesis of MS. Excessive ROS production plays a crucial role in demyelination, axonal/neuronal injury, and BBB integrity modulation.4,6-11 In neurodegenerative diseases, the primary generator of ROS is NADPH-oxidase (NOX), a membrane enzyme composed of several subunits that is activated via p38 MAPK (mitogen-activated protein kinases), extracellular signal-regulated kinase (ERK) 1/2, MEK (MAP kinase) 1/2, PI3K/AKT pathway, and protein kinase C (PKC).12-22 Although the inflammatory process has been extensively researched, modulation, or suppression of oxidative stress is not the focus of immunotherapies currently available to MS patients, possibly due to the lack of translational success in clinical studies. Novel therapeutic targets proposals to MS must take into account the signaling pathways involved in ROS generation. In this context, studies have shown that Edaravone (EDV, 3-methyl-1-phenyl-2-pyrazolin-5-one) effectively reduced oxidative stress in CNS diseases.14,23-28 EDV is a free radical scavenger previously approved in Japan for treating patients who had an acute ischemic stroke, and due to its neuroprotective effect, EDV was also accepted for amyotrophic lateral sclerosis (ALS) treatment.26,29 The scavenging activity of EDV occurs via an electron-donating mechanism over a wide range of radical species.30-33 However, the antioxidant mechanisms of EDV are not fully understood. According to the above, we hypothesize that EDV could modulate oxidative stress by up-regulating the ROS-NOX signaling pathways. The objective of the present study was to evaluate the effects of EDV on resting, phagocytosis, and PKC-activated granulocytes derived from MS patients and healthy controls.
Material and Methods
Study Population
Characteristics of Multiple Sclerosis patients and Control group.

aValues expressed in mean ± standard deviation.
bValues expressed in median (minimum–maximum).
Expanded Disability Status Scale (EDSS)
The EDSS is a method of quantifying disability progression in MS patients based on an examination by a neurologist. The EDSS ranges from 0 to 10.0, with higher scores indicating worse disability. 34
Reagents
The following reagents were purchased from Merk KGaA (Darmstadt, German): Edaravone (3-methyl-1-phenyl-2-pyrazolin-5-one, cat. #M70800), calphostin C from Cladosporium cladosporioides (cat. #C6303), Phorbol 12,13-dibutyrate (PDB, cat. #1269), and zymosan A from Saccharomyces cerevisiae (cat. #Z4250). Isopropyl alcohol was used to dilute 50 mg of Edaravone (37ºC/30min), and the work solution was diluted in saline. Figure 1 shows the dose response curve of Edaravone, no difference was observed between the 3 different concentrations on inhibition of ROS production. The dose of 1 µM of EDV was based on the study from Shi et al.
35
Dose response curve of Edaravone. Values expressed in mean ± standard deviation. n = 3 for each concentration. EDV: Edaravone; G: granulocytes; PBS: phosphate buffered saline; RLU/min: Relative Light Units/minute.
Preparation of Granulocytes
Granulocytes were obtained from peripheral blood, according to Bicalho et al., 36 through a modified version of the Ficoll–Hypaque gradient method. Briefly, samples of heparinized venous blood (10 mL) were applied to double Ficoll–Hypaque gradients of different densities (1.08 and 1.12) to generate 3 interfaces after centrifugation (30–40 minutes). The first interface was rich in peripheral blood mononuclear cells, while the second interface contained granulocytes. The cells were identified and counted based on morphology, granulation, and size using a stereoscopic microscope with 400X magnification. The cellular viability of each sample was determined using the trypan blue exclusion test and was found to be > 90% in all cases.
