Abstract
The objective of this study was to evaluate oral supplements with antioxidants in oocyte and embryonic quality in patients undergoing assisted reproductive treatments. A cohort of 120 patients was included in the study: 60 patients were included in the control group and 60 in the Seidivid group. The medication contained folic acid, myo-inositol, melatonin, and selenium (Seidivid) and was supplied for at least 2 months before the ovarian puncture. The results of the different cycles were compared, evaluating the number of follicles (9.67 vs 9.15, P = .584), the number of metaphase II (8.52 vs 8.53, P = .986), the number of embryos of A + B quality, the number of vitrified embryos (1.93 vs 2.26, P = .505), and biochemical (62.75% vs 66.00%, P = .83) and clinical pregnancy rates (52.94% vs 56.00%, P = .84). A significant fact in our study was that although the supplements with antioxidants were not statistically significant for some aspects, they were significant in terms of obtaining embryos of good quality (A + B).
Keywords
Introduction
Humans are aerobic organisms who use oxygen to perform vital reactions; free radicals, which can cause damage in the cells, 1 are produced as a result. These radicals can be accepted by reducing molecules for elimination, but sometimes, these molecules are saturated and unable to sufficiently accept these radicals, causing an increase in oxidative stress. 2
Reactive oxygen species (ROS) are necessary for essential physiological processes, but sometimes, their high presence can cause damage. 1 They can be associated with miscarriage, idiopathic recurrent miscarriage, and defects in embryogenesis. 3
In assisted reproductive treatment (ART), oocytes are not protected by the follicular fluid or cumulus cells that act protectively when the process occurs in vivo. 4 In the processes of chromosome segregation, extrusion of the polar body, fertilization, and embryonic division, a high amount of energy is used, and oxygen must be used to obtain it, which will produce free radicals. With age, our capacity to eliminate ROS decreases. Thus, considering that many of the techniques used in assisted reproduction are performed for older women, a probable increase in ROS and oxidative stress must be assumed in this group of patients. As a consequence, the mitochondrial DNA damage will increase, causing a reduction of adenosine triphosphate (ATP) production in oocytes that will also reduce oocyte and embryonic quality. 5
We know that the quality of oocytes starts to worsen immediately after ovulation, 6 causing an inflammatory process due to the production of cytokines and proteases that is associated with increased oxidative stress in oocyte maturation.7,8 After 8 hours of ovulation, oxidative stress increases due to so-called aging.6,7 The oocyte is responsible for remodeling the paternal, maternal, and embryonic development genome6,9,10; in addition, the oocyte is also in charge of ATP production and cell processes such as apoptosis in response to appropriate development signals. 11
It has been seen in infertile women that there is high oxidative stress in the follicular fluid. 7 This stress is associated with poor oocyte quality. 7
In recent years, the importance of oxidative stress in ART has become increasingly relevant, particularly for in vitro fertilization (IVF) techniques12,13 and intracytoplasmic sperm injection (ICSI). 14
One way to reduce the effects of oxidative stress is using antioxidants. 15 Several studies have attempted to demonstrate the improvement of female fertility by supplying antioxidants. 16 Some have advocated that the supply of certain antioxidants does improve outcomes in ART.17,18 However, several studies have obtained results similar to our study. Although antioxidants involve a significant improvement in embryonic quality, the remaining parameters tested are not statistically superior. 19
Objective
The primary objective is to evaluate whether an oral supplement containing melatonin, myo-inositol, folic acid, and selenium in women receiving cycles of IVF/ICSI can improve some parameters, specifically, in obtaining a higher number of metaphase II (MII).
The secondary objectives were to establish whether a higher number of follicles (≥16 mm on the day of the trigger shot), a higher number of embryos of good quality, and embryos for vitrification are obtained when using this supplement. These factors will achieve a higher biochemical and clinical pregnancy rate.
Materials and Methods
A double-blind, randomized, prospective study was performed on 120 patients meeting the criteria for performing IVF-ICSI treatment, according to the daily clinical practice protocols, who were divided into 2 groups of 60 patients (study group and control group). Informed consent was obtained from all participants before enrollment.
