Abstract
Aim of the Study:
Parkinson’s disease and schizophrenia are disease end points of dopaminergic deficit and hyperactivity, respectively, in the mid brain. Accordingly, current medications aim to restore normal dopamine levels, overshooting of which results in adverse effects of psychosis and extra-pyramidal symptoms, respectively. There are currently no available laboratory tests to guide treatment decisions or help predict adverse side effects of the drugs. The aim was to therefore explore the possibility of using apolipoprotein E as a biomarker to monitor pharmacological intervention in dopamine dictated states of Parkinson’s disease and schizophrenia for optimum therapy.
Methods:
Naïve and treated, Parkinson’s disease and schizophrenic patients were recruited from neurology and psychiatry clinics. Serum of healthy volunteers was collected as controls. Serum concentrations of apolipoprotein E was estimated by enzyme-linked immunosorbent assay (ELISA). Pathway analysis was carried out to delineate the interactions of apolipoprotein E in Parkinson’s disease and schizophrenia.
Results:
Apolipoprotein E levels are higher in Parkinson’s disease patients as compared with schizophrenic samples (P < .05). Also, post-treatment apolipoprotein E levels in both disease states were at par with levels seen in healthy controls. The interactions of apolipoprotein E validate the results and place the differential expression of the protein in Parkinson’s disease and schizophrenia in the right perspective.
Conclusion:
Apolipoprotein E concentration across the dopaminergic spectrum suggests that it can be pursued not only as a potential biomarker in schizophrenia and Parkinson’s disease, but can also be an effective tool for clinicians to determine efficacy of drug-based therapy.
Keywords
Introduction
Parkinson’s disease and schizophrenia are neurological diseases that are clinically very diverse, but are etiopathologically related to neurotransmitter-dopamine. While motor symptoms of Parkinson’s disease result from death of dopamine-generating neurons, psychotic symptoms of schizophrenia are related to dopaminergic hyperactivity.1,2 Accordingly, pharmacological medications and response in Parkinson’s disease and schizophrenia revolve around the levels of dopamine in brain. 3 Pharmacological interventions in Parkinson’s disease aim to increase availability of dopamine in neurons of brain, while therapy in cases of schizophrenia includes medications that reduce activity of dopamine as a signal between neurons in the brain. Therefore, during the course of Parkinson’s disease therapy, dopamine levels tend to increase above normal levels leading to symptoms of schizophrenia. 4 Likewise, the problem in using neuroleptic drugs in treatment of schizophrenia is that neuroleptic drugs lower the amounts of dopamine and may produce extra-pyramidal side effects. 5 Although extra-pyramidal side effects are slightly reduced with the advent of second-generation anti-psychotic drugs, problems pertaining to side effects of medications continue to be reported across the world. 6
Pharmacological therapy in Parkinson’s disease and schizophrenia often causes overshooting of dopamine during therapy in patients causing complications. This results in patients succumbing to the side effects which is the disease at the other end of the dopaminergic spectrum. 7 There are currently no available laboratory tests to guide treatment decisions or help predict these adverse side effects of the drugs. Clinicians are currently forced to rely completely on patient compliance and symptoms to understand the efficacy of drugs administered.8,9 This causes a lot of inconvenience to patients, patient attendants, and clinicians. This drawback also has an indirect impact on the financial expenditure for the patient, hospital, and the state.
Apolipoprotein E is a lipoprotein of 299 amino acids and expressed in the brain by glia, macrophages, and neurons. 10 Apolipoprotein E exists mainly as a component of lipoprotein complexes along with other apolipoproteins in serum and cerebrospinal fluid. 11 Apolipoprotein E is relevant in brain physiology, neuro-degenerative disorders, and schizophrenia.10–19 Although apolipoprotein E is better known as a diagnostic marker, its value as a therapeutic biomarker in varying phenotypic patients across the brain dopaminergic spectrum has not been investigated. In this study, we explore the possibility of using apolipoprotein E as a biomarker to optimize pharmacological intervention in dopamine dictated states of Parkinson’s disease and schizophrenia. This would help clinicians to titre drug levels, alter the dose, change the drug, and counsel the patient for the imminent side effects.
Methods
Ethics, consent, sample collection
The study was approved by the ethics committee of All India Institute of Medical Sciences, New Delhi (reference: IEC/NP-535/4.11.2013) and procedures followed were in accordance with the ethical standards formulated in the Helsinki declaration. Patients were screened at neurology and psychiatry clinics at the institute as per criteria explained below. Intravenous blood (5 mL) of Parkinson’s disease patients and schizophrenia patients was collected in blood collection tube, allowed to coagulate, centrifuged at 4000 rpm for 5 min at 4°C and supernatant serum was carefully aspirated out and collected in fresh tubes. These were stored at −80°C until further use.
