Abstract
Background:
Symptoms of pseudobulbar affect (PBA) often are misinterpreted as those of a mood disorder. Symptoms seen in patients with PBA can be influenced by a mood disorder and vice versa. Our aim in this study was to determine the occurrence rate of PBA symptoms and comorbid depressive symptoms in a cohort of nursing home patients with different subtypes of neurocognitive disorders (NCD)/dementia.
Methods:
Eighty patients were screened and 61 met the inclusion criteria. These individuals were divided into 5 groups according to NCD subtypes: Alzheimer’s disease, vascular, HIV, unspecified, or combined types. The Center for Neurologic Study–Lability Scale (CNS-LS) and the Geriatric Depression Scale–Short Form (GDS-SF) were used to screen for PBA and depressive symptoms, respectively. The Mini-Mental Status Exam (MMSE) was used to determine patients’ baseline functioning. Fifty-nine patients successfully completed the CNS-LS and 42 completed the GDS.
Results:
Forty-four percent of individuals screened positive for PBA symptoms. Depressive symptoms were found among 23% of these patients. Not only were PBA-symptoms positive patients common among this nursing home residents, the PBA-symptoms positive patients were found to cluster mostly among those with NCD due to HIV (66.0%). PBA symptom-positive patients often received psychotropics.
Conclusion:
Better awareness, screening tools, and treatment approaches for patients with NCD expressing PBA-symptoms—especially those with NCD due to HIV Infection—are needed.
Keywords
Introduction
Pseudobulbar affect (PBA)—also known as affective/emotional lability, involuntary emotional expression disorder, emotionalism, emotional dyscontrol, and emotional incontinence—involves intermittent unpredictable expression of emotions (ie, crying, laughing episodes) that often are unprovoked (sometimes exaggerated) and are incongruent to a triggering situation or context. Known to be partially under voluntary control, these crying/laughing spells can have serious social and emotional effects.
Estimated to affect 1.8 to 7.1 million individuals in the United States,1-4 the pathophysiology of PBA is thought to be secondary to lesions in the cerebro-ponto-cerebellar (CPC) circuitry.5,6 Classified as a disinhibition syndrome thought to stem from a disruption in the serotonin and glutamate pathways,1,2,5-7 PBA can occur in the context of several neurologic disorders.1-4.In healthy individuals the CPC circuitry coordinates appropriate emotional expressions.5,6 In PBA, however, it is thought that damaged CPC circuitry disrupts the ability of the cerebellum to receive critical information and fine-tune an appropriate emotional expression. This leads to symptomatic manifestations observed in patients with PBA.1,2,5-7 As a secondary neurologic disorder, PBA symptoms often manifest in association with several neurologic conditions.1,2,8 The prevalence of PBA is reported to be 29.3% among patients with neurocognitive disorder (NCD) due to Alzheimer’s disease; 26.0% in Parkinson’s disease patients; 45.8% in multiple sclerosis (MS) patients; 44.8% in amyotrophic lateral sclerosis (ALS) patients; 37.8% in stroke patients; and 52.4% in patients with traumatic brain injury (TBI).2,3,8 Although PBA predominantly is associated with these 6 conditions, 4 symptoms also have been reported with viral cerebellitis, multiple system atrophy, supranuclear palsy, as well as adverse effect of medications such as paroxetine, sumatriptan, and ziprasidone.3,9-11