At the first visit, participants were randomized by the nursing staff at a 1:1 ratio into both treatment groups.
Treatment
The medication was based on the daily oral intake of 2 sachets (breakfast and dinner, before or after meals indistinctly) containing 2 g of myo-inositol, 0.975 mg of melatonin, 200 μg of folic acid, and 27.5 μg of selenium (Seidivid), for at least 2 months before the ovarian puncture.
Inclusion criteria
The inclusion criteria were as follows: age 18-41 years at the time of enrollment, body mass index (BMI) from 18 to 29 kg/m2, and normal ovulatory cycle of 24-35 days.
Exclusion criteria
The exclusion criteria were as follows: azoospermia; presence of anatomic abnormalities of the reproductive system that could interfere with implantation or pregnancy; other medical conditions causing ovulatory disorders, such as hyperprolactinemia, hypothyroidism, and adrenal hyperplasia; and hypersensitivity to human gonadotropin preparations or any of the medicines or food supplements used in the study.
Stimulation protocol
The stimulation protocol was performed according to the site criteria for each patient. Two types of protocols were permitted for the purpose of subsequent assessment as follows: short protocol—gonadotropin-releasing hormone (GnRH) antagonists and long protocol—GnRH agonists.
Ovarian stimulation
This stimulation was performed by daily subcutaneous injection of human recombinant follicle–stimulating hormone (follitropin alfa/Gonal F) and/or highly purified human menopausal gonadotropin (hMG-HP/Menopur).
The starting dose ranged from 150 to 300 IU/d and was adjusted on the basis of follicular development, always after a sixth day of stimulation and with a maximum variation of 75 IU for each control. The controls for the sixth day were scheduled according to the daily clinical practice.
Pituitary suppression
Short protocol. Use of GnRH antagonists (Ganirelix 0.25 mg subcutaneous/Orgalutrán) from the day at which follicles were greater than 14 mm in size and until the day before oocyte maturation.
Long protocol. Use of GnRH agonists (nafarelin acetate, 200 μg/12 h intranasal/Synarel), starting in the luteal phase of the cycle prior to the stimulation cycle and until the day before oocyte maturation.
Triggering of ovulation
The oocyte maturation criterion was the ultrasonographical evidence of at least 3 follicles greater than 17 mm. If there were fewer than 15 follicles on the day on which stimulation ended, an injection of 250 μg of recombinant human chorionic gonadotropin was administered subcutaneously (Ovitrelle). If there were more than 15 follicles, an injection of 0.3 mg of triptorelin subcutaneous (Decapeptyl Diario 0.1 mg) was administered, and the transfer was delayed.
The evaluation of the total number of follicles included in the study was performed on the day of human chorionic gonadotropin administration.
Luteal phase treatment
From the night of the day of puncture until at least 7 weeks after transfer, the patient used 200 mg of natural micronized progesterone by vaginal route (Utrogestan 200), which was administered 3 times throughout the day.
Oocyte retrieval
Oocyte retrieval was performed by ultrasound-guided vaginal route between 34 and 38 hours after oocyte maturation and under anesthesia with sedation. The day of oocyte retrieval, another study objective—the total number of oocytes—was evaluated.
Laboratory procedures
The evaluations of oocyte and embryo quality were performed according to the standard operating procedure of the Ginemed laboratory of clinical reproduction.
After 2 to 3 hours of oocyte retrieval, oocyte denudation was performed with hyaluronidase. A count of MII available was performed, and an ICSI was performed at 4 hours after puncture.
At 16-21 hours after microinjection, the first assessment was performed to evaluate embryonic fertilization, which provided another of the secondary study variables, the fertilization rate (total fertilized/total microinjected oocytes).
Subsequently, the embryos were evaluated every 24 hours until the time of transfer was decided; at that time, another study variable—embryo quality—was evaluated.
The embryonic classification criteria were those of ASEBIR (Asebir, 3rd edition, 2015).
