Abstract
Background
Low frequency (≤1 Hz) repetitive transcranial magnetic stimulation (rTMS) has been shown to suppress cortical excitability and is beginning to be trialed for the treatment of refractory epilepsy.
Purpose
As a step toward a larger trial, the current pilot study was aimed to test the tolerability and safety of temporal lobe rTMS using H-coil for the treatment of temporal lobe epilepsy (TLE).
Research Design
1800 pulses of active or sham rTMS were applied 5 days a week for 2 weeks over the temporal lobe of the affected hemisphere.
Results
Nine participants were enrolled and randomized to verum or sham stimulation. One participant dropped out from the sham group after 5 rTMS sessions. In-session, 3 patients had typical seizures during sham stimulation. One patient had seizures also during active stimulation (albeit fewer than during sham). Minor reported adverse events during stimulation otherwise included transient neck pain and headache, and were reported in equal numbers in both groups. Major adverse events were not reported. Our results indicate that H-coil rTMS was well-tolerated.
Conclusion
Given the relatively high prevalence of individuals with TLE who are treatment-resistant and the preliminary results of this study, we suggest that a larger safety and efficacy trial of 1 Hz rTMS for the treatment of TLE is warranted.
Keywords
Introduction
Despite advances in pharmacotherapy, as many as 50% of individuals with epilepsy have persistent seizures, medication-related side effects or both. 1 The majority of treatment-resistant epilepsies have foci in the temporal lobe (i.e., temporal lobe epilepsy [TLE]). Patients with disabling seizures that are refractory to pharmacotherapy, are considered for epilepsy surgery, which has a success rate of ∼20–50%, 1 but comes with the associated risks of a neurosurgical intervention. Additionally, for some, the seizure focus may not be precisely localized or may be close to eloquent cortical structures, and thus may be inoperable.
In recent years, interest in device-based therapies for epilepsy has grown, and trials of implanted stimulators in those with medically uncontrolled epilepsy show favorable results.2-4 Still more recently, methods for noninvasive brain stimulation have emerged as potentially new therapeutic tools for epilepsy. The best studied of these is low frequency (≤1.0 Hz) repetitive transcranial magnetic stimulation (rTMS), which may have the capacity to decrease cortical excitability 5 and thus possess anti-seizure effects without the systemic side effects of pharmacotherapy or the risks of neurosurgery. Over 200 patients have participated in randomized controlled trials evaluating the efficacy of rTMS in epilepsy. 6 Several trials show encouraging results with seizure frequency reduction 7 with maximal efficacy seen in patients with neocortical epilepsy. However, efficacy varied widely within and across trials. Several factors could account for the heterogeneity of published results including inconsistency in patient selection, differing rTMS protocols, as well as differences in the seizure focus depth relative to the skull surface.
The magnetic field produced by the most common form of TMS coil, a standard figure-of-eight, attenuates rapidly such that brain structures deeper than 2 cm beneath the skull are minimally affected by the stimulation. 8 While affected regions in neocortical epilepsy are typically no more than 2 cm deep from the skull, 8 the affected regions of TLE may be considerably deeper. This may explain why rTMS with a standard figure-of-eight coil has been ineffective for patients with mesial temporal lobe seizure foci. 9 Another coil model, namely, the H-coil (Brainsway, Ltd, Jerusalem, Israel), may be able to penetrate deeper into the brain to affect structures 5–6 cm from the coil. 8 rTMS using the H-coil has been successfully evaluated in terms of safety and efficacy of extratemporal cortical stimulation, is Food and Drug Administration (FDA) cleared for treatment of major depression, 10 and is undergoing active evaluation in other diseases.11-14 We employed an H-coil system specifically adapted to target the temporal lobe and report herein the preliminary safety and tolerability of 1 Hz rTMS in a cohort of patients with TLE.
