Abstract
Background:
Knowledge of clinically established factors of physical function such as body composition, bioelectrical phase angle (PhA) and handgrip strength (HGS) with mortality predictive and health-related relevance is limited in prostate cancer survivors (PCS). Therefore, the aim of this study was to characterise and compare body composition data of PCS with extensive reference data as well as to analyse PhA and HGS and the prevalence of critical prognostic values at an early stage of cancer survivorship.
Methods:
One hundred and forty-eight PCS were examined at the start (T1) and end (T2) of a 3-week hospitalised urooncological rehabilitation, which began median 28 days after acute cancer therapy. Examinations included a bioimpedance analysis and HGS test. Comparison of body composition between PCS and reference data was performed using bioimpedance vector analysis (BIVA).
Results:
BIVA of the whole PCS group showed abnormal physiology with a cachectic state and a state of overhydration/oedema, without significant changes between T1 and T2. The age- and BMI-stratified subgroup analysis showed that PCS aged 60 years and older had this abnormal pattern compared to the reference population. HGS (T1: 38.7 ± 8.9 vs T2: 40.8 ± 9.4, kg), but not PhA (T1/T2: 5.2 ± 0.7, °), changed significantly between T1 and T2. Values below a critical threshold reflecting a potentially higher risk of mortality and impaired function were found for PhA in 20% (T1) and 22% (T2) of PCS and in 41% (T1) and 29% (T2) for HGS.
Conclusions:
BIVA pattern and the prevalence of critically low HGS and PhA values illustrate the necessity for intensive continuation of rehabilitation and survivorship care especially in these ‘at risk’ cases. The routine assessment of body composition, PhA and HGS offer the opportunity to conduct a risk stratification for PCS and could help personalising and optimising treatment in rehabilitation and ongoing survivorship care.
Background
Prostate cancer is the most common solid organ cancer in men, with the highest incidence rates recorded in the developed countries of Oceania, North America and Europe.1,2 With a focus on mortality, prostate cancer is the fifth leading cause of death from cancer in men. 2 Despite the high incidence, prognosis and survival rates are good. 3 However, there are well-known side-effects of prostate cancer therapy like urinary incontinence, erectile dysfunction, secondary lymphoedema, loss of muscle strength, muscle mass and bone mineral density4-7 which finally lead to a deterioration of physical functioning. There is evidence that cancer patients with reduced physical function have a shorter life expectancy.8-10 However, every prostate cancer survivor (PCS) in Germany is entitled to take part in a 3-week hospitalised urooncological rehabilitation programme subsequent to acute cancer therapy which is the starting point of a longer-term survivorship care pathway for PCS. Beside coping with the disease, the treatment of disease- and therapy-associated functional limitations and psychological consequences as well as the improvement of reduced physical functioning and health-related quality of life are important rehabilitation goals. 11
In view of parameters of physical functioning handgrip strength (HGS) has been identified as a strong biomarker and also a powerful predictor of mortality.12-15 Body composition is also known as an important factor in physical functioning. Recently, the use of the raw impedance parameters resistance (R), reactance (Xc) and especially the phase angle (PhA) of bioimpedance analysis (BIA) has gained scientific attention. 16 These measures provide valuable information about the hydration status, body cell mass and cell membrane integrity without algorithm-inherent errors associated with the conventional BIA approach, a common scientific method for predicting body composition. The bioimpedance parameter PhA, which also has been shown to be of prognostic value (eg, mortality, disease progression, incidence of post-operative complications), can be interpreted as a global marker of health status and as a biomarker of physical function.16,17 Both are relevant components in the context of recovery, rehabilitation and general health outcome in cancer survivorship. However, the bioelectrical impedance vector analysis (BIVA), a method developed by Piccoli et al., 18 enables a more detailed understanding of body composition compared to PhA alone. 16 BIVA uses the plot of raw impedance data R and Xc normalised per height (H) as a bivariate vector in a RXc vector graph. For instance, a lower R leads to a shortening of the vector and thereby reflects an excess of fluid, such as occurs in the case of oedema. In contrast, a reduced hydration status and dehydration causes a longer vector. High or low Xc indicates an increase or decrease of dielectric mass (membranes and tissue interfaces) of soft tissues, which causes the vector to move up or down. Based on the calculation of PhA from Xc and R, different vector locations in the RXc graph can theoretically be shown with comparable PhA. For this reason, the use of bioimpedance PhA alone is limited in terms of a clear differentiation between athletic (high phase angle, long vector) and obese (high phase angle, short vector) as well as lean (normal phase angle, long vector) and cachectic (low phase angle, long vector) subjects. Longitudinal changes in body cell mass and hydration status can be interpreted much better with the BIVA than with PhA, which makes the BIVA a valuable tool for body composition assessment and monitoring of PCS. In the clinical context, the BIVA method has significant advantages over the conventional BIA approach because no algorithms for conversion of raw impedance data into body-composition compartments are required. Results are therefore not biassed by the choice of regression equation, the accuracy of the criterion method or the selection criteria of the reference population. 