Embryonic transfer
Embryo transfer was performed from 2 to 5 days after oocyte retrieval. The number of embryos transferred ranged from 1 to 3, according to the medical criterion and according to the patient’s wish.
In the cases where vitrification of all embryos for subsequent transfer in a deferred natural or substituted cycle was considered to be adequate as the medical criterion, the vitrified embryos considered in the study were those remaining after performing the first transfer to the patient, including those not devitrified, if any, and those that should be revitrified after the transfer.
Vitrification of embryos
Unused embryos of quality A, B, or C were vitrified; those of quality D were allowed to progress and were only vitrified if they reached the blastocyst stage.
Confirmation of pregnancy
The serum pregnancy test (biochemical pregnancy) was performed 12 to 15 days after the embryonic transfer using a blood test. All tests yielding a result above 50 mU/mL were considered positive. This test represents one aspect of the study’s assessment data.
Follow-up of pregnancy
All patients with a positive pregnancy test underwent a confirmation transvaginal ultrasonography 5 to 6 weeks after oocyte maturation to evaluate the number of gestational sacs and fetal cardiac activity (clinical pregnancy); this procedure evaluated another of the secondary objectives of the study.
Subsequently, pregnancy was monitored by telephone controls, including data from abortions, complications, and malformations.
Statistical analyses
A descriptive analysis was performed of the study data by preparing frequency tables for the nominal variables and central and dispersion trend measures for continuous variables. The 95% confidence intervals were estimated in the case of the latter.
The comparison between qualitative variables was performed using the Fisher exact test, and the comparison between groups and quantitative variables was performed by the Student t test.
In the cases where comparison between continuous variables was necessary by adjusting for baseline assessments, the analysis of variance test for paired data was used.
For all tests, a significance level of P < .05 was established.
This statistical analysis was performed by the following independent site: CROSSDATA, Punta Alta Enric Granados, Barcelona.
Results
Both study groups were homogeneous in terms of eligibility criteria (Table 1). For statistical purposes, 9 patients from the control group and 10 from the Seidivid group were excluded due to noncompliance with the proposed protocol, providing a number of 51 in the control group and 50 in the Seidivid group.
