Abstract
Background
Multiple sclerosis treatment has changed in the last years with the emergence of new disease-modifying therapies (DMTs). Despite a better efficacy profile, these drugs raise concerns about infectious risk, which needs to be mitigated.
Objective
To analyze the results of a systematic collaborative approach between Neurology and Infectious Diseases (ID) Departments in the management of infectious risk and complications in MS patients treated with DMT.
Methods
Retrospective collection of MS patients’ demographic and clinical data from clinical records of MS and ID outpatient clinics (2011–2017).
Results
We included 149 patients: most had evidence of previous contact with Herpesviridae, and half of them were not immune to hepatitis A and B viruses (HAV and HBV). Vaccines for HAV, HBV, and Streptococcus pneumoniae were administered in 91%, 78%, and 88% of non-immune patients, respectively. JC virus serology monitoring prevented natalizumab (NTZ) initiation or prompted its switch in 34/122 patients. Forty patients had latent tuberculosis, in which 88% were treated. Infectious events occurred in 33 patients, mostly mild urinary, respiratory, and herpes virus group infections. Only three patients required inpatient care.
Conclusion
Facing the expansion of the new DMT, we highlight the benefits of an interdisciplinary approach for safer use of the chosen treatment.
Keywords
Introduction
Multiple sclerosis is a chronic immune-mediated disease of the central nervous system (CNS). Although there is still no cure, its natural history may be modified through the use of disease-modifying therapies (DMTs), namely, BRACE drugs (Betaferon®, Rebif®, Avonex®, Copaxone®, Extavia®, that is, interferon beta-1a, interferon beta-1b, and glatiramer acetate). 1 New DMTs were licensed in the last years (non-BRACE therapies), which include oral drugs like fingolimod (FGL), dimethyl fumarate (DMF), teriflunomide (TRF), and cladribine and monoclonal antibodies such as natalizumab (NTZ), alemtuzumab (ALEM), and ocrelizumab (ORC); rituximab (RTX) is also used off-label in selected cases. While being more effective against disease activity, these drugs also raise concerns regarding the risk of infections.2,3
Previous studies, mostly drug approval clinical trials, have reported the most frequent infections related to each DMT and, over time, there has been an effort to revise the most common complications with each therapy.1-5 However, there is still a lack of real-life studies describing the implemented strategies used to mitigate infectious risk, such as vaccination or prophylaxis in the setting of infectious screening before DMT initiation.
In our hospital, a tertiary care center for MS treatment in the north of Portugal, there is a specialized infectious diseases (ID) appointment—Immunomodulation and Infectious Risk Outpatient Clinic (IIROC)—in which patients planning to start non-BRACE immunomodulatory therapies are evaluated at the request of their neurologist, ideally before starting or switching immunomodulatory treatments. At IIROC appointment, patients’ infectious risk is thoroughly assessed, and individual-based preventive measures are implemented, including patient education, diagnostic workup, vaccination, treatment of latent diseases, and, occasionally, prophylactic treatments.
In this study, we aim to introduce the concept of a specialized and routinely performed ID consultation for MS patients and describe the experience and results of a seven-year collaboration between ID and neurology. We present real-world data regarding patients’ infectious risk and its management before and throughout DMT use.
Materials and Methods
We performed a retrospective descriptive study regarding all adult MS patients followed up both in MS clinic and IIROC, between January 1, 2011 and December 31, 2017. The inclusion criteria included patients with 18 years old or more and at least one ID and one CNS inflammatory and demyelinating disorders neurology appointment in our center. We excluded patients who did not have a MS diagnosis, patients who were not planned to start non-BRACE therapies, and patients who had an ID appointment for reasons other than starting immunomodulatory therapies. We collected demographic data (age, sex, and previous medical history), date of diagnosis and treatment history, IIROC data (first IIROC appointment date, antibody screening at first evaluation, tuberculosis screening, vaccination status previously and after IIROC, and prophylactic treatments started), and infectious events throughout the period of the study (date, disease, treatment setting, treatment, and outcome). According to local and international guidelines, patients are screened for tuberculosis with clinical history, chest imaging, tuberculin skin test (TST), and interferon-gamma release assay (IGRA; QuantiFERON-TB Gold). Latent tuberculosis infections were assumed when the TST or the IGRA was positive, in a patient without clinically active disease and without a history of correctly treated tuberculosis. The TST cut-off used was >5 mm in immunosuppressed patients and >10 mm if the test was performed before starting the immunosuppressive treatment. The data were collected from the patient’s electronic medical records. A descriptive statistical analysis was conducted using SPSS® version 24. For analyzing continuous variables, we used the median and interquartile range. Categorical variables were analyzed using absolute and relative frequencies.