19 Nowadays, BIVA is a widely used technique in medicine in different clinical conditions such as sarcopenia and sarcopenic obesity, 20 heart failure, 21 diabetes, 22 renal disease, 23 obesity, 24 COPD, 25 anorexia nervosa 26 and cancer.27-31 Due to existing reference data in healthy populations for BIVA, 32 PhA 19 and HGS, 14 measures can be interpreted and classified in terms of their clinical relevance. For example, bioimpedance values within BIVA that fall outside the 75% tolerance ellipse of the reference population indicate an abnormal physiological situation32,33 and a standardised PhA that falls below the fifth reference percentile appears to be a simple and prognostic relevant cut-off point for impaired functional and nutritional status and increased mortality. 10
To date, however, no scientific data are available on BIVA, PhA and the prevalence of critical prognostic values of physical functional status of PCS in urooncological rehabilitation, reflecting an early stage in cancer survivorship. The first aim of this study was therefore to characterise and compare BIVA data from PCS at the start (T1) and the end (T2) of a 3-week hospitalised urooncological rehabilitation programme with extensive reference data. The second aim was to evaluate PhA, HGS and the prevalence of critical prognostic values in these biomarkers of physical function at T1 and T2.
Methods
Participants and procedures
This observational study with repeated measures was conducted within the Rehabilitation Hospital Vogtland-Klinik Bad Elster, Germany. Between March 2017 and March 2019 a total of 175 PCS were recruited within the research project ‘Return’, which was approved by the ethics committee of Chemnitz University of Technology (V-182-17-AS-Tumor-20012017) and registered at the German Clinical Trials Register (ID: DRKS00014263). All participating PCS gave their written informed consent and fulfilled the inclusion criteria – diagnosis of prostate cancer, no defibrillator or cardiac pacemaker and no orthopaedic restrictions for execution of tetrapolar BIA and HGS assessment. All PCS were evaluated at the start (T1) and the end (T2) of a hospitalised 3-week urooncological rehabilitation programme, which constitutes the standard inpatient cancer rehabilitation duration in Germany 34 before being referred to a community-based rehabilitation programme. The median time interval between admission to hospital for urooncological rehabilitation and the end of acute cancer therapy (surgery, radiotherapy, chemotherapy) was 28 days.
The standardised examination procedure at T1 and T2 began with a single tetrapolar BIA measurement of R and Xc (BIA 5 series multifrequency, EgoFit GmbH, Germany) taken on the subject’s right side between the right wrist and ankle in the supine position (after 10 minutes of supine rest) on a non-conductive surface at a fixed frequency of 50 kHz. Adhesive solid gel electrodes (Ambu® WhiteSensor 0315M, Denmark) were applied to defined anatomical sites on the dorsal surfaces of the hand, wrist, ankle and foot according to the manufacturer’s instructions as follows: the proximal edge of the wrist electrode was applied on an imaginary line at the styloid process of the ulna and the distal edge of the finger electrode was applied on an imaginary line from the centre of the metacarpophalangeal joints of the middle and index finger. The proximal edge of the ankle joint electrode was placed along a imaginary line through the highest points of the outer and inner ankle bones. The distal edge of the toe electrode was placed along an imaginary line through the middle of the metatarsophalangeal joints of the second and third toes. PhA of BIA was calculated by using the following equation:
The HGS test was conducted following the standardised Southampton protocol. 35 Each participant was seated comfortably in a standard chair without arm support, and with their hips flexed at 90° and feet resting on the floor. The elbow of the test arm was flexed to 90°, the forearm and wrist in neutral position, thumb facing upwards. Before the test, the hydraulic hand dynamometer (Baseline®, HIRes™, Gauge ER™, USA) was adjusted to the participant’s hand size so that the device was comfortable in the hand. The examiner supported the base of the dynamometer for testing. Following a demonstration of the protocol, the participant was asked to squeeze the handle with as much force as possible for 3 seconds. Three repeated trials were recorded for both hands with a rest period of at least 15 seconds between trials. The starting hand was chosen randomly with subsequent side-alternating measurements. Only the peak value of HGS assessment was used for statistical analysis. Only PCS with complete measurement data were included in the final analysis.