Homogeneous characteristics of patients.
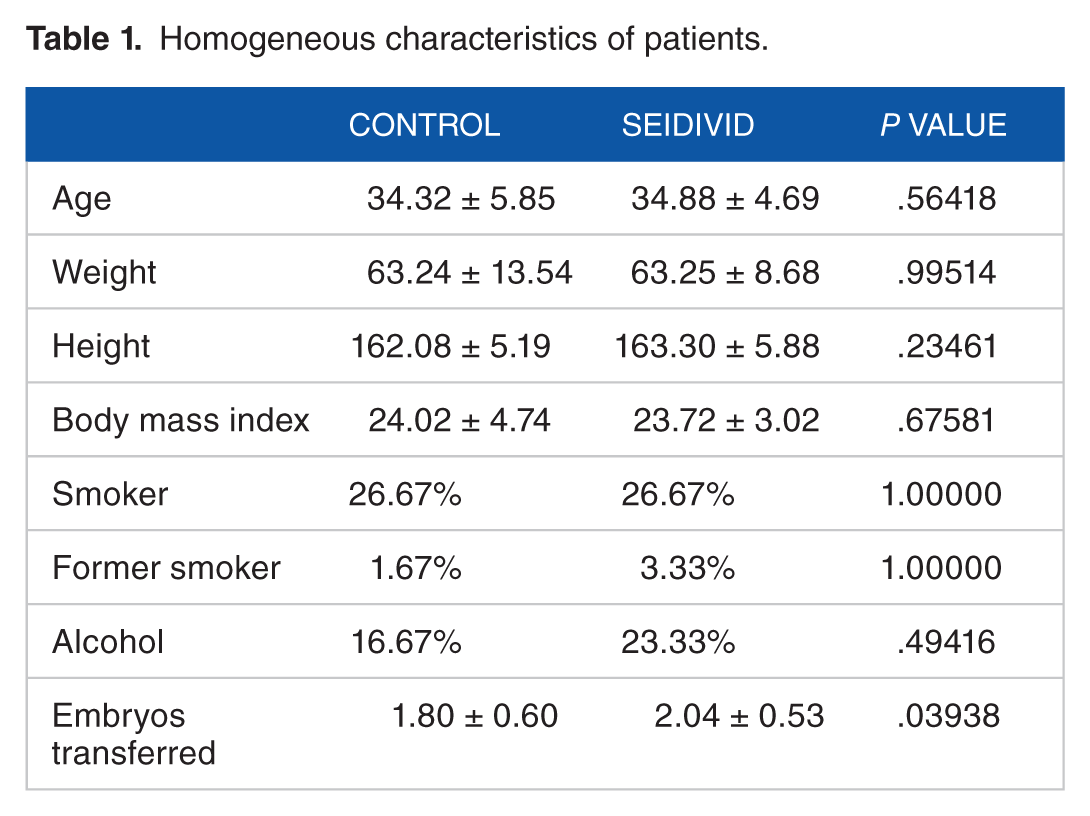
No statistically significant differences were found between the Seidivid and the control groups in terms of number of MII obtained (8.53 vs 8.52, P = .986), number of follicles ≥16 mm on the day of end of stimulation (9.15 vs 9.67, P = .584), or number of total ovules taken in puncture (8.65 vs 8.82, P = .874) (Table 2).
Results in follicle count, number of ovules obtained, stage of the ovules, and embryonic quality.
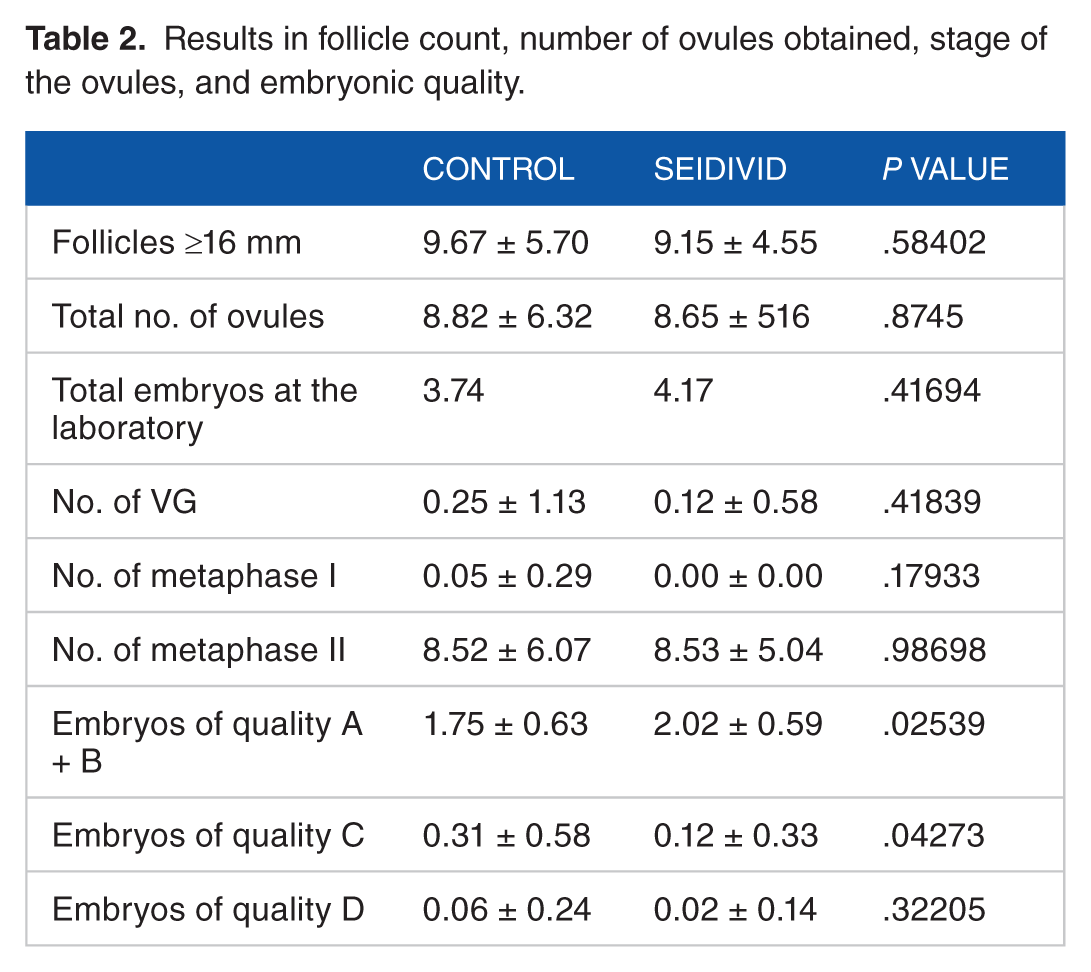
No statistically significant differences were found in the total number of embryos at the laboratory (4.17 vs 3.74, P = .41694) (Table 2), embryos vitrified (2.26 vs 1.93, P = .505), vitrified embryos of A + B quality (2.16 vs 1.72, P = .3341), C quality (1.08 vs 1.20, P = .66229), and D quality (0.10 vs 0.22, P = .26155) (Table 3).
Embryos vitrified by qualities and total number of embryos.
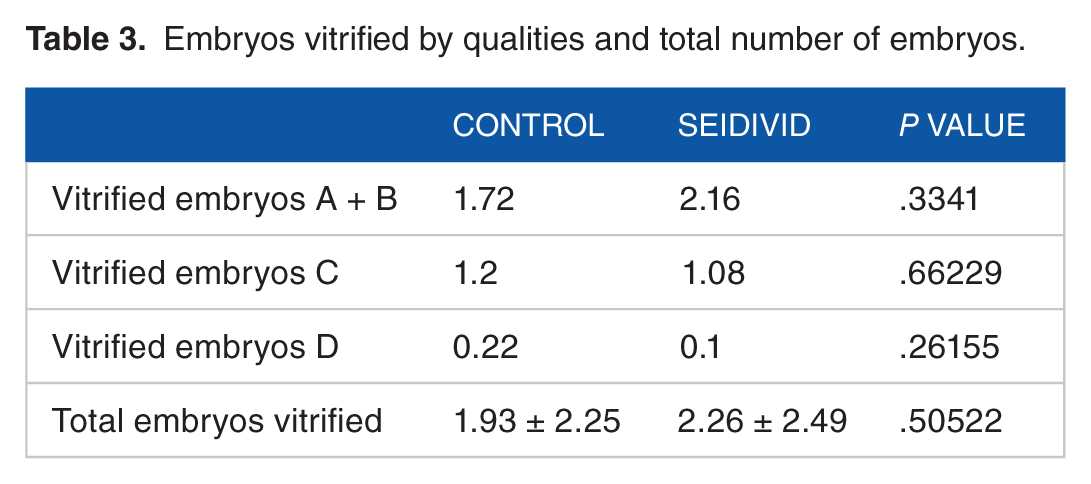
However, we did obtain statistically significant results in obtaining embryos of good quality (2.16 vs 1.72, P = .02539).
