Abstract
Background:
Carbamazepine and valproate are widely used in the treatment of epileptic seizures. However, these agents exhibit certain adverse effects including hematopoietic disorders (carbamazepine) and severe hepatotoxicity (valproate).
Purpose:
To determine the extent of monitoring of the hematologic effects of carbamazepine as well as the extent of monitoring of the hepatic effects of valproate in patients with epilepsy receiving treatment with these agents.
Method:
A cross-sectional antiepileptic drug use study using case notes of patients with epilepsy managed at the neurologic clinics of 2 tertiary medical facilities in Nigeria between January and December 2017.
Results:
Carbamazepine was the most frequently prescribed antiepileptic drug (48.24%), followed by valproate (29.34%) and levetiracetam (9.24%). Pretreatment monitoring of hematologic effect was carried out in only 61.11% of patients placed on carbamazepine therapy while follow-up monitoring was done in 3.7% of these patients. Also, in patients placed on valproate therapy, pretreatment and follow-up monitoring of the hepatic effect was done in only 33.71% and 19.0% of the patients, respectively.
Conclusions:
The extent of monitoring of the hematologic effects of carbamazepine, as well as the hepatic effects of valproate in the cohort studied, is poor.
Introduction
The use of antiepileptic drugs (AEDs) is the first line of treatment in epilepsy. The AEDs prevent seizures without adversely affecting the quality of life of patients with epilepsy. These agents act on different molecular targets to alter the abnormal excitability of neurons selectively. This is achieved by preventing the spread of excitation or reducing the discharges of the focal seizures. Several AEDs are available for the treatment of epilepsy.1,2
The therapeutic goals of AED therapy are to eliminate seizures without interfering with normal physiologic function. Initiation of AED therapy should be done with a single drug, with a gradual increase of the dose until seizures are controlled, adverse effects become unacceptable, or the maximum recommended dose is reached. However, if seizures are not controlled with the initially selected drug at the maximum recommended dose with appropriate plasma concentrations, a second AED should be used as a substitute or added to the initial therapy. However, drug substitution is usually preferred to the concurrent administration of another AED. Drug substitution is also recommended in cases where patients experience intolerable side effects of the initial drug therapy.2-5
Drug therapy for epilepsy should be individualized and selection of the most appropriate pharmacotherapy agent should be based on several important considerations including age, comorbidity, gender, seizure or epilepsy type, potential adverse effects, and potential drug interactions. 6
Although most antiepileptic agents can elicit central nervous system side effects (such as dizziness, drowsiness, and impaired cognition), some agents are more tolerable compared with others. As epilepsy treatment is generally long term, drug tolerability should be considered as being significantly important for the successful treatment of the condition as it affects compliance to therapy.2,4,7 Blood dyscrasias are common with many AEDs, with severe cases occurring in 6 of 10 000 patients. Elevation of the liver enzymes γ-glutamyl transferase and alkaline phosphatase occurs in patients taking enzyme-inducing AEDs.8,9
Carbamazepine is an AED used in epilepsy, trigeminal neuralgia, and bipolar disorder treatment. It blocks sodium channels and inhibits serotonin reuptake.10,11 Carbamazepine is used widely for the treatment of numerous medical disorders. However, the drug exhibits numerous side effects including hematopoietic disorders, such as agranulocytosis, thrombocytopenia, leukocytosis, leukopenia, aplastic anemia, eosinophilia, or pancytopenia. 10 Discontinuation of carbamazepine therapy has been recommended in patients with white blood cells less than 3000/mm3, thrombocytes level less than 100 000/mm3, and neutrophils less than 1500/mm3. 12
Valproate is commonly used in the management of epileptic seizures. 13 Documented side effects of valproate include obesity, insulin resistance, metabolic disorder, and severe forms of hepatotoxicity. Elevated transaminase levels have been reported in up to 5% to 10% of patients taking valproate.13,14
There is a need to routinely monitor liver function and full blood count in patients receiving AED therapy. This study set out to determine the pattern of utilization of AEDs, extent of documentation of adverse drug reactions associated with AEDs, the extent of monitoring of the hematologic effects of carbamazepine, as well as the extent of monitoring of the hepatic effects of valproate in patients with epilepsy receiving treatment with these agents in 2 selected neurologic clinics in Nigeria.