Data analysis
The statistical analyses were conducted using IBM SPSS statistics 26 (Chicago, IL, USA). Statistical group comparisons between T1 and T2 were performed with Student’s paired t-test. The effect size was calculated directly from the t-value and the number of participants using the formula:
BIVA was conducted with BIVA software (Piccoli A & Pastori G, Department of Medical and Surgical Sciences, University of Padova, Padova, Italy, 2002; available by E-mail:
The prognostically relevant fifth PhA reference percentile, identified by Norman et al., 10 served as a cut-off for detection of PCS at risk for impaired function and survival. In view of HGS the extensive normative data published by Steiber 14 were used as the chosen reference data in this study. Steiber 14 suggested a threshold to define a critically weak HGS associated with an increased mortality risk at values that lie ⩾1 standard deviation below the standardised mean HGS. This threshold was also used in the present study to identify PCS with a potential elevated mortality risk.
Results
One hundred and forty-eight PCS were fully assessed at T1 and T2. Table 1 shows the baseline demographics and clinical characteristics.
Baseline demographics and clinical characteristics of N = 148 prostate cancer survivors in urooncological rehabilitation.
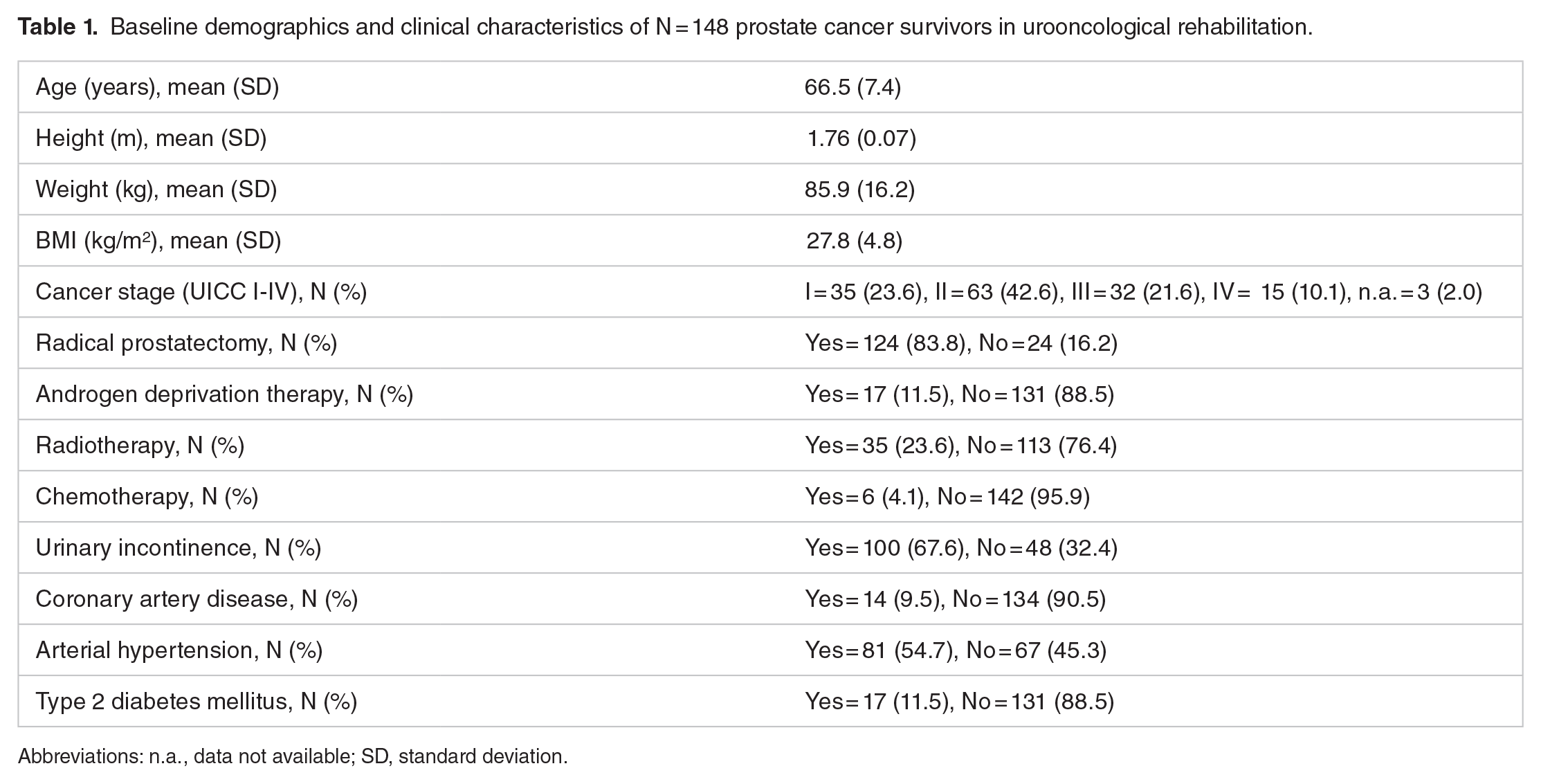
Abbreviations: n.a., data not available; SD, standard deviation.