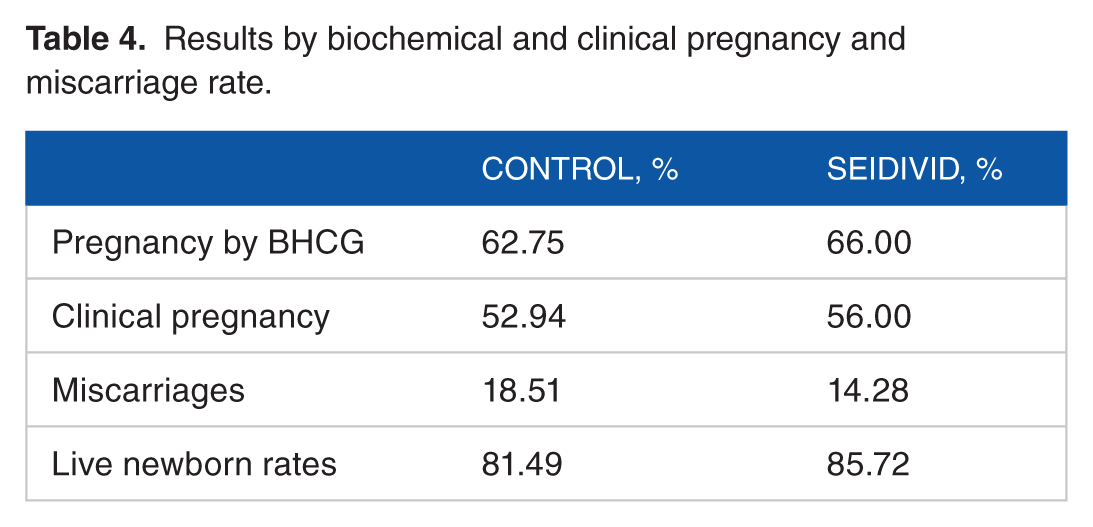
In terms of biochemical and clinical pregnancy, an increased biochemical pregnancy rate (BHCG) (66% vs 62.75%, P = .83) and increased clinical pregnancy rate (LF+) (56.00% vs 52.94%) were shown. Miscarriage rates (18.34% vs 19.21%, P = .64) and live newborn rates (LNB) (81.49% vs 85.72) are shown in Table 4.
Results by biochemical and clinical pregnancy and miscarriage rate.
Discussion
Antioxidants can be classified into 2 types: enzymatic and nonenzymatic. 20 Enzymatic antioxidants are produced endogenously to eliminate oxidative stress; the main enzymatic antioxidants are superoxide dismutase, catalase, and glutathione peroxidase (GPx). They are in charge of detoxifying any excess of oxygen-free radicals by the balance between oxidants/antioxidants.
Regarding nonenzymatic antioxidants, these antioxidants are provided in dietary supplements and contain a number of low-molecular-weight compounds, the most important being reduced glutathione, vitamins E, C, and A, together with flavonoids, phenolic acids, uric acid, bilirubin, some sugars, amino acids, coenzyme Q, ubiquinone, and several derivatives thereof. They also include a hormone, melatonin, and several minerals, such as selenium, copper, and iron, which are part of the active site of enzyme antioxidants, contributing to enzyme-mediated defense without being true antioxidants. 21
Melatonin is a hormone secreted by the pineal gland that naturally acts to eliminate ROS. Several studies have suggested that oral supplements containing melatonin have a direct influence on the improvement of oocyte and embryonic quality, protecting from oxidative stress by activating scavengers and stimulating antioxidant enzymes. 22 It must be considered that excessively high levels of melatonin could reverse these beneficial effects and result in toxicity. 13
Myo-inositol is an isoform of inositol that belongs to the vitamin B complex and is one of the precursors of the synthesis of phosphatidylinositol phosphates, biomolecules that belong to the translation signaling system involving the regulation of several cell processes, such as morphogenesis and cell cytogenesis, the formation of cell membrane, lipid synthesis, and cell growth. 23 Myo-inositol, in fact, is related to oocyte maturation and embryonic development.17,24,25
Myo-inositol plays an important role in the cytoskeleton and in the stabilization of chromatin, characteristics necessary for the correct maturation of the oocyte. It also plays an important role as a precursor of PtdIns involved in the Ca++ intracellular signaling pathway, which has been shown to be critical for proper oocytes maturation. In addition, inositols have a sensitizing effect on insulin. 18
It is thought that inositol plays a critical role in oocyte maturation and embryonic development. Previous studies have shown that oral supplements with inositol can reduce oxidative stress and improve stimulation protocols and pregnancy outcomes in nonfertile women with poor-quality oocytes.17,26
Selenium is a nonmetal chemical element that, despite being toxic at high concentrations, has several essential functions, including its antioxidant action, as it is present in each of the 4 catalytic centers of the GPx enzyme using glutathione to reduce peroxides, thus protecting membranes and other cell structures from the action of lipid peroxides and other free radicals. 27
Folic acid (the anionic form is called folate) is a vitamin that belongs to the B complex. It has coenzyme activity and is involved in the metabolism of several important amino acids, such as methionine synthesis. 28 In the cells, it is essential for DNA synthesis, which transmits genetic traits, and for RNA synthesis, which is necessary to build proteins and tissues and is vital to other cell processes. Therefore, the presence of folic acid in the body is essential for adequate cell division and duplication. 28
Supplementation with antioxidants to improve oocyte quality is a controversial subject that has not yet been fully resolved, and, regarding which, there are several opinions, both favorable18,26 and unfavorable. 29 With this study, we intended to establish the events occurring with a complex of antioxidants selected in the daily practice of a reproduction clinic.