Methods
Study design
A cross-sectional AED use study using case notes of patients with epilepsy managed at the neurologic clinics of 2 tertiary medical facilities in Nigeria between January and December 2017. The case notes of the patients were retrieved for data extraction after physicians’ consultation. Data were extracted from the case notes of the patients using pretested data collection instruments. The first draft of the instrument was pretested with the case notes of 10 patients to assess the completeness and relevance of data capture. The final draft of the instrument was modified based on the results of pretesting, but the data collected during pretesting was not included in the final result. The data collected from patients’ case notes included the following:
Patients’ hospital number;
AEDs prescribed;
Documented adverse drug reactions to AEDs;
Baseline and follow-up laboratory monitoring of white blood cell and platelet counts in patients taking carbamazepine;
Baseline and follow-up laboratory monitoring of liver function in patients taking valproate.
Study setting
The study was conducted in Nigeria at the University of Uyo Teaching Hospital (UUTH) in Uyo-Akwa Ibom State and University of Calabar Teaching Hospital (UCTH) in Calabar-Cross River State. University of Uyo Teaching Hospital is a tertiary health care facility with medical residents located in Uyo, Nigeria. The facility also provides primary and secondary health care services. It is affiliated with the University of Uyo and serves as a major tertiary referral center within the region. University of Uyo Teaching Hospital has service and clinical departments and runs consultative outpatient clinics weekly in several specialties and subspecialties including neurology.
University of Calabar Teaching Hospital is a tertiary health care facility with medical residents located in Calabar, southern Nigeria. It is affiliated with the University of Calabar and serves as a major tertiary referral center within the region. It has services and clinical departments and runs consultative outpatient clinics weekly in several specialties and subspecialties.
The average number of patients with epilepsy managed in the neurologic clinics of each study site per annum is 100 patients. The case notes of patients with epilepsy who presented at the neurologic clinic of the study sites within the study period were included and constituted the sample frame. In total, 83 and 74 case notes of patients with epilepsy were studied in UUTH and UCTH, respectively.
Data analysis
Data were analyzed using the IBM statistical products and services solutions (SPSS) for Windows, version 25.0 (IBM Corp, version 25.0 Armonk, NY, USA). Frequencies and proportions were used to summarize descriptive statistics.
Ethical approval
