Abstract
Neuroblastoma is the most common extracranial tumor derived from neural crest cells in childhood, and treatment of high-risk neuroblastoma is a difficulty in oncology field. The discovery of new treatment strategies to treat pediatric patients with high-risk neuroblastoma is important. Dinutuximab (ch14.18; Unituxin), a chimeric human-mouse monoclonal antibody, is approved by Food and Drug Administration in 2015 to be used specifically in the treatment of high-risk neuroblastoma. It binds the disialoganglioside (GD2) antigen on the surface of neuroblastoma cells and induces lysis of GD2-expressed neuroblastoma cells via antibody-dependent cell-mediated cytotoxicity and complement-dependent cytotoxicity. To enhance its activity, it is used with a combination of granulocyte-macrophage colony-stimulating factor, interleukin 2, and 13-cis-retinoic acid. In this review, we discuss the use of dinutuximab in the treatment of high-risk neuroblastoma.
Keywords
Introduction
Dinutuximab (Unituxin; United Therapeutics Corporation) is a drug that has received Food and Drug Administration approval in 2015 and is used as the first-line therapy for pediatric patients with high-risk neuroblastoma. Dinutuximab is produced in murine myeloma cell line and its mechanism is based on the binding of cell surface glycolipid disialoganglioside (GD2) and lysis of GD2-expressed cells via antibody-dependent cell-mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC). 1 It is almost a new drug against neuroblastoma and is important for the treatment of high-risk neuroblastoma in children.
Neuroblastoma is the most common extracranial tumor that originates from neural crest cells in adrenal medulla and sympathic ganglion during childhood. It constitutes 8% to 10% of all childhood malignancies and is liable for more than 15% of pediatric cancer death. 2 The cause of neuroblastoma derived from primordial neural crest cells is not known exactly. It is thought that neuroblastoma could develop due to neural crest development disorder. Moreover, its genetic basis is associated with ALK (anaplastic lymphoma kinase) gene mutation, PHOX2B gene mutation, and deletion at the 1p36 or 11q14-23 locus.3,4
Neuroblastoma is a heterogeneous disease, and these tumors have the ability to regress or progress spontaneously and are frequently aggressive.2,5 Moreover, pediatric patients with high-risk neuroblastoma have more progression and relapse risk. 2 High-risk neuroblastoma that is seen in 50% of all patients with neuroblastoma is generally related to unfavorable prognosis of neuroblastoma, frequent relapses, and aggressive features, such as loss of heterozygosity of 1p and 11q chromosomes, partial deletions of other chromosomes, tumor amplification of the MYCN oncogene, and metastasis to the liver, bone, and bone marrow.6,7 Rarely, metastasis to lungs and brain parenchyma can occur during relapse and the last period of this disease. 8
The general treatment strategy for high-risk patients with neuroblastoma includes surgery, intensive multiagent induction chemotherapy, radiotherapy, myeloablative consolidation therapy with stem cell rescue and transplantation, 13-cis-retinoic acid therapy, and immunotherapy.9–11 Despite intensive multimodal treatment being included in these therapies, long-term survival rates of pediatric patients with high-risk neuroblastoma are very low. 12 According to the latest cancer statistics, the 5-year survival rate of children with high-risk neuroblastoma is around 23% to 76%. 13 By virtue of the poor outcomes of aggressive treatment and low survival rate, the discovery of new treatment strategies to treat pediatric patients with high-risk neuroblastoma has gained importance.