Oxidative Responses
A luminol-based chemiluminescence method was employed to assess the oxidative responses of granulocytes. In each assay, 200 μL of luminol dissolved in .4 M dimethyl sulfoxide was mixed with a 100 μL aliquot of granulocyte suspension (1 x 106 cells/mL) in phosphate-buffered saline (PBS). Assays to establish the basal level of ROS production in granulocytes were carried out over a 20 min period, and reactions were monitored using a Turner Biosystems (Promega, Madison, WI, USA) model 20/20n luminometer. The effects of modulators on ROS production in granulocytes were assessed in sequential reactions whereby the basal granulocyte level was maintained for 20 min. Subsequently, the modulators were added, and the assay continued for a further 20 min. The modulators employed were EDV (1 μM, 100 uL), opsonized particles (100 μL of a 13.6 mg/mL zymosan-C3b suspension, ZyC3b), PKC-activator phorbol 12,13-dibutyrate (PDB; 10−4 M, 100 μL), and PKC-inhibitor calphostin C (1 μM, 100uL). In order to test the effects of EDV on ROS production in phagocytosis and PKC-activated granulocytes, the associations ZyC3b + EDV, PDB + EDV, and PDB + calphostin C were investigated. EDV and/or calphostin C were added to the corresponding assay mixture in these experiments, and the reaction was monitored for an additional 20 min.
Statistical Analysis