The research protocol was approved by the Health Research Ethics Committees of UUTH and UCTH (Reference numbers: UUTH/AD/S/96/VOL.XIV/571 and UCTH/HREC/33/454).
Results
The sociodemographic/clinical parameters of the patients, pattern of AED utilization, and the outcome of treatment in the cohort studied is as presented in Tables 1 and 2. In 9.55% (15) of the cases studied, a combination of carbamazepine and valproate was prescribed.
Sociodemographic/clinical characteristics of patients.
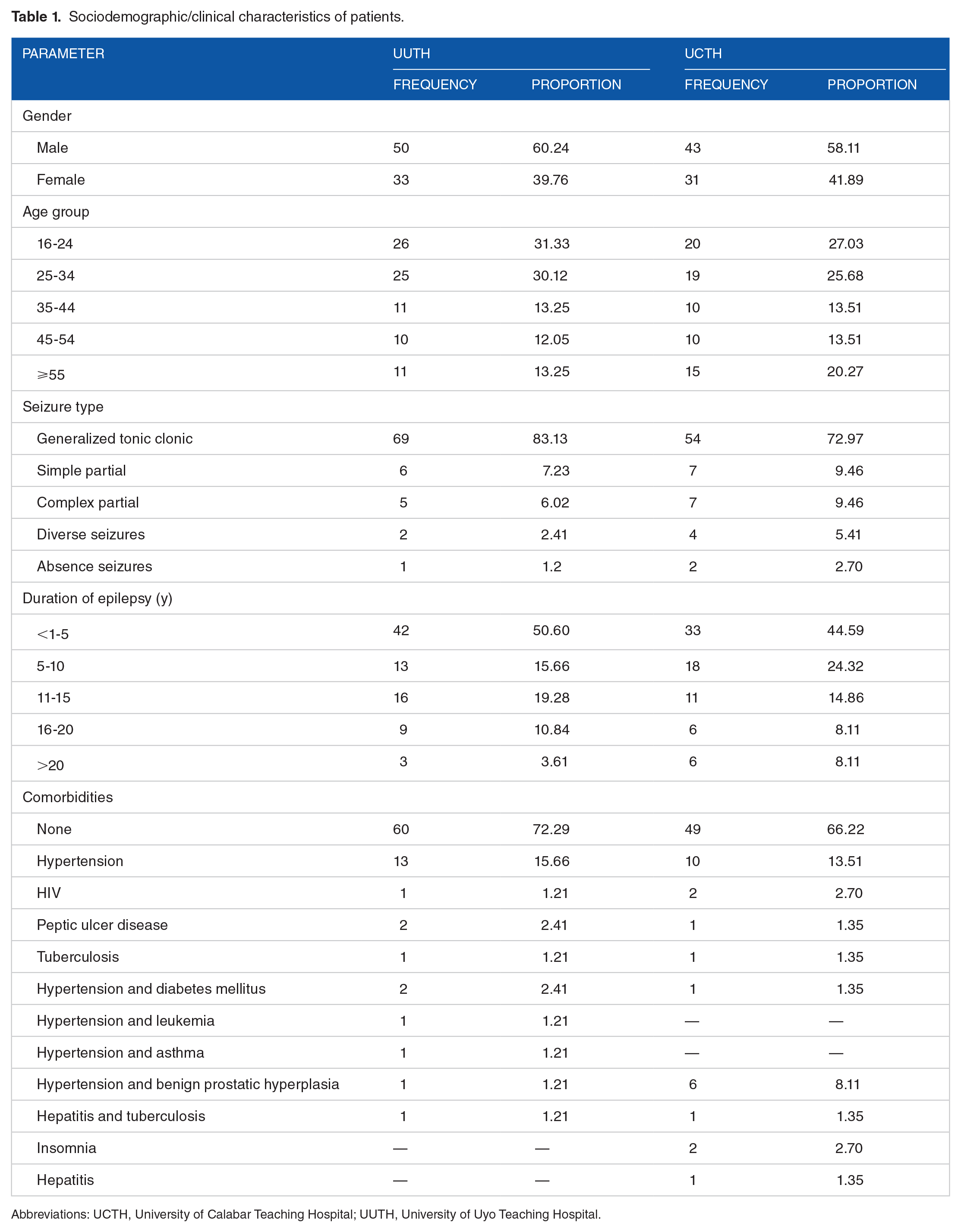
Abbreviations: UCTH, University of Calabar Teaching Hospital; UUTH, University of Uyo Teaching Hospital.
Antiepileptic drug use pattern and treatment outcome.

Abbreviations: AED, antiepileptic drugs; CBZ, carbamazepine; UCTH, University of Calabar Teaching Hospital; UUTH, University of Uyo Teaching Hospital.
The extent of monitoring of liver function in patients placed on valproate prescription and the extent of monitoring of white blood cell and platelet counts in patients placed on carbamazepine prescription in the study sites is shown in Table 3.
Monitoring of adverse effects of carbamazepine and valproate.

Abbreviations: UCTH, University of Calabar Teaching Hospital; UUTH, University of Uyo Teaching Hospital; WBC, white blood cell.
Adverse drug reaction was documented in only 4 (2.55%) of the cases studied (Table 2), namely: 1 case of Steven Johnson syndrome attributed to carbamazepine, 2 cases of suicidal thoughts and attempts attributed to levetiracetam, and a case of peripheral neuropathy attributed to valproate. We found no relationship between treatment outcome and the type or number of AEDs prescribed.
Discussion
We found carbamazepine to be the most commonly prescribed AED in the cohort studied. About 48% of the patients with epilepsy were prescribed carbamazepine, either as a monotherapy or in combination with other AEDs, followed by valproate with about 29% of the prescriptions. This finding agrees with the findings of a study in Jos, Nigeria, which showed that the most frequently prescribed AEDs were carbamazepine and valproate or a combination of the two. 15 However, our finding is at variance with the report of a study carried out in Ethiopia by Gurshaw, Agalu, and Chanie, where phenobarbitone was the most commonly used AED followed by phenytoin, with valproate being prescribed in less than 3% of the cases. 16 However, an Indian study reported valproate as the most commonly used AED, followed by phenytoin and carbamazepine. Treatment with the AED is selected based on several important considerations including age, comorbidity, gender, type of seizure or epilepsy, potential adverse effects, and potential drug interactions. 17 Epilepsy treatment is generally long term, and drug tolerability should be considered as being significantly important for the successful treatment of the condition as it affects compliance to therapy.