Chemotherapy, radiotherapy, surgery, myeloablative consolidation therapy, and immunotherapy are current treatment methods for neuroblastoma. The aim of these treatment methods is to reduce tumor burden. Before the tumor spreads to other parts of the body, surgery is done to remove the tumor. Surgery can be a possible treatment for localized tumor. If tumor spreads to other body parts, chemotherapy or other therapy methods are used. Especially, chemotherapy, that is, killing cancer cells with anticancer drugs, has a major role in the treatment of neuroblastoma, and intensity of chemotherapy increases survival rate proportionally.11,14
To treat neuroblastoma, a chemotherapeutic drug or combination of some chemotherapeutic drugs is used. Common chemotherapeutic drugs, such as cyclophosphamide, cisplatin, vincristine, doxorubicin, etoposide, topotecan, busulfan, and melphalan, are used in the treatment of neuroblastoma.11,15 Mostly, combinations of vincristine, cyclophosphamide, and doxorubicin; carboplatin and etoposide; cisplatin and etoposide; ifosfamide and etoposide; and cyclophosphamide and topotecan are used in the treatment. Recently, dinutuximab with granulocyte-macrophage colony-stimulating factor (GM-CSF), interleukin 2 (IL-2), and 13-cis-retinoic acid has been used for the treatment of patients with high-risk neuroblastoma. 16 This article will review dinutuximab in the treatment of pediatric patients with high-risk neuroblastoma.
Dinutuximab
Dinutuximab (ch14.18; Unituxin), a chimeric human-mouse monoclonal antibody (mAb), is an intravenous drug that is used along with GM-CSF, IL-2, and isotretinoin for the first-line multiagent, multimodality therapy of pediatric patients with high-risk neuroblastoma. 17 It is produced in SP2/0 hybridoma cell consisting of human constant regions for the heavy chain IgG1 and the kappa light chain with mouse variable regions.17–19 It targets specifically human GD2, a surface glycolipid antigen on neurons, peripheral pain fibers, and skin melanocytes. 20 This antigen is highly expressed on neuroblastoma cells, but it is also expressed on normal cells of neuroectodermal origin. Neurons limit the expression of GD2 on normal tissue, and the effect of intravenous mAb is inhibited in normal tissue in this way. 21
In immunotherapy of neuroblastoma, mAbs target tumor-associated antigens which include glycoproteins CD56, L1-CAM and GP95 and gangliosides GD2, GD3 and GM3.22,23 Gangliosides, complex, sialic-acid–containing glycosphingolipids, are located on the outer cell membrane, and they have an important role in signal transduction, cell adhesion, and cell recognition. 24 High expression on several tumors and restricted expression on normal tissues make GD2 an ideal target for mAb-mediated immunotherapy.25,26 GD2, surface glycolipid antigen, is highly expressed on the surface of neuroblastoma, glioma, some melanomas, and sarcomas and is expressed on the surface of neurons, skin melanocytes, and peripheral pain fibers at low levels.20,27
Disialoganglioside is expressed homogeneously in neuroblastoma cells and simplifies the attachment of tumor cells to the extracellular matrix. The mechanism of dinutuximab is binding GD2 antigen on neuroblastoma cells and then triggering ADCC and CDC. 1 Dinutuximab promotes antitumor immunity, and the activation of monocytes, macrophages, and dendritic cells enhances antitumor immunity by increasing ADCC. To enhance ADCC activity, it is combined with GM-CSF and IL-2. Granulocyte-macrophage colony-stimulating factor activates macrophages and neutrophils, and IL-2 activates natural killer cells. Clinical studies showed effects of combination of cytokines with dinutuximab on ADCC activity. 20
Interleukin 2 is necessary for the proliferation and activation of natural killer lymphocytes. Natural killer cells have an important role in ADCC, and their membrane contains IL-2R which consists of α, β, and γ subunits. When IL-2 triggers IL-2R, members of the Janus kinase family cytosolic kinases are required as there is no intrinsic kinase activity of IL2R. 28 Triggered IL-2R activates DNA of natural killer cells to be cytotoxic and secretes interferon gamma (IFN-γ) through MKK1/2/ERK.28,29
Natural killer cells involve common effector mechanisms such as granule exocytosis and the death ligand/death receptor system. Granule exocytosis is induced by secretory granules when natural killer cells are activated. The secretory granules integrate with the plasma membrane of natural killer cells and release their contents, perforin and granzymes. Perforin studies demonstrate that perforin creates polyperforin plasma membrane–specific pores in the target cell. 30 Perforin channels in the plasma membrane expedite the entry of other granule proteins, granzymes. The granzymes are allowed to enter the target cell through a perforin channel. Granzyme induces apoptosis via either granzyme B or granzyme A, and granzyme B leads to caspase-dependent pathway and granzyme A to caspase-independent pathway. 31
In death ligand/death receptor–mediated apoptosis, FasL and TRAIL are expressed on the surface of natural killer cells or released as exosome membrane-bound death ligands. FasL and TRAIL can kill tumor cells through their interaction with their respective death receptors. Activation of these effector mechanisms can be recruited by several mAbs. Monoclonal antibodies bind to Fc receptors expressed by natural killer cells and can lead to ADCC. 32 FasL and TRAIL bind to their particular death receptors, Fas for FasL and TRAIL-R1/2 for TRAIL, on target cell membrane and cause caspase-dependent apoptotic cell death. 32 We suggest that dinutuximab can kill neuroblastoma cells using natural killer cells with the effect of IL-2 (Figure 1). The exact mechanism of dinutuximab in death of neuroblastoma cells may be investigated with experimental and bioinformatics studies.