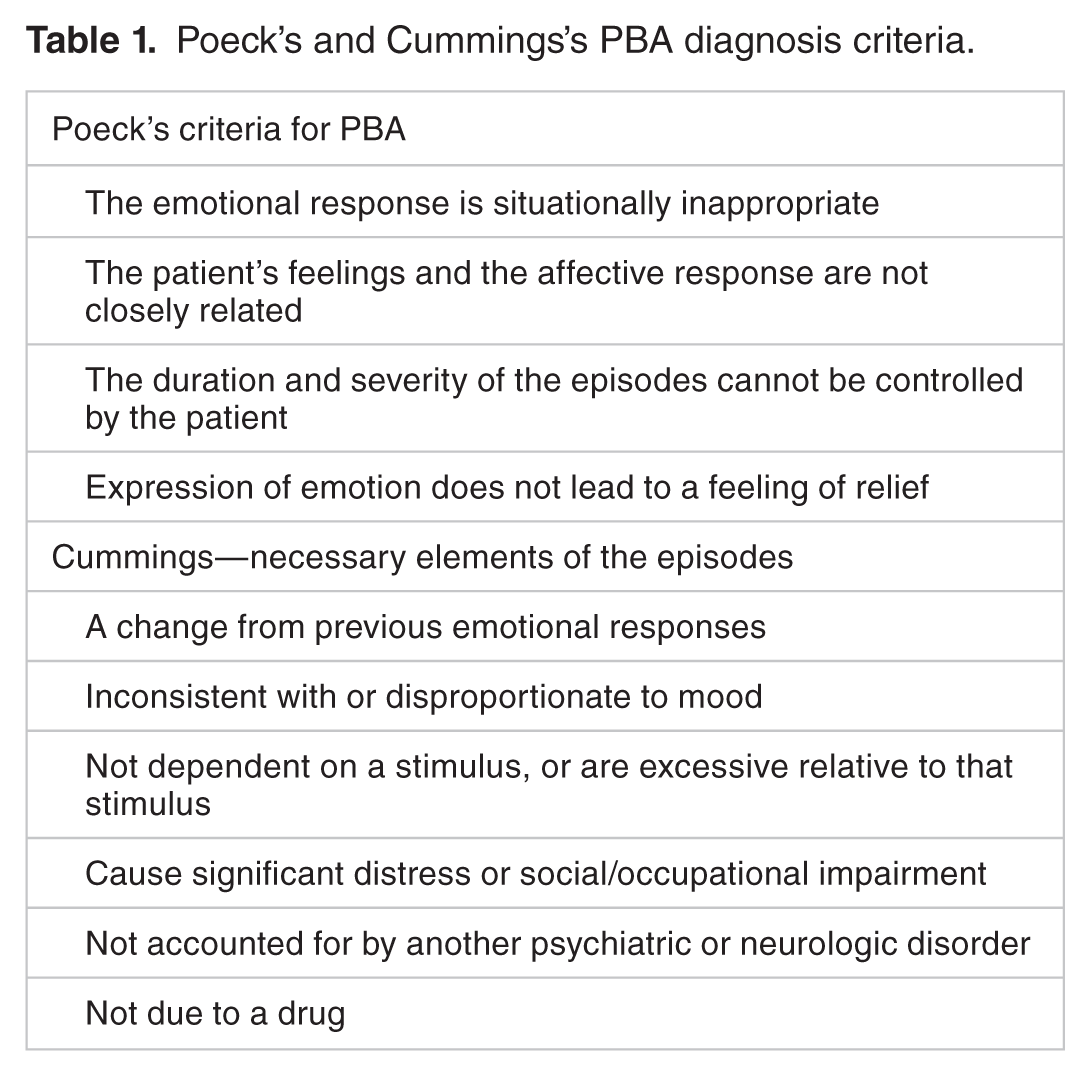
PBA symptoms often are brought to clinical attention by the patient’s caretaker or are observed by the clinician as part of a structured neurologic examination. 1 Most providers use Poeck’s 4 criteria or the revised version by Cummings (Table 1)1,8,12 during the initial diagnostic process. PBA symptoms often are misdiagnosed as a mood disorder. In a survey estimating the prevalence of PBA symptoms, 41% of 937 respondents did not receive any diagnosis for their laughing and crying episodes and most clinicians attributed these symptoms to a depressive episode or as part of the patient’s underlying neurologic illness.1-4 Confusing PBA symptoms with those of a primary mood disorder often results in patients being started on a psychotropic.1,2,13 Depression often is comorbid with PBA, and the patient’s confused and embarrassed reaction to their PBA symptoms can lead to impaired social interaction, depression, diminished quality of life, and increased caregiver burden.1,2,3,8,13 In PBA emotional expression or affect lasts for seconds to a few minutes. Major depressive disorder persists weeks to months, with additional criteria outlined in DSM-5.1,2,4,5,14
Poeck’s and Cummings’s PBA diagnosis criteria.
Many medications have been used off-label for treating PBA. Antidepressants including tricyclic antidepressants and selective serotonin reuptake inhibitors have shown some success.1,2 Dextromethorphan-quinidine (a combination of dextromethorphan and an ultra-low dose quinidine; DMQ) is the only FDA-approved medication for PBA.1,2
Having observed many missed PBA cases in an outpatient psychiatric clinic during psychiatric residency, we want to see how common PBA was among our nursing home residents. We evaluated how many individuals in this community nursing home: (1) were experiencing PBA symptoms, and (2) how often their PBA symptoms were recognized or misdiagnosed. Our hypotheses were that: (1) the prevalence of PBA symptomatology among patients with NCD at our nursing home would be similar to previously reported rates of 29.3% 2 ; (2) that there would be a high rate of comorbid PBA symptoms and depressive symptoms; and (3) that PBA symptom-positive patients would be receiving several psychotropic medications.1,2
Methods
This study focused on patients with PBA symptoms—defined as experiencing emotional responses to situations that were inappropriate to triggering events or situations and represent a variation from the patient’s baseline emotional functioning. A PBA diagnosis indicated assessment by a neurologist using standard diagnostic practices.