The D'Agostino and Pearson test was used to assess the normality of the continuous data. Normally distributed data were expressed as mean ± standard error (SE) and nonparametric data as median (minimum-maximum). The differences in the samples were compared using the unpaired Student t-test or the Mann–Whitney U-test and, in some cases, the χ2 test. P < .05 was considered statistically significant. All analyses were performed using GraphPad Prism 5 (GraphPad Software, Inc).
Results
Table 1 shows the detailed profile of the studied populations, which comprised patients diagnosed with multiple sclerosis and healthy individuals (control group). The median duration of the disease was 5 years (minimum 1 and maximum 20 years). Twenty-three MS patients were diagnosed in a relapsing-remitting course, and 2 in progressive relapsing. According to the EDSS scale, 8 MS patients with no-minimal disability were classified in EDSS 0–1.5, and seventeen patients were in the EDSS 2–8, moderate to severe disability.
EDV Inhibited ROS Production in Phagocytosis-Stimulated Granulocytes
EDV inhibited ROS production during phagocytosis of opsonized ZC3b particles in granulocytes from Multiple Sclerosis patients and control group.
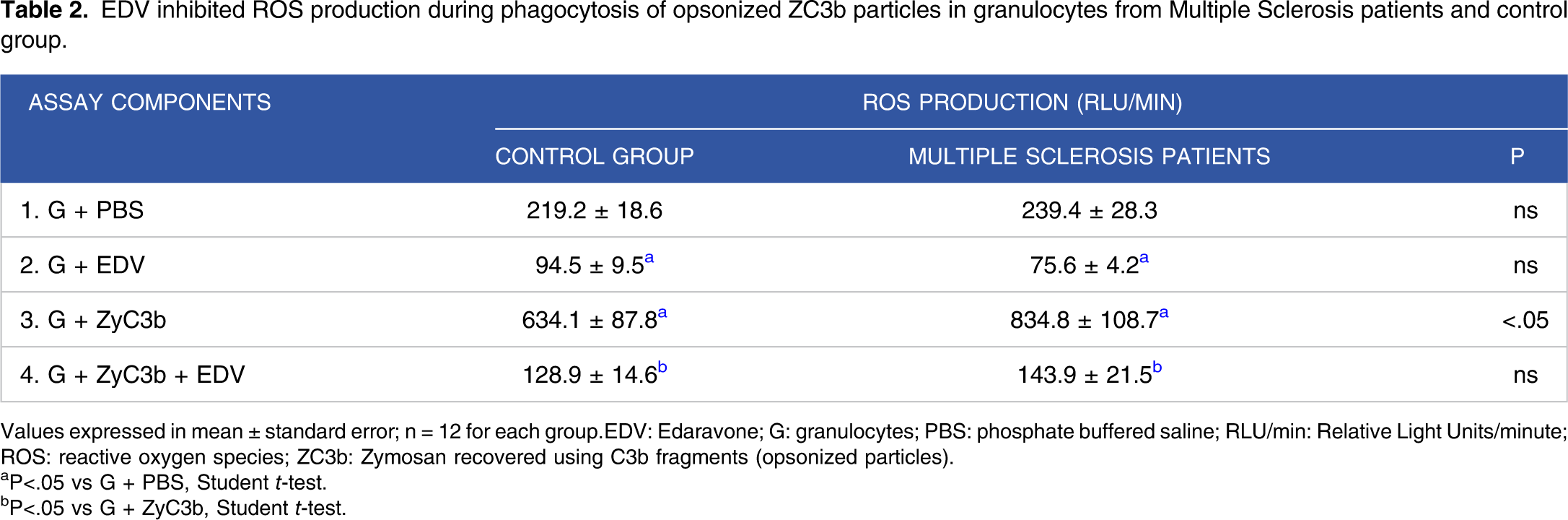
Values expressed in mean ± standard error; n = 12 for each group.EDV: Edaravone; G: granulocytes; PBS: phosphate buffered saline; RLU/min: Relative Light Units/minute; ROS: reactive oxygen species; ZC3b: Zymosan recovered using C3b fragments (opsonized particles).