Bioelectrical impedance vector analysis
The statistical comparison of raw impedance parameters and raw impedance parameters normalised for height at T1 and T2 is displayed in Table 2. Significant differences were found for R and R/H (both P < .05), whereas the effect size was small.
Raw data of bioimpedance analysis of prostate cancer survivors in urooncological rehabilitation.

Abbreviations: H, height; PhA, phase angle; R, resistance; T1, start of rehabilitation; T2, end of rehabilitation; Xc, reactance.
Bioimpedance values presented as mean (standard deviation).
Figure 1 shows the BIVA RXc graphs with 95% confidence ellipses for the total group and the age- and BMI-stratified subgroups of PCS in relation to the reference impedance vector with 50%, 75% and 95% tolerance ellipses. At T1 and T2 the total group of PCS presented a mean impedance vector with 95% confidence ellipses located outside the 95% tolerance ellipse in the cachectic quadrant and a state of overhydration/oedema. Based on the graphical overlapping, no significant difference was detected between T1 and T2. For reasons of clarity and because of no noteworthy graphical or statistical differences of the age- and BMI-stratified BIVA graphs between T1 and T2 data are only presented for T1. With focus on the different subgroups the BIVA RXc graphs show significant differences between PCS and the reference population (Figure 1). No significant differences could be found in PCS between the different age groups. PCS from the age of 60 years and above with BMI of 19 to 25 kg/m2 or >25 to 30 kg/m2 were located outside the 75% tolerance ellipse in the cachectic quadrant and a state of over-hydration/oedema. All age groups of PCS with BMI >30 to 35 kg/m2 showed an impedance vector position outside the 75% tolerance ellipse.

The BIVA RXc graphs of (A) the total group of prostate cancer survivors (PCS) at the start (T1) and the end (T2) of urooncological rehabilitation, (B) PCS with BMI 19 to 25 kg/m2 stratified by age, (C) PCS with BMI >25 to 30 kg/m2 stratified by age and (D) PCS with BMI >30 to 35 kg/m2 stratified by age. All reference graphs are based on data of Piccoli et al 32 with sex-specific 50%, 75% and 95% tolerance ellipses (grey). Optimal body composition is located at the centre (within 50% and 75% tolerance ellipses). Bioimpedance values that fall outside the 75% tolerance ellipse of the reference population indicate an abnormal physiological situation.
Biomarkers for physical function
Data of PhA and handgrip strength are presented in Table 3.
Biomarkers of physical function of prostate cancer survivors in urooncological rehabilitation.