Patient inclusion and exclusion criteria
Inclusion criteria
Patients suspected of Parkinson’s disease were screened based on Unified Parkinson’s disease Rating Scale (UPDRS). While a score of 0 represents no disability, a score of 199 on the UPDRS scale represents complete disability. 20 international classification of diseases (ICD-10) scale was used to score the extent and progression of schizophrenia. 21
Exclusion criteria
Patients with disease and other co-existing pathology were not included for the sample collection.
Enzyme-linked immunosorbent assay
Serum concentration of apolipoprotein E was estimated using ELISA Kit from RayBiotech (RayBiotech Inc., Norcross, GA, USA). The methodology used was as per the manufacturer’s instruction protocol. Concentrations of apolipoprotein E were extrapolated from the standard curve. The detectable range of apolipoprotein E was 1.5 to 400 ng/mL. Two technical replicates of each sample were done twice. The values were plotted on a graph using GraphPad Prism 6 (GraphPad Software Inc., San Diego, CA, USA).
Statistical analysis
Data analysis was carried out using statistical software SPSS IBM version 20.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics such as mean, median, standard deviation, and range values were calculated. More than two group medians were compared using Kruskal-Wallis test. Frequencies of categorical variables were compared using chi-square. Bi-variate Spearman rank correlation coefficient was calculated to study variables of age and disease score on apolipoprotein E concentrations. Mann-Whitney U test was used to compare median values of apolipoprotein E between the two genders. For all statistical tests, a two-sided probability of P < .05 was considered as statistically significant. To rule out age as a confounding factor, univariable logistic regression analysis was carried out by taking disease status as a dependent variable and age and apolipoprotein E as independent variables.
Pathway analysis
Freely available data sources like UniProt, Gene, and PubMed databases were used to retrieve the information on the genes corresponding to identified proteins relating to the Parkinson’s disease and schizophrenia. The gene interactions in Parkinson’s disease and schizophrenia pathways were extracted using KEGG and Schizo-Pi database. 22 The Cytoscape v2.8.0 software was used to visualize the interaction and pathways of identified proteins and its interactions.23,24 The Michigan Molecular Interactions (MiMI) plug-in tool on Cytoscape was used to gather the human gene regulatory interactome extracted from the public databases including STRING, MINT, MENTHA, and HPRD to merge the information.25–30 From this complete network, sub-networks for Parkinson’s disease and schizophrenia were extracted up to the first neighbouring nodes using the plug-in BiNoM v2.5. The resulting networks were merged using Cytoscape. Venn/Euler diagram was used to delineate unique and common pathways between Parkinson’s disease and schizophrenia.
Results
Clinical profile
A total of 52 serum samples from 23 Parkinson’s disease patients, 17 schizophrenic patients and 12 normal individuals were collected. The patient profile details are provided in Tables 1 to 3. Among the Parkinson’s disease patient samples, 5 patients were naïve, and 18 of them received dopaminergic treatment for at least 2.8 years duration. Out of 17 schizophrenic patients, 5 were drug naïve and rest 12 were on anti-dopaminergic therapy. 12 serum samples of volunteers were collected as healthy controls.
Profile of serum samples collected from Parkinson’s disease patients.

Abbreviation: UPDRS, Unified Parkinson’s disease Rating Scale.
Profile of serum samples collected from schizophrenia patients.

Abbreviation: BPRS, brief psychiatric rating scale.
Profile of serum samples from healthy volunteers.

Demographic and statistical analysis
Parkinson’s disease and the schizophrenia groups had higher male participants as compared with females (Table 4). This is as per the fact that incidence of these two dopamine dictated clinical states largely affects the male population. 31 The mean and median age of Parkinson’s disease group and schizophrenia groups were varying significantly in the study. It may however be noted that the incidence of Parkinson’s disease rapidly increases over the age of 60 years, with only 4% of them being under the age of 50 years. 32 Whereas, the incidence of schizophrenia is at the age of 16 to 25 years. 33 In this study, the mean age of 23 Parkinson’s disease patients is 56 years and the mean age of 17 schizophrenia patients is 30.8 years (Table 5).
Gender distribution of patients in the three study groups.

Age profile of patients in the three study groups.