aP<.05 vs G + PBS, Student t-test.
bP<.05 vs G + ZyC3b, Student t-test.

EDV-induced down-regulation on ROS generation in resting (A), phagocytosis-stimulated cells (B), and PDB-stimulated granulocytes (C) from healthy control group and Multiple Sclerosis patients. Typical curves obtained in kinetic studies of 5 experimental protocols for each group. EDV: Edaravone; G: granulocytes; PDB: Phorbol Dibutyrate; ZC3b: Zymosan recovered using C3b fragments (opsonized particles).
Inhibition of ROS Production by EDV Involves PKC
EDV inhibited ROS production in PKC-stimulated granulocytes from Multiple Sclerosis patients and control group.
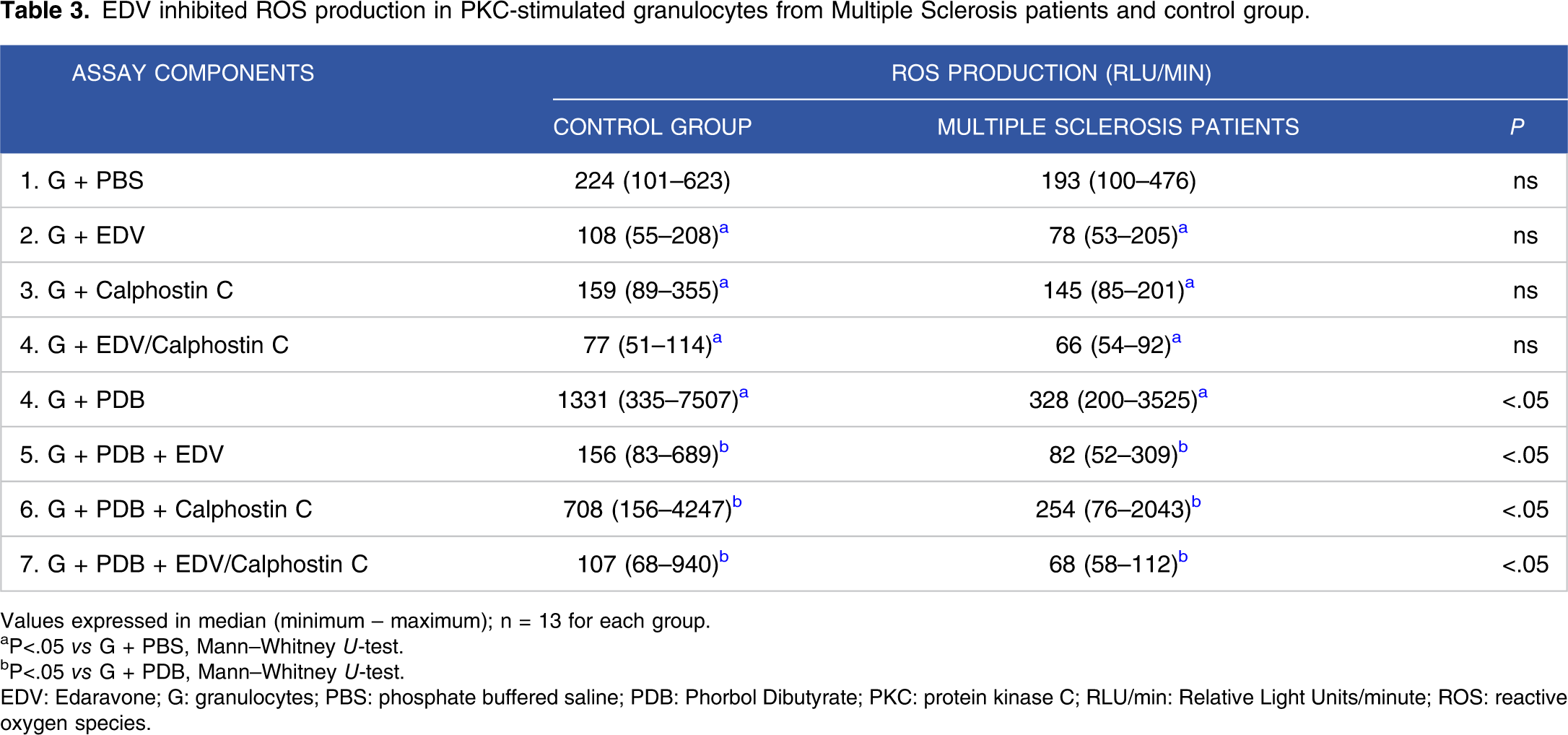
Values expressed in median (minimum – maximum); n = 13 for each group.
aP<.05 vs G + PBS, Mann–Whitney U-test.
bP<.05 vs G + PDB, Mann–Whitney U-test.
EDV: Edaravone; G: granulocytes; PBS: phosphate buffered saline; PDB: Phorbol Dibutyrate; PKC: protein kinase C; RLU/min: Relative Light Units/minute; ROS: reactive oxygen species.
Discussion
Although EDV is not yet used in MS treatment, drug repositioning is increasing in therapeutic applications. Moriya et al. 23 and Zhao et al. 14 suggested that EDV may also apply to neurodegenerative disorders treatment in which oxidative stress has been primarily implicated. In the current study, EDV inhibits ROS generation in resting, phagocytosis, and PKC-stimulated granulocytes from MS patients and healthy controls (Figure 2, Tables 2 and 3).