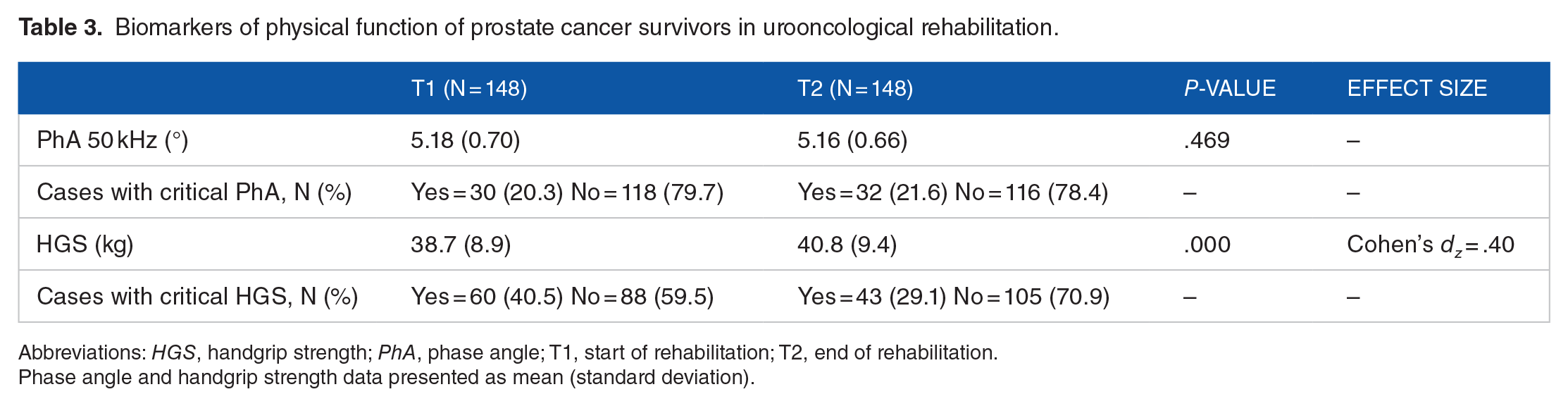
Abbreviations: HGS, handgrip strength; PhA, phase angle; T1, start of rehabilitation; T2, end of rehabilitation.
Phase angle and handgrip strength data presented as mean (standard deviation).
Between T1 and T2 a significant improvement, with a small effect size, was obtained for handgrip strength (P < .001) but not for PhA. Small to medium correlations were found between PhA and handgrip strength at T1 (r = .41, P < .001) and T2 (r = .23, P < .01). In 20% to 22% of PCS a critical PhA was detected at T1 and T2. At T1, 41% of PCS presented a critical handgrip strength, which decreased to 29% at T2. Thirteen percent of PCS showed a combination of both critical conditions (PhA and handgrip strength) at T1 which decreased to 9% at T2.
Discussion
In the present study, for the first-time, PCS were examined with regard to their BIVA pattern and PhA in the early stage of cancer survivorship. Our main findings provide evidence that BIVA data of PCS in inpatient urooncological rehabilitation differ significantly from population-based reference data, with no statistical significance (within-group) change over time. Independently of the BMI BIVA values of PCS from the age of 60 years felt outside the BIVA reference ellipse of 75% (indicating an abnormal physiology), whereas the mean vector was situated in the cachectic quadrant (low body cell mass) and a state of fluid overload (apparent oedema). These findings can be explained by several factors. Firstly, prostate cancer specific treatments like radical prostatectomy, radiation therapy and androgen deprivation therapy (ADT) may cause secondary lymphoedema. Secondly, many of the PCS in the present study suffered from comorbidities from the cardiovascular system (eg, arterial hypertension in 55% of PCS), which necessitate the use of potentially oedema-causing drugs. For instance, 22% of PCS took calcium channel blockers (eg, Amlodipine) which are used to treat high blood pressure and cause oedema by increasing capillary filtration of fluid and may reduce lymphatic drainage. 37 The lack of a statistically relevant change in the RXc graph over time could possibly be explained by the short duration of the rehabilitation hospital stay, which is probably too short for corresponding adaptations of the fluid load or dielectric mass of soft tissues. In view of this, various studies showed a change in the R and Xc values with respect to PhA after 12 weeks of progressive resistance training in older woman.38,39 However, in order to avoid post-operative complications due to the short time difference of approx. 1 month between surgery (radical prostatectomy in 84%) and onset of rehabilitation, the majority of global muscle strengthening exercises as part of the medical exercise therapy in 3-week urooncological rehabilitation can only be carried out with a low load. Ongoing ADT may also have contributed to this. In this context, a meta-analysis by Chen et al. 40 provided evidence that low- to moderate-intensity resistance and aerobic training is effective for improvement of muscle strength but may not be sufficient for affecting muscle mass. Another reason could be the chronic use of medication with oedema-causing effects (eg, calcium channel blockers, nonsteroidal anti-inflammatory drugs, corticosteroids, anti-depressants) which maintained the oedema state during the rehabilitation phase.