Enzyme-linked immunosorbent assay
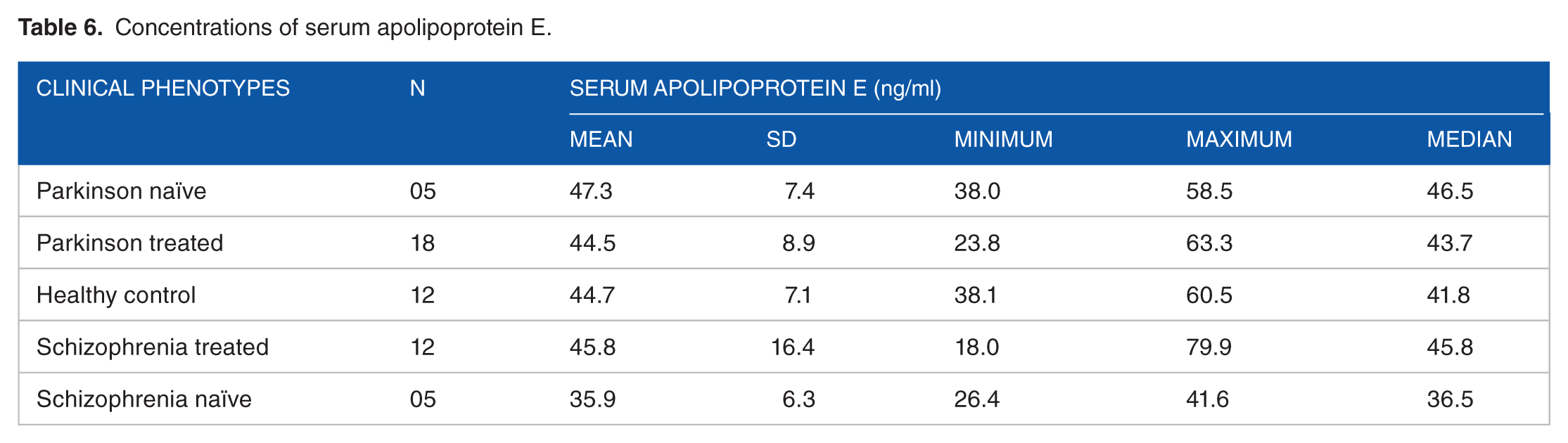
Enzyme-linked immunosorbent assay (ELISA) results on the 52 serum samples are shown in Figure 1. The results indicate higher apolipoprotein E levels in the group comprising of naїve Parkinson’s disease patients as compared with naïve schizophrenic samples (P = .03). Also, apolipoprotein E levels exhibit a post-treatment change, in both Parkinson’s disease and schizophrenic patients. Interestingly, the mean of apolipoprotein E in the treated patients is almost equal to the levels that are observed in healthy controls (Table 6). There is therefore a linear inverse pattern in apolipoprotein E concentrations across the dopaminergic spectrum of Parkinson’s disease, healthy controls, and schizophrenia. Although age may be seen as a confounding factor, age-matched analysis had no bearing on the correlation between apolipoprotein E and age (Table 7). Apolipoprotein E concentrations for males and females in Parkinson group were 45.8 and 43.2 ng/mL, respectively, while the apolipoprotein E concentrations for males and females in schizophrenia were estimated to be 41.6 and 36 ng/mL, respectively. The distribution of patients by gender, duration of disease, and extent of disease for scoring had no bearing on the results pertaining to apolipoprotein E expression among the phenotypes (Table 7). After adjusting for age, the adjusted odds ratio for apolipoprotein E being associated with Parkinson’s disease is 1.03 (P = .73), and with schizophrenia is 1.2 (P = .88).

Scatter plot showing the levels of apolipoprotein E (mean ± SD) in serum. The concentrations plotted are the average of duplicate readings of each sample. Diagrammatic representation of the dopamine concentration in cerebrospinal fluid is shown along the x-axis.34,35 Kruskal-Wallis non-parametric test was performed.
Concentrations of serum apolipoprotein E.
Spearman rank correlation between apolipoprotein E concentrations and disease scoring, gender, and age.

Apolipoprotein E interactions and pathway analysis
A total of 51 proteins are shown to be interacting with apolipoprotein E (Figure 2). Of these, 15 are unique to Parkinson’s disease, 21 are unique to schizophrenia, and the rest 15 are common to both pathways. Apolipoprotein E directly interacts with 3 proteins in Parkinson’s disease network, 4 proteins in schizophrenia network, and 7 proteins in the common pathway.