Method
Participants and study design
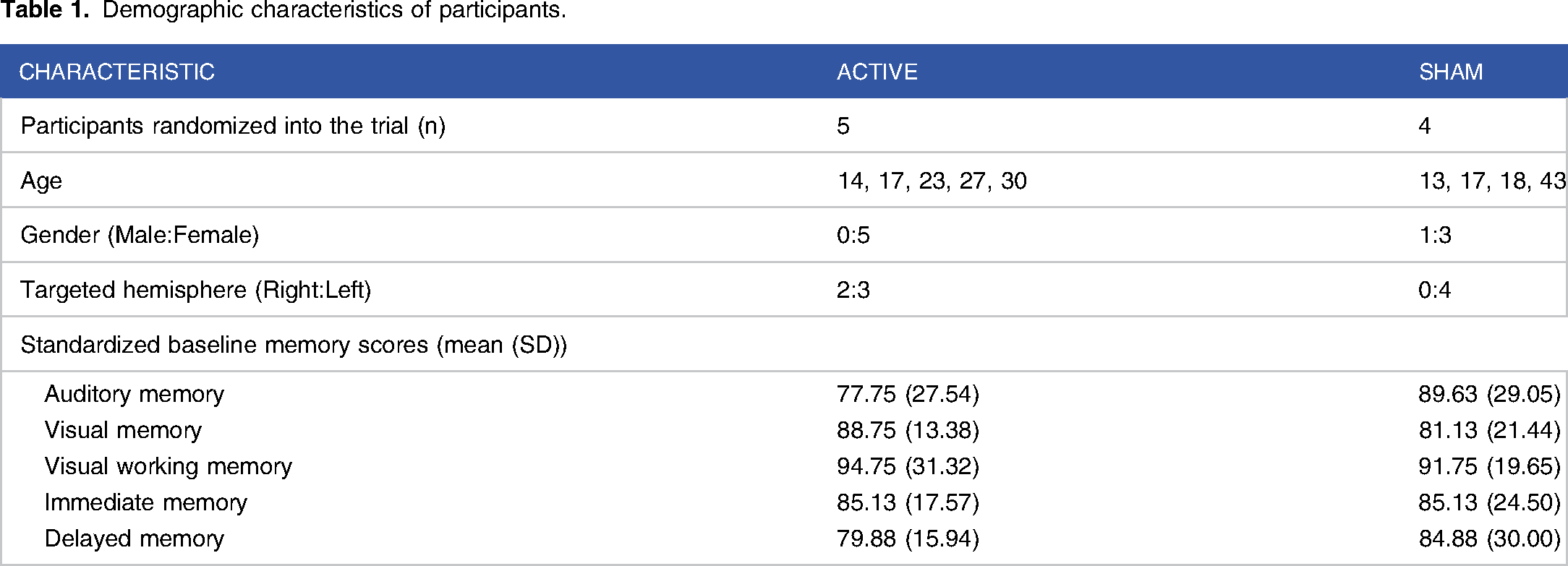
Demographic characteristics of participants.
Epilepsy etiology and focus.

A single-blind modified cross-over design, with only investigators being aware of randomization, was chosen to ensure the safety of the participant. All participants were unblinded after completing the randomized (blinded) phase of the trial, post-treatment behavioral evaluation, and the 4-week post-treatment seizure diary. After unblinding, all participants were given the option to continue with open-label active stimulation.
Three out of the four participants randomized to the Sham condition continued with the open-label phase. The fourth participant withdrew participation prior to completing the series of sham sessions. Additionally, two out of the five participants randomized to the Active condition completed additional open-label treatments (see tolerability section).
Eligibility was determined based on a clinical diagnosis of treatment-resistant TLE as determined by the referring clinician. Participants who were taking antiepileptic drugs or other neurotropic medications continued their regimen. All participants were given a comprehensive neurological exam by a board-certified neurologist with specific training in child neurology and clinical neurophysiology. The hemisphere targeted for stimulation was chosen based on the identified focus of seizure activity. Two participants (both randomized to active treatment) had seizure foci in the right hemisphere while 7 participants had seizure foci in the left hemisphere.
All participants provided written informed consent to participate in the study. For those under the age of 18, a parent or guardian was required to consent. The study protocol was reviewed and approved by the Institutional Review Board at Boston Children’s Hospital (IRB-P00001917). All stimulation sessions were conducted on the Epilepsy and Clinical Neurophysiology Unit under the supervision of trained physicians and registered nurses prepared to respond to any adverse event.