PCS in the present study showed a mean PhA of 5.2° at the start and the end of the 3-week urooncological rehabilitation whereas a fifth of PCS presented a value below the fifth percentile of sex-, age- and BMI-stratified PhA reference values published by Bosy-Westphal et al. 19 Norman et al. 10 found that patients with a solid or haematological tumour disease which showed a PhA below the fifth reference percentile exhibited a significantly higher 6-month mortality risk (OR: 4.0; 95% CI: 2.4, 6.8; P < .001) and a 37.4% probability of death. Referring to the data of Bosy-Westphal et al., 19 PhA in healthy adults usually range between 5° and 7° on average, depending on their age and BMI. Barbosa-Silva et al. 41 presented population-specific PhA reference values for men above 6°. In summary, PCS in the present study on average demonstrated a PhA at the lower spectrum of available reference values which reflects lower cellularity, cell membrane integrity and poorer cell function. However, further research is needed to evaluate if the PhA could also be increased in PCS by longer term rehabilitation interventions in survivorship care (eg, strength training). Further studies should also focus on PhA as a potential prognostic indicator for an increased mortality risk in PCS, especially in those with an identified PhA below the fifth reference percentile.
In addition, PCS showed a significant improvement of HGS after rehabilitation, albeit the effect size was small. Referring to the threshold for critically weak HGS obtained by Steiber, 14 two fifths of PCS presented critical values at the beginning of inpatient rehabilitation. After completion, critical values were still present in almost a third of PCS. Based on survival analysis, Steiber 14 showed that men whose HGS fell below the critical threshold had an 86% greater risk of death within 8 years of the HGS test. Besides these findings numerous other studies consistently confirm that low HGS increases the risk for mortality.12,13,15 When considering the mean age of PCS in our study (66.5 years), the found HGS on average was lower than the mean value (44 kg) of the age-specific reference population (60-69 years) described by Steiber. 14
Our study has some limitations. We could not include a follow up analysis after the initial 3-week hospitalised urooncological rehabilitation programme. Therefore, our results can only give an early picture of BIVA pattern and physical functional status in the rehabilitation process and survivorship pathway. Another limitation was the small sample size in some age and BMI related subgroups. Studies with larger sample sizes are necessary to confirm the current results. Moreover, because of a missing control group our study design does not allow to draw firm conclusions about the effectiveness of urooncological rehabilitation.
Conclusions
This study presents initial findings about a BIVA pattern in PCS and the occurrence of critical prognostic values of physical function at an early stage of cancer survivorship. The BIVA pattern differ significantly from population-based reference data and indicate an abnormal physiology. Combined with the prevalence of critically low HGS and PhA values our findings illustrate the necessity for intensive continuation of rehabilitation and survivorship care especially in these ‘at risk’ cases. Moreover, our data has shown that an inpatient 3-week rehabilitation programme improves muscular strength but without affecting the body cell mass. The routine assessment of BIVA, PhA and handgrip strength offer the opportunity to conduct a risk stratification for PCS and could help personalising and optimising treatment in rehabilitation and ongoing survivorship care.
Supplemental Material
sj-docx-1-rpo-10.1177_11795727211064156 – Supplemental material for Bioelectrical Impedance Vector Pattern and Biomarkers of Physical Functioning of Prostate Cancer Survivors in Rehabilitation
Supplemental material, sj-docx-1-rpo-10.1177_11795727211064156 for Bioelectrical Impedance Vector Pattern and Biomarkers of Physical Functioning of Prostate Cancer Survivors in Rehabilitation by Alexander Stäuber, Marc Heydenreich, Peter R Wright, Steffen Großmann, Niklas Grusdat, Dirk-Henrik Zermann and Henry Schulz in Rehabilitation Process and Outcome
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The publication of this article was funded by Chemnitz University of Technology.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AS performed data management and analysis, manuscript writing and edition; MH performed data collection and data management; SG helped in data analysis; NG helped in manuscript editing; DHZ, PRW and HS provided editorial assistance and scientific oversight for the manuscript. All authors have read and approved the final manuscript.
Ethical Approval and Consent to Participate
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of University of Technology (V-182-17-AS-Tumor-20012017). Written informed consent was obtained from all individual participants included in this study.
Availability of Data and Materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