Pathway analysis shows apolipoprotein E along with its respective interactions in Parkinson’s disease and schizophrenia pathway. Apolipoprotein E is shown in white node, its interacting nodes in Parkinson’s disease pathway are highlighted in green, interacting nodes in schizophrenia pathway are highlighted in pink, and nodes that are common to both the pathways are in blue colour. First node interactions are shown circled by bold lines; and interactions from there on are shown circled by normal lines. Interactions that are connecting the nodes are shown by grey lines.
Discussion
The present study focuses on the serum apolipoprotein E expression profiles of naïve and pharmacologically treated patients with Parkinson’s disease and schizophrenia. ELISA experiment clearly establishes apolipoprotein E to be having higher level of expression in Parkinson’s disease as compared with schizophrenia. This result is in accordance with previous studies which have shown elevated levels of apolipoprotein E in the neurons and cerebro-spinal fluid of Parkinson’s disease patients, and a decreased expression of apolipoprotein E in the serum of schizophrenia patients.36–39 Even though adjusted odd ratio is more than 1, it is not statically significant. It is likely that it is due to sample size limitation. It is likely that a 47.3 ng/mL concentration of apolipoprotein E is indicative of Parkinson’s disease state and a 35.9 ng/mL concentration of apolipoprotein E is indicative of schizophrenia. This preliminary study is a good platform for further studies on a larger size cohort.
Apolipoprotein E has interactions with proteins that are implicated in crucial pathways in Parkinson’s disease and schizophrenia. Some of the interactions that are relevant from this study’s point of view are as follows: (1) low-density lipoprotein receptor-related protein (LRP) is an important receptor for apolipoprotein E in the brain and is responsible for apolipoprotein E trafficking in the neuronal cells. 36 LRP is co-expressed with Apo E since it is the primary receptor for lipidated form of Apo E; (2) voltage-dependent ion channel interaction affects intra-neuronal Ca2+ concentration, thereby affecting dopamine release to into the synapse in Parkinson’s disease; (3) α-synuclein is involved in aggregation to form inclusion bodies called as Lewy body and is a pathological hall mark of Parkinson’s disease.40–42 There is association between α-synuclein-related neurotoxicity and increased apolipoprotein E levels in Parkinson’s disease. 40
The neuro-transmitter dopamine, along with dopamine receptors and dopamine transporters (DATs), has a significant influence on dopaminergic neurotransmission in Parkinson’s disease and schizophrenia. 43 A pictorial depiction of dopaminergic system and its relation to apolipoprotein E is shown in Figure 3 and the corresponding discussion is provided below.
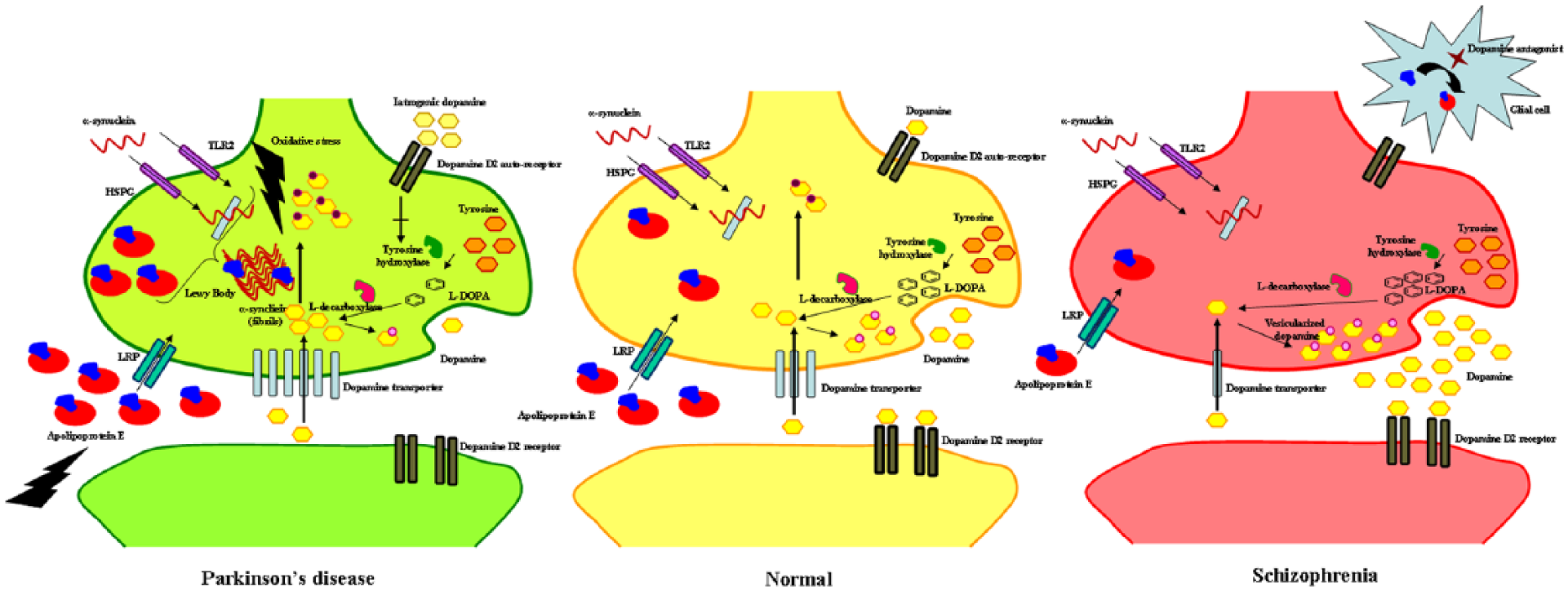
Diagrammatic representation of neuronal synapse depicting experimental result-based hypothesis that explain molecular events in Parkinson’s disease, neurological controls, and schizophrenia.