The study was conducted in accordance with the Declaration of Helsinki and approved by Ethics Committee of São João Hospital Centre/Faculty of Medicine of University of Porto with the number CE66-2020, who waived the patient’s informed consent.
Results
Patient characteristics
During the study period, a total number of 445 patients followed up in MS clinic started a non-BRACE therapy for MS. Of these, 215 were evaluated both in MS clinic and IIROC. We included 149 MS patients planned to start a new non-BRACE DMT, after exclusion of 66 patients who met exclusion criteria (Figure 1); median age was 37 years old (IQR 29–46) and 72% were female (107/149). Median MS duration was 8 years (IQR 4–12). Selection of enrolled patients. ID - infectious diseases; BRACE - Betaferon®, Rebif®, Avonex®, Copaxone®, Extavia® (i.e., interferon beta-1a, interferon beta-1b, and glatiramer acetate).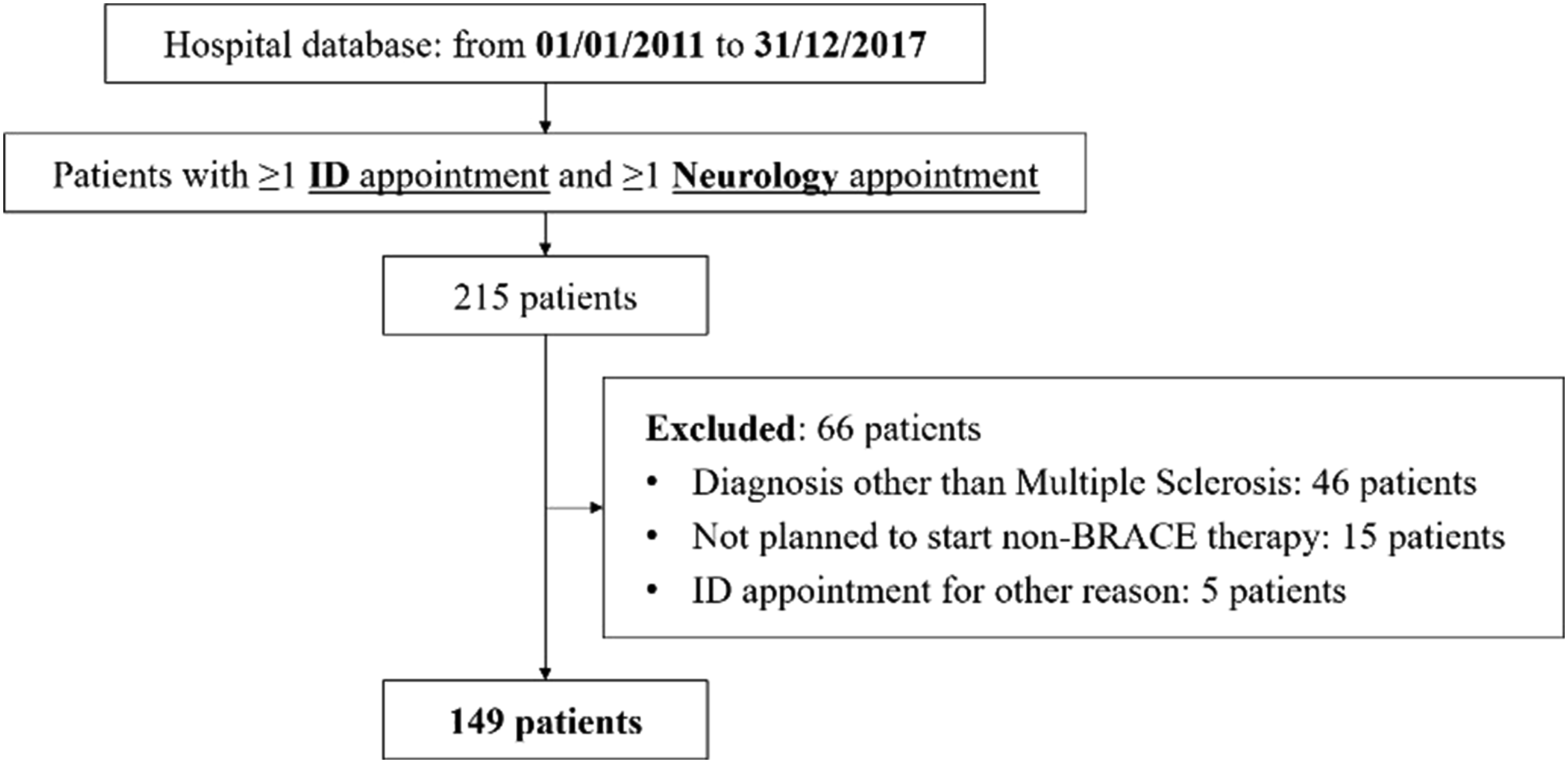
Details of included patients at baseline.