In about 80% of the cases studied, AEDs were prescribed as monotherapy. An AED utilization study in India showed that only 19% of the cases were prescribed AEDs as monotherapy, with 55% of the cases placed on dual therapy while 26% of the cases were managed with triple therapy. 17 In Jos, Nigeria, AEDs were used as monotherapy in 54% of the cases and as polytherapy in 46% of the cases. 15 Polytherapy in epilepsy management has been reported to offer no advantage over monotherapy. It rather increases the potential for drug-drug interactions, fails to evaluate the individual drugs, can increase the risk of chronic toxicity (including neurocognitive problems), may affect compliance, and is associated with a higher cost of medication and the need for therapeutic drug monitoring. 18 On the contrary, the use of monotherapy is associated with better compliance, fewer side effects, and the absence of drug-drug interactions among AEDs. 16
Antiepileptic drug-associated adverse effect was documented in less than 3% of the cases studied. Although this finding is similar to that reported earlier in India, 19 it may not be reflective of the true occurrence of adverse reactions in these patients as poor institutionalized adverse drug reaction reporting system limits proper documentation of adverse reactions in a health care setting. A similar study in Ethiopia showed that adverse reactions to AEDs were documented in about 33% of the patient medical records reviewed. 16 The prevalence of adverse reactions to AEDs in a previous study in Jos, Nigeria, was reported to be about 28%. 15 Antiepileptic drugs can cause various adverse effects, and this is an important factor to consider when selecting therapy. Although most AEDs can cause common central nervous system side effects, some AEDs are more tolerable compared with others. 17
The seizure was either controlled or substantially reduced in about 62% of the cases studied, thus indicating a good response to AED therapy. Regarding epilepsy treatment outcome in terms of seizure freedom, Gurshaw, Agalu, and Chanie in their study reported that almost 57% of patients were found to be seizure-free for a consecutive 3-year follow-up period. Their study also showed that conventional AEDs were still safe and effective for seizure control among a substantial segment of patients with epilepsy in the resource-poor setting reviewed. 16 Another study carried out in Cleveland, USA, reported that 64% of the cases were completely seizure-free in 12 months. 20 Antiepileptic drugs may be withdrawn after at least 2 years of seizure freedom. Tapering of dose should be done to avoid relapse, increased frequency, and severity of seizures. Seizure relapse is seen in about 20% to 40% of patients. The age at the onset of epilepsy appears to be a significant predictor. 17
In about 38% of the cases we studied, there was no significant reduction in seizure frequency. This may be cases of drug-resistant epilepsy, a term that has been used to define failure to achieve and sustain seizure freedom in a patient with epilepsy after the use of 2 tolerated and appropriate AEDs, whether as monotherapies or in combination. 21 Most cases of newly diagnosed epilepsy respond well to AEDs. Poor response to AEDs may be due to an inaccurate diagnosis of epilepsy, a wrong choice of drug for the epilepsy syndrome, failure to take the prescribed medication, an underlying cerebral neoplasm, metabolic condition, or immune process, concurrent drug or alcohol misuse. 22
On the contrary, poor response to AEDs as identified in 38% of the cases in this study may be due to patients’ poor adherence to antiepileptic therapy. Medication nonadherence usually results in poor clinical outcomes, an increase in morbidity and mortality, and an increase in health care expenditure. Reports indicate that about 50% to 60% of patients are nonadherent to the medicine prescribed by their physician, particularly patients with chronic diseases. 23 Nonadherence to antiepileptic medications has been reported to be high. Studies showed a high prevalence of seizure (21%-45%) in patients who did not adhere to their antiepileptic medications. 24 More than half of patients with epilepsy have poor seizure control due to nonadherence to medications. 25 More than 30% of people with epilepsy do not attain full seizure control even with the best available treatment regimen. Failure to have a controlled seizure in such a significant proportion of patients with epilepsy is attributed to poor adherence to medications. 26
The patients’ beliefs about the cause of epilepsy and preference to the treatment modality are important factors influencing epilepsy treatment. Patients’ attitudes toward the treatment are also equally important in ensuring the success of treatment and adherence. 27 Failure to adhere through forgetfulness, misunderstanding, or uncertainty about clinician’s recommendations, or intentionally due to their expectations of treatment, side effects, and lifestyle choices are found to be the reasons for nonadherence. 28
Therapeutic drug monitoring was not carried out in any of the cases studied. This is worrisome considering the high percentage of cases with poor seizure control. The measurement and interpretation of serum concentrations of AEDs can be of profound benefit in the treatment of uncontrollable seizures. Therapeutic drug monitoring enables a more decisive and effective optimization of therapy and disease management. 29 The lack of therapeutic drug monitoring as revealed in this study may be due to the pervasive problem of nonavailability of the required facilities to carry out such investigations, a problem that appears to be common in resource-poor settings.