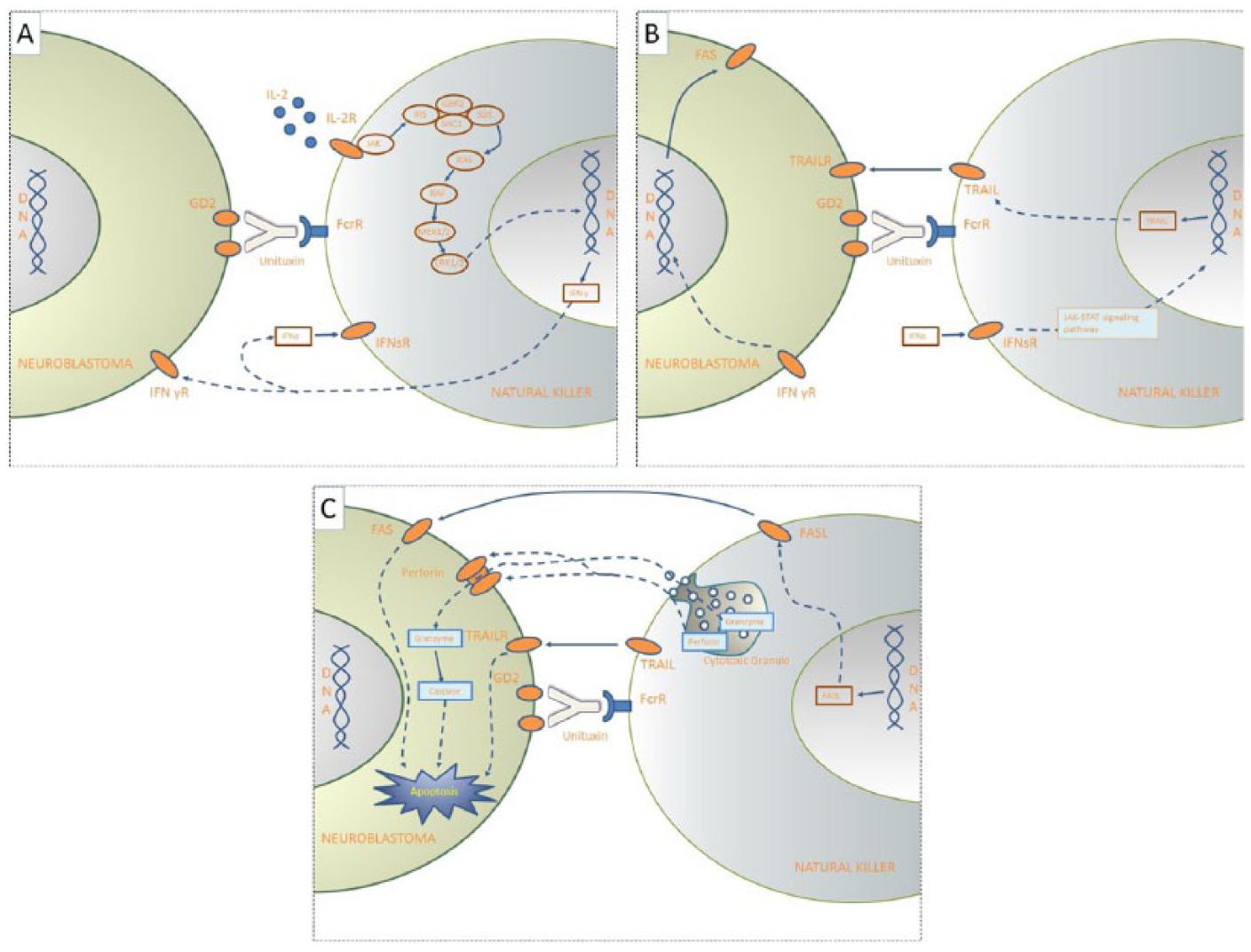
Our predicted death mechanism of neuroblastoma with dinutuximab (Unituxin). (A) Dinutuximab connects neuroblastoma (NB) cells to surface disialoganglioside (GD2) receptors and makes NB recognizable by natural killer (NK) cells. NK cells bind to GD2 via FcrR. Activation of NK cells was triggered by interleukin 2 (IL-2). IL-2 molecules activate NK to produce interferon gamma (IFN-γ) in the signaling pathway shown. IFN-γ stimulates IFN-γR on the surface of NB cells and IFN receptor on NK. (B) IFN-γR activates NB DNA to generate FAS receptor. Besides, induced IFN receptor activates the NK DNA to produce TRAIL by pathway of Janus kinase-signal transducer and activator of transcription (JAK-STAT). TRAIL connects to the NK membrane and effect TRAILR on the NB cells. (C) Cytotoxic granules of NK cells including perforin and granzyme are secreted, and after perforin molecules constitute pore on the NB membrane, the granzymes enter these cells and activate caspase 3. In addition, FasL connects to the NK membrane and effect FAS receptor on the membrane. Granzyme, FAS, and TRAIL cause cell death through apoptotic pathways.
Dinutuximab is completely bioavailable and administered via intravenous injection. 1 Its recommended dose is 17.5 mg/m2/day and is dosed more than 10 to 20 hours for 4 consecutive days for a maximum of 5 cycles. 17 In the study that included 27 children with high-risk neuroblastoma, patients received 5 cycles of treatment with dinutuximab at a dose of 17.5 mg/m2/day via intravenous infusion for more than 10 to 20 hours for 4 consecutive days every 28 days in combination with GM-CSF, IL-2, and retinoic acid, and pharmacokinetic properties of dinutuximab were assessed. This study showed that the maximum plasma concentration of dinutuximab was 11.5 µg/mL, mean volume of distribution at steady state was 5.2 L, clearance was 0.025 L/h, and terminal half-life was 10 days. 20 Administration of a high dose of this drug was associated with an increased uptake of the mAb by the liver and lymphoid tissue and lymph node hyperplasia. No formal metabolism, excretion, or pharmacokinetic drug interaction studies have been conducted. 1