IIROC: Immunomodulation and Infectious Risk Outpatient Clinic; BRACE: Betaferon®, Rebif®, Avonex®, Copaxone®, Extavia®, that is, interferon beta-1a, interferon beta-1b, and glatiramer acetate; MS: multiple sclerosis; DMT: disease-modifying therapies.
Tuberculosis screening
Based on a complete tuberculosis screening—clinical symptoms, TST, IGRA, chest radiography, previous known relevant contact without tuberculosis treatment, and history of untreated or incorrectly treated tuberculosis—40 cases of LTBI were diagnosed. LTBI TST and IGRA were positive in 33 (84%) and 13 (33%) patients, respectively; previous significant contact was found in 9% (N = 3), and no cases had suspicious changes in chest radiography, nor a previous history of tuberculosis. Thirty-five of these patients were treated mostly with a 9-month course of isoniazid, in accordance with Portuguese guidelines. In three patients, treatment was postponed until a new DMT was effectively planned to be started; one patient had also the treatment postponed due to the potential risk of liver toxicity, in view of a low risk of TB reactivation related to the new DMT; one patient refused treatment despite the recommendation from the ID specialist. No cases of active tuberculosis were diagnosed throughout the study.
Serologic screening
Initial screening, vaccination, and treatment.
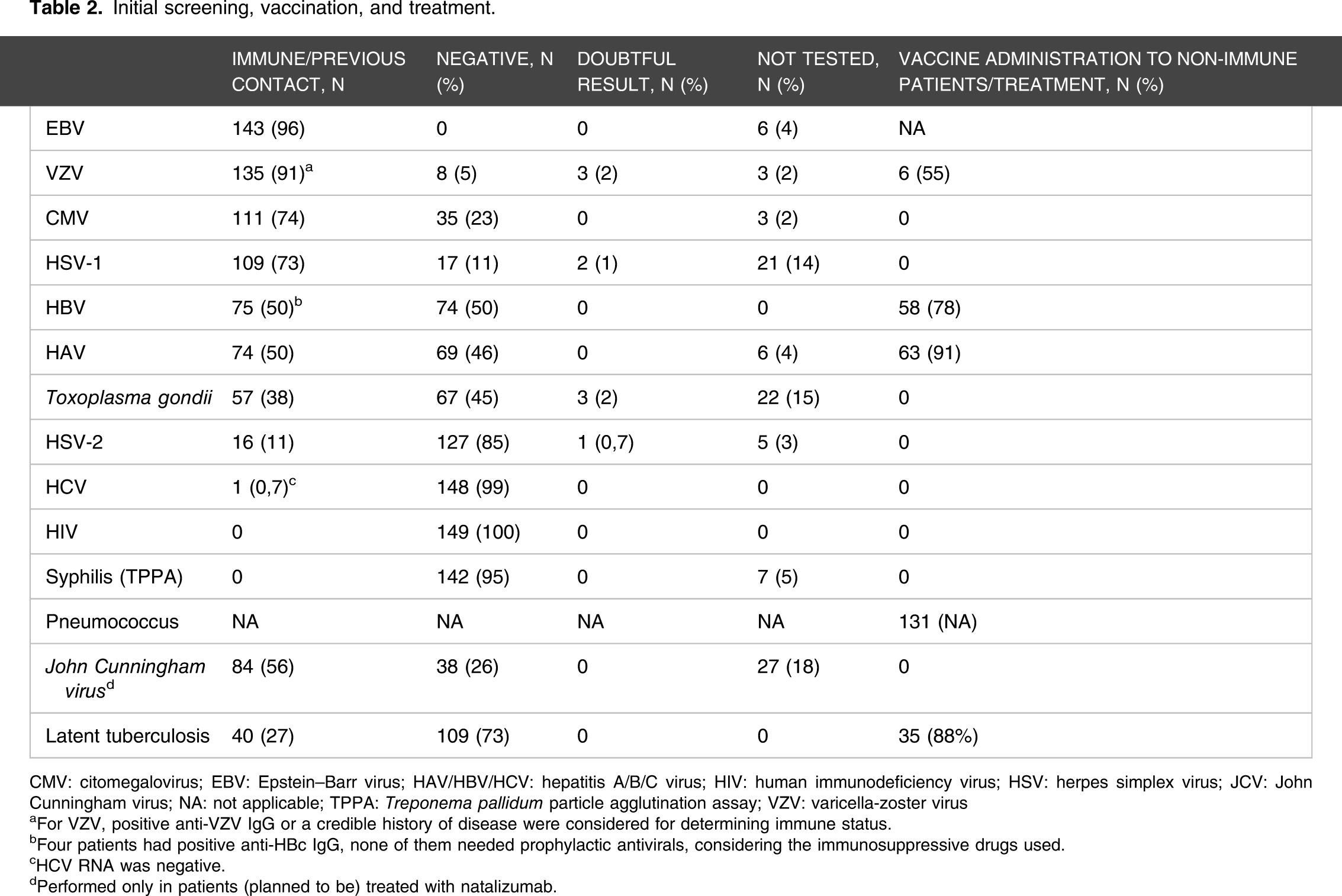
CMV: citomegalovirus; EBV: Epstein–Barr virus; HAV/HBV/HCV: hepatitis A/B/C virus; HIV: human immunodeficiency virus; HSV: herpes simplex virus; JCV: John Cunningham virus; NA: not applicable; TPPA: Treponema pallidum particle agglutination assay; VZV: varicella-zoster virus
aFor VZV, positive anti-VZV IgG or a credible history of disease were considered for determining immune status.