We conducted a cross-sectional study to determine the rate of PBA symptom occurrence, and comorbidity with depressive symptoms in a cohort of patients with NCD/dementia in an inner-city community hospital nursing home. A convenience sample of 40 residents on the non-Alzheimer’s NCD/dementia unit (ie, dementia due conditions other than Alzheimer’s disease) and 40 residents from a NCD due to Alzheimer’s disease (Alzheimer’s dementia) unit were screened; 61 meet inclusion criteria. None of the patients chosen for this study had a previous PBA diagnosis. The nursing home was a nonprofit, voluntary, residential health care unit affiliated with a community hospital. The NCD subtypes seen at the nursing home include NCD due to Alzheimer’s disease, unspecified NCD, vascular NCD, frontotemporal NCD, NCD with Lewy bodies, NCD due to TBI, NCD due to HIV infection, NCD due to another medical condition (such as Parkinson’s disease), and NCD due to multiple etiologies. Psychiatric disorders among our sample included depression, bipolar disorder, schizophrenia, and schizoaffective disorder. The hospital’s institutional review board approved the study and data collection occurred from October 2015 to March 2016. The project’s inclusion criteria were: age ⩾18, in residence at the nursing home, having a documented primary diagnosis of NCD regardless of type, and having the ability to provide informed consent or through a surrogate medical decision maker. Non-residents of the nursing home, those with inability to provide informed consent (from either the patients or his/her surrogate), and those having no documented diagnosis of NCD were excluded.
Patients’ information was collected using forms designed for the project. All 61 patients who met the inclusion criteria were evaluated for depression and PBA symptoms with screening questionnaires. Not all patients were able to complete the questionnaires because of behavioral issues and/or severe limitation caused by NCD. The self-administered Center for Neurologic Study–Lability Scale (CNS-LS) is suggestive—but not diagnostic—of the condition and has been used to measure PBA symptom frequency and severity.1,2,4,15,16 A PBA diagnosis is suspected with a score ⩾13.1,2,15,16 Only 59 patients completed the CNS-LS scale. We used the 15-item rater-administered Geriatric Depression Scale—Short Form (GDS-SF), which is validated for institutionalized older adults,17,18 to assess for depression. However, only 42 patients were able to complete the scale. The Mini-Mental Status Exam (MMSE), already documented in the patients’ chart, was used to define the patient’s baseline cognitive status and impairment severity. Individuals with mild cognitive impairment (MMSE ⩾20) consented to participate in the study; surrogate decision makers provided consent for patients with a MMSE score <20 (considered moderate-to-severe cognitive impairment). Although these scales were used to rule in conditions in question (PBA and depressive symptoms), neither the CNS-LS (for PBA) nor the GDS-SF (for depressive symptoms) was used as a substitute for a formal diagnosis.
The identifying variables included in the data collection and analysis were the patient’s sex, age, primary diagnosis, medications, race/ethnicity, medical record number, psychiatric illnesses, medical comorbidities, CNS-LS score, MMSE score, and GDS-SF score. All participants were de-identified and assigned a research number attached to the data collection forms. All data collected were placed in an encrypted, password-protected spreadsheet accessible only by the principal investigator and the project coordinators with the corresponding patient’s initial matching the assigned research number. At the end of the data collection, patients found to be positive for PBA symptoms (CNS-LS ⩾13) were informed of the outcome of the screening test (via either the patient or the surrogate decision maker) and their primary care providers were informed for additional diagnostic evaluations and treatments. A descriptive analysis of the data obtained on the PBA symptom-positive patients and depressive symptom-positive patients was performed.
Results
A total of 61 participants initially were enrolled in the study. Of these, 29 participants (47.5%) were male, 48 (78.7%) were African American, 11 (18%) were Hispanic, and 2 (3.3%) were white. Based on a review of the participants’ medical records, the breakdown of primary diagnoses was: NCD due to HIV infection in 30 individuals (49%), NCD due to Alzheimer’s disease in 18 individuals (29%), vascular NCD in 4 individuals (7%), NCD due to multiple etiologies (combined type) in 1 individual (2%), and unspecified NCD for 8 individuals (13%). The patients’ age ranged from 40 to 79 years. After initial enrollment, 2 patients withdrew from the study because of NCD-associated behavioral difficulties. Of the remaining 59 participants, 26 (44%) scored ⩾13 on the CNL-LS, suggesting PBA symptomatology. Among the NCD due to HIV infection group, 20 participants (66%) screened positive for PBA symptomatology compared with 5 individuals (29.4%) from the NCD due to Alzheimer’s disease group and 1 patient from the unspecified NCD group. No participants from the vascular NCD type nor the NCD due to multiple etiologies (combined-type) type groups screened positive for PBA symptomatology.