Patient randomization into 2 groups was performed so that all had homogeneous characteristics (Table 1) for age, BMI, lifestyle, alcohol intake, and smoking. Groups were also homogeneous in terms of number of embryos transferred (Table 1).
The assessment of oocyte maturation remains an unexplored field; for the moment, the only indication we have to evaluate whether or not an oocyte is mature, that is, whether it is in the optimal stage to be fertilized, is by extrusion of the first polar body. In this stage, the oocyte is known as MII. 30
Our study intended to increase the number of MII in the group treated with Seidivid and indirectly increase the number of follicles greater than 16 mm (Table 2); however, no increase was seen in these variables. Therefore, antioxidant supply does not increase ovarian response and is therefore reserved to stimulation.
Oocyte quality, therefore, must be evaluated based on the effects of supplementation, that is, the embryo quality obtained. On evaluating the oocyte quality through embryonic quality, the bias caused by fertilization must be considered. We have found no differences in the fertilization rate or in the number of embryos obtained.
In contrast, a positive statistically significant effect is seen in relation to embryonic quality, and in the group of patients who have taken supplements with antioxidants, there is an improvement in embryonic quality, more embryos of type A + B according to ASEBIR classification and, therefore, a higher number of vitrified embryos of qualities A + B in the Seidivid group (Table 2).
On evaluating the results based on pregnancy rates, this difference is not marked, given the effect of embryo selection in the results in terms of biochemical and clinical pregnancy (Table 3). With the follow-up of the patients included in the study, we expect to provide further data on the effect of antioxidants in oocyte quality by assessing the existence of miscarriages and their possible relationship with the embryos transferred (Table 4), evaluating the final result shown in the total embryos per cycle (Table 2). The positive effects of the antioxidant supplementation are seen when there is an increase in clinical pregnancy rate and LNB (Table 4). In terms of results by miscarriage rate and LNB, no statistically significant differences are seen between the control group and the Seidivid group (Table 4). These groups are within the normal ranges and can be assigned to the risk of possible aneuploidies linked to the age of the participant. 31
Conclusions
After this study, it can be stated that oral supply of antioxidants does not significantly improve the results in ART (number of follicles, mature oocytes, good-quality vitrified embryos, and biochemical and clinical pregnancy). However, it significantly improves embryonic quality.
We consider appropriate to continue performing studies with a higher number of patients to evaluate the effects of supplying melatonin, myo-inositol, selenium, and folic acid in ART to continue progressing and include it as routine treatment in ART.
Footnotes
Acknowledgements
This article has been translated and corrected by American Journal Experts.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the funding provided by Laboratorio SEID S.A.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
FSM contributed to the conception and design, analysis and interpretation of data; JMJT, PPT, MGM, MHD, BMP, PSM, and FSM to the acquisition of data; and FSM and RS-B to the drafting the article, revision for intellectual content, and final approval of the completed article. All authors saw and approved the final version of the manuscript.