bFour patients had positive anti-HBc IgG, none of them needed prophylactic antivirals, considering the immunosuppressive drugs used.
cHCV RNA was negative.
dPerformed only in patients (planned to be) treated with natalizumab.
Antibodies to John Cunningham virus (JCV) and subsequent anti-JCV antibody indices were routinely determined in the serum using the STRATIFY assay in every patient planned to start NTZ, according to the guidelines for PML risk assessment. 6 Initial screening—which was positive in 84 (69%) patients—and the reassessment of anti-JCV antibody index throughout the study prevented NTZ initiation or led to NTZ switch in 34 patients (28%).
Measles IgG screening was only available in our center since 2018, so although it is routinely performed nowadays, the enrolled patients were not tested.
Vaccination and chemoprophylaxis
Vaccines for HAV, HBV, and Streptococcus pneumoniae were recommended for those without documented immunity and/or previous vaccination. VZV vaccination was performed in 6/11 susceptible patients, but ongoing immunosuppressive therapy at the time of the first IIROC appointment precluded live varicella vaccine administration in the other 5 patients. No serious reactions to vaccination or secondary MS flares were reported.
Throughout the follow-up period, two patients started cotrimoxazole for primary prophylaxis of Pneumocystis jirovecii pneumonia due to a low CD4 + count under DMF or FGL treatment.
Infectious events
Number of patients with infectious events while on treatment with each DMT.

SSTI: skin and soft tissue infection
Note. Since some patients experienced more than one episode or type of infection with a single drug, and some of them had infectious events while under different treatments, a single patient may be represented in more than one drug.