Using the GDS-SF test scoring guideline, a score ⩾10 was an indicator of “definite” depression; while a score of ⩾5 was determined to be “possible” depression. Of the 42 patients who successfully completed the questionnaire, 18 (43%) screened positive for depressive symptoms: 6 (14%) scored in the “definite depression” range and 12 (29%) scored in the “possible depression” range. The remaining 24 patients (57%) had a negative screen for depressive symptoms. Although on average the GDS-SF scores for individuals with PBA symptoms were similar to those without PBA symptoms, patients who had an additional psychiatric diagnosis (ie, a mood disorder or a psychotic spectrum disorder) PBA symptomatology was more prevalent (65.4%) compared with those who did not (36.4%) (Table 2). A significant difference was found between the use of antidepressants, antipsychotics, and/or mood stabilizers in PBA symptom-positive patients compared with PBA symptom-negative patients, similar to previously reported findings.1-3 Psychotropic use was higher among the PBA symptom-positive group compared with the PBA symptom-negative group. More specifically, there was a higher number of PBA symptom-positive neurocognitively disordered patients taking mood stabilizers (Table 2).
Prevalence of PBA symptoms with comorbid psychiatric disorder and psychotropic medications.
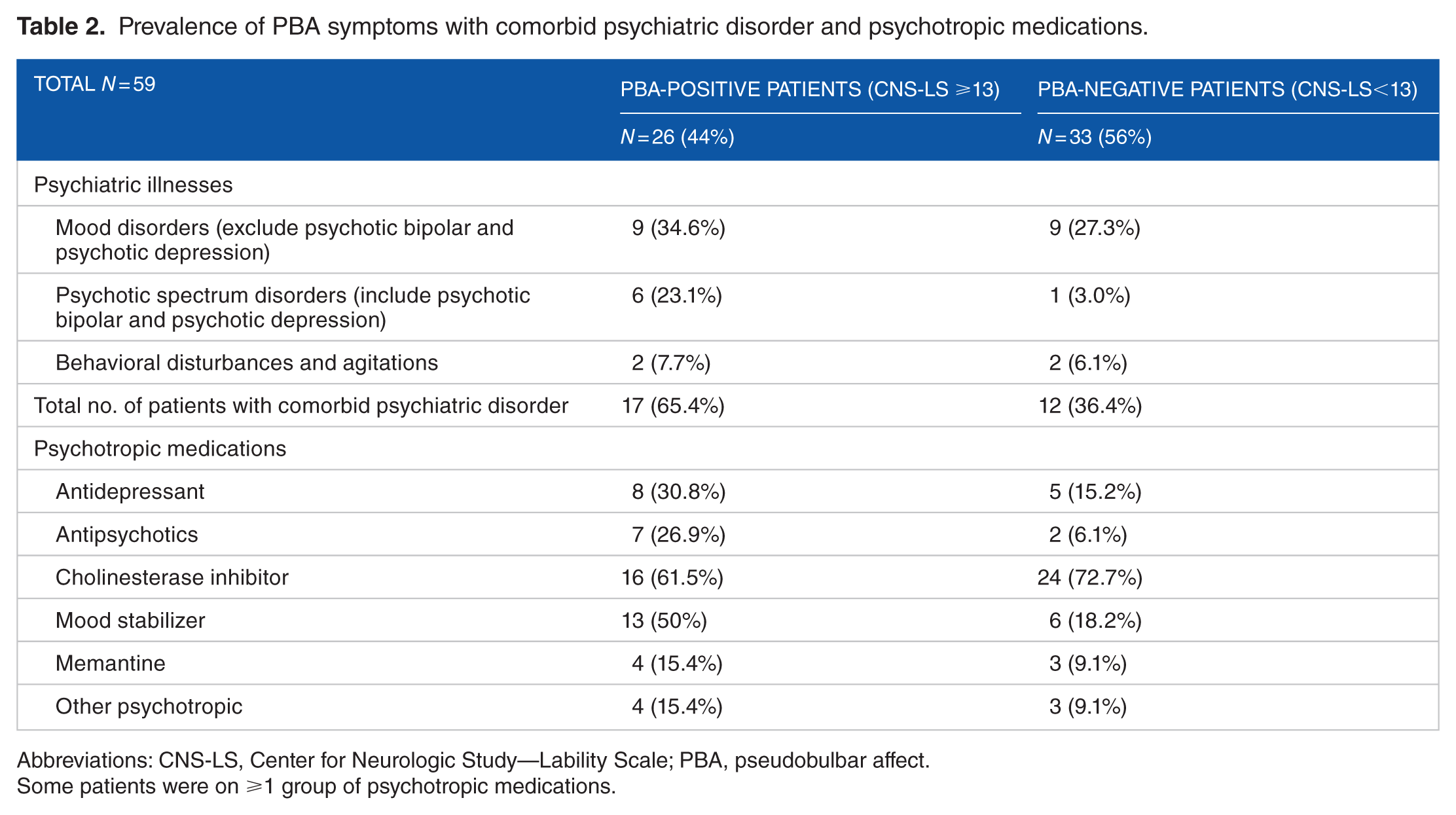
Abbreviations: CNS-LS, Center for Neurologic Study—Lability Scale; PBA, pseudobulbar affect.