Our study revealed a poor laboratory monitoring of the hematologic adverse effect of carbamazepine. Pretreatment monitoring of white blood cell and platelet counts was conducted in only 61% of the patients taking carbamazepine, while follow-up monitoring of blood counts was carried out in less than 4% of these patients. Aplastic anemia is considered to be the most worrisome idiosyncratic adverse reaction of carbamazepine. 30 Carbamazepine is also associated with dose-independent transient leucopenia, which occurs in about 10% of patients with carbamazepine therapy.30,31
The risk of serious hematologic reactions to carbamazepine can be minimized by patient education and laboratory monitoring. 31 It has been recommended that when carbamazepine therapy is initiated, patients should be counseled to seek urgent medical attention for the incidence of high fever, infection, petechiae, or unusual fatigue. Carbamazepine therapy should be discontinued immediately if the diagnosis of aplastic anemia is confirmed.30,31 Recommended laboratory monitoring in patients placed on carbamazepine therapy includes complete blood counts before initiation of treatment and every 2 months of therapy.30,31 In cases where no abnormalities are detected, hematological monitoring should continue at intervals of 3 months or when the patient develops signs or symptoms of myelosuppression. However, in cases where mild leucopenia develops, complete blood counts should be evaluated fortnightly until the blood counts return to baseline values. However, carbamazepine therapy should be discontinued if absolute neutrophil count drops below 1500/mm3 or if infection occurs. 31
Monitoring of liver function among patients taking valproate was less than optimal. About 40% of the patients placed on valproate therapy did not receive pretreatment evaluation of liver function. Furthermore, follow-up monitoring for potential hepatotoxic effects of valproate therapy was conducted in less than 4% of these patients. Reports show that approximately 40% of patients treated with valproate will experience a dose-related increase in serum transaminase. This abnormality is usually asymptomatic and responds rapidly to dose reduction or discontinuation of drug therapy.30,32
It is recommended that physicians discontinue valproate therapy in cases where there is an elevation in hepatic enzymes 3 times above baseline values, in cases of abnormalities in laboratory tests of hepatic synthesis or metabolism (such as elevated bilirubin or prothrombin time or decreased serum albumin concentration; and also in cases where the development of clinical symptoms of hepatitis is evident). Baseline laboratory values, including liver enzymes and hepatic function tests, should be determined before therapy is initiated.30,32
Before initiation of AED therapy, patients should be educated on the potential adverse effects. Patients should also be counseled on the need to seek medical attention immediately for early signs of adverse drug reactions such as somnolence with vomiting, rash, or bruising particularly in the first weeks following the commencement of AED therapy.
Conclusions
Carbamazepine and valproate were the most frequently prescribed AEDs. The extent of documentation of adverse drug reactions associated with the use of AEDs in the study sites is poor. The extent of monitoring of the hematologic effects of carbamazepine, as well as the hepatic effects of valproate in the cohort studied, is poor. This poses a great risk to the patients. There is a need for the implementation of suitable interventions geared toward promoting the safe use of these drugs among patients with epilepsy.
Footnotes
Funding:
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interest:
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
This work was carried out in collaboration among all authors. UIE designed the study, performed the statistical analysis, wrote the protocol and wrote the first draft of the manuscript. CMU and CVU managed the analyses of the study and the literature searches. All authors read and approved the final.