Infectious events by patient and by affected system of organs.
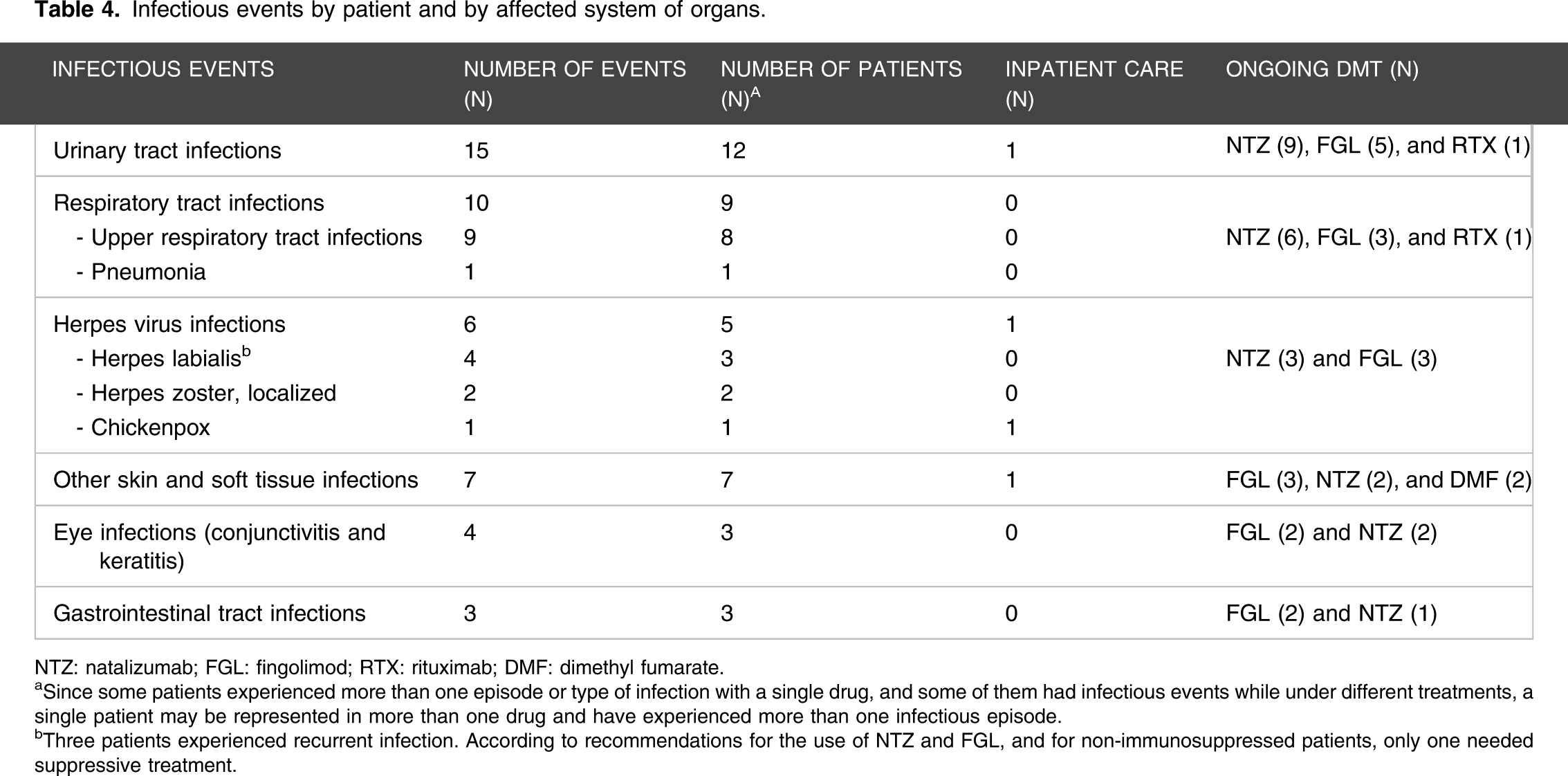
NTZ: natalizumab; FGL: fingolimod; RTX: rituximab; DMF: dimethyl fumarate.
aSince some patients experienced more than one episode or type of infection with a single drug, and some of them had infectious events while under different treatments, a single patient may be represented in more than one drug and have experienced more than one infectious episode.
bThree patients experienced recurrent infection. According to recommendations for the use of NTZ and FGL, and for non-immunosuppressed patients, only one needed suppressive treatment.