The clinical study that includes 226 pediatric patients with high-risk neuroblastoma evaluated the safety and efficacy of dinutuximab. This clinical study showed that dinutuximab-containing regimen reduces relapse, progression, and secondary malignancy risk according to regimen containing only retinoic acid. 20 This is one of the better outcomes of dinutuximab treatment because high-risk neuroblastoma is characterized with frequent relapses and progression. Although better outcomes in high-risk neuroblastoma treatment were obtained from clinical study, this therapy has some adverse reactions. The most prevalent reason that patients discontinue this therapy is these adverse reactions. Adverse reactions of dinutuximab are pain, pyrexia, thrombocytopenia, lymphopenia, infusion reactions, hypotension, hyponatremia, increased alanine aminotransferase, anemia, vomiting, diarrhea, hypokalemia, capillary leak syndrome, neutropenia, urticaria, hypoalbuminemia, increased aspartate aminotransferase, and hypocalcemia. 17 Moreover, infections, infusion reactions, hypokalemia, hypotension, fever, pain, and capillary leak syndrome are the most common serious adverse reactions of this therapy. Granulocyte-macrophage colony-stimulating factor and IL-2 are responsible for most of these adverse effects. Especially, IL-2 can cause serious adverse reactions such as fever, capillary leak syndrome, and infections. 33 Clinical studies showed that most of the adverse reactions are also seen in regimen containing only retinoic acid with low possibility. 17
Furthermore, dinutuximab can cause neuropathic pain because this mAb can bind GD2 receptors that are found on the surface of myelin and peripheral nerve fibers, and this binding may induce neuropathic pain. 17 In clinical studies, peripheral sensory and motor neuropathy are seen in patients who are treated with dinutuximab-containing regimen, but they are not experienced by patients who are treated with retinoic acid alone.17,20
Future Direction
In recent studies, it was found that some natural products, such as β-carotene, Dunaliella salina, and oleuropein, have both selective cytotoxic activity against neuroblastoma and numerous other biological and pharmacologic activities.34-43
β-carotene, vitamin A, and retinoic acid precursor inhibit differentiation of cancer stem cells and neuroblastoma tumorigenesis and show cytotoxic activity against neuroblastoma.34,35 Also, it plays an important role in immune system by increasing natural killer cell activity.36,37 Moreover, D salina has both selective cytotoxicity against human neuroblastoma cells and an important role in the immune system.38,39 It enhances IFN-γ and IL-2 levels and increases phagocytosis of macrophages and cytotoxicity of natural killer cells. 39