Behavioral measures
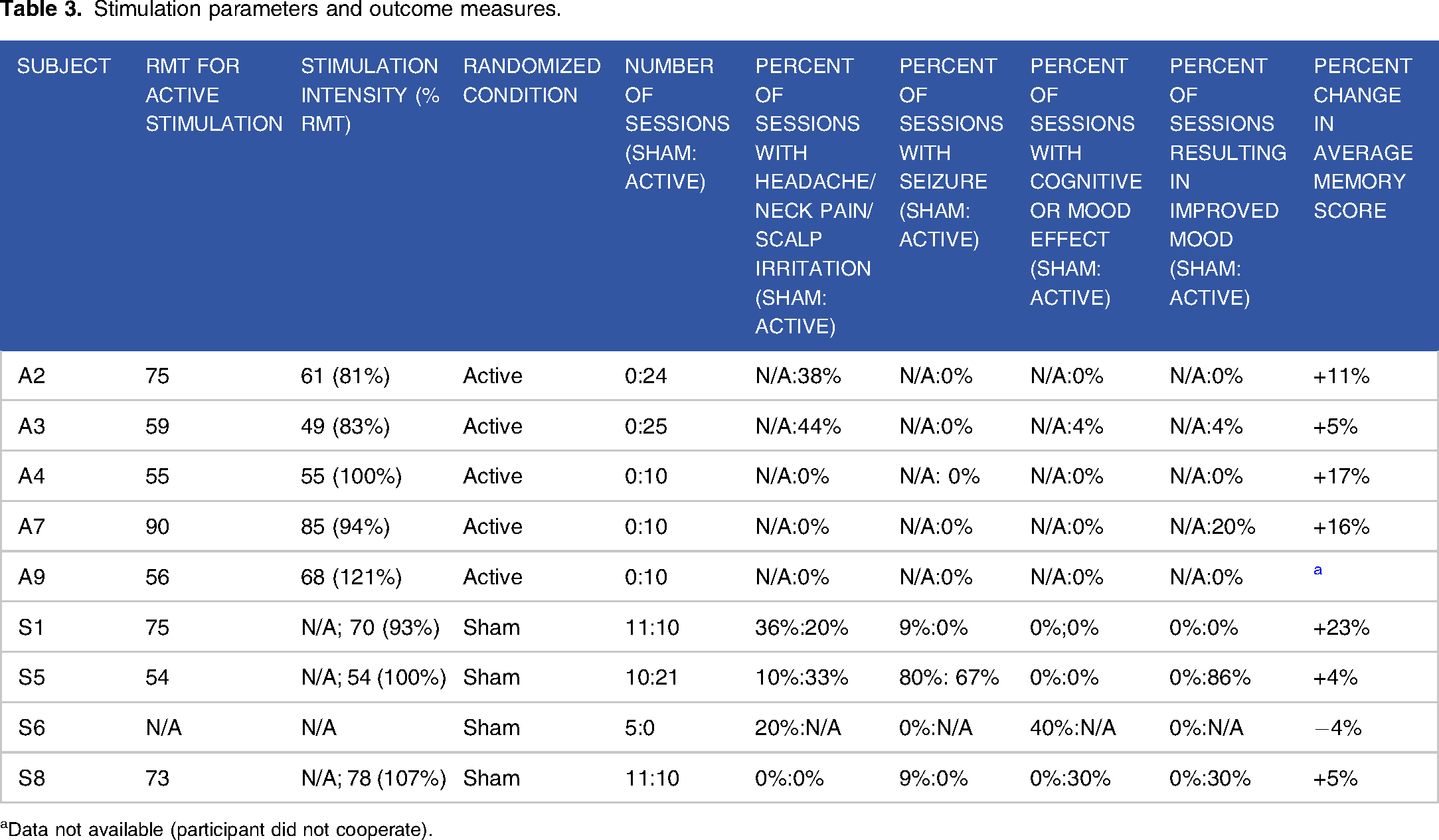
Stimulation parameters and outcome measures.
aData not available (participant did not cooperate).
Stimulation
As previously described, 15 participants received 1800 pulses of rTMS applied at a frequency of 1 Hz and titrated to 120% of resting motor threshold (RMT) for the hand contralateral to the seizure focus or maximal tolerable intensity for a total of 30 minutes every week day for 2 weeks (10 sessions or more if necessary to complete the interrupted sessions). Stimulation was applied using the Brainsway H12-coil and a Magstim Rapid 2 stimulator (Magstim Co., Ltd, Wales, UK). The H-coil was positioned such that the maximal stimulation was centered over the temporal region of either the right or left hemisphere, according to seizure focus localization. Under the Sham setting, the coil delivered an audible and palpable stimulus with negligible intracranial current induction. The participant’s RMT was determined by visual inspection of the contralateral hand during stimulation over the primary motor cortex (see Table 3 for RMT and stimulation intensities).
Data analysis
There was a total of 120 active sessions and 37 sham sessions available for analysis (see Table 3). Mann Whitney U test was performed to assess the difference in side effects between active and sham groups. A mixed factorial ANOVA was performed to assess change in memory scale scores between the active and sham groups with condition (Active, Sham) as a between subjects variable and time point (pre, post) as a within subjects variable. A significance level (α) of P < .05 was set for all statistical analyses.