Discussion
In this study, we present the results of a seven-year follow-up of MS patients in IIROC in a MS tertiary care center, providing real-world data about the infectious screening and infectious complications in patients treated with newer DMTs, which up to now is an issue scarcely addressed in the literature.
The majority of patients were referred to IIROC before initiating a second-line therapy, namely, NTZ and FGL, which may reflect a greater physicians’ awareness of infectious complications secondary to these therapies. Unfortunately, by the time of the first IIROC, 23% of patients were already on non-BRACE DMT, which happened mostly at the beginning of this collaboration between the two specialties. Timely referral may have several implications since several tests performed under immunomodulation can be less sensitive and harder to interpret (e.g., serologies, TST, or IGRA) 7 ; some preventive measures are contraindicated in the immunocompromised patient, as in the case of VZV and other live vaccines administration2,3; and other attitudes may be less effective or implemented too late, such as TBL treatment or HBV prophylaxis.
One of the most important procedures performed after IIROC was the vaccination of patients who were not immune to pathogens that could complicate treatment with DMT. Checking the individual vaccination scheme is an important part of ID evaluation and, whenever needed, missing vaccines are recommended, according to Portugal’s national vaccination program and current recommendations on immunosuppressed patients. Anti-pneumococcal immunization was one of the most prominent interventions and was performed in 131 patients (88%), with a plausible relevant impact on subsequent infectious events, considering that none of the observed respiratory tract infections required inpatient care and that no CNS infections were recorded.
In our center, we perform a pretreatment serologic screening which includes HSV-1, HSV-2, EBV, CMV, and VZV to check for previous contact with these viruses, which allows an individual counseling regarding the need of VZV vaccination and/or other prophylactic measures to reduce infectious risk while on DMT. In our cohort, the majority of patients were not only immune to EBV—as would be expected considering the high prevalence of EBV seropositivity in the general population and MS patients 8 —but also to CMV, VZV, and HSV-1. Despite the high number of patients with positive anti-VZV IgG or a plausible history of previous contact, 11 patients were not immune. Live vaccines are contraindicated in patients already under immunosuppressive treatment, namely, NTZ, which precluded vaccination in 5 of them.2-4 Most herpes virus infections reported in our cohort were mild (recurrent herpes labialis and localized herpes zoster infections), easily manageable, and did not require suspension of DMT. However, we report a case of chickenpox in one of the patients who could not be vaccinated because he was already under NTZ on IIROC evaluation, reinforcing the need for early infectious risk management, before starting DMT.
A high proportion of non-immune patients were promptly immunized against HAV and HBV—91% and 78%, respectively. Hepatitis B and C screening and management are of special importance with anti-CD20 therapies, 1 although it is considered universal for other therapies, as well as HIV screening.3,4 In our cohort, no cases of active viral hepatitis were recorded and vaccination might have contributed to this.
Seropositivity for JCV was documented in 72% of tested patients, which is in line with previous seroprevalence studies in MS Portuguese patients. 9 JCV testing is of particular relevance in patients treated with NTZ since there is an increased risk of PML, a CNS infectious disorder with no efficacious therapy available, which leads to significant morbidity and mortality. Patients taking NTZ for more than two years, with JCV index values of >1.5 and previous immunosuppression, are at increased risk and should be monitored closely for this complication. 4 In our cohort, this evaluation, along with the IgG antibody index, motivated the switch or prevented NTZ initiation in 34 patients and, importantly, no cases of PML were reported.