DAT is involved in uptake of dopamine from synaptic cleft to pre-synaptic cell. 44 Within the cell, dopamine is stored in vesicles by vesicular monoamine transporter 2 (VMAT 2) or it is metabolized by monoamine oxidase (MAO). 45 Exogenous dopamine is involved in activation of dopamine D2 auto-receptor causing inhibition of tyrosine hydroxylase, which is a rate-limiting enzyme in dopamine synthesis pathway. 46 Soluble α-synuclein, a regulator of dopamine, enters the cell through toll-like receptor 2 (TLR2) and heparan sulphate proteoglycan (HSPG).47,48 This soluble α-synuclein binds to DAT, thereby reducing the number of DAT molecules present on the plasma membrane for the uptake of dopamine from the synaptic cleft. 49 In Parkinson’s disease, aggregated α-synuclein in the form of Lewy body does not bind to intra-cellular DAT, thereby increasing dopamine uptake by DAT that is localised on the plasma membrane. 50 VMAT2 which is involved in vesicular uptake of dopamine is down-regulated in Parkinson’s disease and therefore, there is less vesicular uptake of dopamine resulting in higher concentration of free dopamine in cytoplasm. 51 This free dopamine is rapidly oxidized resulting in formation of cytotoxic dopamine metabolites and reactive oxygen species (ROS). 52 This triggers the production of apolipoprotein E which combats the oxidative stress and has a neuroprotective effect. 53 In addition, apolipoprotein E is an important cholesterol transporter, which interacts with the cholesterol-binding domains of α-synuclein facilitating its aggregation.54,55 Iatrogenic dopamine activates the dopamine D2 auto-receptor to cause inhibition of tyrosine hydroxylase, thereby decreasing the availability of free dopamine for production of ROS in the cytosol. 56 This explains the normalization of apolipoprotein E in treated Parkinson’s disease group as seen in our ELISA experiment. In case of schizophrenia, increased dopaminergic activity at the post-synaptic membrane causes psychotic symptoms in schizophrenia. 57 Anti-dopaminergic drugs given as therapy in schizophrenia up-regulate the expression of apolipoprotein E by activation of sterol regulatory element-binding protein (SREBP) transcription factors through liver-X-receptor (LXR) pathway in the glial cells.58,59 Apolipoprotein E is then taken up by neuronal cells by LRP.36,60 This explains the increase in apolipoprotein E in schizophrenic patients who received pharmacotherapy.
Limitations of the study
(1) Low recruitment of naïve patients and (2) difficulty in procuring cerebrospinal fluid for experiments.
Conclusion
Serum apolipoprotein E has an inverse correlation along the clinical phenotypes of low dopaminergic Parkinson’s disease state to high dopaminergic schizophrenia state. The dynamic variation of this protein along the spectrum is ideal for them to be pursued as pharmacotherapeutic biomarkers to monitor pharmacological efficacy in Parkinson’s disease and schizophrenia. Detailed pathway analysis convincingly establishes plausible hypothesis for its regulation in the pathogenesis of Parkinson’s disease and schizophrenia. This study will be of immense help to clinicians and patients to monitor and improve efficacy of pharmacotherapy in Parkinson’s disease and schizophrenia.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Department of Science and Technology, Government of India: SO/BB-0122/2013.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AKG and KR contributed equally to this work.