Results
Side effects
Seizures
Of the 120 active sessions, there were 14 where seizures occurred during the session. All of these seizures were in 1 individual (who received a total of 21 active rTMS sessions) who typically experienced multiple seizures per day. The same participant also had seizures during 8 of the 10 sham rTMS sessions. No other participant experienced a seizure during active stimulation. In addition to this participant, there were 2 other seizures (1 each in two individuals) that occurred during sham stimulation sessions. All these seizures were the participants’ typical seizure presentation.
Headache and neck pain and scalp irritation
Of the 120 active rTMS sessions, there were 6 reports of headache (2 individuals reported headache on 3 sessions), eighteen reports of neck pain (3 individuals), and 6 reports of scalp irritation (3 individuals). Of the 37 sham sessions, there were 6 reports of headache (3 individuals), one report of neck pain and 2 reports of scalp irritation (in one individual). There was not a significant difference between the active and sham groups in the reported number of sessions with headache, neck pain, or scalp irritation (25% vs 24.3%).
Cognitive and mood effects
Of the 120 active rTMS sessions, there was only 1 instance where the subject reported anxiety and 3 sessions where an individual reported feeling more tired after stimulation. One participant in the sham group reported “spaceyness” following 2 sessions. There was not a significant difference between the active and sham groups in the reported number of cognitive or mood adverse side-effects (3.3% vs 5.4%). Additionally, 4 participants reported improved mood following a proportion (ranging from 4% in 1 participant to 86% in another participant) of the active sessions. This was not reported during any of the sham sessions.
Mini-mental state exam (MMSE)
In 7 out of 9 cases, the participants’ score in this assessment was within the normal range (27 or above out of 30) immediately following each session. The only exceptions were a participant with intellectual disability and limited verbal ability and another participant who did not speak English and therefore could not complete the task. The MMSE score was not affected by either Sham or Active stimulation and remained stable across the whole study in both groups.
Other minor side effects
One participant in the active stimulation group bit her tongue while talking during 1 of the stimulation sessions and another participant reported ringing in the ears following a single session. One participant in the sham stimulation group reported dizziness on 2 sessions and nausea following another session. There was no significant difference between the active and sham groups in the reported other minor side effects.
Tolerability
One individual randomized to the sham condition did not tolerate the stimulation and withdrew from the study prior to completion. All other participants completed all their sessions. Additionally, three out of four of the participants randomized to sham and two out of five of the participants randomized to active stimulation returned for additional open-label active sessions. It should be noted that half of the participants (four out of the nine) who received active treatment, were not able to tolerate stimulation intensities above 100% RMT.
Discussion
Results from this study provide positive preliminary evidence for the safety and tolerability of 1 Hz rTMS in patients with TLE. Only minor side effects were reported and there were no group differences between the active and sham groups. The quality and frequency of side effects that were reported are in-line with other reported side effects reported in clinical trials using the H-coil. 10 Regarding headache and neck pain, it is likely that this was a result of the induced magnetic field generated by the coil leading to facial and neck muscle contraction as has been reported in other trials.10,16 Only 1 participant dropped out of the study after initiating the rTMS sessions (from Sham group) and after completing the randomized phase of the study, the majority of the participants elected to continue to receive additional open-label treatment sessions. Regarding overall tolerability, though most participants remained in the study, only half were able to tolerate suprathreshold intensities. Notably, the motor thresholds may have been high given that all participants were taking anti-seizure medications. As stimulation intensities increase, there will likely be a corresponding reduction in tolerability and increase in the number of adverse events.
Given the small sample size in our report (this study’s major limitation), we are unable to test H-coil rTMS anti-seizure efficacy. The high anticipated inter-participant variability of response to rTMS will necessitate participant numbers that are beyond the scope of this pilot. However, we have reported a case study from a participant in the current trial suggesting that seizure suppression can be evident after rTMS H-coil stimulation in TLE patients. 15 Additionally, even though the H-coil was specifically designed to target the temporal lobe and has been validated with computational models and phantom brain analyses, the exact spatial distribution of the field in each individual’s brain cannot be determined. Nonetheless, we believe that the lack of spatial specificity may have acted as a benefit for temporal lobe epilepsy patients where the exact seizure focus is not always well-defined. In this context, the H-coil device by design has less spatial specificity as compared to figure-of-eight coils and may have affected as much as the entire temporal cortex. 8
In conclusion, the safety and tolerability of 1 Hz rTMS in this small cohort of patients with TLE is encouraging. Studies to verify the efficacy of this intervention are warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RG is presently employed by BrainsWay; AR received research support from Brainsway
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: BrainsWay (AR), Epilepsy Therapy Project (AR), Boston Children's Hospital Translational Research Program (AR).