Some patients were on ⩾1 group of psychotropic medications.
Discussion
The goal of our study was to determine the prevalence of PBA symptoms among nursing home residents with NCD using the CNS-LS scale. We also aimed to establish the existence of comorbid PBA symptom-positive and depressive symptom-positive (using the GDS-SF) in this cohort of nursing home residents with different subtypes of NCD. Our initial hypothesis that there was a high prevalence of PBA symptom-positive patients with NCD among these nursing home residents was confirmed. We found that PBA symptom-positive patients were common among these nursing home residents, and these patients were clustered among those with NCD due to HIV infection. The study also found that 23.1% of PBA symptom-positive patients in this cohort experienced depressive symptoms, a finding that is in accord with previous reports. 19 In other words, many patients with NCD have coexisting PBA and depressive symptoms. That these comorbid symptoms were seen mostly among patients with NCD due to HIV infection is an area for further investigation.
This study has limitations. As a descriptive study, the available data cannot be used to make deeper and more complex statistical inferences. This study can only confirm a high rate of NCD patients also have symptoms of PBA and a depressive disorder, and that this link was more prevalent among those with NCD due to HIV infection. The small sample size limits the power of our data and our ability to make more generalized conclusions. The overall finding might not be representative of all patients with NCD. An additional limitation is the lack of a comparison groups that could have facilitated broader and more specific statistical analysis and correlation studies among the NCD subtypes. This impedes our ability to perform deeper statistical analyses of the results. Although the CNS-LS questionnaire was used to rule in PBA symptomatology, this scale is validated only for ALS 15 and MS. 16 It is possible that the limitations of these screening tools are reproduced in our research findings.
Additional issues include the specificity of our screening data for ruling in PBA symptom-positive patients because of our cutoff score of 13. The CNS-LS and the GDS are both are self-report measures with their own limitations. Further segregating the data to ascertain the difference in medication use between those with vs those without PBA by removing those with comorbid psychiatric conditions from the sample and re-running the analyses would have rendered our sample size too small to be useful. Although this exercise could have given us a clearer picture about which agents are prescribed to PBA-symptom positive patients, the small sample- size was a limiting factor. Recall bias could be a factor as well because the screening scales asked the caregiver and/or patient to remember behavior in the past weeks or months. Inter-rater reliability of the MMSE results obtained from the patient’s medical records and the limitations of a self-report to inform on lability could call into question the overall reliability of the data.
There is a high prevalence of PBA misdiagnosis as a primary mood disorder among patients with subtypes of NCD. Because the symptom path to these 2 conditions might not be clearly elucidated, patients often are misdiagnosed and inappropriately treated with psychotropics, which could lead to adverse effects among geriatric patients.
Our results confirm that PBA symptoms are underdiagnosed and undertreated among nursing home residents with different subtypes of NCD. Patients with NCD due to HIV infection were found to express PBA symptoms at a higher rate (66.0%) compared with patients with other forms of NCD. Additionally, PBA symptom-positive patients often were treated with psychotropics. To our knowledge, this is the first research attempting to identify PBA symptom-positive patients according different dementia subtypes. Most significant research in this area has focused on the rate of expressed PBA symptoms in patients with NCD due to Alzheimer’s disease.2,19 More focused studies describing the etiology of the high prevalence of PBA symptomatology among patients with NCD due to HIV infection, as well as of the high rate of depressive symptoms among PBA symptom-positive patients are needed. More refined screening tools aimed at patients with NCD exhibiting PBA symptoms also are required.
Footnotes
Acknowledgements
The authors wish to thank the Mueen Ahmad, MD, Sheeima Paramatmuni, MD, and Carolyn Springer, PhD for their contributions to this project.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