EDV, an antioxidant that crosses BBB, has been used to treat acute ischemic stroke and ALS.26,29 In ALS patients, the use of EDV delayed the progression of functional motor disturbances by reducing oxidative stress. 37 Experimental studies have been shown that EDV generates neuroprotective effects,28,38,39 ameliorates the clinical severity of experimental autoimmune encephalomyelitis (EAE) by reducing the lymphocytes infiltration and the expression of inducible nitric oxide synthase (iNOS) 23 , attenuates oxidative stress induced by chronic cerebral hypoperfusion injury 40 , protects against retinal damage caused by oxidative stress in streptozotocin-induced diabetic mice, 41 and decreases the levels of different isoforms of PKC and mitogen-activated protein kinase (MAPK) signaling proteins in experimental autoimmune myocarditis. 42 The signaling pathways of EDV have been associated with the inhibition of AKT, AMP-activated protein kinase (AMPK), and MAPKs, such as ERK1/2.14,42,43 In contrast, studies also demonstrated that EDV increases the antioxidant system by activating ERK/Nrf2/HO-1, 40 alleviates neuronal injury, and has anti-apoptotic effects via a pathway involving activation of ERK1/2.44,45
The production of ROS is necessary for cell activity, proliferation, and the effectiveness of phagocyte cells. Nevertheless, increased ROS production can participate in demyelination, axonal/neuronal injury, BBB integrity modulation, secretion of pro-inflammatory cytokines, and reacts with lipids, proteins, and nucleic acids, leading to functional disabilities.4-11,44-49 The results presented here indicated that ROS produced through phagocytosis can be downregulated by EDV (Table 2). Phagocytosis of the myelin sheath is an important mechanism to eliminate myelin debris, preventing the accumulation of neurotoxic lipid peroxidation products, even though it causes damage in the CNS and the stimulation of ROS production are toxic to oligodendrocytes and axons.50-54 A considerable body of evidence suggests that phagocytosis and generation of ROS seem to be altered in granulocytes from MS patients, and oxidative stress, one of the most significant harmful conditions for the CNS, may be involved directly in several processes underlying disease pathogenesis.4,6,7,9,10,55-58
The presence of infiltrating T cells in CNS mediates the influx and activity of granulocytes that initiated axonal demyelination and represent a major source of ROS.5,59,60 PKC, a serine/threonine kinase family with at least 11 isoforms involved in different intracellular effects signal transduction in various cell types, stimulates ROS production through the phosphorylation of NOX subunits.61,62 PDB, a membrane-permeable activator of PKC, activated ROS generation in cells from both studied groups, although the ROS production was significantly lower in cells from MS patients than in the control group (Table 3). Similar results have been reported with PMA (also an activator of PKC) in MS patients with a severe course and during bouts of MS.63,64 Both calphostin C (an inhibitor of PKC) and EDV inhibited ROS production in PDB-stimulated granulocytes either from MS patients or healthy control (Table 3). Our findings suggest EDV could act on the PKC, but other signaling pathways are possibly involved.
Targeting the ROS-generating pathway may be a possible treatment of CNS disorders. Apocynin and DPI (diphenyliodonium chloride) are chemical compounds with NOX-inhibitory properties. The activities of those NOX inhibitors have been studied in EAE, showing reduction of BBB permeability,65,66 inhibition of ROS formation and blockage of myelin phagocytosis, 58 prevention of activated microglia from killing oligodendrocytes, 67 reduction of demyelination, infiltration of immune cells, and reduction of clinical symptoms. 66 The inhibition of NOX assembly in EAE by blockage or deletion of NOX subunits, such as p47phox, attenuated ROS production and neuroinflammation, 68 decreased EAE severity, 69 reduced toxicity to oligodendrocytes, prevented the weight loss, attenuated oligodendrocyte loss, and reduced microglia reactivity. 67
The trigger of MS remains unknown. Nonetheless, the activation of innate immune response is well characterized by inflammation and oxidative damage. Although inflammation can lead to oxidative stress and vice versa, oxidative stress precedes the inflammatory response in MS patients. 70 Hence, the generation of ROS can be considered as an inducer phase in MS pathology.
This study has some limitations, including the precise molecular mechanisms by which edaravone inhibits ROS production is unknown, and the evaluation of other signaling pathways such as NOX complex and intracellular oxidative production.
Collectively, these results indicate that EDV is effective as a ROS inhibitor in various in vitro models, including those involving resting, phagocytosis, and PKC-activated granulocytes. We suggest that EDV acts on the ROS-PKC-NOX signaling pathways modulating oxidative stress in MS. Thus, EDV might be considered as a possible complementary option to MS treatment.
Conclusion
Due to its use in other neurologic pathologies and the downregulating ROS generation, we suggest that EDV can be considered a promising medication for auxiliary treatment for MS. Therefore, further investigations are necessary to elucidate the precise activity of EDV in the modulation of oxidative stress.
Footnotes
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