Furthermore, oleuropein, a main phenolic active component in olive leaves, has many biological properties, such as anticancer, antioxidant, antipyretic, anti-inflammatory, and neuroprotective.40,41 Oleuropein shows cytotoxicity against neuroblastoma and decreases invasion, migration, and colony formation in neuroblastoma cells. 42 Also, it serves a function in hematopoiesis, and this function can be useful to treat hematologic disorder. 43 Combination studies of these natural products with dinutuximab in vitro and in vivo may lead to the development of new therapy strategies, decreasing the side effects of dinutuximab.
Antibody-dependent cell-mediated cytotoxicity mediated by natural killer cells, monocytes, macrophages, and neutrophils is an important mechanism against tumor in immunotherapy, and immunotherapy regimens mostly include cytokines to enhance this cytotoxic activity. 44 Especially, IL-2 that is produced by activated T cells has antitumor effect and critical role in immune regulation and is used in clinical experience.45-47 Several studies showed that it increases ADCC activity by activating natural killer cells. 47
Dinutuximab, anti-GD2 mAb, induces ADCC and CDC in neuroblastoma cells by binding to GD2 surface antigen. Moreover, this drug is used together with IL-2 in high-risk neuroblastoma treatment, but it can cause serious side effects, such as fever, capillary leak syndrome, and hypersensitivity reaction. Due to these serious side effects, another cytokine that has the same function and fewer side effects may be used with dinutuximab instead of IL-2.
Interleukin 12 (IL-12) can be a potent prospective cytokine for tumor immunotherapy due to its ability to stimulate natural killer and T cells. It is a proinflammatory cytokine produced by antigen-presenting cells and plays a critical role in immune regulation and anticancer defense.48-50 It regulates natural killer and T-cell function and induces IFN-γ production.50,51 Similar to IL-2 function, IL-12 enhances ADCC activity by stimulating growth and cytotoxicity in T cells and natural killer cells. After assessing the toxicity, side effects, and therapeutic efficacy of IL-12 in combination with dinutuximab, it may be used in this combination therapy instead of IL-2.
Footnotes
Peer review:
Six peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1096 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
The authors contributed equally to this work.