Prevention of tuberculosis was another essential intervention. A recent study by Graf et al. 10 in German patients with MS and neuromyelitis optica found LTBI to be the most prominent infectious risk factor (besides JCV, which was more commonly tested), with a positive IGRA in 7.5%. Thus, although in low prevalence regions TB screening is recommended only before starting specific DMTs, a routinely performed universal screening may reveal a non-negligible percentage of LTBI.10,11 In our cohort, LTBI diagnosis was made in 27% of patients, which may reflect a higher incidence of TB in our region, 12 as well as a more accurate screening. Although the reactivation risk of LTBI is not the same for different DMT, screening is strongly advisable before starting immunomodulating therapies, particularly if patients may be considered for high-dose pulsed corticosteroids and when they live, lived, or travel from an endemic tuberculosis region.1,2,4,5 In 2017, the tuberculosis notification rate in the northern region of Portugal was 21 cases per 100,000 inhabitants. 12 Considering this, along with the fact that all MS patients may eventually need high-dose pulsed corticosteroids, LTBI screening is part of our IIROC routine. In our cohort, 88% of patients with LTBI were treated to prevent further TB reactivation. As previously stated, we use both TST and IGRA to screen for LTBI. TST has some limitations, particularly in a population highly vaccinated with BCG. However, the positive and negative predictive values of this test are affected by several factors, such as the patient’s age when TST is performed and when BCG was administered and, most importantly, the tuberculosis prevalence in a given region. 13 In our region, there is still a considerable burden of tuberculosis, which was even higher in the past, during most of the living time of our adult patients. On the other hand, immunosuppressed patients, particularly those under corticosteroid therapies and under some specific biologic treatments, have not only a high risk of developing active tuberculosis but also a high risk of developing severe and rapidly progressing disease. That said, and keeping in mind that we do not have infallible tests—which means that some patients are going to be diagnosed with LTBI despite not having it—we advocate that both TST and IGRA should be performed in these patients, and that LTBI should be diagnosed and treated, even if discordant results (i.e., positive TST and negative IGRA) occur. We believe that it is safer to overdiagnose and overtreat patients with drugs that mostly are well tolerated and whose side effects we can monitor and manage easily, than to risk the development of active TB in this population. The relevant number of LTBI diagnoses and the fact that no TB reactivations occurred reinforce the importance of this strategy.
Although infectious events were reported in a significant number of patients during the period of the study (33, 22%), we highlight that the vast majority presented mild and easily manageable disease, and that none needed DMT withdrawal. Only 3 patients required inpatient care (pyelonephritis, cervicitis, and chickenpox) and all patients recovered with no sequels. We believe that IIROC contributed to this good outcome, mostly through three important preventive measures: patient education, meticulous screening, and opportune management (e.g., immunization and prophylaxis). The fact that patients are aware of their new condition concerning infectious risk is key in the avoidance of risk factors, in the recognition of alarm signals, and in reaching for timely care when needed. Although cotrimoxazole is not the standard of care in patients treated with FGL and DMF, due to persistent lymphopenia with low CD4 + T-cell counts registered in two patients receiving these drugs, a preventive approach with prophylactic cotrimoxazole to mitigate the risk of infection with Pneumocystis jirovecii was implemented, as there is an increased theoretical risk of Pneumocystis jirovecii pneumonia if persistent decrease of CD4 + T-cell counts is observed. 3
This study has some limitations: it is a retrospective study and we have probably missed some clinical details, some infectious events, as well as some preventive measures not reported in clinical records. Nevertheless, we believe that all severe infections, which could lead to treatment modifications or inpatient care, have been reported. Most infections occurred under NTZ and FGL therapies, but these drugs were the most frequently used and, thus, their associated complications are probably overrepresented, considering the low number of patients with other therapies such as DMF, RTX, or TRF.
As additional future considerations, we consider that it would be interesting to compare the rate of testing, immunization, and infectious complications of patients who attended the IIROC with those who did not. Moreover, some drugs currently used—such as ALEM, cladribine, or ORC—were not represented in our study, so it would be interesting to include them in future analysis.
Conclusion
We believe that IIROC is of great value, especially with a timely referral—either before starting DMT or even at the time of diagnosis. In our cohort, we highlight the high number of patients who were not immune to HAV and HBV and the significant proportion of patients who were diagnosed with LTB, leading to prompt vaccination and treatment in most of these cases, respectively. An interdisciplinary approach with ID professionals, who are capable of individually educate, screen, and institute chemoprophylaxis and immunization when needed, in a population of patients receiving DMT, allows for a reduction of infectious risk and, hence, more confident use of the chosen drug.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
